Supplemental Digital Content is available in the text.
Keywords: baroreflex, digoxin, orthostasis, modified oxford method, postural tachycardia syndrome, pyridostigmine
Abstract
Upright postural tachycardia syndrome (POTS) resembles hemorrhage with reduced central blood volume, parasympathetic withdrawal, and sympathetic activation. Baroreflex dysfunction causes low heart rate variability, enhanced blood pressure variability, and decreased maximum baroreflex gain (Gmax) putatively measured by spontaneous fluctuation of blood pressure and heart rate. We investigated whether/how cardiovagal baroreflex in POTS differ from control, supine, and upright by comparing indices of spontaneous baroreflex function to that measured using the reference standard modified Oxford method. This uses sodium nitroprusside and phenylephrine to generate the sigmoidal cardiovagal baroreflex curve. Baroreflex in POTS was evaluated supine and upright untreated and then treated to determine whether pyridostigmine or digoxin (a vagotonic agent) corrects baroreflex deficits. Supine, Gmax was reduced by 25% in POTS compared with controls, and descriptors of this sigmoidal relationship showed a reduction, downward shift, and left shift of the response to the pharmacological decrease and increase in blood pressure. Digoxin normalized supine cardiovagal baroreflex while pyridostigmine resulted in partial normalization as Gmax, and other descriptors of these relationships were similar to control. Upright, cardiovagal curves were distorted and displaced in untreated POTS, while digoxin and pyridostigmine left shifted the cardiovagal curves due to sympathetic activity. Cardiovagal baroreflex deficits in POTS relate to parasympathetic withdrawal while supine, remediated completely by digoxin, and sympathetic activation upright through alteration of baroreflex responsivity. Since these baroreflex effects resemble those measured following microgravity/chronic bedrest, vagotonic/sympatholytic treatment combined with aerobic exercise might normalize the cardiovagal baroreflex and provide therapeutic benefit in patients with POTS.
See Editorial, pp 1245–1247
Standing upright (orthostasis) produces central hypovolemia by translocating blood to the dependent body. Orthostatic intolerance is defined by signs and symptoms, such as lightheadedness, exercise intolerance, tachycardia, hypotension, hypertension, headache, fatigue, cognitive deficits, and nausea, while upright relieved by recumbence.1,2
Postural tachycardia syndrome (POTS) is defined in adults by orthostatic intolerance symptoms plus an excessive increase in heart rate exceeding 30 beats/min within 10 minutes of standing or upright tilt.3 Systolic arterial pressure (SAP) is maintained and may increase.4 Larger heart rate increments by 40 beats/min or more are required to diagnose POTS in younger patients <19 years old.5–7 More than 85% are women aged 14 to 50 years.8
POTS resembles hypovolemia with reduced systemic venous return and reduced cardiac output (CO).9 In some instances, absolute hypovolemia is found10; in others, a redistributive central hypovolemia occurs when upright.11 The latter is designated neuropathic POTS and results from decreased adrenergic vasoconstriction in the legs,12 or splanchnic vasculature5,13 causing baroreflex unloading, decreased reflex cardiovagal stimulation, increased reflex sympathetic excitation,14 and reflex tachycardia.15 An alternative form of POTS in which sympathetic adrenergic function is primarily increased is denoted hyperadrenergic POTS.
In both cases, baroreflex regulation is affected. Changes in blood pressure (BP) during orthostasis are primarily buffered by arterial baroreflexes, which are negative feedback inhibitory stretch reflexes.16 An increase in arterial pressure causes decreased heart rate mediated primarily by parasympathetic (cardiovagal) efferent activity, decreased peripheral vasoconstriction mediated primarily by reduced vascular synaptic sympathetic activity, and suppression of adrenal medullary epinephrine secretion POTS patients have normal muscle sympathetic nerve activity while supine, but increased muscle sympathetic nerve activity when upright compared with healthy controls17 suggesting a reflex response with intact arterial baroreflexes afferents.
Since POTS is characterized by tachycardia, investigations have frequently focused on cardiovagal baroreflex impairments. Methods, such as transfer function analysis, are often used that measure small spontaneous fluctuations of HR (or RR interval) and SAP to obtain a linear frequency domain relationship between RR variability and SAP variability near 0.1 Hz, low frequency in which the slope, ΔRR/ΔSAP, is thought to represent the sensitivity or gain characterizing the cardiovagal baroreflex.18,19 Low-frequency SAP oscillations are thought to reflect sympathetic activity,20 while low-frequency RR interval oscillations result from cardiovagal baroreflex transduction of SAP oscillations.21 The slope of ΔRR/ΔSAP is typically reduced in POTS21 compared with controls supine and even more when upright.
Since spontaneous fluctuations generate information over a very narrow range of arterial pressures their ability to accurately represent baroreflex function, particularly during orthostatic stress, is questionable.22 This is because the cardiovagal baroreflex is often characterized by its slope (sensitivity, gain) at a single point referred to as the operating point, the particular SAP and its corresponding RR interval defining the state of the system at which the slope is measured. But cardiovagal baroreflex cannot be defined at a single point. Rather, it is defined by the nonlinear, functional relationship between the RR interval and the SAP, closely fit by a sigmoidal (logistic) function over a wide range of arterial pressures.23
Slopes obtained at operating points cannot define the cardiovagal baroreflex, but rather represent a changeable aspect of the current state of the system, corresponding to the variation of RR interval with variation of SAP deriving from spontaneous fluctuations of RR interval and SAP. Results are only applicable near that particular operating point24 and not elsewhere; the assumption that the slope at the operating point estimates the maximum cardiovagal baroreflex gain, Gmax, may be erroneous. As shown below in Figure 1, if the operating point is near the centering point where the curve is nearly linear (ie, operating point 1), its slope will closely approximate Gmax; otherwise, the slope is less than Gmax and can neither accurately reflect Gmax nor assist in constructing the baroreflex relationship.24 Thus, for example, if the SAP corresponds to operating point 2, the slope at that point is much smaller than Gmax.
Figure 1.
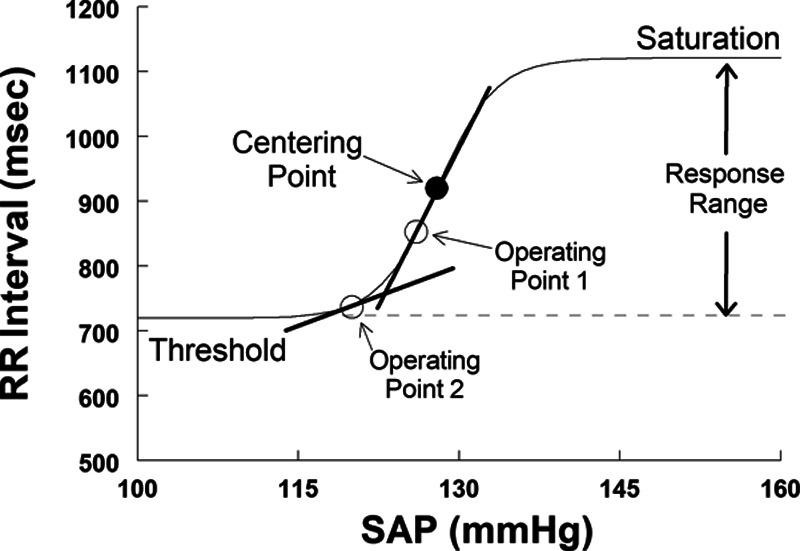
It shows an idealized cardiovagal baroreflex as a sigmoidal relationship between systolic arterial pressure (SAP) and RR interval over a wide range of pressures. The sigmoid curve can be uniquely specified by 4 parameters: the systolic blood pressure (BP; SAP) at the centering point, the slope at the centering point (maximum gain or baroreflex sensitivity, Gmax), the curve’s threshold, and its response range. Operating points define the current state of the system; local slopes at operating points and correspond to the variation of RR interval with variation of BP (BPV) near 0.1 Hz near that particular operating point.
We have shown previously in healthy volunteers that orthostasis shifts or resets the centering point, saturation, and threshold en bloc, while Gmax and response range are unchanged.25 BP can be modestly affected resulting in different operating points. Resetting is attributable to the modulating effects of cardiopulmonary baroreflexes on arterial baroreflexes with decreased cardiac filling in the upright position.25 Often, operating points are almost directly displaced downwards, and the local slope at OP-upright may not adequately estimate Gmax.
The purpose of this investigation was, therefore, to examine whether and how cardiovagal baroreflex relationships in POTS differ from healthy controls in both the supine and upright positions. We used the so-called modified Oxford method to construct the sigmoidal baroreflex curve and tested whether parasympathetic stimulation with pyridostigmine (Mestinon)26 or digoxin27 can correct baroreflex deficits.
Methods
The data that support the findings of this study can be made available from the corresponding author upon reasonable request.
Subjects
We enrolled 36 female patients with POTS aged 15 to 30 (mean=21.7±7.7 SD) and 20 healthy female volunteer subjects aged 18 to 24 years (median=21.3±5.2 SD years) of similar weight, height, and body mass index. POTS subjects fulfilled POTS criteria defined previously. Symptoms of orthostatic intolerance were present on a daily basis for >6 months and were relieved once supine. Symptoms were replicated during a prior 10 minutes, 70° upright tilt showing excessive tachycardia without hypotension or syncope. A similar upright tilt test on control subjects showed the absence of orthostatic intolerance. Only female subjects were employed because we could not sufficiently power a study of male patients with POTS as the majority of patients with POTS are female.8 All subjects were free of systemic or infectious disease. Subjects were either therapeutically naive or weaned off drugs for at least 2 weeks. Only nonpharmaceutical birth control was employed by subjects during the study. Subjects did not use nicotine. Subjects refrained from caffeine for at least 72 hours before testing. Subjects fasted for a minimum of 4 hours before testing. There were no trained athletes or bedridden subjects. Studies were performed in the midluteal phase when autonomic tone is maximized.28 Informed consent was obtained from all participants. All protocols were approved by the Committee for the Protection of Human Subjects of New York Medical College.
Protocol
Testing began at 9:30 am. Subjects were instrumented for ECG, beat-to-beat AP using a Finometer (FMS, Amsterdam) calibrated against an oscillometric BP cuff. CO was estimated using the Finometer which models the circulation as an adaptive Windkessel. Systemic vascular resistance was computed using the formula systemic vascular resistance = mean arterial pressure/CO (mm Hg/L per minute). This was initially calibrated against an inert gas rebreathing (Innocor, Innovision, Denmark) while supine before experiments began. An intravenous catheter was placed in the left antecubital vein.
Supine and upright cardiovagal baroreflex relationships were obtained using the modified Oxford method described below, and estimate of the slope ΔRR/ΔSAP (spontaneous baroreflex sensitivity) at the operating point were calculated by Fourier based transfer function analysis, when squared coherence >0.5, and by autoregressive modeling.19
Following a 30-minute rest period, data were recorded throughout a 10-minute supine baseline period. During this and all subsequent study periods, HR and AP data were used for the calculation of RR interval variability, SAP variability, and baseline spontaneous baroreflex sensitivity.
Following baseline measurements, the modified Oxford method was performed in the supine position. For this and all subsequent uses, performance of the modified Oxford method consisted of an intravenous bolus injection of 100 µg sodium nitroprusside (SNP) was administered followed 1 minute later by an intravenous bolus injection of 150 µg phenylephrine.29 Subjects were then allowed to recover to resting levels over a 30-minute period, and the method was repeated.
Next was a 2-hour period for drug absorption (no drug given in control or POTS on day 1=untreated).
A second baseline data collection for spontaneous fluctuation variabilities was obtained after 2 hours and a second supine modified Oxford method was performed.
After a 30 minutes recovery, subjects were tilted to 40° upright for 10 minutes. Data from minutes 2 to 7 were used for the calculation of upright spontaneous baroreflex sensitivity. The modified Oxford method was repeated once at 8 minutes to avoid syncope. After upright HR and AP returned to near pre-Oxford levels, subjects were returned to the supine position. All subjects were able to tolerate a 40° tilt + modified Oxford.
Control subjects underwent 1 day of testing without medication, while patients with POTS repeated the protocol during 3 additional days during which they received oral placebo, oral pyridostigmine 60 mg, or oral digoxin 500 mg. One of the 3 drugs was given just before step 3. Digoxin was given last because of its long (≈36 hours) half-life. The intervening 2 days were used to administer placebo and pyridostigmine in random order. The dose of pyridostigmine corresponds to a short-acting therapeutic dose. The dose of digoxin corresponds to a half loading dose for an adult and plasma levels reach the therapeutic range in 2 hours.30 A flow sheet of the course of study is shown in Figure 2.
Figure 2.
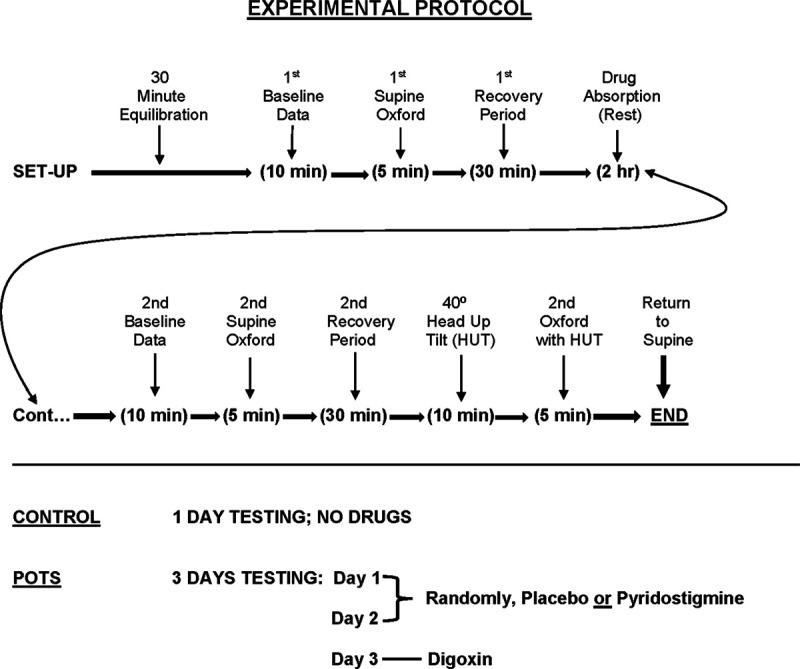
It shows the flow sheet and design of the described studies. The experimental protocol is shown on top and the administration of drugs is shown on the bottom.
Data Analysis
Approximately 5 minutes of baseline data were analyzed preceding the modified Oxford method in the supine position. Upright HR and AP data were obtained during minutes 2 to 7 of head-up tilt (HUT) to avoid the hemodynamic equilibration phase that occurs during the first minutes of tilt.31 Data were collected continuously during the hypotensive and hypertensive phases of the modified Oxford method in both supine and upright positions and included ECG, CO, systolic (SAP), diastolic AP, and mean arterial pressure calculated using the Finometer calibrated against oscillometric cuff pressure. RR interval and HR were calculated from the ECG. Data were sampled at 200 Hz using custom signal processing software and analyzed offline.
Cardiovagal Baroreflex Determined by the Modified Oxford Method
RR intervals were plotted as a function of SAP to generate the cardiovagal baroreflex response during the modified Oxford method, and the methods for calculation of the cardiovagal baroreflex is described in the Data Supplement using established methods.23,24,32
Spontaneous Cardiovagal Baroreflex Indices
To measure spontaneous cardiovagal baroreflex function, the slope corresponding to spontaneous fluctuations in SAP and RR intervals were calculated during supine baseline, supine 2 hours after drug administration, and during 40° upright tilt before the modified Oxford method, as described in the Data Supplement using established methods.23,24,33,34
Statistics
A repeated-measures analytic approach was used. We used a multilevel approach using linear mixed models to account for the within-subject repeated measurements,35,36 as described in the Data Supplement.
Results
The following is a summary of the hemodynamic data in patients with POTS while supine before (supine premed), and after (supine postmed) treatment with placebo, pyridostigmine or digoxin, or controls, and during upright tilt (tilt) and are shown in Table S1 of the Data Supplement. Data averaged over 15 seconds is reported for pre-SNP. Data at the time of minimum SAP are reported for SNP and at the time of maximum SAP is reported for phenyl.
Hemodynamics Before and During the Modified Oxford Method
HR Presodium Nitroprusside
Supine HR tends to be higher in POTS,37 and in this study was higher in POTS-untreated and POTS-placebo after the 2-hour period (supine postmed). HR became similar to control following administration of either pyridostigmine or digoxin while supine. HR subsequently increased on tilt in all (including control), although POTS-pyridostigmine increased less than other POTS and POTS-digoxin. All POTS except POTS-pyridostigmine had a HR during tilt higher than control. This corresponds to a reduction in RR interval and a downward shift in the baroreflex curve compared with control.
HR Sodium Nitroprusside
SNP-premed increased HR in all groups but most in patients with POTS. A proposed nitrergic nitric oxide deficiency in POTS may produce a potentiated response to exogenous nitric oxide.38 The addition of pyridostigmine blunted the SNP-mediated HR increase in both POTS-pyridostigmine postmed and POTS-pyridostigmine tilt. This may indicate ganglionic potentiation of nitrergic parasympathetic nerves.
HR Phenyl
There was a relative reduced bradycardic response to phenylephrine in POTS-tilt groups except for POTS-pyridostigmine.
SAP Presodium Nitroprusside
In contrast, SAP was reduced when supine in POTS unmedicated, increasing with the addition of pyridostigmine or digoxin to control levels while remaining supine. With tilt, SAP was reduced in all POTS compared with control regardless of treatment. This corresponds to a left shift of the operating point of baroreflex curves for POTS compared with control independent of medication.
SAP Sodium Nitroprusside
SNP produced similar reductions of systolic BP when supine, independent of medication. Systolic BP increased during tilt in control but remained unchanged in POTS treatment groups.
SAP Phenyl
SAP response to phenylephrine was similar for all POTS compared with control except for POTS-pyridostigmine which blunted systolic BP increase.
Diastolic AP Pre-SNP
This was reduced in all POTS compared with control pre-SNP when tilted.
CO and Systemic Vascular Resistance
CO during tilt in POTS was similar to control pre-SNP, CO during tilt was lower in POTS than control during SNP, except for POTS-digoxin, which was similar to control, and higher than control during phenyl in POTS except for POTS-digoxin. CO was enhanced by phenylephrine when upright. Systemic vascular resistance was decreased in POTS supine and upright compared with control.
Sigmoidal Baroreflex Results From the Modified Oxford Method
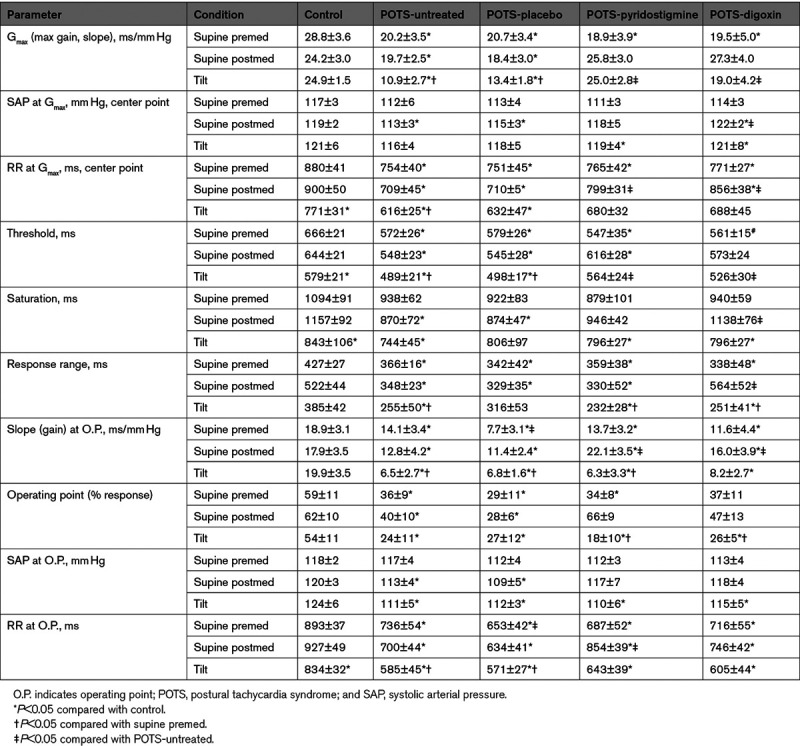
In addition to the fitted cardiovagal baroreflex parameters (Gmax, SAP, and RR interval at center point, threshold, saturation, and response range), we tabulated the SAP and RR interval at operating points (Table).
Table.
Baroreflex Results From Sigmoidal Curves
Supine and Supine Postmed
As shown in the Table, Gmax supine in POTS and control subjects was smaller in POTS compared with control. Gmax was unchanged in untreated or placebo-treated POTS but increased, becoming similar to control after either pyridostigmine or digoxin were administered.
Centering Point, Response Range
Based on the results shown in the Table and depicted in Figures 3 through 5, Gmax, which by definition occurs at the centering point, was displaced downwards (smaller RR interval) and to the left (lower SAP) in POTS compared with control. This is not a simple resetting because the shape of the sigmoidal function is not preserved; rather, Gmax and the response range are both reduced while supine in POTS. Administration of either pyridostigmine or digoxin repaired the BP location of the centering point as well as Gmax, but only digoxin corrected its displacement along the RR interval axis and the response range. Thus, digoxin repairs the supine cardiovagal baroreflex in POTS.
Figure 3.
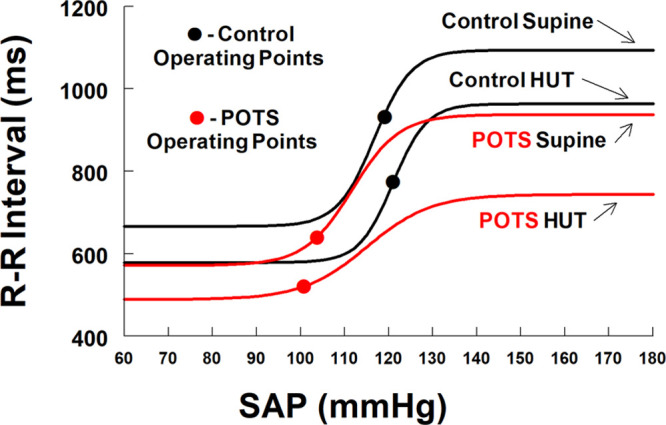
It compares untreated patients with postural tachycardia syndrome (POTS; red lines) with control (black lines) supine and during head-up tilt (HUT). Operating points are shown by red and black circles. Gmax and the response range remained unchanged with tilt in control but were reduced in POTS, and further reduced in POTS when tilted. Additional key features of POTS include left lateral and downwards shifts of centering points and operating points and decreased response range with tilt.
Figure 5.
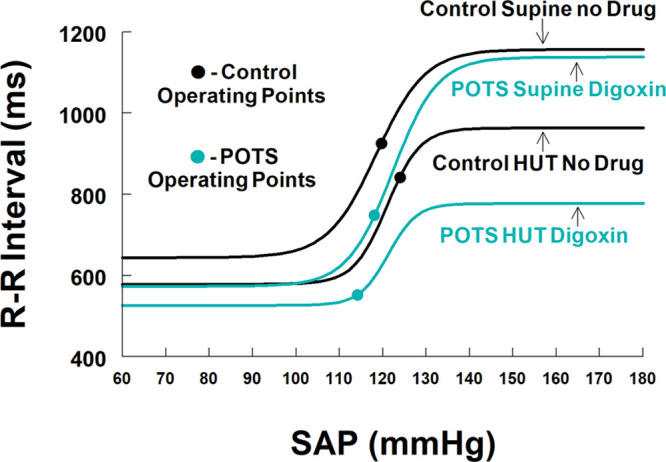
It compares patients with postural tachycardia syndrome (POTS) treated with digoxin (teal lines) with control (black lines) supine and during head-up tilt (HUT). Operating points are shown by cyan and black circles. Digoxin returns the centering point and Gmax of patients with POTS to similar to control values. The response range supine postdigoxin is greater in treated POTS than control. After tilt, threshold was more in POTS while systolic arterial pressure (SAP) at centering point and Gmax were sustained. However, response range decreased markedly with tilt in POTS and contributed to the left shift of the operating point for digoxin and consequently reduced operating point slope.
Operating Point and Operating Point Slope
The operating point response is expressed as a percentage of the response range from the threshold. Relative to control, untreated or placebo-treated POTS shifted the operating point towards lower percentile, that is, towards the knee of the threshold where the sigmoidal nonlinear and flat. Control remained near the center. RR interval was reduced in POTS. The operating point slope was much smaller in premed POTS than in control. This corresponds to a reduction in the alpha index estimate of operating point gain (see below), and, therefore, a marked reduction of heart rate variation for given degree of BP variation (the sigmoid curve is flat) and a general loss in the buffering effects of HR on BP. Treatment with either pyridostigmine or digoxin brought the operating point closer to the centering point and the operating point slope increased.
Tilt
Gmax
With tilt Gmax remained similar to control when treated with pyridostigmine or digoxin but fell for untreated or placebo-treated POTS.
Centering Point, Response Range
The centering point for control was shifted downwards (higher HR when upright) and slightly to the right. The response range remained unchanged and the operating point moved closer to the centering point optimizing BP buffering. Operating point slope (sensitivity) thus more closely approximates Gmax for control. Centering points for POTS-untreated or POTS-placebo moved downwards (higher HR) and to the left (higher SAP) but response range remained reduced compared with control. The operating point moved sharply to the left and thus the operating point slope was greatly reduced. Pyridostigmine or digoxin shifted the centering point to lower RR interval but response range and operating point moved sharply to the left such that the slope at the operating point was comparable to POTS-untreated and much lower than control.
The sigmoidal baroreflex results from the modified Oxford Method were used to generate Figures 3 through 5 using mean values for sigmoidal parameters averaged over all subjects, from data in the Table. In all determinations, POTS-placebo results were similar to POTS-untreated and are not depicted graphically.
Figure 3 shows POTS-untreated both supine and following HUT. Gmax and the response range remained unchanged with tilt in control, is reduced in POTS, and further reduced in POTS when tilted. Additional key features of POTS include left lateral and downwards shifts of centering points and operating points and decreased response range with tilt. Placebo-treated results were not different from untreated POTS.
Figure 4 compares control with POTS-pyridostigmine. Gmax is restored to near control values supine and following HUT. Supine SAP at centering and operating points are similar to control, but threshold is displaced downward and response range is curtailed. Centering point SAP and Gmax were sustained but response range and operating point were greatly reduced during HUT and contributed to the left shift of the operating point for pyridostigmine with consequently reduced slope. It appears, therefore, that a reduction in response range with unchanged centering point and Gmax may account for left-shifted operating points.
Figure 4.
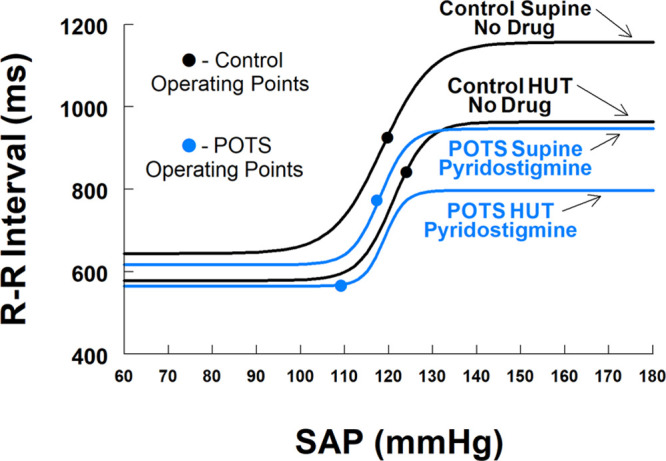
It compares patients with postural tachycardia syndrome (POTS) treated with pyridostigmine (blue lines) with control (black lines) supine and during head-up tilt (HUT). Operating points are shown by blue and black circles. Gmax is restored to near control values supine and upright. Supine systolic arterial pressure (SAP) at centering and operating points are similar to control, but threshold is displaced downwards and response range is curtailed. During tilt centering point, SAP and Gmax were sustained but response range was greatly reduced during tilt contributing to the left shift of the operating point.
Figure 5 compares control with POTS-digoxin supine and following HUT. Digoxin sustains the centering point and Gmax at close to control values. Digoxin, alone, increases response range supine because of a reduced threshold. Tilted, threshold was further reduced. SAP at centering point and Gmax were sustained but response range fell precipitously with tilt and contributed to the left shift of the operating point for digoxin and consequently reduced slope.
Spontaneous Fluctuations—Comparison With Modified Oxford Method Results
Table S2 shows total RR interval and SAP variability, and variability measures over low-frequency and high-frequency bands. Baroreflex gain (local slope) is presented for the sequence method and for the α-index method obtained from transfer function analysis.
Baroreflex Sensitivity (Gain)
Alpha index and sequence methods yielded similar results. These indices were larger in control than in POTS, increased by pyridostigmine and digoxin comparable to control when supine postmed, and decreased once tilted. Indices in control decreased to a lesser extent. Pyridostigmine ameliorated the decrease with tilt. Slopes from sigmoidal baroreflex at the operating point (Table) were similar to spontaneous fluctuation alpha indices.
Discussion
Our data are the first to show deficits in both supine and upright cardiovagal baroreflex in POTS, measured using the reference standard modified Oxford method. While supine, deficits were primarily reflective of severely reduced vagal activity. Although Gmax was reduced that did not account for the majority of vagal loss. Rather, the response range was reduced, and centering points shifted to lower SAP and RR. This combination resulted in movement of operating points to the flat-curvilinear portion of the baroreflex function with consequent profound reductions in operating point slope. Comparing the responses shown in Figures 3 through 5, in patients with POTS, pyridostigmine ameliorated and digoxin corrected these deficits while supine: Gmax was similar to control, centering point SAP was similar to control although the RR interval at the centering point SAP was shifted towards lower RR intervals. The response range exceeded control for digoxin.
Upright posture in POTS elicited more marked baroreflex impairment compared with supine. For POTS-untreated Gmax decreased, response range decreased while centering points were conserved (being shifted to the left and downwards as when supine). Operating slope was on the flat portion of the curve, and operating point slope was very low. Both digoxin and pyridostigmine shifted the operating point to the vicinity of the knee of the sigmoid curve where the second derivative is maximum. Because of these shifts, an increase in SAP can give rise to a large increase in RR interval while a small decrease in SAP fails to appreciably lower RR interval. The system then responds asymmetrically and buffers increases in SAP with a decrease in HR but with little buffering capacity when SAP decreases.
Impaired upright cardiovagal baroreflex is not corrected by pyridostigmine or digoxin, although Gmax and centering point are reset to a lower RR interval while SAP is reasonably well sustained. Also, SAP at the operating points is only slightly diminished compared with supine. However, although SAP only changes slightly, the operating points for both pyridostigmine and digoxin are shifted to the left onto the flat portion of the curve where slope is quite small. This is the result of reduced response range. Thus, for example, if the response range is small while Gmax and centering points remain unchanged, the linear portion of the sigmoidal curve occurs between narrow SAP limits. Then, even a small left shift in SAP can shift the operating point to the flat part of the sigmoidal curve where response to change is reduced.
Sympathetic activation reduces the response range to baroreflex stimulation.39 POTS has a much larger increase in sympathetic activity when upright compared with control,17 SAPV-low frequency is an indicator of sympathetic activation20 which increase in SAPV-low frequency in POTS when upright. The relative reduction of the bradycardic response to phenylephrine during tilt in POTS except for POTS-pyridostigmine is consistent with anti-α1 adrenergic antibodies recently reported.40
Cardiovagal deficits are described by changes in the functional sigmoidal relationship between SAP and RR interval. A resetting of the baroreflex (centering point translation) accommodates changes in posture, BP, or activity by changing the location of the sigmoidal curve without changing its shape. Apart from resetting, Gmax and response range suffice to define the sigmoidal curve. The position along the curve (the operating point) offers an additional degree of freedom and can be defined by determining the extant SAP or RR interval. The cardiovagal baroreflex cannot be defined by the slope at a single point but can only be usefully defined as a functional relationship between SAP and RR interval over a wide range of BPs.22,41,42 The cardiovagal baroreflex helps to buffer beat-to-beat changes in BP by changing HR in the opposite direction. If the operating point is uniformly placed on a flat portion of the sigmoidal curve, then HR cannot vary much with changing SAP, and changes in HR inadequately compensate for either increasing or decreasing BP. As a consequence, BP variability is enhanced at low frequency with tilt. We have previously shown that this interferes with cerebral autoregulation and cognitive capability.43,44
Local slope at the operating point may characterize the local change in RR interval with BP and may, therefore, be considered to represent local baroreflex functioning24 but it cannot predict the sigmoid baroreflex relationship. Rather, centering point location which is reset with orthostasis,25 and the distance of the operating point from the centering point, determined by the relations of Gmax with threshold and response range yield operating point locations. These may be on the linear portion of the curve close to the centering point, as in control, in which case the local slope approximates Gmax, or on the flat portion of the curve distant from the centering point, as in untreated POTS, in which case the local slope is far smaller than Gmax.
Pyridostigmine and digoxin each have vagotonic actions26,27 and repair the supine cardiovagal baroreflex in POTS by normalizing Gmax, correcting the centering point, and reexpanding the response range. The resulting operating point falls on the linear portion of the curve and slopes become good estimates of Gmax. Digoxin is also sympatholytic45 while pyridostigmine may also exert sympathotonic effects.46
When tilted upright, SAPV is elevated, specifically in the low-frequency range. This is consistent with increased sympathetic activity.20,47 Downward resetting of the centering point and reduced response range conforms to increased sympathetic activity which may explain their abnormalcy in POTS in which sympathetic activation is excessive when upright. Neither pyridostigmine nor digoxin maintain the baroreflex once upright because increased cardiac sympathetic activity reduces the response range which left shifts the operating point without important changes in SAP. Potential causes of excessive sympathetic activation in POTS include absolute or positional central hypovolemia,5 intrinsically increased sympathetic CNS outflow as proposed in hyperadrenergic POTS, or increased central command in the face of postural muscle atrophy.48 Sympatholytic agents, in addition to digoxin or pyridostigmine, should increase the response range and normalize baroreflex control.
Administration of either pyridostigmine or digoxin repaired the SAP of the centering point as well as Gmax but only digoxin corrected its displacement along the RR interval axis and the response range. Pyridostigmine enhances acetylcholine and nitrergic parasympathetics, resulting in increased nitrergic nitric oxide which acts at prejunctional and postjunctional sites to reduce adrenergic sympathetic transduction.49 Reduction of Gmax, reduced response range, leftwards and downwards translation of the centering point are also evident in microgravity deconditioning, which can be simulated by bed rest deconditioning50,51 and to a lesser degree by cardiovascular deconditioning.52
Many of our patients with POTS are bed rested and cardiovascular deconditioned. Our findings are, therefore, highly consistent with studies53,54 indicating that exercise reconditioning may be the ideal treatment for POTS because it increases parasympathetic and decreases sympathetic cardiac activity. In addition, since digoxin was able to repair the SAP of the centering point and Gmax and correct its displacement along the RR interval axis and response range, the use of this drug in combination with directed exercise/reconditioning may be a beneficial treatment modality for patients with POTS.
Limitations
Least squared fits of threshold, saturation, centering point, and response range are model-dependent and obtained after nonlinear fitting of data to the sigmoid curve. We always captured the threshold and most linear portion of the curve (the center) and some portion of the saturation in every subject.
The mean age of our study subjects was relatively young, and results may not generalize to older age groups. Data were collected only from female subjects since POTS is predominantly a female illness from menarche to menopause. Male and female physiology may differ; there was no way we could sufficiently power a study of POTS in males as well as females.
Medication was not delivered in a completely random manner and, therefore, its sequential delivery, combined with the training effect of repeated tilt testing may have introduced bias. Also, both pyridostigmine and digoxin have additional effects other than increasing vagal tone. Administration of drugs that elicit multiple drug effects can be problematic when studying systemic outcomes, however, we used these because both have well known vagotonic effects.
Perspectives
Orthostasis produces central hypovolemia by translocating blood to the dependent body. POTS is defined by orthostatic intolerance and increased in heart rate and maintenance or increase in arterial pressure. Orthostatic changes in BP are buffered by arterial baroreflexes. The cardiovagal baroreflex regulation is deficient in POTS by reducing the local slope at the operating point estimated by spontaneous fluctuations of heart rate and BP over a very narrow range of arterial pressures; local slopes may not represent true baroreflex function which can be assessed by reference standard modified Oxford method to generate the nonlinear sigmoidal relationship between the RR interval and the SAP over a wide range of arterial pressures. We describe the determinants of abnormal cardiovagal baroreflex response in POTS, supine and upright, compared with control. Supine baroreflex abnormalities were corrected by digoxin and ameliorated by Mestinon, although sympathetic stimulation of orthostasis produced further baroreflex deficits.
Acknowledgments
We thank Tyler Fialkoff and Ms. Brianna Tuma-Marcella for their technical help in performing experiments and data collection.
Sources of Funding
The funding for this project was provided by grants RO1 HL 112736 and RO1 HL 134674 from the National Heart Lung and Blood Institute (NHLBI).
Disclosures
None.
Supplementary Material
Nonstandard Abbreviations and Acronyms
- BP
- blood pressure
- CO
- cardiac output
- HUT
- head-up tilt
- POTS
- postural tachycardia syndrome
- SAP
- systolic arterial pressure
- SNP
- sodium nitroprusside
The Data Supplement is available with this article at https://www.ahajournals.org/doi/suppl/10.1161/HYPERTENSIONAHA.120.16113.
For Sources of Funding and Disclosures, see page 1243.
Contributor Information
Irfan A. Warsy, Email: irfan_warsy@bchphysicians.org.
Paul Visintainer, Email: paul.visintainer@baystatehealth.org.
Courtney Terilli, Email: courtney_terilli@nymc.edu.
Marvin S. Medow, Email: marvin_medow@nymc.edu.
Novelty and Significance
What Is New?
We showed differences in characteristics of the cardiovagal baroreflex in patients with postural tachycardia syndrome (POTS) compared with control both supine and while upright during an orthostatic challenge.
Parasympathetic stimulation with digoxin remediated the baroreflex deficit in POTS while supine.
Parasympathetic stimulation with both pyridostigmine and digoxin partially corrected the baroreflex deficit in POTS while upright.
What Is Relevant?
Patients with POTS have baroreflex deficits while upright shown using the nonlinear modified Oxford method.
Parasympathetic stimulation with pyridostigmine and digoxin partially corrected this deficit.
Since many patients with POTS are bed rested and physically deconditioned, a combination of exercise reconditioning which increases parasympathetic activity and reduces sympathoexcitation, plus pyridostigmine/digoxin may prove beneficial for treatment of POTS.
Summary
Cardiovagal baroreflex deficits in POTS relate to parasympathetic withdrawal while supine, remediated completely by digoxin, and to sympathetic activation while upright distorting the baroreflex response range. Since these baroreflex effects resemble those measured following microgravity/chronic bedrest, vagotonic/sympatholytic treatment combined with aerobic exercise might normalize the cardiovagal baroreflex and provide therapeutic benefit in patients with POTS.
References
- 1.Robertson D. The epidemic of orthostatic tachycardia and orthostatic intolerance. Am J Med Sci. 1999; 317:75–77. doi: 10.1097/00000441-199902000-00001 [DOI] [PubMed] [Google Scholar]
- 2.Stewart JM, Medow MS, DelPozzi A, Messer ZR, Terilli C, Schwartz CE. Middle cerebral O2 delivery during the modified Oxford maneuver increases with sodium nitroprusside and decreases during phenylephrine. Am J Physiol Heart Circ Physiol. 2013; 304:H1576–H1583. doi: 10.1152/ajpheart.00114.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology. 1993; 43:132–137. doi: 10.1212/wnl.43.1_part_1.132 [DOI] [PubMed] [Google Scholar]
- 4.Grubb BP, Kosinski DJ, Boehm K, Kip K. The postural orthostatic tachycardia syndrome: a neurocardiogenic variant identified during head-up tilt table testing. Pacing Clin Electrophysiol. 1997; 209 Pt 12205–2212. doi: 10.1111/j.1540-8159.1997.tb04238.x [DOI] [PubMed] [Google Scholar]
- 5.Stewart JM, Medow MS, Glover JL, Montgomery LD. Persistent splanchnic hyperemia during upright tilt in postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2006; 290:H665–H673. doi: 10.1152/ajpheart.00784.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tanaka H, Yamaguchi H, Matushima R, Tamai H. Instantaneous orthostatic hypotension in children and adolescents: a new entity of orthostatic intolerance. Pediatr Res. 1999; 46:691–696. doi: 10.1203/00006450-199912000-00022 [DOI] [PubMed] [Google Scholar]
- 7.Singer W, Sletten DM, Opfer-Gehrking TL, Brands CK, Fischer PR, Low PA. Postural tachycardia in children and adolescents: what is abnormal? J Pediatr. 2012; 160:222–226. doi: 10.1016/j.jpeds.2011.08.054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goldstein DS, Holmes C, Frank SM, Dendi R, Cannon RO, III, Sharabi Y, Esler MD, Eisenhofer G. Cardiac sympathetic dysautonomia in chronic orthostatic intolerance syndromes. Circulation. 2002; 106:2358–2365. doi: 10.1161/01.cir.0000036015.54619.b6 [DOI] [PubMed] [Google Scholar]
- 9.Barcroft H, McMichael JE, Scarpey-Schafer EP. Posthaemorrhagic fainting. Study by cardiac outputand forearm flow. Lancet. 1944; 1:489–491 [Google Scholar]
- 10.Raj SR, Robertson D. Blood volume perturbations in the postural tachycardia syndrome. Am J Med Sci. 2007; 334:57–60. doi: 10.1097/MAJ.0b013e318063c6c0 [DOI] [PubMed] [Google Scholar]
- 11.Stewart JM, Montgomery LD. Regional blood volume and peripheral blood flow in postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2004; 287:H1319–H1327. doi: 10.1152/ajpheart.00086.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stewart JM. Pooling in chronic orthostatic intolerance: arterial vasoconstrictive but not venous compliance defects. Circulation. 2002; 105:2274–2281. doi: 10.1161/01.cir.0000016348.55378.c4 [DOI] [PubMed] [Google Scholar]
- 13.Tani H, Singer W, McPhee BR, Opfer-Gehrking TL, Haruma K, Kajiyama G, Low PA. Splanchnic-mesenteric capacitance bed in the postural tachycardia syndrome (POTS). Auton Neurosci. 2000; 86:107–113. doi: 10.1016/S1566-0702(00)00205-8 [DOI] [PubMed] [Google Scholar]
- 14.Bonyhay I, Freeman R. Sympathetic nerve activity in response to hypotensive stress in the postural tachycardia syndrome. Circulation. 2004; 110:3193–3198. doi: 10.1161/01.CIR.0000147280.90339.E9 [DOI] [PubMed] [Google Scholar]
- 15.Stewart JM, Weldon A. Reflex vascular defects in the orthostatic tachycardia syndrome of adolescents. J Appl Physiol (1985). 2001; 90:2025–2032. doi: 10.1152/jappl.2001.90.6.2025 [DOI] [PubMed] [Google Scholar]
- 16.Sagawa K. Baroreflex control of systemic arterial pressure and vascular bed. Handbook of Physiology, The Cardiovascular System. 1983, Am Physiol Soc:453–496 [Google Scholar]
- 17.Muenter Swift N, Charkoudian N, Dotson RM, Suarez GA, Low PA. Baroreflex control of muscle sympathetic nerve activity in postural orthostatic tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2005; 289:H1226–H1233. doi: 10.1152/ajpheart.01243.2004 [DOI] [PubMed] [Google Scholar]
- 18.Goldstein DS, Bentho O, Park MY, Sharabi Y. Low-frequency power of heart rate variability is not a measure of cardiac sympathetic tone but may be a measure of modulation of cardiac autonomic outflows by baroreflexes. Exp Physiol. 2011; 96:1255–1261. doi: 10.1113/expphysiol.2010.056259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Robbe HW, Mulder LJ, Rüddel H, Langewitz WA, Veldman JB, Mulder G. Assessment of baroreceptor reflex sensitivity by means of spectral analysis. Hypertension. 1987; 10:538–543. doi: 10.1161/01.hyp.10.5.538 [DOI] [PubMed] [Google Scholar]
- 20.Okamoto LE, Raj SR, Gamboa A, Shibao CA, Arnold AC, Garland EM, Black BK, Farley G, Diedrich A, Biaggioni I. Sympathetic activation is associated with increased IL-6, but not CRP in the absence of obesity: lessons from postural tachycardia syndrome and obesity. Am J Physiol Heart Circ Physiol. 2015; 309:H2098–H2107. doi: 10.1152/ajpheart.00409.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jacob G, Diedrich L, Sato K, Brychta RJ, Raj SR, Robertson D, Biaggioni I, Diedrich A. Vagal and sympathetic function in neuropathic postural tachycardia syndrome. Hypertension. 2019; 73:1087–1096. doi: 10.1161/HYPERTENSIONAHA.118.11803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lipman RD, Salisbury JK, Taylor JA. Spontaneous indices are inconsistent with arterial baroreflex gain. Hypertension. 2003; 42:481–487. doi: 10.1161/01.HYP.0000091370.83602.E6 [DOI] [PubMed] [Google Scholar]
- 23.Hunt BE, Farquhar WB. Nonlinearities and asymmetries of the human cardiovagal baroreflex. Am J Physiol Regul Integr Comp Physiol. 2005; 288:R1339–R1346. doi: 10.1152/ajpregu.00038.2004 [DOI] [PubMed] [Google Scholar]
- 24.Schwartz CE, Medow MS, Messer Z, Stewart JM. Spontaneous fluctuation indices of the cardiovagal baroreflex accurately measure the baroreflex sensitivity at the operating point during upright tilt. Am J Physiol Regul Integr Comp Physiol. 2013; 304:R1107–R1113. doi: 10.1152/ajpregu.00559.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schwartz CE, Stewart JM. The arterial baroreflex resets with orthostasis. Front Physiol. 2012; 3:461. doi: 10.3389/fphys.2012.00461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nóbrega AC, dos Reis AF, Moraes RS, Bastos BG, Ferlin EL, Ribeiro JP. Enhancement of heart rate variability by cholinergic stimulation with pyridostigmine in healthy subjects. Clin Auton Res. 2001; 11:11–17. doi: 10.1007/BF02317797 [DOI] [PubMed] [Google Scholar]
- 27.Ferrari A, Gregorini L, Ferrari MC, Preti L, Mancia G. Digitalis and baroreceptor reflexes in man. Circulation. 1981; 63:279–285. doi: 10.1161/01.cir.63.2.279 [DOI] [PubMed] [Google Scholar]
- 28.Minson CT, Halliwill JR, Young TM, Joyner MJ. Influence of the menstrual cycle on sympathetic activity, baroreflex sensitivity, and vascular transduction in young women. Circulation. 2000; 101:862–868. doi: 10.1161/01.cir.101.8.862 [DOI] [PubMed] [Google Scholar]
- 29.Rudas L, Crossman AA, Morillo CA, Halliwill JR, Tahvanainen KU, Kuusela TA, Eckberg DL. Human sympathetic and vagal baroreflex responses to sequential nitroprusside and phenylephrine. Am J Physiol. 1999; 2765 Pt 2H1691–H1698. doi: 10.1152/ajpheart.1999.276.5.h1691 [DOI] [PubMed] [Google Scholar]
- 30.Sumner DJ, Russell AJ. Digoxin pharmacokinetics: multicompartmental analysis and its clinical implications. Br J Clin Pharmacol. 1976; 3:221–229. doi: 10.1111/j.1365-2125.1976.tb00596.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wieling W, Krediet CT, van Dijk N, Linzer M, Tschakovsky ME. Initial orthostatic hypotension: review of a forgotten condition. Clin Sci (Lond). 2007; 112:157–165. doi: 10.1042/CS20060091 [DOI] [PubMed] [Google Scholar]
- 32.Studinger P, Goldstein R, Taylor JA. Mechanical and neural contributions to hysteresis in the cardiac vagal limb of the arterial baroreflex. J Physiol. 2007; 583pt 31041–1048. doi: 10.1113/jphysiol.2007.139204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Marquardt DW. An algorithm for least-squares estimation of nonlinear parameters. J Soc Industrial Appl Math. 1963; 11:431–41 [Google Scholar]
- 34.Triedman JK, Saul JP. Blood pressure modulation by central venous pressure and respiration. Buffering effects of the heart rate reflexes. Circulation. 1994; 89:169–179. doi: 10.1161/01.cir.89.1.169 [DOI] [PubMed] [Google Scholar]
- 35.Singer JD, Willet JB. Applied Longitudinal Data Analysis. 2003, Oxford University press [Google Scholar]
- 36.Fitzmaurice GM, Ware JH. Applied Longitudinal Analysis. 2004, John Wiley & Sons [Google Scholar]
- 37.Ross AJ, Ocon AJ, Medow MS, Stewart JM. A double-blind placebo-controlled cross-over study of the vascular effects of midodrine in neuropathic compared with hyperadrenergic postural tachycardia syndrome. Clin Sci (Lond). 2014; 126:289–296. doi: 10.1042/CS20130222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Medow MS, Minson CT, Stewart JM. Decreased microvascular nitric oxide-dependent vasodilation in postural tachycardia syndrome. Circulation. 2005; 112:2611–2618. doi: 10.1161/CIRCULATIONAHA.104.526764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nosaka S, Murata K, Kobayashi M, Cheng ZB, Maruyama J. Inhibition of baroreflex vagal bradycardia by activation of the rostral ventrolateral medulla in rats. Am J Physiol Heart Circ Physiol. 2000; 279:H1239–H1247. doi: 10.1152/ajpheart.2000.279.3.H1239 [DOI] [PubMed] [Google Scholar]
- 40.Fedorowski A, Li H, Yu X, Koelsch KA, Harris VM, Liles C, Murphy TA, Quadri SMS, Scofield RH, Sutton R, et al. Antiadrenergic autoimmunity in postural tachycardia syndrome. Europace. 2017; 19:1211–1219. doi: 10.1093/europace/euw154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fisher JP, Ogoh S, Junor C, Khaja A, Northrup M, Fadel PJ. Spontaneous baroreflex measures are unable to detect age-related impairments in cardiac baroreflex function during dynamic exercise in humans. Exp Physiol. 2009; 94:447–458. doi: 10.1113/expphysiol.2008.044867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Parati G, Di Rienzo M, Mancia G. How to measure baroreflex sensitivity: from the cardiovascular laboratory to daily life. J Hypertens. 2000; 18:7–19 [PubMed] [Google Scholar]
- 43.Medow MS, Del Pozzi AT, Messer ZR, Terilli C, Stewart JM. Altered oscillatory cerebral blood flow velocity and autoregulation in postural tachycardia syndrome. Front Physiol. 2014; 5:234. doi: 10.3389/fphys.2014.00234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Stewart JM, Medow MS, Messer ZR, Baugham IL, Terilli C, Ocon AJ. Postural neurocognitive and neuronal activated cerebral blood flow deficits in young chronic fatigue syndrome patients with postural tachycardia syndrome. Am J Physiol Heart Circ Physiol. 2012; 302:H1185–H1194. doi: 10.1152/ajpheart.00994.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Watanabe AM. Digitalis and the autonomic nervous system. J Am Coll Cardiol. 1985; 55 suppl A35A–42A. doi: 10.1016/s0735-1097(85)80461-7 [DOI] [PubMed] [Google Scholar]
- 46.Singer W, Opfer-Gehrking TL, McPhee BR, Hilz MJ, Bharucha AE, Low PA. Acetylcholinesterase inhibition: a novel approach in the treatment of neurogenic orthostatic hypotension. J Neurol Neurosurg Psychiatry. 2003; 74:1294–1298. doi: 10.1136/jnnp.74.9.1294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Just A, Wagner CD, Ehmke H, Kirchheim HR, Persson PB. On the origin of low-frequency blood pressure variability in the conscious dog. J Physiol. 1995; 489pt 1215–223. doi: 10.1113/jphysiol.1995.sp021043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Michelini LC, O’Leary DS, Raven PB, Nóbrega AC. Neural control of circulation and exercise: a translational approach disclosing interactions between central command, arterial baroreflex, and muscle metaboreflex. Am J Physiol Heart Circ Physiol. 2015; 309:H381–H392. doi: 10.1152/ajpheart.00077.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Storgaard T, Nedergaard OA. Prejunctional modulation by angiotensins of noradrenaline release from sympathetic neurons in isolated rabbit aorta. Naunyn Schmiedebergs Arch Pharmacol. 1997; 356:706–711. doi: 10.1007/pl00005109 [DOI] [PubMed] [Google Scholar]
- 50.Eckberg DL, Fritsch JM. Human autonomic responses to actual and simulated weightlessness. J Clin Pharmacol. 1991; 31:951–955. doi: 10.1002/j.1552-4604.1991.tb03655.x [DOI] [PubMed] [Google Scholar]
- 51.Fritsch-Yelle JM, Charles JB, Jones MM, Beightol LA, Eckberg DL. Spaceflight alters autonomic regulation of arterial pressure in humans. J Appl Physiol (1985). 1994; 77:1776–1783. doi: 10.1152/jappl.1994.77.4.1776 [DOI] [PubMed] [Google Scholar]
- 52.Smith SA, Querry RG, Fadel PJ, Welch-O’Connor RM, Olivencia-Yurvati A, Shi X, Raven PB. Differential baroreflex control of heart rate in sedentary and aerobically fit individuals. Med Sci Sports Exerc. 2000; 32:1419–1430. doi: 10.1097/00005768-200008000-00010 [DOI] [PubMed] [Google Scholar]
- 53.Fu Q, Vangundy TB, Galbreath MM, Shibata S, Jain M, Hastings JL, Bhella PS, Levine BD. Cardiac origins of the postural orthostatic tachycardia syndrome. J Am Coll Cardiol. 2010; 55:2858–2868. doi: 10.1016/j.jacc.2010.02.043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fu Q, Vangundy TB, Shibata S, Auchus RJ, Williams GH, Levine BD. Exercise training versus propranolol in the treatment of the postural orthostatic tachycardia syndrome. Hypertension. 2011; 58:167–175. doi: 10.1161/HYPERTENSIONAHA.111.172262 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


