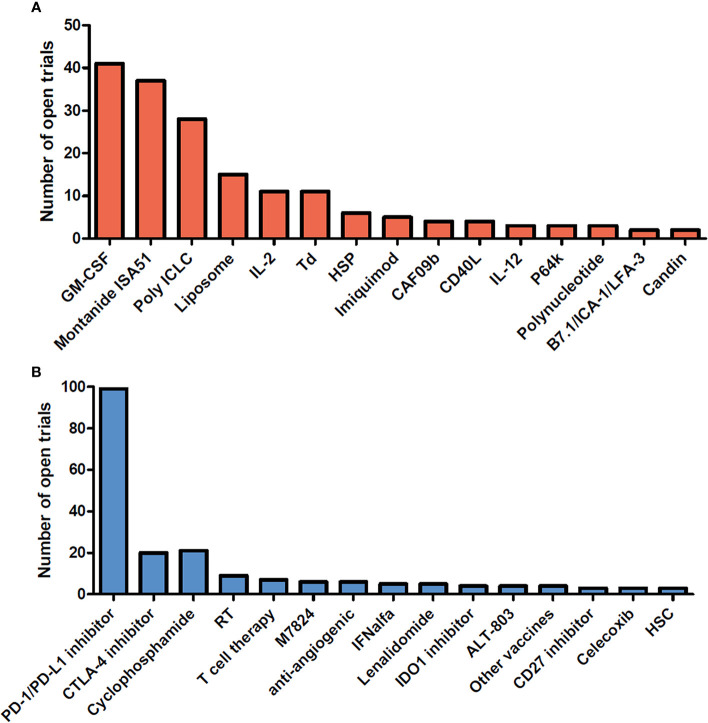
Figure 2.
Adjuvants and combinatorial immunomodulatory therapies being used in cancer vaccine trials. Cancer vaccine trials listed as open at ClinicalTrials.gov on August 2020. The number of trials using each adjuvant (A) and associating each immunomodulatory therapy with the cancer vaccine (B) are shown in the bar graph. Adjuvants and combinatorial therapies used in less than 2 clinical trials are not shown. GM-CSF, Granulocyte-macrophage colony-stimulating factor; IL-2, interleukin-2; Td, Tetanus/diphtheria toxoid; HSP, heat shock protein; CAF09b, cationic liposomes (DDA-MMG1) with complex bound synthetic double-stranded RNA (Poly(I:C)2); IL-12, Interleukin- 12; P64k, Neisseria meningitides protein; PD-1, Programmed cell death 1; PD-L1, Programmed cell death ligand 1; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; RT, radiotherapy; M7824, fusion protein composed of a human IgG1 monoclonal antibody against PD-L1 fused with 2 extracellular domains of TGF-βRII; IFNalfa, Interferon alfa; IDO1, indoleamine 2,3-dioxygenase 1; ALT-803, IL-15 superagonist; Other vaccines, Salmonella, pneumococcal vaccines; HSC, hematopoietic stem cells.