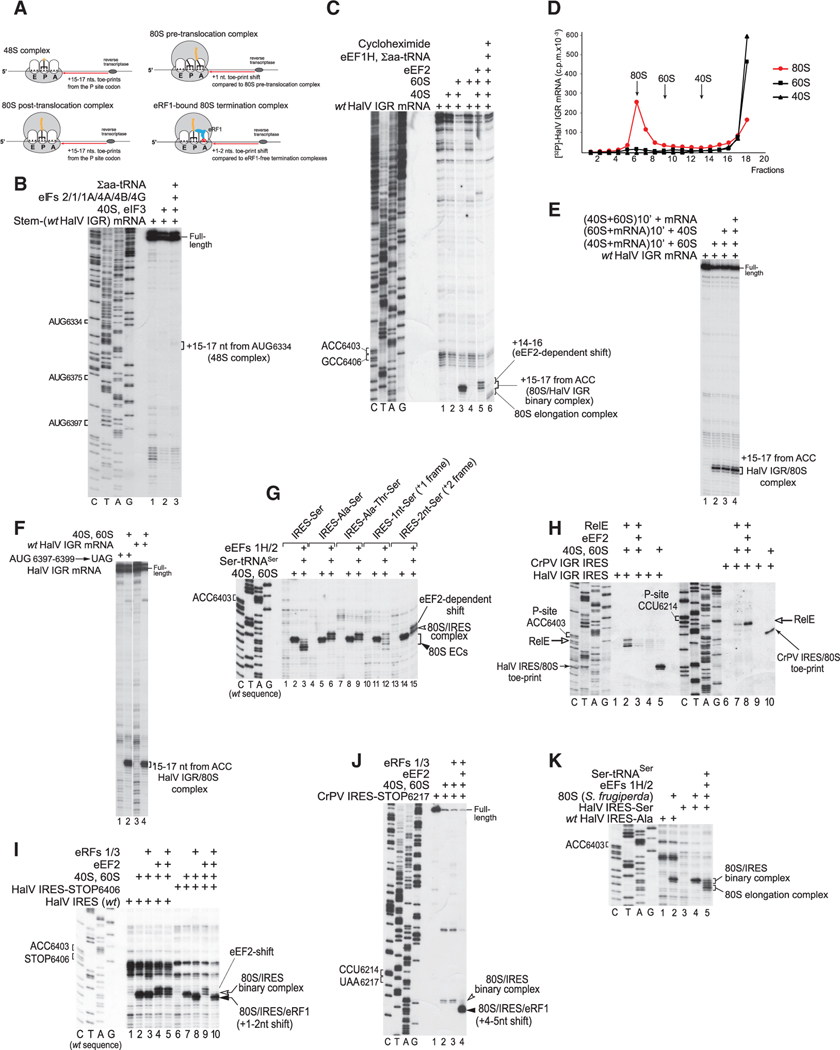
Figure 1. The Mechanism of Initiation on the HalV IGR IRES.
(A) Schematic representation of various ribosomal complexes showing the relative positions of their toe-prints.
(B) 48S complex formation on HalV IGR mRNA comprising a 5′-terminal stem (ΔG = −25.80 kcal/mol) followed by nucleotides 6211–7460 of the HalV genome in the presence of 40S subunits, Σaa-tRNA, and canonical eIFs, as indicated, assayed by toe-printing. The positions of AUG codons in the HalV IGR and assembled ribosomal complexes are indicated.
(C) Direct binding of the HalV IGR mRNA to 80S ribosomes followed by elongation upon addition of eEF1H, eEF2, Σaa-tRNA, and cycloheximide, assayed by toeprinting. The positions of the A site and P site codons of the HalV IGR IRES are shown on the left. The positions of ribosomal complexes and the eEF2-mediated toe-print shift are indicated on the right. Separation of lanes by white lines indicates that they were juxtaposed from the same gel.
(D) Association of 32P-labeled HalV IGR-containing mRNA with individual 40S and 60S ribosomal subunits and 80S ribosomes, assayed by sucrose density gradient centrifugation.
(E) Toe-printing analysis of ribosomal association of the HalV IGR IRES depending on the order of incubation of mRNA, 40S ribosomal subunits, and 60S ribosomal subunits. The position of IRES/80S complexes is indicated.
(F) Ribosome-binding activity of the AUG6397-6399→UAG stop codon HalV IGR mRNA mutant, assayed by toe-printing. The position of IRES/80S complexes is indicated.
(G) The fidelity of reading frame selection on the HalV IGR IRES investigated by the ability of 80S/IRES complexes formed on GCC6406-6408(Ala)→UCU(Ser), ACU6409-6411(Thr)→UCU(Ser), and AUU6412-6414(Ile)→UCU(Ser) HalV IGR variants and IGR mutants with UCU(Ser) placed in the +1 or +2 reading frame by insertion of one (G) or two (GC) nucleotides. between ACC6403–6405 and UCU6406–6408(Ser) codons to undergo one-cycle elongation in the presence of eEF1H, eEF2, and Ser-tRNASer, assayed by toe-printing. The positions of IRES/80S binary complexes and 80S elongation complexes (80S ECs) are shown on the right.
(H) Comparison of the eEF2 dependency of the A site accessibility in 80S ribosomal complexes assembled on HalV and CrPV IGR IRESs, assayed by RelE cleavage. Sites of RelE cleavages were determined by primer extension. The positions of P site codons, RelE cleavages, and IRES/80S control toe-prints are indicated.
(I and J) Comparison of the eEF2 dependency of the A site accessibility in 80S ribosomal complexes formed on HalV and CrPV IGR IRESs by binding of eRF1 and eRF3 to 80S ribosomes assembled on (I) HalV IRES-STOP(UAA) and (J) CrPV IRES-STOP(UAA) mutant mRNAs, assayed by toe-printing. The positions of ribosomal complexes and P site and stop codons are indicated.
(K) The ability of HalV IGR IRES to form elongation-competent complexes with insect (Spodoptera frugiperda) 80S ribosomes, assayed by toe-printing. The positions of the A site codon and ribosomal complexes are indicated. (B, C, and G–K) Lanes C, T, A, and G depict CrPV or HalV sequences, as indicated.