Table 2.
Arene substituent scope and variable tether lengtha
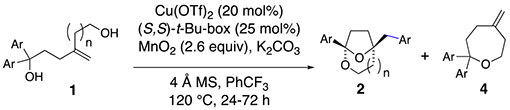 | |||||
|---|---|---|---|---|---|
| Entry | Ar | n | Product | yield (%)b | ee (%)c |
| 1 | Ph | 1 | 2a | 64 | 92 |
| 2d | 4-MeC6H4 | 1 | 2b | 40 | 87 |
| 3d | 3-MeC6H4 | 1 | 2c | 42 | 92 |
| 4e | 4-CF3C6H4 | 1 | 2d | 71 | >95 |
| 5f | 4-CF3C6H4 | 1 | 2d | 51 | >95 |
| 6 | 4-MeOC6H4 | 1 | 4e | 61 | -- |
| 7d,g | 4-MeOC6H4 | 1 | 2e | 40 | 70 |
| 8 | 4-ClC6H4 | 1 | 2f | 53 | 88 |
| 9g | 3-ClC6H4 | 1 | 2g | 58 | 94 |
| 10 | 4-TsMeNC6H4 | 1 | 2h | 73 | 90 |
| 11 | 4-PhC6H4 | 1 | 2i | 59 | 20 |
| 12h | 4-PhC6H4 | 1 | 2i | 64 | 56 |
| 13 | Ph | 2 | 2j | 68 | 93 |
Table 1, entry 1 conditions were applied with 0.1 mmol of substrate 1.
Isolated yield of major product following chromatography on silica gel.
Enantiomeric excess measured by chiral HPLC.
30-40% of an SN1 product 4 was also formed.
25 mol% Cu(OTf)2 and 31 mol% (S,S)-t-Bu-Box was used.
1 mmol of 1d was used. 34% of starting 1d was recovered.
Cu(NTf2)2 was used.
(S,S)-i-Pr-Box was used, reaction temperature was 105 °C (at 120 °C, 58% of 2i was obtained in 50% ee).
