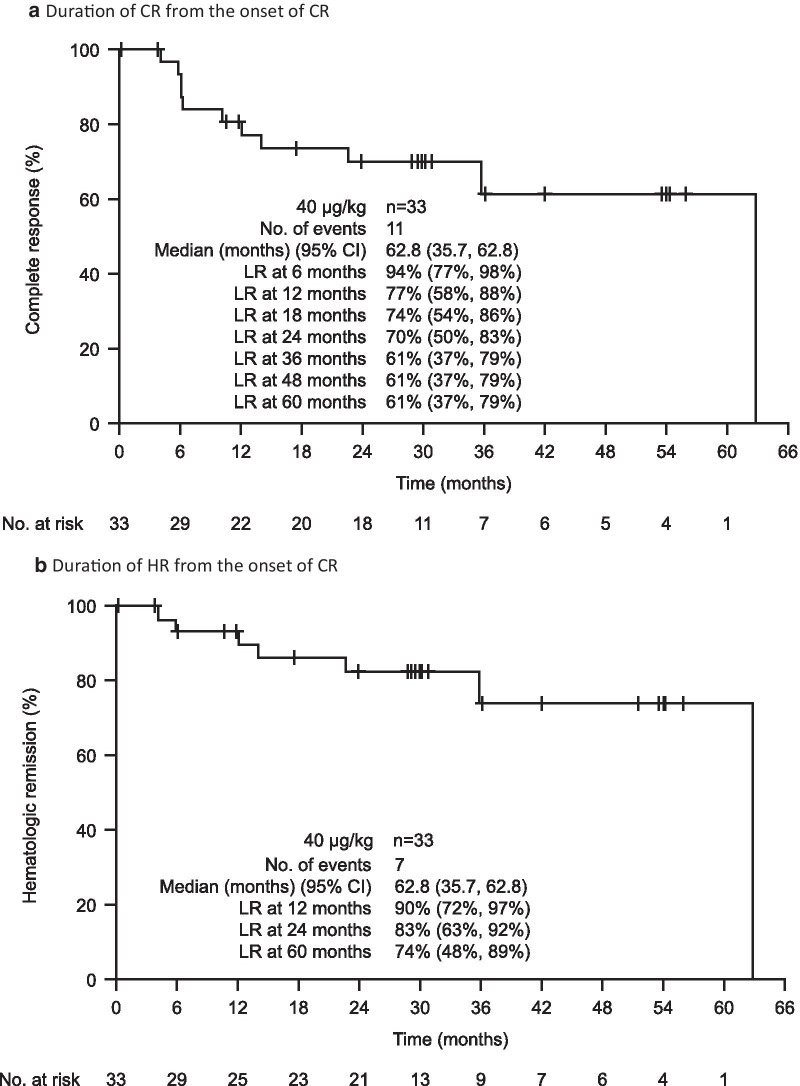
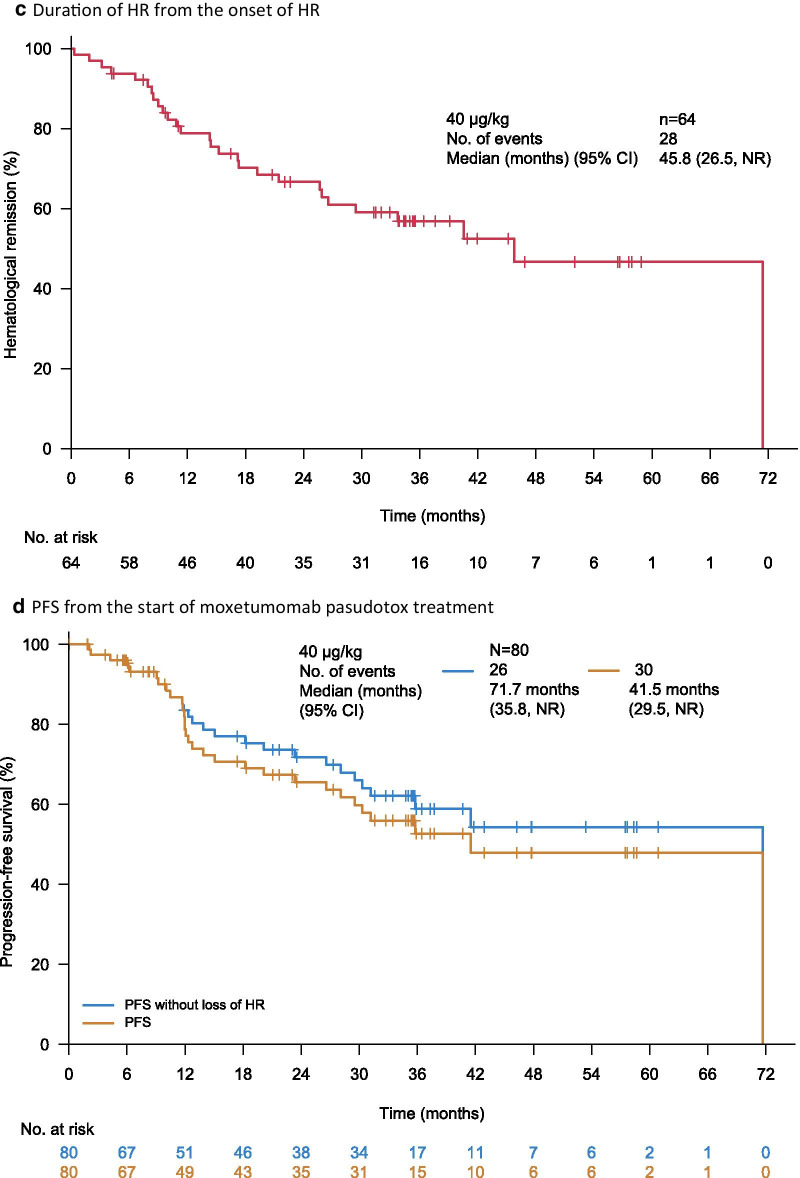
Fig. 1.
Moxetumomab pasudotox provided durable responses in heavily pre-treated patients. As assessed by BICR in the ITT population, Kaplan–Meier plots of a duration of CR and b duration of HR, in patients with CR, c duration of HR in all patients with HR, and d PFS. HR is defined as hemoglobin ≥ 11.0 g/dL, absolute neutrophil count ≥ 1.5 × 103/µL, and platelet count ≥ 100 × 103/µL, without receiving transfusions or growth factors within the preceding 4 weeks of assessment. BICR blinded independent central review, CI confidence interval, CR complete response, HR hematologic remission, ITT intent-to-treat population, LR landmark rate, NE not evaluable, NR not reported, OR objective response, PFS progression-free survival