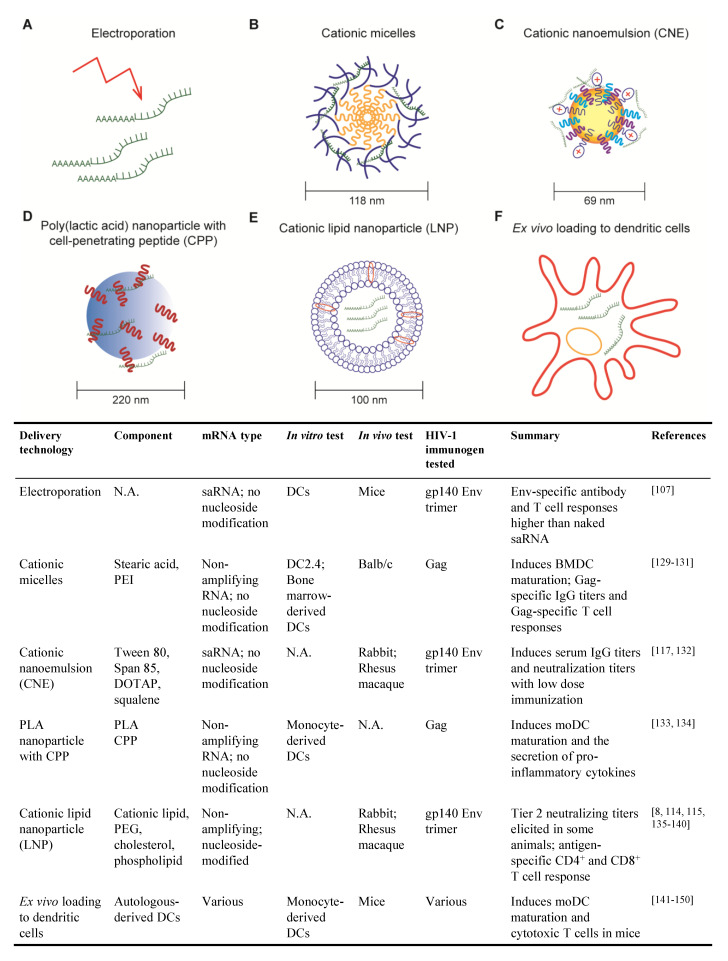
Figure 2.
Delivery methods for HIV mRNA vaccine. (A) Electroporation; (B) Cationic micelles composed of stearic acid (yellow) and Polyethylenimine (PEI) (blue); (C) Cationic nanoemulsion (CNE); The yellow core shows squalene. Surfactant such as Tween 80 and Span 85 are shown in blue and purple. Cationic lipid DOTAP (Dioleoyl-3-trimethylammonium propane) is shown with red ‘+’ mark; (D) poly(lactic acid) (PLA) nanoparticle (blue core) with cell penetrating peptide (CPP) (shown in red); (E) Cationic lipid nanoparticle (LNP); figure shows a lipid bilayer with other components that can be included, such as Polyethylene glycol (PEG), cholesterol or phospholipid; (F) Ex vivo loading of dendritic cell (DC). The table below summarizes the HIV mRNA vaccines that have been tested, detailing the delivery method, the type of mRNA, the in vitro and in vivo system used for testing, the mRNA-encoded immunogen and a short note on the results.