Abstract
Type 2 innate lymphoid cells (ILC2s) are important innate immune cells that are involved in type 2 inflammation, in both mice and humans. ILC2s are stimulated by factors, including interleukin (IL)-33 and IL-25, and activated ILC2s secrete several cytokines that mediate type 2 immunity by inducing profound changes in physiology, including activation of alternative (M2) macrophages. M2 macrophages possess immune modulatory, phagocytic, tissue repair and remodeling properties, and can regulate ILC2s under infection. The present review summarizes the role of ILC2s as innate cells and M2 macrophages as anti-inflammatory cells, and discusses current literature on their important biological significance. The present review also highlights how the crosstalk between ILC2s and M2 macrophages contributes to lung development, induces pulmonary parasitic expulsion, exacerbates pulmonary viral and fungal infections and allergic airway diseases, and promotes the development of lung diseases, such as pulmonary fibrosis, chronic obstructive pulmonary disease and carcinoma of the lungs.
Keywords: type 2 innate lymphoid cells, alternative macrophages, crosstalk, lung development, lung diseases
1. Introduction
Lung development initiates in utero and continues until infancy, and involves a complex process regulated by different types of cells, factors and mediators, such as macrophages, dendritic cells and lymphocytes (1). Abnormal lung development can be harmful to respiratory health, which may result in bronchopulmonary dysplasia, neonatal respiratory distress syndrome, asthma and chronic obstructive emphysema (2–4). Type 2 immune response is important for pulmonary development and several types of pulmonary diseases, such as asthma, lung infection and pulmonary fibrosis (5–7).
Interleukin (IL)-4, IL-5, IL-9 and IL-13 are important cytokines that play key roles in type 2 immunity, and are usually involved in allergic diseases or during helminthic parasitic infections (8,9). Th2 cells and certain myeloid cells are considered the primary source of these type 2 cytokines (10,11); however, recent studies have reported that a rare subpopulation of innate lymphocytes are the predominant source (12–14). Type 2 innate lymphoid cells (ILC2s), which were first discovered as non-T and B cells (15,16), play a defensive role in the initial stage of helminthic infestation (17), and are considered a major component of type 2 immunity in lungs (18,19).
Several types of cells, including eosinophils, mast cells, basophils and alternative (M2) macrophages, activated by IL-4 and IL-13 that are involved in type 2 immune response, also regulate the repair response following tissue injury (20). M2 macrophages initiate different responses to parasites, tissue remodeling, angiogenesis and allergic diseases (21–23). Therefore, it may be hypothesized that M2 macrophages can crosstalk with ILC2s during pulmonary development and in different pulmonary diseases.
2. ILC2s
ILCs are innate immune cells that regulate mucosal immune response (24). ILCs are important effector cells in the innate immune system (25). In addition to acting as first-line defense against pathogen invasion and infection, ILCs are also involved in lymphoid organ formation, tissue repair and mucosal homeostasis (26).
ILC2s are a subset of ILCs, and activation of these produce several Th2 cytokines, including IL-4, IL-5, IL-9 and IL-13, and/or dual-regulatory proteins, such as amphiregulin (AREG) (27). ILC2s depend on transcription factors, GATA binding protein 3 and retinoid acid receptor related orphan receptor α, for their development and function, but lack antigen-specific receptors (28,29). ILC2s are distributed throughout the body and are abundant on mucosal surfaces, such as the lungs, gastrointestinal tract and skin, in both humans and mice (30). ILC2s account for a major proportion in mouse pulmonary innate lymphocytes, and <3% of human lung innate lymphocytes (31,32).
Lung ILC2s are rapidly activated when exposed to epithelial-derived alarmin proteins and other inflammatory mediators, including IL-33, IL-25 and thymic stromal lymphopoietin (TSLP) (33). A previous study demonstrated that IL-25 reactive lung ILC2s can change into IL-33 reactive lung ILC2s, both in vivo and in vitro (34). IL-33 and IL-25 both promote the enrichment of ILC2s in lung in vivo; however, only IL-33 can directly induce the migration of ILC2s in vitro (35). Similar effects of IL-33 are observed in skin (36), while TSLP and IL-25 exhibit relatively poor chemotaxis, although they can be detected at high concentrations in lungs (37,38).
Although ILC2s secrete IL-9, autocrine IL-9 maintains homeostasis of pulmonary ILC2s (37,38). IL-2 was the first cytokine reported to promote the secretion of IL-9 by ILC2s (39). IL-2 is also important for activating and culturing ILC2s in vitro (39,40). Another study demonstrated that IL-4 can increase IL-9 expression by stimulating ILC2s (41). Suppression of IL-9 production inhibits IL-33-induced eosinophilic airway inflammation, highlighting its role in effectively proliferating and activating ILC2s (42). In addition, the synergistic effects of TSLP and IL-33 markedly effect the production of IL-9 via ILC2s (37).
ILC2s express corresponding receptors, including suppression of tumorigenicity 2 (ST2), IL-25R (IL-17RB), TSLPR and AREG receptor, as well as toll-like receptors (TLRs) 2 and 4 (28,29,43–45). Upon activation, excluding Th2-type cytokines and/or AREG, ILC2s also secrete other factors, including granulocyte-macrophage colony stimulating factor (GM-CSF), IL-6 and IL-10 (46–48). In addition to stimulators, there are also inhibitors of ILC2s. For example, the neuropeptide calcitonin gene-related peptide and its receptor can inhibit the secretion and enrichment of pulmonary ILC2s and Th2 cytokines driven by alarmin, both in vitro and in vivo (49).
Elevated numbers of ILC2s in patients with asthma and chronic sinusitis suggest that ILC2s are detrimental to chronic inflammation (50). However, intrahepatic ILC2s can exacerbate fibrosis in liver diseases by secreting AREG (51). Thus, the roles of ILC2s vary in different tissues and diseases, and involve complex molecular mechanisms.
Recently, ILC2s have become the research focus in different tissue and organ diseases. It has been reported that intestinal helminthic infection induces activation of ILC2s, proliferation of IL-13 dependent goblet cells and increases mucin production at distal sites, including the lungs (52). In severe cases, increased mucus secretion via alveoli and the lungs inhibits lung metastasis (52). This suggests that the innate immunity of ILC2s is not only limited to certain tissues, but also influences and interacts with different organs. According to a previous study, aging influences innate immunity (53). ILC2s in elderly lungs are not uniform in transcription and function, and cannot produce cytokines during influenza infection and homeostasis in vivo (53). The transfer of ILC2s in the lungs of young mice strengthens the immunity of old mice to influenza infection (53). Notably, ILC2s in neonatal lungs involve distinct pro-inflammatory and tissue repair subgroups (54). Neonatal endogenous IL-33 stimulates ILC2s in the pulmonary, which may ‘train’ ILC2s for implantation into the lungs following birth, thus becoming resident cells that respond more effectively to future challenges (55). Thus, by secretion of a plethora of mediators, ILC2s play vital roles in inducing and supporting type 2 immune responses in lung tissues.
3. M2 macrophages
Macrophages, which act as myeloid cells, are among the first cells that respond to pathogens and tissue damage (56). They not only have innate immune function, which acts by phagocytizing and killing pathogens directly to exert innate immunity, but also initiate adaptive immunity by presenting pathogens to T lymphocytes (57,58). Tissue macrophages, which are important immune cells, are produced by yolk sac or fetal liver and their function is guided by resident tissues (59). Thus, it is important to study the macrophages that reside in the lung to understand the role of macrophages in lung diseases. There are two subtypes based on anatomical position of pulmonary resident macrophages, alveolar macrophages (AMs) and interstitial macrophages (60).
AMs, which are the most important resident macrophages in the lung, act as immune barriers in the alveoli against several pathogens of the respiratory tract (61). Alveolar macrophages are highly heterogeneous and exhibit unique phenotypes and functions in the complex microenvironment of the body (62). They are non-polarized under normal conditions (63). However, macrophages are induced and polarized into classical activation (M1) or alternate activation (M2) phenotypes under the stimulation of inflammation or in different immune development stages (64,65). These also play a role in producing different chemokines and cytokines in the local microenvironment (66).
M2 macrophages are predominantly induced by cytokines, including IL-4, IL-10 and IL-13, glucocorticoids and immune complexes TLRs (67). Similar to ILC2s, they can also induce typical Th2 cytokines to decrease inflammatory response by promoting angiogenesis, tissue repairing, remodeling and wound healing (68). In addition, excessive tissue repair and remodeling results in fibrosis, which can aggravate the condition (69). M2 macrophages highly express type I arginase encoding genes (arginase-1, Arg1) and mannose receptor (CD206), and thus the expression and activity of Arg1 and CD206 are used to identify M2 macrophages (70). Under the induction of memory Th2 cells, M2 macrophages, which are important immune effector cells, can scavenge pathogens, which is associated with Arg1 activity (71). M2 macrophages have a weak antigen-presenting capacity compared with M1 macrophages, and downregulate the immune response by secreting inhibitory cytokines, such as IL-10 and/or tumor growth factor β (TGF-β) (72). A different type of M2 macrophage exists in the tumor site, which can be induced by IL-10 and is affected by chemokines, including CCL2, M-CSF and vascular endothelial growth factor (58).
4. Crosstalk between ILC2s and M2 macrophages during lung development
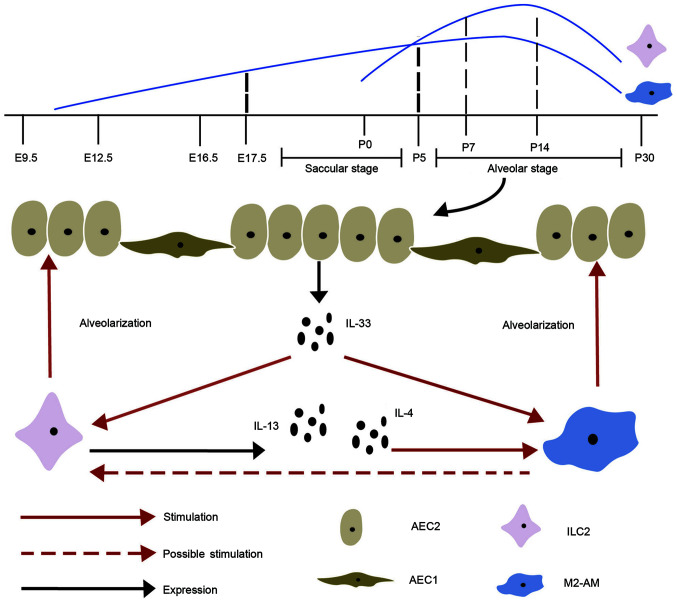
The developmental process of lungs involves complex steps in humans/mice, and is subdivided into five stages, embryonic, pseudoglandular, canalicular, saccular and alveolar (73). Among these, the vesicle [Embryo day(E)16-E266/E17.5-Postnatal day (P)5] and alveolar (E252-2 years/P5-P30) stages are important as they affect the development and maturity of lungs (Fig. 1) (73). Macrophages first appear on day 10 of pregnancy and can be continuously detected during fetal lung development (74), which then increases with alveolarization (75,76). The perinatal period is a critical window for transferring and distributing congenital immune cells to all the tissues and organs during lung development (77). ILC2s, which are similar to tissue macrophages, also appear during pregnancy, but at a later stage, and most of the peripheral ILC2 pools are produced de novo following birth (77). Several studies have confirmed that rapid amplification and activation of ILC2s in pulmonary occur during the early postnatal period (78–80). Pulmonary resident ILC2s are minimal at birth, increase during alveolarization, reach peak at 7–14 days and subsequently decrease in adulthood, similar to AMs (76,81–83). Thus, the interactions between ILC2s and macrophages most likely occur during the vesicle and alveolar stages. Gradually, fewer ILC2s in lung tissues are replaced by newly generated ILC2s, but the expanded ILC2s during the early postnatal period account for the majority of adult lung ILC2s (77).
Figure 1.
Quantitative changes of ILC2s and M2 macrophages in lung development and the effect of crosstalk on alveolarization. ILC2, type 2 innate lymphoid cell; M2-AM, alternative macrophage; IL, interleukin; AM, alveolar macrophage; AEC1, type 1 alveolar epithelial cell; AEC2, type 2 alveolar epithelial cell.
From the very start, the lung is exposed to the external environment (84). The microenvironment of the lung undergoes a notable change within a short period of time and requires rapid regulation to avoid inflammatory response caused by environmental stimulation (84). After being stimulated during labor, IL-33 rapidly increases and activates ILC2s in the fluid filled lung and begins to promote the formation of type 2 immune environment in pulmonary tissues (76). Type 2 immunity involves type 2 cytokines, eosinophilia, mucogenesis, IgE and M2 macrophages (85). The presence of AMs is consistent with that of IL-13-secreting ILC2s, which exhibit IL-13 dependent anti-inflammatory M2-type in the early stage of lung development (76). It has also been reported that IL-4 receptor α (IL-4R α), including IL-4 and IL-13, can promote AMs to polarize into M2 macrophages, suggesting that the crosstalk between ILC2s and M2 macrophages plays a role in regulating type 2 immunity (86,87). Another study demonstrated that the addition of ILC2s can make AMs express more M2 macrophages-related markers in vitro (88). Postnatal adaptation to breathing depends on pulmonary surfactant being synthesized and secreted by type 2 alveolar epithelial cells (AEC2) (89). Promoted by M2 macrophages, AEC2 continuously proliferate and differentiate to accelerate alveolar formation (90).
A previous study revealed that M2 macrophages are enriched in lung tissues and AEC2 proliferated rapidly following pneumonectomy (91). ILC2s increase and become the main source of IL-13, which induces AMs to differentiate into M2 phenotype (91). Both IL-4R α-expressing ILC2s and M2 macrophages, which are necessary for optimal lung regeneration, promote the regeneration of lung tissues by stimulating the growth of AEC2 (91). Rindler et al (92) reported that M2 macrophages are clustered together and localized in the site of AEC2 multiplication during regeneration.
It has been reported that activation of IL-33 can promote type 2 immunity in pulmonary development by amplifying and activating ILC2s during the perinatal period (81). IL-4, IL-5 and IL-13 exhibit upregulation after activation of ILC2s, which constitutively express ST2. In addition to activating ILC2s, IL-33 also stimulate the expression and polarization of AMs by basophils during alveolar formation (93,94). Thus, it is hypothesized that IL-33 promotes proliferation and activation of ILC2s and M2 macrophages during lung development, and crosstalk between ILC2s and M2 macrophages promotes alveolarization. This is consistent with the IL-33-ST2 axis regulating regeneration of epithelial through activation of monocyte differentiation into reparative M2 macrophages and ILC2s-mediated M2 macrophages (95). In summary, ILC2s promote the polarization of M2 macrophages via IL-4/13. In addition to IL-4/13, there may be other associations between ILC2s and M2 macrophages in the complex process of embryonic development, which have not been fully investigated. Thus, future studies are required to determine how M2 macrophages directly affect ILC2s, and how their crosstalk promotes fetal and preterm lung development.
5. Crosstalk between ILC2s and M2 macrophages in lung diseases
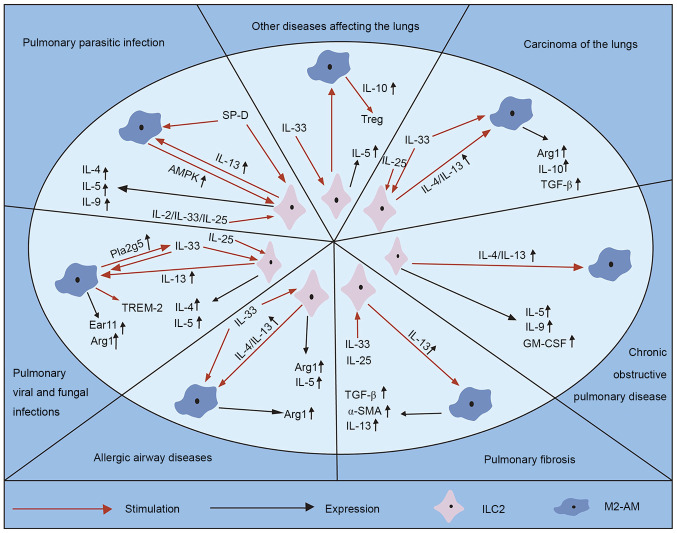
The arrest of alveolar development or disruption of alveolar structure is not only associated with neonatal respiratory distress syndrome, bronchopulmonary dysplasia and persistent pulmonary hypertension, but also chronic lung diseases, such as asthma, allergic diseases and chronic obstructive pulmonary emphysema (2–4). Pulmonary epithelial barrier dysfunction is an important pathological component of lung injury, which is mainly caused by damage of epithelial cell migration (96). ILC2s participate in the regulation of AEC2 and different lung diseases (37). M2 macrophages are a subgroup of macrophages whose polarization is important for AEC2 regulation and inflammatory response (97). Thus, the crosstalk between increased ILC2s and upregulated M2 macrophages may regulate lung development, and modulate the processes of several lung diseases (Fig. 2).
Figure 2.
Crosstalk between ILC2s and M2 macrophages regulates type 2 immune response in lung diseases. ILC2, type 2 innate lymphoid cell; M2-AM, alternative macrophage; IL, interleukin; AMPK, Amp activated protein kinase; SP-D, surfactant protein D; TGF-β, tumor growth factor β; TREM-2, triggering receptors on myeloid cell 2; Pla2g5, group V phospholipase A2; Ear11, eosinophil-associated ribonuclease 11; GM-CSF, granulocyte-macrophage colony stimulating factor.
Pulmonary parasitic infection
Several parasites, namely pulmonary parasitic diseases, spread to other parts of the human body via blood circulation, and often reside in the lungs, causing pathological changes (98). The host cells of helminth mega parasites are involved in type 2 immune response, including Th2 cells and type 2 cytokines (IL-4, IL-5, IL-9 and IL-13), which are required to fight these pathogens (99,100). Recently, it has been reported that the relative abundance of these macrophages and the rare ILC2s have a swift and strong response to helminth antigen and helminth induced injury, activating damaged epithelial cells and recruiting other effector factors (101). Immunocompromised larvae of helminths have a significant morphological defect, which is affected by aggregation of IL-13-secreting ILC2s and CD4+ T cells, and the polarization of M2 macrophages (102). Application of IL-2 or IL-33 can bypass the requirement of T cells, resulting in proliferation of IL-13 and secretion of ILC2s and death of larvae, and exhaustion of ILC2s inhibits larval death in mice by transferring IL-2 (102). Thus, it is not surprising that ILC2s are the key factor during infection and are maintained by CD4+ T cells, which not only ensure rapid activation of IL-13 dependent M2 macrophages, but also maintain their immune function in lung tissues (102).
Amp activated protein kinase (AMPK) is a significant driving factor of cellular energy, which exists in AMs (103). Deletion of AMPK decreases the secretion of IL-13 and impairs the expansion of ILC2s in lung tissues from mice that are selectively deprived of α 1 subunit, thereby exacerbating lung injury following ancylostoma infection (103). Surfactant protein D (SP-D) is an important epithelial product (104). Increased levels of pulmonary SP-D before infection can enhance parasite excretion and type 2 immune response, including the increase of IL-13-producing ILC2s, M2 macrophages and the cytokines, IL-4 and IL-13 (104). Thus, it is speculated that AMs and ILC2s assist in coordinating the regulation of mucosal tissue damage through metabolic enzyme function (103,104).
Pulmonary viral and fungal infections
Several studies have confirmed that the intensity of infection is affected by type 1 immune response and polarization of M1 macrophages, while type 2 immunity and polarization of M2 macrophages are closely associated with disease progression and adverse outcomes (105–107). In infected lungs, the number of ILC2s significantly increase following induction of type 2 response (108). ILC2-deficient mice exhibit a notable declination in type 2 immune response 14 days after infection, which is characterized by decreased expression levels of IL-4, IL-5 and IL-13, as well as the number of M2 macrophages (108).
The change in polarization of pulmonary macrophages in ILC2-deficient mice is frequently associated with better control of fungi and prolongation of survival time of infected mice (108). Rhinovirus (RV) infection also induces IL-25, IL-33, IL-4, IL-5, IL-13 and ILC2s expansion, mucus metaplasia and airway hyperresponsiveness (109). IL-1 β of pulmonary macrophages inhibits type 2 inflammation and mucus metaplasia following RV infection by decreasing ILC2s and cytokines (109).
Group V phospholipase A2 (Pla2g5) is a lipid-producing enzyme that is required for macrophage functioning in lung inflammation (110). Macrophages also assist in regulating IL-33 induction and free fatty acids (FFAs)-driven ILC2s activation via Pla2g5, significantly contributing to type-2 immunity (110). In addition, mass spectrometry analysis demonstrated significant reduction of FFAs in Pla2g5 deficient lung tissues and BM-macrophages in Alternaria-exposed wild-type mice (110).
Another study reported that type 2 immunoregulatory neutrophil infiltration is influenced by mouse eosinophil associated ribonuclease 11, and is secreted by M2 macrophages downstream of ILC2s that are stimulated by IL-25 (111). Furthermore, neutrophils can promote type 2 immune response without aggravating inflammation (111).
Chronic post viral disease is characterized by excessive airway mucus formation and multiplication of M2 differentiated pulmonary macrophages, requiring expression of macrophages for triggering receptors on myeloid cell 2 (TREM-2) (112). With increasing levels of IL-13, virus replication increases the levels of macrophages and TREM-2 in the lung tissues, preventing macrophage apoptosis in acute diseases (112). Following infection clearance, IL-13 promotes cleavage of TREM-2 into the soluble form, STREM-2, which prevents macrophage apoptosis (112). These results may explain how crosstalk between ILC2s with M2 macrophages in acute infection results in chronic inflammatory diseases.
Recruitment of neutrophils, eosinophils and inflammatory chemokines (KC, eotaxin-1, MIP1a and MIP1b), Th2 cytokines (IL-4/5), arginase-1 (M2 macrophage marker) and IL-33R+ ILC2s cells are significantly elevated in adenovirus Oncostatin M (OSM) mice, while these responses are significantly attenuated in IL-33-/- mice (113). In vitro, IL-33 upregulates OSM expression in RAW264.7 macrophage cells and bone marrow-derived macrophages (113). Thus, IL-33 is a key mediator of OSM-driven lung inflammation, induction of type 2 immune responses and M2 macrophages in mice, which contributes via activation of ILC2s (113).
Allergic airway diseases (AAD)
In addition to the common tissue tropism, AAD also have obvious inflammatory patterns, including eosinophils, M2 macrophages, ILC2s, IgE secreting B cells and Th2 cells, and cytokines, including IL-33, IL-4, IL-5 and IL-13 (114,115). Reduction of Th2 cytokines (IL-4, IL-5 and IL-13), macrophages, ILC2s and other cells in lung tissues, and alveolar lavage fluid, can improve allergic airway inflammation in mice, which may be a potential way to treat allergic asthma (58,116).
Arg1, produced by M2 macrophages, can regulate asthma and allergic inflammation (117). A study demonstrated that compared with M2 macrophages expressing Arg1 after activation of STAT6 mediated by IL-4/13, ILC2s constitutively express Arg1 in a STAT6-independent manner (117). IL-33 can affect Arg1 in lung tissues by promoting the proliferation of ILC2s and indirectly activating macrophages via STAT6 (117). These results further highlight that ILC2s and M2 macrophages have a synergistic regulatory effect on asthma and allergic inflammation via Arg1.
During allergic response, the selective depletion of E3 ligase VHL in innate lymphoid progenitor cells increases hypoxia inducible factor-1α (HIF-1α) expression, which in turn decreases ST2 and inhibits the development of ILC2s induced by IL-33 via epigenetic modification (118). HIF-1α affects glycolysis and phenotype of macrophages (119), suggesting that HIF-1α acts through the regulation of ILC2s and macrophages during allergic reaction.
Lung ILC2s exhibit an inverse correlation with MHC-IIhigh resident macrophages (M1), and a positive correlation with MHC-IIlow resident macrophages (M2), and their contribution to AAD induced by HDM may also be affected by heredity (120). Notably, ILC2s, which are amateur antigen presenting cells (121), cooperate with macrophages to form and regulate adaptive immunity to allergens and helminth (121).
Pulmonary fibrosis
Idiopathic pulmonary fibrosis is characterized by fibroblast aggregation, collagen deposition and extracellular matrix remodeling, in which myofibroblasts are considered effector cells (72). In the pulmonary fibrosis model, AMs were recruited into the alveoli, and the phenotype involves M2 macrophages, which upregulates CD206 on the cell surface (72). In vitro, 264.7 cells treated with IL-4 were used as M2 macrophages, and the TGF-β levels in the supernatant significantly increased. α-SMA expression increased following co-culturing of lung epithelial cells (MLE-12) with M2 macrophages, suggesting that M2 macrophages regulate pulmonary fibrosis by inducing epithelial-to-mesenchymal transition (72).
In addition to the increase of M2 macrophages, the increase of IL-33, IL-13, TGF-β1 and inflammatory chemokines are also observed during pulmonary fibrosis (122). IL-13 and TGF-β1 are produced by M2 macrophages, and IL-13 is secreted by ILC2s, both in vivo and in vitro, and induced by IL-33 (122). As IL-13 can induce the polarization of M2 macrophages (123), a cycle where IL-13 can be produced by M2 macrophages and promotes polarization of M2 macrophages is formed. IL-33 sends signals through ST2, and recruits and guides inflammatory cell function in ST2- and macrophage-dependent manners, and enhances the generation of pro-fibrosis cytokines, thus promoting the occurrence and development of pulmonary fibrosis (122).
Zhao et al (124) reported that bone marrow-derived ILC2s accumulate in the fibrotic lung and activated fibroblasts to promote pulmonary fibrosis by inducing the IL-33/ST2 signaling pathway. In addition, ILC2s are induced by IL-25, which results in significant changes in the pathological process of pulmonary fibrosis through the production of IL-13 (125). Thus, the application of anti-IL-33 antibody and depletion of AMs or ILC2s may be potential therapeutic methods for pulmonary inflammation and fibrosis.
Chronic obstructive pulmonary disease (COPD)
A clinical study demonstrated that normal AMs are mainly nonpolarized (63). However, the polarization of M1 and M2 macrophages significantly enhances, and the co-expression of M1 and M2 markers in the same AMs also significantly increases, with the aggravation of smoking and COPD severity (63).
In human COPD, ILCs accumulate in lung tissues, with increasing signature cytokines, such as IL-5 and GM-CSF (126). The levels of neutrophil elastase and IL-5 increase in patients with acute exacerbation of COPD (127), and the levels of IL-13 mRNA in eosinophils and endothelial cells in the sputum also increase to about 30 times (128). In addition, Th2 cytokine IL-9 can also aggravate lung injury by activating STAT3 in COPD mice and increasing inflammation and oxidative stress (129).
For the interaction of STIP1 homology and U-box-1 (STUB1), IL-4R α is used as the target, which prevents IL-4 or IL-13 signal transduction via ubiquitination mediated proteasome degradation (130). In STUB1-deficient mice, spontaneous airway inflammation increases IL-4R α expression, STAT6 is continuously activated, M2 macrophages are activated and serum IgE increases (130). The level of STUB1 in the airway of patients with asthma or COPD increases, suggesting that upregulation of STUB1 may be an important feedback mechanism for inhibiting IL-4R signal transduction in airway inflammation (130).
Carcinoma of the lungs
In different tumors, type 2 immune responses induce polarization of M2 macrophages, which in turn enhances the invasion and migration of tumor cells by secreting Arg1, IL-10 and TGF-β (107,131,132). The progression of lung cancer is associated with poor patient prognosis and high mortality (133). The survival rate of tumor-bearing mice with vitamin A deficiency diet is low, and the tumor size increases with increasing number of type 2 cytokines, ILC2s and M2 macrophages in BALF of mice, suggesting that ILC2s and polarized M2 macrophages play a synergistic role in promoting cancer progression (133). This synergistic effect may be accomplished via two pathways, the co-promotion of ILC2s and M2-type macrophages by IL-33 (134–136), and the promotion of M2 macrophage polarization by type 2 cytokines (123,137), such as IL-4 and IL-13, secreted by ILC2s (138). This is consistent with the fact that both M2 subtype macrophages (M2a and M2b) and IL-25-stimulated ILC2s favor cancer progression (139). Notably, other substances that inhibit the polarization of M2 macrophages by IL-4/13 can change the tumor microenvironment (140). However, further studies are required to understand the crosstalk between ILC2s and M2 macrophages in lung cancer and determine their underlying molecular mechanisms.
Other diseases affecting the lungs
Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection (141). The lung is an extremely fragile organ that is prone to sepsis (142). In sepsis model with cecal ligation and puncture, IL-33 upregulates IL-5 in ILC2s, whereas IL-5 inhibits neutrophil and monocyte infiltration, suggesting that this axis is involved in lung injury early after sepsis (142). Survivors of sepsis will have chronically low immune functions (143). IL-33, which is produced following sepsis, activates ILC2s and promotes the polarization of M2 macrophages, thus accelerating the proliferation of Treg cells through IL-10 (143). Subsequently, increased ILC2s, M2 macrophages, IL-10 and Treg cells result in immunosuppression (143).
6. Conclusions and perspective
Lung resident ILC2s are important immunoregulatory cells that are involved in metabolism, tissue repair and multiple organ remodeling, outlining a previously unanticipated role of type 2 immunity in regulating basal homeostasis. Similarly, macrophages are a group of pluripotent and plasticity immune cells, that also regulate type 2 immune response. In lungs, AMs and interstitial macrophages differentiate into different cell phenotypes at different stages of development, including M1 and M2 macrophages.
The proliferation and activation of ILC2s and M2 macrophages are consistent, and are not only involved in lung development, but also in lung diseases. In addition, ILC2s and M2 macrophages interact to regulate the lung microenvironment, which is effective in pulmonary development and pulmonary diseases. The crosstalk between IL-4R α-expressing ILC2s and upregulated M2 macrophages produces remarkable effects in lung inflammation, allergy, tumor and fibrosis responses. Further studies are required to better understand the development, activation, turnover and interaction between ILC2s and M2 macrophages in lung tissues. Targeting the IL-33/ILC2s/M2-macrophage axis may be an effective novel approach for the treatment of several lung diseases.
Acknowledgements
Not applicable.
Funding Statement
The present review was financially supported by the Natural Science Foundation of Jiangsu Province (grant no. BK20201226) and the Social Development Foundation of Zhenjiang, China (grant no. SH2020037).
Funding
The present review was financially supported by the Natural Science Foundation of Jiangsu Province (grant no. BK20201226) and the Social Development Foundation of Zhenjiang, China (grant no. SH2020037).
Availability of data and materials
Not applicable.
Authors' contributions
LLM and HYL conceived the present study and performed the literature review. LLM and YZ collected and reviewed the literature, and drafted the initial manuscript. All authors confirm the authenticity of all the raw data and critically revised the manuscript for important intellectual content. LLM and YZ produced the figures. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Domingo-Gonzalez R, Zanini F, Che X, Liu M, Jones RC, Swift MA, Quake SR, Cornfield DN, Alvira CM. Diverse homeostatic and immunomodulatory roles of immune cells in the developing mouse lung at single cell resolution. Elife. 2020;9:e56890. doi: 10.7554/eLife.56890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Martinez FD. Early-life origins of chronic obstructive pulmonary disease. N Engl J Med. 2016;375:871–878. doi: 10.1056/NEJMra1603287. [DOI] [PubMed] [Google Scholar]
- 3.Lange P, Celli B, Agusti A, Boje Jensen G, Divo M, Faner R, Guerra S, Marott JL, Martinez FD, Martinez-Camblor P, et al. Lung-function trajectories leading to chronic obstructive pulmonary disease. N Engl J Med. 2015;373:111–122. doi: 10.1056/NEJMoa1411532. [DOI] [PubMed] [Google Scholar]
- 4.McGeachie MJ, Yates KP, Zhou X, Guo F, Sternberg AL, Van Natta ML, Wise RA, Szefler SJ, Sharma S, Kho AT, et al. Patterns of growth and decline in lung function in persistent childhood asthma. N Engl J Med. 2016;374:1842–1852. doi: 10.1056/NEJMoa1513737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Loering S, Cameron GJM, Bhatt NP, Belz GT, Foster PS, Hansbro PM, Starkey MR. Differences in pulmonary group 2 innate lymphoid cells are dependent on mouse age, sex and strain. Immunol Cell Biol. 2020 Dec 8; doi: 10.1111/imcb.12430. (Epub ahead of print). doi: 10.1111/imcb.12430. [DOI] [PubMed] [Google Scholar]
- 6.Lan F, Zhang N, Holtappels G, De Ruyck N, Krysko O, Van Crombruggen K, Braun H, Johnston SL, Papadopoulos NG, Zhang L, Bachert C. Staphylococcus aureus induces a mucosal type 2 immune response via epithelial cell-derived cytokines. Am J Respir Crit Care Med. 2018;198:452–463. doi: 10.1164/rccm.201710-2112OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sciurba JC, Gieseck RL, Jiwrajka N, White SD, Karmele EP, Redes J, Vannella KM, Henderson NC, Wynn TA, Hart KM. Fibroblast-specific integrin-alpha V differentially regulates type 17 and type 2 driven inflammation and fibrosis. J Pathol. 2019;248:16–29. doi: 10.1002/path.5215. [DOI] [PubMed] [Google Scholar]
- 8.Hajimohammadi B, Athari SM, Abdollahi M, Vahedi G, Athari SS. Oral administration of acrylamide worsens the inflammatory responses in the airways of asthmatic mice through agitation of oxidative stress in the lungs. Front Immunol. 2020;11:1940. doi: 10.3389/fimmu.2020.01940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ryan NM, Oghumu S. Role of mast cells in the generation of a T-helper type 2 dominated anti-helminthic immune response. Biosci Rep. 2019;39:BSR20181771. doi: 10.1042/BSR20181771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Choi JP, Kim YM, Choi HI, Choi SJ, Park HT, Lee WH, Gho YS, Jee YK, Jeon SG, Kim YK. An important role of tumor necrosis factor receptor-2 on natural killer T cells on the development of dsRNA-enhanced Th2 cell response to inhaled allergens. Allergy. 2014;69:186–198. doi: 10.1111/all.12301. [DOI] [PubMed] [Google Scholar]
- 11.Sun L, Cornell TT, LeVine A, Berlin AA, Hinkovska-Galcheva V, Fleszar AJ, Lukacs NW, Shanley TP. Dual role of interleukin-10 in the regulation of respiratory syncitial virus (RSV)-induced lung inflammation. Clin Exp Immunol. 2013;172:263–279. doi: 10.1111/cei.12059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Helou DG, Shafiei-Jahani P, Lo R, Howard E, Hurrell BP, Galle-Treger L, Painter JD, Lewis G, Soroosh P, Sharpe AH, Akbari O. PD-1 pathway regulates ILC2 metabolism and PD-1 agonist treatment ameliorates airway hyperreactivity. Nat Commun. 2020;11:3998. doi: 10.1038/s41467-020-17813-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Leyva-Castillo JM, Galand C, Mashiko S, Bissonnette R, McGurk A, Ziegler SF, Dong C, McKenzie ANJ, Sarfati M, Geha RS. ILC2 activation by keratinocyte-derived IL-25 drives IL-13 production at sites of allergic skin inflammation. J Allergy Clin Immunol. 2020;145:1606–1614.e4. doi: 10.1016/j.jaci.2020.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Miller MM, Patel PS, Bao K, Danhorn T, O'Connor BP, Reinhardt RL. BATF acts as an essential regulator of IL-25-responsive migratory ILC2 cell fate and function. Sci Immunol. 2020;5:eaay3994. doi: 10.1126/sciimmunol.aay3994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fort MM, Cheung J, Yen D, Li J, Zurawski SM, Lo S, Menon S, Clifford T, Hunte B, Lesley R, et al. IL-25 induces IL-4, IL-5, and IL-13 and Th2-associated pathologies in vivo. Immunity. 2001;15:985–995. doi: 10.1016/S1074-7613(01)00243-6. [DOI] [PubMed] [Google Scholar]
- 16.Hurst SD, Muchamuel T, Gorman DM, Gilbert JM, Clifford T, Kwan S, Menon S, Seymour B, Jackson C, Kung TT, et al. New IL-17 family members promote Th1 or Th2 responses in the lung: In vivo function of the novel cytokine IL-25. J Immunol. 2002;169:443–453. doi: 10.4049/jimmunol.169.1.443. [DOI] [PubMed] [Google Scholar]
- 17.Oliphant CJ, Hwang YY, Walker JA, Salimi M, Wong SH, Brewer JM, Englezakis A, Barlow JL, Hams E, Scanlon ST, et al. MHCII-mediated dialog between group 2 innate lymphoid cells and CD4(+) T cells potentiates type 2 immunity and promotes parasitic helminth expulsion. Immunity. 2014;41:283–295. doi: 10.1016/j.immuni.2014.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.She L, Alanazi HH, Yan L, Brooks EG, Dube PH, Xiang Y, Zhang F, Sun Y, Liu Y, Zhang X, Li XD. Sensing and signaling of immunogenic extracellular RNAs restrain group 2 innate lymphoid cell-driven acute lung inflammation and airway hyperresponsiveness. PLoS One. 2020;15:e0236744. doi: 10.1371/journal.pone.0236744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Entwistle LJ, Gregory LG, Oliver RA, Branchett WJ, Puttur F, Lloyd CM. Pulmonary group 2 innate lymphoid cell phenotype is context specific: Determining the effect of strain, location, and stimuli. Front Immunol. 2019;10:3114. doi: 10.3389/fimmu.2019.03114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gieseck RL, III, Wilson MS, Wynn TA. Type 2 immunity in tissue repair and fibrosis. Nat Rev Immunol. 2018;18:62–76. doi: 10.1038/nri.2017.90. [DOI] [PubMed] [Google Scholar]
- 21.Katsura Y, Harada N, Harada S, Ishimori A, Makino F, Ito J, Kamachi F, Okumura K, Akiba H, Atsuta R, Takahashi K. Characteristics of alveolar macrophages from murine models of OVA-induced allergic airway inflammation and LPS-induced acute airway inflammation. Exp Lung Res. 2015;41:370–382. doi: 10.3109/01902148.2015.1044137. [DOI] [PubMed] [Google Scholar]
- 22.Fang SB, Zhang HY, Meng XC, Wang C, He BX, Peng YQ, Xu ZB, Fan XL, Wu ZJ, Wu ZC, et al. Small extracellular vesicles derived from human MSCs prevent allergic airway inflammation via immunomodulation on pulmonary macrophages. Cell Death Dis. 2020;11:409. doi: 10.1038/s41419-020-2606-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Su B, Han H, Gong Y, Li X, Ji C, Yao J, Yang J, Hu W, Zhao W, Li J, et al. Let-7d inhibits intratumoral macrophage M2 polarization and subsequent tumor angiogenesis by targeting IL-13 and IL-10. Cancer Immunol Immunother. 2020 Nov 25; doi: 10.1007/s00262-020-02791-6. (Epub ahead of print). doi: 10.1007/s00262-020-02791-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.De Salvo C, Buela KA, Pizarro TT. Cytokine-mediated regulation of innate lymphoid cell plasticity in gut mucosal immunity. Front Immunol. 2020;11:585319. doi: 10.3389/fimmu.2020.585319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Silver J, Humbles AA, Ohne Y. Isolation, culture, and induction of plasticity in ILC2s. Methods Mol Biol. 2020;2121:115–127. doi: 10.1007/978-1-0716-0338-3_11. [DOI] [PubMed] [Google Scholar]
- 26.Vacca P, Chiossone L, Mingari MC, Moretta L. Heterogeneity of NK cells and other innate lymphoid cells in human and murine decidua. Front Immunol. 2019;10:170. doi: 10.3389/fimmu.2019.00170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li S, Bostick JW, Ye J, Qiu J, Zhang B, Urban JF, Jr, Avram D, Zhou L. Aryl hydrocarbon receptor signaling cell intrinsically inhibits intestinal group 2 innate lymphoid cell function. Immunity. 2018;49:915–928.e5. doi: 10.1016/j.immuni.2018.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Klose CS, Artis D. Innate lymphoid cells as regulators of immunity, inflammation and tissue homeostasis. Nat Immunol. 2016;17:765–774. doi: 10.1038/ni.3489. [DOI] [PubMed] [Google Scholar]
- 29.Kabata H, Moro K, Koyasu S. The group 2 innate lymphoid cell (ILC2) regulatory network and its underlying mechanisms. Immunol Rev. 2018;286:37–52. doi: 10.1111/imr.12706. [DOI] [PubMed] [Google Scholar]
- 30.Pasha MA, Patel G, Hopp R, Yang Q. Role of innate lymphoid cells in allergic diseases. Allergy Asthma Proc. 2019;40:138–145. doi: 10.2500/aap.2019.40.4217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Monticelli LA, Sonnenberg GF, Abt MC, Alenghat T, Ziegler CG, Doering TA, Angelosanto JM, Laidlaw BJ, Yang CY, Sathaliyawala T, et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat Immunol. 2011;12:1045–1054. doi: 10.1038/ni.2131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Simoni Y, Fehlings M, Kloverpris HN, McGovern N, Koo SL, Loh CY, Lim S, Kurioka A, Fergusson JR, Tang CL, et al. Human innate lymphoid cell subsets possess tissue-type based heterogeneity in phenotype and frequency. Immunity. 2018;48:1060. doi: 10.1016/j.immuni.2018.04.028. [DOI] [PubMed] [Google Scholar]
- 33.Camelo A, Rosignoli G, Ohne Y, Stewart RA, Overed-Sayer C, Sleeman MA, May RD. IL-33, IL-25, and TSLP induce a distinct phenotypic and activation profile in human type 2 innate lymphoid cells. Blood Adv. 2017;1:577–589. doi: 10.1182/bloodadvances.2016002352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Huang Y, Guo L, Qiu J, Chen X, Hu-Li J, Siebenlist U, Williamson PR, Urban JF, Jr, Paul WE. IL-25-responsive, lineage-negative KLRG1(hi) cells are multipotential ‘inflammatory’ type 2 innate lymphoid cells. Nat Immunol. 2015;16:161–169. doi: 10.1038/ni.3078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li Y, Chen S, Chi Y, Yang Y, Chen X, Wang H, Lv Z, Wang J, Yuan L, Huang P, et al. Kinetics of the accumulation of group 2 innate lymphoid cells in IL-33-induced and IL-25-induced murine models of asthma: A potential role for the chemokine CXCL16. Cell Mol Immunol. 2019;16:75–86. doi: 10.1038/s41423-018-0182-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Salimi M, Barlow JL, Saunders SP, Xue L, Gutowska-Owsiak D, Wang X, Huang LC, Johnson D, Scanlon ST, McKenzie AN, et al. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med. 2013;210:2939–2950. doi: 10.1084/jem.20130351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mohapatra A, Van Dyken SJ, Schneider C, Nussbaum JC, Liang HE, Locksley RM. Group 2 innate lymphoid cells utilize the IRF4-IL-9 module to coordinate epithelial cell maintenance of lung homeostasis. Mucosal Immunol. 2016;9:275–286. doi: 10.1038/mi.2015.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Moretti S, Renga G, Oikonomou V, Galosi C, Pariano M, Iannitti RG, Borghi M, Puccetti M, De Zuani M, Pucillo CE, et al. A mast cell-ILC2-Th9 pathway promotes lung inflammation in cystic fibrosis. Nat Commun. 2017;8:14017. doi: 10.1038/ncomms14017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wilhelm C, Hirota K, Stieglitz B, Van Snick J, Tolaini M, Lahl K, Sparwasser T, Helmby H, Stockinger B. An IL-9 fate reporter demonstrates the induction of an innate IL-9 response in lung inflammation. Nat Immunol. 2011;12:1071–1077. doi: 10.1038/ni.2133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bartemes KR, Kephart GM, Fox SJ, Kita H. Enhanced innate type 2 immune response in peripheral blood from patients with asthma. J Allergy Clin Immunol. 2014;134:671–678.e4. doi: 10.1016/j.jaci.2014.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Motomura Y, Morita H, Moro K, Nakae S, Artis D, Endo TA, Kuroki Y, Ohara O, Koyasu S, Kubo M. Basophil-derived interleukin-4 controls the function of natural helper cells, a member of ILC2s, in lung inflammation. Immunity. 2014;40:758–771. doi: 10.1016/j.immuni.2014.04.013. [DOI] [PubMed] [Google Scholar]
- 42.Matsuki A, Takatori H, Makita S, Yokota M, Tamachi T, Suto A, Suzuki K, Hirose K, Nakajima H. T-bet inhibits innate lymphoid cell-mediated eosinophilic airway inflammation by suppressing IL-9 production. J Allergy Clin Immunol. 2017;139:1355–1367.e6. doi: 10.1016/j.jaci.2016.08.022. [DOI] [PubMed] [Google Scholar]
- 43.Zhang K, Jin Y, Lai D, Wang J, Wang Y, Wu X, Scott M, Li Y, Hou J, Billiar T, et al. RAGE-induced ILC2 expansion in acute lung injury due to haemorrhagic shock. Thorax. 2020;75:209–219. doi: 10.1136/thoraxjnl-2019-213613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ishii T, Muroi M, Horiguchi K, Tanamoto KI, Nagase T, Yamashita N. Activation through toll-like receptor 2 on group 2 innate lymphoid cells can induce asthmatic characteristics. Clin Exp Allergy. 2019;49:1624–1632. doi: 10.1111/cea.13490. [DOI] [PubMed] [Google Scholar]
- 45.Maggi L, Montaini G, Mazzoni A, Rossettini B, Capone M, Rossi MC, Santarlasci V, Liotta F, Rossi O, Gallo O, et al. Human circulating group 2 innate lymphoid cells can express CD154 and promote IgE production. J Allergy Clin Immunol. 2017;139:964–976.e4. doi: 10.1016/j.jaci.2016.06.032. [DOI] [PubMed] [Google Scholar]
- 46.Gury-BenAri M, Thaiss CA, Serafini N, Winter DR, Giladi A, Lara-Astiaso D, Levy M, Salame TM, Weiner A, David E, et al. The spectrum and regulatory landscape of intestinal innate lymphoid cells are shaped by the microbiome. Cell. 2016;166:1231–1246.e13. doi: 10.1016/j.cell.2016.07.043. [DOI] [PubMed] [Google Scholar]
- 47.Robinette ML, Fuchs A, Cortez VS, Lee JS, Wang Y, Durum SK, Gilfillan S, Colonna M, Immunological Genome Consortium Transcriptional programs define molecular characteristics of innate lymphoid cell classes and subsets. Nat Immunol. 2015;16:306–317. doi: 10.1038/ni.3094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kim HS, Jang JH, Lee MB, Jung ID, Kim YM, Park YM, Choi WS. A novel IL-10-producing innate lymphoid cells (ILC10) in a contact hypersensitivity mouse model. BMB Rep. 2016;49:293–296. doi: 10.5483/BMBRep.2016.49.5.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wallrapp A, Burkett PR, Riesenfeld SJ, Kim SJ, Christian E, Abdulnour RE, Thakore PI, Schnell A, Lambden C, Herbst RH, et al. Calcitonin gene-related peptide negatively regulates alarmin-driven type 2 innate lymphoid cell responses. Immunity. 2019;51:709–723.e6. doi: 10.1016/j.immuni.2019.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ho J, Bailey M, Zaunders J, Mrad N, Sacks R, Sewell W, Harvey RJ. Group 2 innate lymphoid cells (ILC2s) are increased in chronic rhinosinusitis with nasal polyps or eosinophilia. Clin Exp Allergy. 2015;45:394–403. doi: 10.1111/cea.12462. [DOI] [PubMed] [Google Scholar]
- 51.Jeffery HC, McDowell P, Lutz P, Wawman RE, Roberts S, Bagnall C, Birtwistle J, Adams DH, Oo YH. Human intrahepatic ILC2 are IL-13positive amphiregulinpositive and their frequency correlates with model of end stage liver disease score. PLoS One. 2017;12:e0188649. doi: 10.1371/journal.pone.0188649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Campbell L, Hepworth MR, Whittingham-Dowd J, Thompson S, Bancroft AJ, Hayes KS, Shaw TN, Dickey BF, Flamar AL, Artis D, et al. ILC2s mediate systemic innate protection by priming mucus production at distal mucosal sites. J Exp Med. 2019;216:2714–2723. doi: 10.1084/jem.20180610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.D'Souza SS, Shen X, Fung ITH, Ye L, Kuentzel M, Chittur SV, Furuya Y, Siebel CW, Maillard IP, Metzger DW, Yang Q. Compartmentalized effects of aging on group 2 innate lymphoid cell development and function. Aging Cell. 2019;18:e13019. doi: 10.1111/acel.13019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ghaedi M, Shen ZY, Orangi M, Martinez-Gonzalez I, Wei L, Lu X, Das A, Heravi-Moussavi A, Marra MA, Bhandoola A, Takei F. Single-cell analysis of RORα tracer mouse lung reveals ILC progenitors and effector ILC2 subsets. J Exp Med. 2020;217 doi: 10.1084/jem.20182293. jem.20182293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Steer CA, Matha L, Shim H, Takei F. Lung group 2 innate lymphoid cells are trained by endogenous IL-33 in the neonatal period. JCI Insight. 2020;5:e135961. doi: 10.1172/jci.insight.135961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lindquist RL, Bayat-Sarmadi J, Leben R, Niesner R, Hauser AE. NAD(P)H oxidase activity in the small intestine is predominantly found in enterocytes, not professional phagocytes. Int J Mol Sci. 2018;19:1365. doi: 10.3390/ijms19051365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vellozo NS, Pereira-Marques ST, Cabral-Piccin MP, Filardy AA, Ribeiro-Gomes FL, Rigoni TS, DosReis GA, Lopes MF. All-trans retinoic acid promotes an M1- to M2-phenotype shift and inhibits macrophage-mediated immunity to leishmania major. Front Immunol. 2017;8:1560. doi: 10.3389/fimmu.2017.01560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Moreira AP, Cavassani KA, Hullinger R, Rosada RS, Fong DJ, Murray L, Hesson DP, Hogaboam CM. Serum amyloid P attenuates M2 macrophage activation and protects against fungal spore-induced allergic airway disease. J Allergy Clin Immunol. 2010;126:712–721.e7. doi: 10.1016/j.jaci.2010.06.010. [DOI] [PubMed] [Google Scholar]
- 59.Wu Y, Hirschi KK. Tissue-resident macrophage development and function. Front Cell Dev Biol. 2020;8:617879. doi: 10.3389/fcell.2020.617879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liu G, Zhai H, Zhang T, Li S, Li N, Chen J, Gu M, Qin Z, Liu X. New therapeutic strategies for IPF: Based on the ‘phagocytosis-secretion-immunization’ network regulation mechanism of pulmonary macrophages. Biomed Pharmacother. 2019;118:109230. doi: 10.1016/j.biopha.2019.109230. [DOI] [PubMed] [Google Scholar]
- 61.Li R, Shang Y, Hu X, Yu Y, Zhou T, Xiong W, Zou X. ATP/P2X7r axis mediates the pathological process of allergic asthma by inducing M2 polarization of alveolar macrophages. Exp Cell Res. 2020;386:111708. doi: 10.1016/j.yexcr.2019.111708. [DOI] [PubMed] [Google Scholar]
- 62.Ke X, Chen C, Song Y, Cai Q, Li J, Tang Y, Han X, Qu W, Chen A, Wang H, et al. Hypoxia modifies the polarization of macrophages and their inflammatory microenvironment, and inhibits malignant behavior in cancer cells. Oncol Lett. 2019;18:5871–5878. doi: 10.3892/ol.2019.10956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bazzan E, Turato G, Tine M, Radu CM, Balestro E, Rigobello C, Biondini D, Schiavon M, Lunardi F, Baraldo S, et al. Dual polarization of human alveolar macrophages progressively increases with smoking and COPD severity. Respir Res. 2017;18:40. doi: 10.1186/s12931-017-0522-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lin F, Song C, Zeng Y, Li Y, Li H, Liu B, Dai M, Pan P. Canagliflozin alleviates LPS-induced acute lung injury by modulating alveolar macrophage polarization. Int Immunopharmacol. 2020;88:106969. doi: 10.1016/j.intimp.2020.106969. [DOI] [PubMed] [Google Scholar]
- 65.Soliman E, Elhassanny AE, Malur A, McPeek M, Bell A, Leffler N, Van Dross R, Jones JL, Malur AG, Thomassen MJ. Impaired mitochondrial function of alveolar macrophages in carbon nanotube-induced chronic pulmonary granulomatous disease. Toxicology. 2020;445:152598. doi: 10.1016/j.tox.2020.152598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Nenasheva T, Gerasimova T, Serdyuk Y, Grigor'eva E, Kosmiadi G, Nikolaev A, Dashinimaev E, Lyadova I. Macrophages derived from human induced pluripotent stem cells are low-activated ‘Naive-Like’ cells capable of restricting mycobacteria growth. Front Immunol. 2020;11:1016. doi: 10.3389/fimmu.2020.01016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang L, Wang Y, Wu G, Xiong W, Gu W, Wang CY. Macrophages: Friend or foe in idiopathic pulmonary fibrosis? Respir Res. 2018;19:170. doi: 10.1186/s12931-018-0864-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bronte V, Zanovello P. Regulation of immune responses by L-arginine metabolism. Nat Rev Immunol. 2005;5:641–654. doi: 10.1038/nri1668. [DOI] [PubMed] [Google Scholar]
- 69.Grabarz F, Aguiar CF, Correa-Costa M, Braga TT, Hyane MI, Andrade-Oliveira V, Landgraf MA, Camara NOS. Protective role of NKT cells and macrophage M2-driven phenotype in bleomycin-induced pulmonary fibrosis. Inflammopharmacology. 2018;26:491–504. doi: 10.1007/s10787-017-0383-7. [DOI] [PubMed] [Google Scholar]
- 70.de Campos GY, Oliveira RA, Oliveira-Brito PK, Roque-Barreira MC, da Silva TA. Pro-inflammatory response ensured by LPS and Pam3CSK4 in RAW 264.7 cells did not improve a fungistatic effect on Cryptococcus gattii infection. PeerJ. 2020;8:e10295. doi: 10.7717/peerj.10295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Anthony RM, Urban JF, Jr, Alem F, Hamed HA, Rozo CT, Boucher JL, Van Rooijen N, Gause WC. Memory T(H)2 cells induce alternatively activated macrophages to mediate protection against nematode parasites. Nat Med. 2006;12:955–960. doi: 10.1038/nm1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zhu L, Fu X, Chen X, Han X, Dong P. M2 macrophages induce EMT through the TGF-beta/Smad2 signaling pathway. Cell Biol Int. 2017;41:960–968. doi: 10.1002/cbin.10788. [DOI] [PubMed] [Google Scholar]
- 73.Loering S, Cameron GJ, Starkey MR, Hansbro PM. Lung development and emerging roles for type 2 immunity. J Pathol. 2019;247:686–696. doi: 10.1002/path.5211. [DOI] [PubMed] [Google Scholar]
- 74.Blackwell TS, Hipps AN, Yamamoto Y, Han W, Barham WJ, Ostrowski MC, Yull FE, Prince LS. NF-kappaB signaling in fetal lung macrophages disrupts airway morphogenesis. J Immunol. 2011;187:2740–2747. doi: 10.4049/jimmunol.1101495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jones CV, Williams TM, Walker KA, Dickinson H, Sakkal S, Rumballe BA, Little MH, Jenkin G, Ricardo SD. M2 macrophage polarisation is associated with alveolar formation during postnatal lung development. Respir Res. 2013;14:41. doi: 10.1186/1465-9921-14-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Saluzzo S, Gorki AD, Rana BMJ, Martins R, Scanlon S, Starkl P, Lakovits K, Hladik A, Korosec A, Sharif O, et al. First-breath-induced type 2 pathways shape the lung immune environment. Cell Rep. 2017;18:1893–1905. doi: 10.1016/j.celrep.2017.01.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Schneider C, Lee J, Koga S, Ricardo-Gonzalez RR, Nussbaum JC, Smith LK, Villeda SA, Liang HE, Locksley RM. Tissue-resident group 2 innate lymphoid cells differentiate by layered ontogeny and in situ perinatal priming. Immunity. 2019;50:1425–1438.e5. doi: 10.1016/j.immuni.2019.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Huang Y, Mao K, Chen X, Sun MA, Kawabe T, Li W, Usher N, Zhu J, Urban JF, Jr, Paul WE, Germain RN. S1P-dependent interorgan trafficking of group 2 innate lymphoid cells supports host defense. Science. 2018;359:114–119. doi: 10.1126/science.aam5809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Steer CA, Martinez-Gonzalez I, Ghaedi M, Allinger P, Matha L, Takei F. Group 2 innate lymphoid cell activation in the neonatal lung drives type 2 immunity and allergen sensitization. J Allergy Clin Immunol. 2017;140:593–595.e3. doi: 10.1016/j.jaci.2016.12.984. [DOI] [PubMed] [Google Scholar]
- 80.Nussbaum JC, Van Dyken SJ, von Moltke J, Cheng LE, Mohapatra A, Molofsky AB, Thornton EE, Krummel MF, Chawla A, Liang HE, Locksley RM. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature. 2013;502:245–248. doi: 10.1038/nature12526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.de Kleer IM, Kool M, de Bruijn MJ, Willart M, van Moorleghem J, Schuijs MJ, Plantinga M, Beyaert R, Hams E, Fallon PG, et al. Perinatal activation of the interleukin-33 pathway promotes type 2 immunity in the developing lung. Immunity. 2016;45:1285–1298. doi: 10.1016/j.immuni.2016.10.031. [DOI] [PubMed] [Google Scholar]
- 82.Ghaedi M, Steer CA, Martinez-Gonzalez I, Halim TYF, Abraham N, Takei F. Common-lymphoid-progenitor-independent pathways of innate and T lymphocyte development. Cell Rep. 2016;15:471–480. doi: 10.1016/j.celrep.2016.03.039. [DOI] [PubMed] [Google Scholar]
- 83.Sahoo D, Zaramela LS, Hernandez GE, Mai U, Taheri S, Dang D, Stouch AN, Medal RM, McCoy AM, Aschner JL, et al. Transcriptional profiling of lung macrophages identifies a predictive signature for inflammatory lung disease in preterm infants. Commun Biol. 2020;3:259. doi: 10.1038/s42003-020-0985-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ubags NDJ, Alejandre Alcazar MA, Kallapur SG, Knapp S, Lanone S, Lloyd CM, Morty RE, Pattaroni C, Reynaert NL, Rottier RJ, et al. Early origins of lung disease: Towards an interdisciplinary approach. Eur Respir Rev. 2020;29:200191. doi: 10.1183/16000617.0191-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Obata-Ninomiya K, Ishiwata K, Tsutsui H, Nei Y, Yoshikawa S, Kawano Y, Minegishi Y, Ohta N, Watanabe N, Kanuka H, Karasuyama H. The skin is an important bulwark of acquired immunity against intestinal helminths. J Exp Med. 2013;210:2583–2595. doi: 10.1084/jem.20130761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Minutti CM, Jackson-Jones LH, Garcia-Fojeda B, Knipper JA, Sutherland TE, Logan N, Ringqvist E, Guillamat-Prats R, Ferenbach DA, Artigas A, et al. Local amplifiers of IL-4Rα-mediated macrophage activation promote repair in lung and liver. Science. 2017;356:1076–1080. doi: 10.1126/science.aaj2067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chen S, Kammerl IE, Vosyka O, Baumann T, Yu Y, Wu Y, Irmler M, Overkleeft HS, Beckers J, Eickelberg O, et al. Immunoproteasome dysfunction augments alternative polarization of alveolar macrophages. Cell Death Differ. 2016;23:1026–1037. doi: 10.1038/cdd.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kim J, Chang Y, Bae B, Sohn KH, Cho SH, Chung DH, Kang HR, Kim HY. Innate immune crosstalk in asthmatic airways: Innate lymphoid cells coordinate polarization of lung macrophages. J Allergy Clin Immunol. 2019;143:1769–1782.e11. doi: 10.1016/j.jaci.2018.10.040. [DOI] [PubMed] [Google Scholar]
- 89.King SD, Chen SY. Recent progress on surfactant protein A: cellular function in lung and kidney disease development. Am J Physiol Cell Physiol. 2020;319:C316–C320. doi: 10.1152/ajpcell.00195.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Buckley S, Bui KC, Hussain M, Warburton D. Dynamics of TGF-beta 3 peptide activity during rat alveolar epithelial cell proliferative recovery from acute hyperoxia. Am J Physiol. 1996;271:L54–L60. doi: 10.1152/ajplung.1996.271.1.L54. [DOI] [PubMed] [Google Scholar]
- 91.Lechner AJ, Driver IH, Lee J, Conroy CM, Nagle A, Locksley RM, Rock JR. Recruited monocytes and type 2 immunity promote lung regeneration following pneumonectomy. Cell Stem Cell. 2017;21:120–134.e7. doi: 10.1016/j.stem.2017.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rindler TN, Stockman CA, Filuta AL, Brown KM, Snowball JM, Zhou W, Veldhuizen R, Zink EM, Dautel SE, Clair G, et al. Alveolar injury and regeneration following deletion of ABCA3. JCI Insight. 2017;2:e97381. doi: 10.1172/jci.insight.97381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kurowska-Stolarska M, Stolarski B, Kewin P, Murphy G, Corrigan CJ, Ying S, Pitman N, Mirchandani A, Rana B, van Rooijen N, et al. IL-33 amplifies the polarization of alternatively activated macrophages that contribute to airway inflammation. J Immunol. 2009;183:6469–6477. doi: 10.4049/jimmunol.0901575. [DOI] [PubMed] [Google Scholar]
- 94.Cohen M, Giladi A, Gorki AD, Solodkin DG, Zada M, Hladik A, Miklosi A, Salame TM, Halpern KB, David E, et al. Lung single-cell signaling interaction map reveals basophil role in macrophage imprinting. Cell. 2018;175:1031–1044.e18. doi: 10.1016/j.cell.2018.09.009. [DOI] [PubMed] [Google Scholar]
- 95.Dagher R, Copenhaver AM, Besnard V, Berlin A, Hamidi F, Maret M, Wang J, Qu X, Shrestha Y, Wu J, et al. IL-33-ST2 axis regulates myeloid cell differentiation and activation enabling effective club cell regeneration. Nat Commun. 2020;11:4786. doi: 10.1038/s41467-020-18466-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Silva JD, Su Y, Calfee CS, Delucchi KL, Weiss D, McAuley DF, O'Kane C, Krasnodembskaya AD. MSC extracellular vesicles rescue mitochondrial dysfunction and improve barrier integrity in clinically relevant models of ARDS. Eur Respir J. 2020 Dec 17; doi: 10.1183/13993003.02978-2020. (Epub ahead of print). doi: 10.1183/13993003.02978-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Duan F, Guo L, Yang L, Han Y, Thakur A, Nilsson-Payant BE, Wang P, Zhang Z, Ma CY, Zhou X, et al. Modeling COVID-19 with human pluripotent stem cell-derived cells reveals synergistic effects of anti-inflammatory macrophages with ACE2 inhibition against SARS-CoV-2. Res Sq. 2020 Aug 20; (Epub ahead of print). doi: 10.21203/rs.3.rs-62758/v1. [Google Scholar]
- 98.Sersar SI, Elnahas HA, Saleh AB, Moussa SA, Ghafar WA. Pulmonary parasitosis: Applied clinical and therapeutic issues. Heart Lung Circ. 2006;15:24–29. doi: 10.1016/j.hlc.2005.04.004. [DOI] [PubMed] [Google Scholar]
- 99.Miller MM, Reinhardt RL. The heterogeneity, origins, and impact of migratory iILC2 cells in anti-helminth immunity. Front Immunol. 2020;11:1594. doi: 10.3389/fimmu.2020.01594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Meiners J, Reitz M, Rudiger N, Turner JE, Heepmann L, Rudolf L, Hartmann W, McSorley HJ, Breloer M. IL-33 facilitates rapid expulsion of the parasitic nematode Strongyloides ratti from the intestine via ILC2- and IL-9-driven mast cell activation. PLoS Pathog. 2020;16:e1009121. doi: 10.1371/journal.ppat.1009121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Webb LM, Tait Wojno ED. The role of rare innate immune cells in Type 2 immune activation against parasitic helminths. Parasitology. 2017;144:1288–1301. doi: 10.1017/S0031182017000488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bouchery T, Kyle R, Camberis M, Shepherd A, Filbey K, Smith A, Harvie M, Painter G, Johnston K, Ferguson P, et al. ILC2s and T cells cooperate to ensure maintenance of M2 macrophages for lung immunity against hookworms. Nat Commun. 2015;6:6970. doi: 10.1038/ncomms7970. [DOI] [PubMed] [Google Scholar]
- 103.Nieves W, Hung LY, Oniskey TK, Boon L, Foretz M, Viollet B, Herbert DR. Myeloid-restricted AMPKα1 promotes host immunity and protects against IL-12/23p40-dependent lung injury during hookworm infection. J Immunol. 2016;196:4632–4640. doi: 10.4049/jimmunol.1502218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Thawer S, Auret J, Schnoeller C, Chetty A, Smith K, Darby M, Roberts L, Mackay RM, Whitwell HJ, Timms JF, et al. Surfactant protein-D is essential for immunity to helminth infection. PLoS Pathog. 2016;12:e1005461. doi: 10.1371/journal.ppat.1005461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Snietura M, Brewczynski A, Kopec A, Rutkowski T. Infiltrates of M2-like tumour-associated macrophages are adverse prognostic factor in patients with human papillomavirus-negative but not in human papillomavirus-positive oropharyngeal squamous cell carcinoma. Pathobiology. 2020;87:75–86. doi: 10.1159/000505522. [DOI] [PubMed] [Google Scholar]
- 106.Yan C, Wu J, Xu N, Li J, Zhou QY, Yang HM, Cheng XD, Liu JX, Dong X, Koda S, et al. TLR4 deficiency exacerbates biliary injuries and peribiliary fibrosis caused by clonorchis sinensis in a resistant mouse strain. Front Cell Infect Microbiol. 2021;10:526997. doi: 10.3389/fcimb.2020.526997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Wang H, Zhang CS, Fang BB, Hou J, Li WD, Li ZD, Li L, Bi XJ, Li L, Abulizi A, et al. Dual role of hepatic macrophages in the establishment of the echinococcus multilocularis metacestode in mice. Front Immunol. 2021;11:600635. doi: 10.3389/fimmu.2020.600635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kindermann M, Knipfer L, Obermeyer S, Muller U, Alber G, Bogdan C, Schleicher U, Neurath MF, Wirtz S. Group 2 innate lymphoid cells (ILC2) suppress beneficial type 1 immune responses during pulmonary cryptococcosis. Front Immunol. 2020;11:209. doi: 10.3389/fimmu.2020.00209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Han M, Ishikawa T, Bermick JR, Rajput C, Lei J, Goldsmith AM, Jarman CR, Lee J, Bentley JK, Hershenson MB. IL-1β prevents ILC2 expansion, type 2 cytokine secretion, and mucus metaplasia in response to early-life rhinovirus infection in mice. Allergy. 2020;75:2005–2019. doi: 10.1111/all.14241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Yamaguchi M, Samuchiwal SK, Quehenberger O, Boyce JA, Balestrieri B. Macrophages regulate lung ILC2 activation via Pla2g5-dependent mechanisms. Mucosal Immunol. 2018;11:615–626. doi: 10.1038/mi.2017.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Panova V, Gogoi M, Rodriguez-Rodriguez N, Sivasubramaniam M, Jolin HE, Heycock MWD, Walker JA, Rana BM, Drynan LF, Hodskinson M, et al. Group-2 innate lymphoid cell-dependent regulation of tissue neutrophil migration by alternatively activated macrophage-secreted Ear11. Mucosal Immunol. 2020;14:26–37. doi: 10.1038/s41385-020-0298-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wu K, Byers DE, Jin X, Agapov E, Alexander-Brett J, Patel AC, Cella M, Gilfilan S, Colonna M, Kober DL, et al. TREM-2 promotes macrophage survival and lung disease after respiratory viral infection. J Exp Med. 2015;212:681–697. doi: 10.1084/jem.20141732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Botelho F, Dubey A, Ayaub EA, Park R, Yip A, Humbles A, Kolbeck R, Richards CD. IL-33 mediates lung inflammation by the IL-6-type cytokine oncostatin M. Mediators Inflamm. 2020;2020:4087315. doi: 10.1155/2020/4087315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Pei W, Zhang Y, Li X, Luo M, Chen T, Zhang M, Zhong M, Lv K. LncRNA AK085865 depletion ameliorates asthmatic airway inflammation by modulating macrophage polarization. Int Immunopharmacol. 2020;83:106450. doi: 10.1016/j.intimp.2020.106450. [DOI] [PubMed] [Google Scholar]
- 115.Cai H, Wang J, Mo Y, Ye L, Zhu G, Song X, Zhu M, Xue X, Yang C, Jin M. Salidroside suppresses group 2 innate lymphoid cell-mediated allergic airway inflammation by targeting IL-33/ST2 axis. Int Immunopharmacol. 2020;81:106243. doi: 10.1016/j.intimp.2020.106243. [DOI] [PubMed] [Google Scholar]
- 116.Nagashima R, Kosai H, Masuo M, Izumiyama K, Noshikawaji T, Morimoto M, Kumaki S, Miyazaki Y, Motohashi H, Yamamoto M, Tanaka N. Nrf2 suppresses allergic lung inflammation by attenuating the type 2 innate lymphoid cell response. J Immunol. 2019;202:1331–1339. doi: 10.4049/jimmunol.1801180. [DOI] [PubMed] [Google Scholar]
- 117.Bando JK, Nussbaum JC, Liang HE, Locksley RM. Type 2 innate lymphoid cells constitutively express arginase-I in the naive and inflamed lung. J Leukoc Biol. 2013;94:877–884. doi: 10.1189/jlb.0213084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Li Q, Li D, Zhang X, Wan Q, Zhang W, Zheng M, Zou L, Elly C, Lee JH, Liu YC. E3 Ligase VHL promotes group 2 innate lymphoid cell maturation and function via glycolysis inhibition and induction of interleukin-33 receptor. Immunity. 2018;48:258–270.e5. doi: 10.1016/j.immuni.2017.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Liu J, Qiu P, Qin J, Wu X, Wang X, Yang X, Li B, Zhang W, Ye K, Peng Z, Lu X. Allogeneic adipose-derived stem cells promote ischemic muscle repair by inducing M2 macrophage polarization via the HIF-1α/IL-10 pathway. Stem Cells. 2020;38:1307–1320. doi: 10.1002/stem.3250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Scoville DK, Nolin JD, Ogden HL, An D, Afsharinejad Z, Johnson BW, Bammler TK, Gao X, Frevert CW, Altemeier WA, et al. Quantum dots and mouse strain influence house dust mite-induced allergic airway disease. Toxicol Appl Pharmacol. 2019;368:55–62. doi: 10.1016/j.taap.2019.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Schuijs MJ, Hammad H, Lambrecht BN. Professional and ‘Amateur’ Antigen-presenting cells in type 2 immunity. Trends Immunol. 2019;40:22–34. doi: 10.1016/j.it.2018.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Li D, Guabiraba R, Besnard AG, Komai-Koma M, Jabir MS, Zhang L, Graham GJ, Kurowska-Stolarska M, Liew FY, McSharry C, Xu D. IL-33 promotes ST2-dependent lung fibrosis by the induction of alternatively activated macrophages and innate lymphoid cells in mice. J Allergy Clin Immunol. 2014;134:1422–1432.e11. doi: 10.1016/j.jaci.2014.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Park HJ, Chi GY, Choi YH, Park SH. Lupeol suppresses plasminogen activator inhibitor-1-mediated macrophage recruitment and attenuates M2 macrophage polarization. Biochem Biophys Res Commun. 2020;527:889–895. doi: 10.1016/j.bbrc.2020.04.160. [DOI] [PubMed] [Google Scholar]
- 124.Zhao Y, De Los Santos FG, Wu Z, Liu T, Phan SH. An ST2-dependent role of bone marrow-derived group 2 innate lymphoid cells in pulmonary fibrosis. J Pathol. 2018;245:399–409. doi: 10.1002/path.5092. [DOI] [PubMed] [Google Scholar]
- 125.Hams E, Armstrong ME, Barlow JL, Saunders SP, Schwartz C, Cooke G, Fahy RJ, Crotty TB, Hirani N, Flynn RJ, et al. IL-25 and type 2 innate lymphoid cells induce pulmonary fibrosis. Proc Natl Acad Sci USA. 2014;111:367–372. doi: 10.1073/pnas.1315854111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.De Grove KC, Provoost S, Verhamme FM, Bracke KR, Joos GF, Maes T, Brusselle GG. Characterization and quantification of innate lymphoid cell subsets in human lung. PLoS One. 2016;11:e0145961. doi: 10.1371/journal.pone.0145961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Pouwels SD, Zijlstra GJ, van der Toorn M, Hesse L, Gras R, Ten Hacken NH, Krysko DV, Vandenabeele P, de Vries M, van Oosterhout AJ, et al. Cigarette smoke-induced necroptosis and DAMP release trigger neutrophilic airway inflammation in mice. Am J Physiol Lung Cell Mol Physiol. 2016;310:L377–L386. doi: 10.1152/ajplung.00174.2015. [DOI] [PubMed] [Google Scholar]
- 128.Hershenson MB. Rhinovirus-induced exacerbations of asthma and COPD. Scientifica (Cairo) 2013;2013:405876. doi: 10.1155/2013/405876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zou SC, Pang LL, Mao QS, Wu SY, Xiao QF. IL-9 exacerbates the development of chronic obstructive pulmonary disease through oxidative stress. Eur Rev Med Pharmacol Sci. 2018;22:8877–8884. doi: 10.26355/eurrev_201812_16656. [DOI] [PubMed] [Google Scholar]
- 130.Wei Q, Sha Y, Bhattacharya A, Abdel Fattah E, Bonilla D, Jyothula SS, Pandit L, Khurana Hershey GK, Eissa NT. Regulation of IL-4 receptor signaling by STUB1 in lung inflammation. Am J Respir Crit Care Med. 2014;189:16–29. doi: 10.1164/rccm.201305-0874OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Saha J, Sarkar D, Pramanik A, Mahanti K, Adhikary A, Bhattacharyya S. PGE2-HIF1α reciprocal induction regulates migration, phenotypic alteration and immunosuppressive capacity of macrophages in tumor microenvironment. Life Sci. 2020;253:117731. doi: 10.1016/j.lfs.2020.117731. [DOI] [PubMed] [Google Scholar]
- 132.Lu Q, Wang X, Zhu J, Fei X, Chen H, Li C. Hypoxic tumor-derived exosomal Circ0048117 facilitates M2 macrophage polarization acting as miR-140 sponge in esophageal squamous cell carcinoma. Onco Targets Ther. 2020;13:11883–11897. doi: 10.2147/OTT.S284192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Cui W, Zhang W, Yuan X, Liu S, Li M, Niu J, Zhang P, Li D. Vitamin A deficiency execrates Lewis lung carcinoma via induction of type 2 innate lymphoid cells and alternatively activates macrophages. Food Sci Nutr. 2019;7:1288–1294. doi: 10.1002/fsn3.961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Robbins SM, Senger DL. To promote or inhibit glioma progression, that is the question for IL-33. Cell Stress. 2020;5:19–22. doi: 10.15698/cst2021.01.240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Mai S, Liu L, Jiang J, Ren P, Diao D, Wang H, Cai K. Oesophageal squamous cell carcinoma-associated IL-33 rewires macrophage polarization towards M2 via activating ornithine decarboxylase. Cell Prolif. 2021;54:e12960. doi: 10.1111/cpr.12960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Li J, Razumilava N, Gores GJ, Walters S, Mizuochi T, Mourya R, Bessho K, Wang YH, Glaser SS, Shivakumar P, Bezerra JA. Biliary repair and carcinogenesis are mediated by IL-33-dependent cholangiocyte proliferation. J Clin Invest. 2014;124:3241–3251. doi: 10.1172/JCI73742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Yang Y, Xia S, Zhang L, Wang W, Chen L, Zhan W. MiR-324-5p/PTPRD/CEBPD axis promotes papillary thyroid carcinoma progression via microenvironment alteration. Cancer Biol Ther. 2020;21:522–532. doi: 10.1080/15384047.2020.1736465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.You Y, Zhang X, Wang X, Yue D, Meng F, Zhu J, Wang Y, Sun X. ILC2 Proliferated by IL-33 stimulation alleviates acute colitis in Rag1(−/-) Mouse through promoting M2 macrophage polarization. J Immunol Res. 2020;2020:5018975. doi: 10.1155/2020/5018975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Della Valle L, Gatta A, Farinelli A, Scarano G, Lumaca A, Tinari N, Cipollone F, Paganelli R, Di Gioacchino M. Allergooncology: An expanding research area. J Biol Regul Homeost Agents. 2020;34:319–326. doi: 10.23812/19-418-63-E. [DOI] [PubMed] [Google Scholar]
- 140.Park HJ, Chi GY, Choi YH, Park SH. The root bark of Morus alba L. regulates tumor-associated macrophages by blocking recruitment and M2 polarization of macrophages. Phytother Res. 2020;34:3333–3344. doi: 10.1002/ptr.6783. [DOI] [PubMed] [Google Scholar]
- 141.Esposito S, De Simone G, Boccia G, De Caro F, Pagliano P. Sepsis and septic shock: New definitions, new diagnostic and therapeutic approaches. J Glob Antimicrob Resist. 2017;10:204–212. doi: 10.1016/j.jgar.2017.06.013. [DOI] [PubMed] [Google Scholar]
- 142.Xu H, Xu J, Xu L, Jin S, Turnquist HR, Hoffman R, Loughran P, Billiar TR, Deng M. Interleukin-33 contributes to ILC2 activation and early inflammation-associated lung injury during abdominal sepsis. Immunol Cell Biol. 2018;96:935–947. doi: 10.1111/imcb.12159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Nascimento DC, Melo PH, Pineros AR, Ferreira RG, Colon DF, Donate PB, Castanheira FV, Gozzi A, Czaikoski PG, Niedbala W, et al. IL-33 contributes to sepsis-induced long-term immunosuppression by expanding the regulatory T cell population. Nat Commun. 2017;8:14919. doi: 10.1038/ncomms14919. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.