ABSTRACT
The apical junctional complexes (AJCs) of airway epithelial cells are a key component of the innate immune system by creating barriers to pathogens, inhaled allergens, and environmental particles. AJCs form between adjacent cells and consist of tight junctions (TJs) and adherens junctions (AJs). Respiratory viruses have been shown to target various components of the AJCs, leading to airway epithelial barrier dysfunction by different mechanisms. Virus-induced epithelial permeability may allow for allergens and bacterial pathogens to subsequently invade. In this review, we discuss the pathophysiologic mechanisms leading to disruption of AJCs and the potential ensuing ramifications. We focus on the following viruses that affect the pulmonary system: respiratory syncytial virus, rhinovirus, influenza viruses, immunodeficiency virus, and other viruses such as coxsackievirus, adenovirus, coronaviruses, measles, parainfluenza virus, bocavirus, and vaccinia virus. Understanding the mechanisms by which viruses target the AJC and impair barrier function may help design therapeutic innovations to treat these infections.
KEYWORDS: Respiratory Syncytial Virus (RSV), epithelial cells, actin cytoskeleton, viral infection, apical junctional complex, tight junction, adherens junction, barrier dysfunction, trans-epithelial electrical resistance, permeability, epithelial barrier
Introduction
The human airway is in direct contact with the outside world, and therefore, is an integral member of the innate immune system and maintaining inner homeostasis. The airway acts as a barrier, comprised of surface liquids, mucus, and epithelium with its apical junctional complexes (AJCs).1,2 AJCs form between adjacent cells and consist of tight junctions (TJs) and adherens junctions (AJs), maintaining cell polarity and attachment between neighboring cells. Likewise, AJCs control molecule, microorganism, and particle trafficking throughout the paracellular space in a semipermeable manner (Figure 1).1,2 The apical TJ is a dynamic structure and undergoes continuous turnover.3 It includes three main subsets of proteins: 1. The claudin family, comprising claudin-1 through 27 in mouse and human;4 2. The tight junction-associated Marvel domain-containing (TAMPs) family, containing occludin, tricellulin, and MARVEL domain-containing protein 3 (MARVELD3); and 3. The immunoglobulin family, consisting of Junctional Adhesion Molecule-A (JAM-A), JAM-B, JAM-C, and the Coxsackie and Adenovirus Receptor (CAR).5,6 Immunohistological experiments have shown that claudins differ in their intracellular localization. Claudin-3, −5, and −8 have been detected exclusively in TJ complexes, while claudin-1 and −4 localized throughout the lateral membranes and the apico-lateral TJ complexes.7–9 The localization patterns have implications for viral targeting of these proteins, as discussed in subsequent sections.
Figure 1.
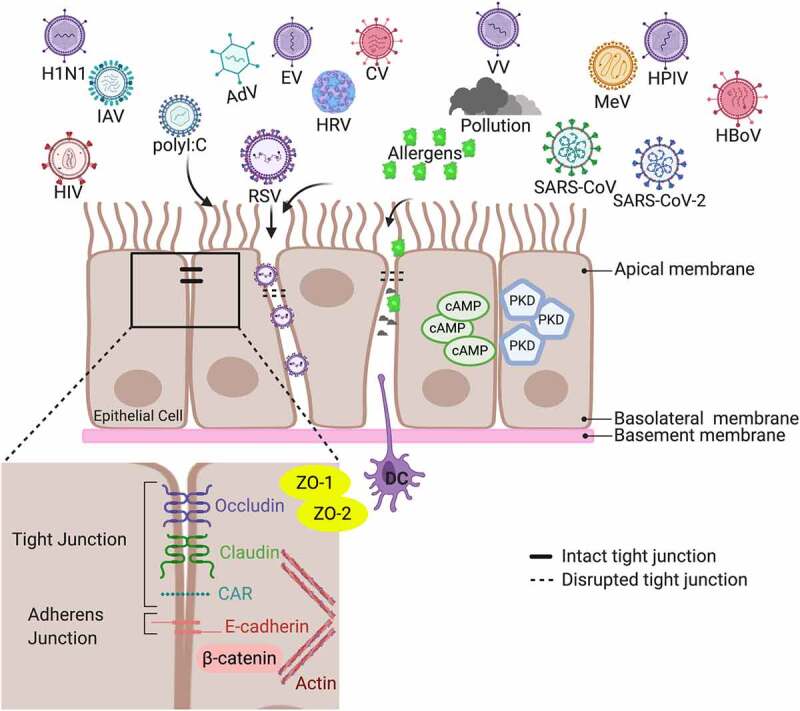
Structure of the airway epithelial cell apical junction complexes. The human airway comprises epithelium, surface mucus layers, and underlying immune cells, such as dendritic cells (DC). AJC form between neighboring cells and comprise TJ and AJ, which are associated with an underlying perijunctional actin cytoskeleton. Respiratory viruses can lead to AJC disassembly and disruption. Subsequently, virus-induced epithelial permeability may allow for allergens, environmental insults, like pollution and cigarette smoke, and bacterial pathogens to invade. An immune response, represented here with DC, is activated to combat the viral infection and reestablish the AJC and eliminate the bacterial pathogens and allergens that gained access to the internal system. Multiple pathways have been implicated in restoring the AJC barrier. Following RSV infection, cyclic adenosine cAMP and PKD-inhibitors have reduced viral replication and attenuated AJC disruption to restore the epithelial barrier. This image was created with BioRender.com
Abbreviations used in the image: AJC (apical junctional complex), AJ (adherens junction), TJ (tight junction), ZO (zonula occludens), DC (dendritic cell), cAMP (cyclic adenosine monophosphate), PKD (protein kinase D), RSV (respiratory syncytial virus), HRV (human rhinovirus), COVID-19 (coronavirus disease 2019, also known as SARS-CoV-2), SARS-CoV (severe acute respiratory syndrome coronavirus), HIV (human immunodeficiency virus), MeV(measles virus), IAV (influenza A virus), EV (enterovirus), CV (coxsackievirus), AdV (human adenovirus), H1N1 (2009 pandemic Influenza A/H1N1pdm09 strain), HPIV (human parainfluenza virus), HBoV (human bocavirus), VV (vaccinia virus), CAR (coxsackie and adenovirus receptor).
The AJ is located more basally than the TJ and contains proteins within the Cadherin family, most notably E-cadherin, and the nectin sub-family.10 They maintain and stabilize cell-cell adhesions and regulate the actin cytoskeleton. Moreover, E-cadherin binds to α – and β-catenin, which are involved in gene expression, intracellular signaling, and transcriptional regulation. Cytosolic plaque proteins and polarity proteins both interact with TJ proteins and play crucial parts in maintaining TJ function. TJ proteins bind to zonula occludens (ZO) family plaque proteins, which link the TJ with actin-binding proteins. Together, these proteins regulate paracellular transport of ions and small molecules, as well as maintain cellular structure by interacting with the actin cytoskeleton. Polarity proteins play a vital role in the maintenance of apical and basolateral polarization. Some major polarity proteins include Crumbs complex, comprised of Crumbs, Proteins Associated with Lin Seven 1 (PALS1), PALS1-Associated Tight Junction protein (PATJ), and the Scribble complex, containing scribble, Dlg, and Lgl.1,2
Viruses are obligate intracellular parasites and require the use of living cellular machinery for survival.11 This means that viruses have to enter cells, transport their genome to the cytosol or nucleus, and then replicate and produce their progeny. Viral infections enter epithelial cells either via binding to specific AJC targets as receptors for subsequent internalization or by causing disassembly of distinct AJC proteins through regulating the expression or localization of the protein, thus obtaining unencumbered access to the underlying tissue.12,13 Many viruses bind to receptors on the apical side of the epithelial cells. Others, however, target basolateral receptors, which are ordinarily inaccessible due to the TJ barrier. These viruses have developed mechanisms to cause TJ disassembly, typically either through regulation of protein expression or localization, in order to reach their receptors.14 While some viral infections lead to barrier dysfunction by direct cytotoxicity, others cause AJC disruption without inhibiting cell viability, which may represent an evolutionary strategy that facilitates viral replication.1 Not only does AJC dysfunction allow for viral infectivity, but virus-induced epithelial permeability may allow for allergens and bacterial pathogens to subsequently invade.2 In this review, we discuss the pathophysiologic mechanisms of viral entry resulting in disruption of the AJC, subsequent ramifications, and restoration of the epithelial barrier. We focus on the following viruses that affect the pulmonary system: respiratory syncytial virus (RSV), human rhinovirus (HRV), influenza viruses, human immunodeficiency virus (HIV), and multiple other viruses such as coxsackievirus (CV), adenovirus (AdV), coronavirus family, measles virus (MeV), human parainfluenza virus (HPIV), human bocavirus (HBoV), and vaccinia virus (VV). Uncovering the mechanisms by which viruses impact the AJC and impair barrier function will help design therapeutic innovations to treat these infections.
Viral disruption of tight junctions
Emerging evidence has shown that different viruses cause AJC dysfunction by targeting several TJ and AJ proteins (Table 1). The majority of these studies have been conducted on transformed airway epithelial cell lines, which form polarized, tight barriers, while others have utilized primary airway epithelial cells (AECs) and murine models. Below, we discuss recent studies that have investigated the impact of viral infection on airway epithelial barrier integrity.
Table 1.
Viral disruption of tight junction proteins
| Virus |
Virus Type (RNA/ DNA) |
In Vitro |
In Vivo |
TJ/AJ proteins |
Citation |
| Respiratory Syncytial Virus (RSV) | enveloped, negative-sense, single-stranded, RNA virus | 16HBE | wildtype C57BL/6 mice | Disassembly of ZO-1, occludin, E-cadherin, β-catenin ↓ claudin-1 and claudin-4 |
15–20 |
| AECs | |||||
| Nasal Epithelial Cells | wildtype BALB/6 mice | ||||
| Human Rhinovirus (HRV) | non-enveloped, positive-sense, single stranded, RNA virus |
16HBE | wildtype C57BL/6 mice |
↓ ZO-1, claudin-1, occludin, E-cadherin |
21–24 |
| AECs | |||||
| Primary Human Nasal Epithelial Cells | |||||
|
Coxsackievirus (CV-A16) & Enterovirus (EV-A71) |
non-enveloped, positive-sense, single-stranded, RNA virus |
16HBE | N/A | ↓ claudin-4, E-cadherin, ZO-1 |
25–28 |
|
Human Adenovirus (AdV) |
non-enveloped, double-stranded, DNA virus |
AECs | N/A | ↓ PALS1 and ZO-2 | 14 |
| Influenza A (IAV) | enveloped, negative-sense, single-stranded, RNA virus | ISO-HAS-1 Cells | N/A | ↓ claudin-4 | 29,30 |
| NC1-H441 Cells | |||||
| Influenza H1N1 virus | enveloped, negative-sense, single-stranded, RNA virus | AECs | N/A | ↓ ZO-1 and F-actin cytoskeleton | 31,32 |
| Human Immunodeficiency Virus (HIV) | enveloped, positive-sense, single-stranded, RNA virus |
AECs | HIV-1 transgenic and wild-type Fischer 344 rats | ↓ ZO-1, occludin, claudin-1, claudin-2, claudin-4, claudin-18, E-cadherin |
33–36 |
|
Severe Acute Respiratory Syndrome (SARS-CoV) |
enveloped, positive-sense, single-stranded, RNA virus |
MDCKII | N/A | ↓ E-cadherin and ZO-1 |
37,38 |
|
Severe Acute Respiratory Syndrome 2 (SARS-CoV-2) |
enveloped, positive-sense, single-stranded, RNA virus |
AECs | N/A | ↓ ZO-1 | 39 |
|
Measles Virus (MeV) |
enveloped, negative-sense, single-stranded, RNA virus |
AECs | N/A | Disassembly of F-actin ↓ nectin-4 |
40,41 |
|
Human Parainfluenza Virus (HPIV) |
enveloped, negative-sense, single-stranded, RNA virus |
A549 | N/A | ↓ claudin-1 | 42–44 |
| AECs | |||||
|
Human Bocavirus (HBoV) |
non-enveloped, positive-sense, single-stranded, DNA virus |
AECs | N/A | Disassembly of ZO-1 ↓ claudin-1 |
45–47 |
| A549 | |||||
| Vaccinia Virus (VV) | Eenveloped, double-stranded, DNA virus |
AECs | N/A | ↓ ZO-1 | 48 |
Respiratory Syncytial Virus (RSV)
The family Pneumoviridae was recently reclassified as a family with two genera, orthopneumovirus and metapneumovirus. Human RSV is a representative member of the orthopneumovirus family, and human metapneumovirus (hMPV) is an example within the metapneumovirus family.49 These viruses are large, enveloped, negative-sense, single-stranded, RNA viruses. There are multiple reports of the effect of RSV on the airway epithelial barrier, while the effect of hMPV has not been studied. RSV primarily infects airway epithelial cells and is the leading cause of lower respiratory tract infections (LRTI) among young children and a significant cause of morbidity and mortality among the elderly and immunocompromised individuals.50–52 It is transmitted by either direct or indirect contact with respiratory droplets.53,54 Multiple studies have shown a strong association between RSV infection, persistent wheezing, and childhood asthma.55–57 Environmental exposures are considered to be major driving factors for exacerbations of viral infection and may increase the susceptibility of the airway epithelial barrier to RSV infection.58
Our group previously showed that RSV infection of both 16HBE14o – human bronchial epithelial (16HBE) cell lines and primary AECs led to disassembly and disruption of TJ and AJ proteins, such as ZO-1, occludin E-cadherin, β-catenin, and claudin-1 and 4 independent of cell death and necrosis. Moreover, RSV infection did not affect the expression and total quantity of AJC proteins, indicating that the changes in the assembly of the AJC could be explained by rearrangements and internalization of the TJs, a process called endocytosis.15 These results were consistent with other studies that showed RSV induced a decrease in epithelial resistance and increased permeability of the monolayers.16 While these studies in 16HBE and AECs cells obtained from lower airways showed marked disruption of claudin-4, studies done in nasal epithelial cells showed an induction of claudin-4, which is possibly explained by the differences in cellular and anatomic properties between nasal and bronchial epithelial cells.17
Protein kinase D (PKD) is involved in regulating cell motility, polarity, and shape by controlling the phosphorylation of cortactin.59,60 Cortactin is a ubiquitously expressed actin-binding protein that regulates F-actin dynamics between polymerization and depolymerization states during plasma membrane remodeling.61,62 Studies showed that PKD colocalizes with E-cadherin, indicating a close role in AJC.63 Other investigators showed that activation of PKD could decrease the expression of claudin-1 in bronchial epithelial cells.64 Our group showed that in both 16HBE and AECs, RSV infection triggered PKD-dependent TJ disassembly.60 In another study, we showed similar effect of double-stranded RNA (dsRNA) mimetic, poly(I:C), induced barrier dysfunction, which was also prevented by PKD antagonists.59 TJ and AJ are associated with cortical F-actin bundles, which are essential for their stabilization in steady-state epithelial cell monolayers.5 Taken together, RSV-induced AJC disorganization and dysfunction is likely a result of cortical F-actin cytoskeletal remodeling, which is regulated by PKD phosphorylation of cortactin. Likewise, RSV-induced relocalization of cortactin was significantly reduced by pretreatment of epithelial cell monolayers with cyclic adenosine monophosphate (cAMP) analogs. Cyclic cAMP is an essential second messenger required for many critical homeostatic cellular functions such as hormonal regulation, cell differentiation and proliferation, and gene expression.65 Elevated cAMP levels in epithelial cells modified RSV-induced airway barrier disruption and reduced viral replication.15 Interestingly, this study showed a barrier-protective role for cAMP even when added up to 24 h after viral inoculation.15
In wild-type C57BL/6 mice, RSV infection induced downregulation of ZO-1, occludin, and claudin-1 proteins.18 Another study using wild-type BALB/c mouse also found that RSV infection led to decreased mRNA expression of claudin-1 and occludin in lung samples.66 There is evidence that ZO-1 and occludin act as positive regulators of epithelial barrier integrity, and therefore, these changes likely contribute to the leakiness of the airway epithelial barrier following infection. The loss of ZO-1 has been shown to be associated with increases in paracellular permeability to ions and large uncharged molecules.19,20 Similarly, decreased occludin attenuated the formation of barrier structure in vitro, 67 and downregulation of claudin-1 was associated with increased permeability of airway epithelial monolayers.64 There was an increase in inhaled fluorescent dextran uptake and evidence of increased flux of substances across the respiratory membranes in the mice, thus suggesting the important role of ZO-1, occludin, and claudin-1 in barrier integrity. Intriguingly, RSV infection induced marked upregulation of claudin-2 expression.18,21 Claudin-2 belongs to a subset of TJ proteins called pore-forming TJ that prevent epithelial barrier formation, and claudin-2-deficient mice showed decreased permeability in renal proximal tubules.68 Likewise, upregulation of claudin-2 by proinflammatory cytokines was shown to increase the permeability of epithelial barriers.69
Human rhinovirus
HRV is a member of the Enterovirus genus and family Picornaviridae and is a non-enveloped, positive-sense, single-stranded RNA virus.70,71 HRVs are divided into three distinct groups: HRV A, HRV B, and HRV C .72,73 Transmission of HRV occurs through either direct contact or through small aerosol droplets. It is the leading cause of the common cold and upper respiratory tract infections.71 Previous studies have revealed that infection of AECs and 16HBE with HRV causes a decrease in trans-epithelial electrical resistance (TEER) and an increase in paracellular permeability. In both in vivo and in vitro studies, HRV caused disassembly of ZO-1. Some studies have found a dose-dependent increase in apoptosis, 69 while others have found no evidence of cell death.22,74 Looi et al. observed a decrease in claudin-1, occludin, and ZO-1 protein expression in AECs obtained from asthmatic children compared to non-asthmatic.23 Interestingly, there was sustained barrier dysfunction in the AECs of asthmatic children, suggesting a continued compromise of the airway. In addition to the same AJC changes that Looi et al. detected, Faris et al. observed a reduction in expression of claudin-4, E-cadherin, and Crumbs3 in polarized, undifferentiated AECs from healthy nonsmokers.74 Similarly, in primary human nasal epithelial cells, HRV decreased ZO-1, occludin, claudin-1, and E-cadherin expression, thereby increasing permeability for viral entry.21,24
Understanding the underlying mechanism is crucial in determining possible therapeutic targets. HRV-induced epithelial cell dysfunction was not modified by neutralizing chemokines, such as tumor necrosis factor (TNF)-α, IFN-γ, and IL-1β, suggesting a cytokine-independent process.22 NADPH oxidase (NOX), a multiunit enzyme that mediates the release of reactive oxygen species (ROS), called the oxidative burst, during viral and bacterial infections, 75 has been shown to contribute to the inflammatory pathway and barrier disruption in multiple viruses, such as RSV, influenza, and Hepatitis C .76–78 Comstock et al. showed, in 16HBE, that HRV-induced ZO-1 and occludin dissociation from the cell periphery was due to the generation of ROS by Rac1-dependent NOX1 activity.79 Rac1 is a GTPase protein that regulates many cellular processes, such as cell-cell adhesion and motility.80 These effects were also replicated by using poly(I:C), which suggests that HRV exerts its barrier disrupting effects through the generation of dsRNA. Inhibitors of Rac1 attenuated HRV – and poly(I:C) – induced disruptions of barrier function. In nonpolarized AECs, HRV was shown to still stimulate ROS generation, but instead through activation of xanthine oxidase.79 Further studies are warranted to understand the role of antioxidants in viral clearance and bacterial superinfection prevention.
Coxsackievirus (CV) and other enteroviruses
CV and other enteroviruses are also other members of the Enterovirus genus and family Picornaviridae. These viruses cause a range of diseases, like poliomyelitis, hepatitis A, hand, foot, and mouth disease, and the common cold, and can also trigger severe respiratory infections.81,82 Transmission usually happens through the respiratory droplets, but can alsooccur through fecal-oral contamination.25,26,81,83,84
The effect of enterovirus-A71(EV-A71) and coxsackievirus-A16 (CV-A16) on airway barrier have been studied. Epithelial permeability increased in 16HBE cells infected with either EV-A71 and CV-A16 infections, suggesting a disruption in TJ assembly.25 There was decreased claudin-4, E-cadherin, and ZO-1 levels due to a decrease in the poliovirus receptor related protein 1 (PVRL1).25 PVRL1, also known as nectin1, serves as a cell adhesion molecule (CAM) at the AJ level. This epithelial barrier breakdown allows exposure of the CAR, a member of the immunoglobulin TJ family, as it is located more basolateral within the TJ.25–27 As a result, these viruses bind to CAR, leading to infection of the epithelial cell and spread to neighboring cells. In addition, coxsackie B internalization is coupled with the endocytosis of occludin, and studies have shown their co-localization in vesicles inside cells.14,28 Occludin enters the cell via macropinocytosis, while coxsackie enters through caveolin-mediated endocytosis. Despite this difference, drugs that impair the macropinocytosis of occludin also impair virus internalization, and vice versa.
Adenovirus
AdVs are members of the Adenoviridae family, which are non-enveloped, double-stranded, DNA viruses.85–88 Human AdVs are common causes of upper and lower respiratory tract infections, as well as conjunctival and gastrointestinal tract infections.86–88 Transmission usually occurs through aerosol droplets, either by direct contact or surface contact.87,88 Like many EVs and CVs, AdVs also bind to CAR for viral entry, and therefore, similarly, TJ disruption is required in order for AdVs to reach the receptor. AdV 9 E4-ORF, as a representative example, binds to PATJ and ZO-2 proteins, which causes a disruption in the TJ and cell polarity without comprising the AJ.14 This allows for AdV binding of CAR, and subsequent binding of more basolateral molecules called integrins in order to enter the cell. Recent studies have hypothesized a different mechanism in which AdV enters cells. A splice isoform of CAR, CAREX8, localizes to the apical surface of polarized epithelial cells and thus allows for AdV entry.14 Further investigation is needed to determine which mechanisms AdV employs to gain access into the cells.
Influenza viruses
Influenza viruses are members of the Orthomyxoviridae family and are enveloped, negative-sense, single-stranded, RNA viruses.29,89,90 Influenza virus is mainly transmitted by droplets or respiratory secretions of infected persons.29,91 It causes upper respiratory infections, which present with fever, runny nose, cough and sore throat. It may progress to the lower respiratory system and present with shortness of breath, pneumonia, and respiratory distress. Acute respiratory distress syndrome (ARDS) and multi-organ failure are uncommon but potentially fatal developments of influenza.92,93 Secondary bacterial pneumonia is a frequent complication of influenza infection, particularly in patients with chronic diseases, which may result in sepsis and significant morbidity and mortality. Influenza viruses are divided into two major categories: A (IAV) and B .94,95 The IAV viruses are further classified to different subtypes and strains based on their hemagglutinin (H) and neuraminidase (N) surface proteins. Influenza H1N1 virus caused a pandemic in the summer of 2009.
Influenza viruses primarily infect the airway epithelium and are known to damage the AJC by altering the cytoskeleton of AECs.30,96,97 Short et al. used an in vitro co-culture model of human epithelial cells and endothelial cells infected with IAV (H1N1 and H5N1 subtypes). They demonstrated that the infection induced disruption of the epithelial cell barrier, leading to a significant decrease in TEER and increase in permeability. There was loss of claudin-4, but no changes in ZO-1, β-catenin and E-cadherin.29,30 The barrier disruption was not due to cell death, which is in contrast with previous studies showing cell death in influenza infected cells.98 Interestingly, infection was only detected inside epithelial cells, and not endothelial cells, suggesting that epithelial cells may be more permissive to IAV infection.30 Pro-inflammatory cytokines were upregulated by endothelial cells but did not play a role in epithelial barrier dysfunction.30
In another study, differentiated AECs cells were infected with 2009 pandemic Influenza A/H1N1pdm09 strain.31 There was evidence of a decline in barrier function, with a marked decrease in TEER and increase in permeability. They showed a decrease of ZO-1 and the F-actin cytoskeleton, as well as cell death following viral infection and barrier dysfunction.98 Many cytokines and chemokines were secreted in infected cells, with a significant increase in IL-6, TNF-α, and IL-8, suggesting activation of anti-viral inflammatory responses.31 Further research is needed to determine if the increased cytokine production is independent of or a result of AJC disruption. Another study used two strains of H1N1pdm09 viruses, A/California04/09 (CA04), and A/New York/1682/09 (NY1682), to infect human alveolar type II epithelial cells. The study, similarly, found a disruption of the airway barrier with a decrease in TEER and increase in permeability. NY1682 had a higher infectivity rate compared to CA04 and resulted in significantly more epithelial barrier injury, but it stimulated less cytokine production.32
Human Immunodeficiency Virus (HIV)
HIV is a member of the Retroviridae family and an enveloped, positive-sense, single-stranded, RNA virus that, upon entry into the target cell, undergoes reverse transcription and converts to double-stranded DNA.21 In 2019, there were approximately 38 million people worldwide living with HIV.99 The virus is primarily transmitted through sexual contact, infected blood, and perinatally.100 Pulmonary infection remains the leading reason for hospitalization among individuals with HIV, and multiple previous studies have also identified noninfectious pulmonary complications, such as chronic obstructive pulmonary diseases (COPD) and poor lung function.33,34,101 HIV infects the airway epithelium and induces secretions of mucus and pro-inflammatory cytokines.33 Brune et al. studied the role of HIV on primary AECs monolayers by infecting the cells with two different HIV strains, X4 tropic and R5 tropic. Cells exposed to HIV X4 tropic strain increased paracellular permeability within 24 hours, compared to control cells or HIV R5 tropic cells. This was not due to cell death or apoptosis.33 There was a statistically significant decrease in E-cadherin mRNA, and protein levels following exposure to the X4 tropic virus. Furthermore, lysosomal inhibitors attenuated E-cadherin degradation following virus exposure. Therefore, this reduction in E-cadherin protein levels was a result of both decreased transcription and lysosomal degradation. Interestingly, AECs cells exposed to HIV R5 tropic showed no changes in E-cadherin.33 X4 tropic virus uses the CXCR4 co-receptor for cell entry, while R5 tropic virus uses the CCR5 co-receptor. Due to the differences in co-receptors, the X4 tropic virus was internalized by AECs, but R5 virus was not.33 This perhaps explains the differences in E-cadherin degradation between these two strains.
Nuclear factor erythroid 2 (Nrf2) activates antioxidant response elements, thereby protecting cells from oxidative stress. Fan et al. obtained primary AECs from HIV-1 transgenic and wild-type Fischer 344 rats to study the role of Nrf2-mediated antioxidants on the TJ.34 They found that inhibiting Nrf2 expression triggered a significant decrease in TEER and an increase in paracellular permeability. This was due to decreased expression of ZO-1, occludin and claudin-18. Furthermore, the overexpression of Nrf2 had a more robust effect on enhancing the barrier structure and function in cells isolated from HIV-1 transgenic rats than wild-type.34,35 Lassiter et al. showed an increase in oxidative stress in the lung lavage fluids of HIV transgenic rats, in comparison to wild-type rats. This was correlated with the relocalization of ZO-1 and occludin into the cytoplasm of cells, as well as overall decreases in protein expression levels, leading to decreased TEER and increased paracellular permeability.36 Next, alveolar epithelial monolayers from wild type rats were exposed to gp120 and Tat, HIV-related proteins, which, similar to the HIV transgenic rats, led to decreased ZO-1 and occludin protein levels at the TJ. Taken together, this suggests that gp120 and Tat produce oxidative stress, disrupting ZO-1 and occludin and resulting in barrier dysfunction. Alcohol consumption can exacerbate HIV-induced oxidative stress. It has been shown that the combination of alcohol and HIV decreased ZO-1 and occludin drastically, making alveolar epithelial cells more susceptible toward the virus.35,36
Coronavirus family
The Coronaviridae family are enveloped, positive-sense, single-stranded, RNA viruses.102,103 Coronaviruses are enteric and respiratory tract pathogens, which usually cause a mild disease. However, the Middle East respiratory syndrome (MERS-CoV), severe acute respiratory syndrome coronavirus (SARS-CoV), and SARS-CoV-2, known as Coronavirus disease 2019 (COVID-19), are well-recognized members of the Coronaviridae family that cause severe respiratory and systemic diseases. MERS and SARS-CoV caused large outbreaks in 2002–2003 and 2012, respectively, while COVID-19, which was first discovered in Wuhan city, Hubei province in China in December 2019, has caused a massive global pandemic, infecting more than 95 million people and resulting in more than two million deaths worldwide to date.104 Pulmonary symptoms associated with these viruses include dry cough, dyspnea, hypoxemia, pneumonia, and ARDS, and have led to serious complications such as rapid, progressive respiratory failure and mortality.105,106 To date, there are two vaccines available for coronavirus prophylaxis, Pfizer-BioNTech and Moderna COVID-19 Vaccine, which the Food and Drug Administration (FDA) granted authorization of emergency use (EUA). Both of these vaccines use mRNA technology and require two doses. The mRNA is taken up by cells and provides the instructions to produce a spike protein, which is then displayed on the cell’s surface. Because of the spike protein, the immune system recognizes that there is a threat and initiates an immune response by making antibodies.107 Additionally, many other vaccines candidates against COVID-19 are in pre‐clinical, early, and late phases of development.
The pathophysiology of these viruses causing inflammation, especially their effect on AECs, is not well understood. Presently, only a few studies have investigated the impact of these viruses on the AEC barrier. A study investigating SARS-CoV in Madin-Darby Canine Kidney II (MDCKII) epithelial cells showed that binding of SARS-CoV’s envelope (E) protein to PALS1 PDZ domain caused a rearrangement of PALS1, leading to disruption of TJ proteins, such as ZO-1 and E-cadherin, loss of barrier function, an increase in paracellular permeability, and further infiltration of SARS-CoV virions into the subepithelial space.37 Similarly, De Maio et al. highlighted that binding of the E protein to PALS1 could play a role in the pathogenesis of COVID-19.38 Zhu et al. showed that COVID-19 is able to replicate in human airway epithelium, leading to cytopathic effects. However, the effect of the virus on the airway barrier was not studied.108 Robinol et al. published a preliminary, non-peer-reviewed study that showed evidence of COVID-19 affecting the tight junction. The study revealed that COVID-19 decreased TEER, increased paracellular permeability, and induced disruption of ZO-1 in primary AECs.39 Infected cells were forced out from the apical side, indicating a role for apoptosis.39 As this novel virus is rapidly spreading across the world, further investigation is needed to understand its pathogenesis, especially the impact on tight junction barriers in both cell cultures and animal models.
Measles virus (MeV)
MeV is a member of the Paramyxoviridae family and is an enveloped, non-segmented, single-stranded, negative-sense RNA virus.40 MeV is highly contagious, as it is transmitted through respiratory aerosols and continues to affect individuals worldwide.40,109,110 MeV’s target is commonly lymphoid organs, but epithelial tissues of different organs are susceptible to infection. MV infects macrophages and dendritic cells in the airway to then gain access to the lymphatic organs, where it replicates. The virus then crosses back into the airway in order to be expelled and infect the next host. Until recently, the mechanism of MeV infection of epithelial cells was unknown, as there was a limited number of cell lines that support the virus, and MeV usually infects AECs from the basolateral surface, which makes the study design more challenging.109,110
Muhlebach et al. used primary AECs grown in air-liquid interface and showed that MeV needs nectin-4, an AJ protein of the immunoglobulin family, in order to infect and spread basolaterally.41 They found that strongly nectin-4 positive cells were located immediately adjacent to infectious centers. However, nectin-4 was downregulated within infected epithelial cells.41 Therefore, it is believed that nectin-4 is the epithelial cell receptor for MeV within the airway epithelium. The downregulation of nectin-4 allowed MeV to exit into the airway and be released into the air through coughing and sneezing, thereby leading to infection of the next host.41 Other AJC proteins may be involved in MeV pathogenesis, but further research is warranted to confirm this. In one study, well-differentiated primary AECs were infected with MeV and there was disassembly of the F-actin filaments on day three of infection.40 Ludlow et al. found no changes in ZO-1, with ZO-1 remaining intact at the cell membrane, in both in vitro and in vivo models, suggesting that barrier breakdown is not necessary for MeV spread to the adjacent cells.111
Human parainfluenza virus (HPIV)
HPIV is another member of the Paramyxoviridae family and is an enveloped, non-segmented, negative-strand, RNA virus.42 The virus is subdivided into HPIV 1–5. They are common respiratory tract pathogens and are known to cause bronchiolitis, croup, pneumonia, and tracheobronchitis in children.42–44 Transmission occurs through direct or indirect contact with infectious fluids, either through contaminated fomites on surfaces or through inhalation of airborne droplets.112
A limited number of studies have investigated the role of tight junction integrity on HPIV infectivity. Both HPIV3 and HPIV5 caused a decrease in TEER and increased permeability in AECs, indicating disruption of the epithelial barrier.42 However, this study did not investigate which AJC proteins were disrupted. In another study, human alveolar epithelial cells (A549 cells) were infected with HPIV2. There was an upregulation of claudin-1 mRNA expression but no changes in claudin-3, claudin-4, claudin-7, ZO-1, ZO-2, and occludin levels.43 In order to further investigate the importance of claudin-1 upregulation, the researchers used A549 cell lines that overexpress claudin-1. They found decreased HPIV2 growth and protein expression following viral infection in these cells and, therefore, that HPIV2 cell-to-cell spread is inhibited by claudin-1. It is thought that claudin-1 mRNA and protein expression following HPIV infection is induced by multiple cytokines, such as TNFα and IL-1β.43 HPIV has developed mechanisms in order to decrease claudin-1 expression, thereby promoting its own spread throughout the body. One of HPIV’s viral proteins, the V protein, reduced claudin-1 expression during infection, allowing for both viral entry and dissemination through cell-to-cell spread.43 It is proposed that the V protein suppresses the production of IL-1β, thereby inhibiting claudin-1 expression. There is currently no vaccine or antiviral treatments that can eliminate HPIV from the body.113 Understanding the mechanisms of viral spread can aid in developing treatment options.
Human Bocavirus (HBoV)
HBoV is a member of the Parvoviridae family and is a non-enveloped, positive-sense, single-stranded DNA virus.45,46 It is categorized into HBoV 1–4 and is a common respiratory virus that causes acute respiratory infections and wheezing in young children.45,46 The transmission mechanism of HBoV is still unknown. However, other viruses in this family are transmitted through inhalation or contact with infectious surfaces.114 In primary human AECs infected with human bocavirus type 1 (HBoV1), TEER slowly started to decrease after 4 days’ post infection and continued to do so even after 48 days, which is consistent with the persistent virus’s release kinetics.45 Multiple studies have shown that HBoV1 causes airway epithelial damage through disassembly of ZO-1 proteins.45,46 Furthermore, there is a decrease in β-tubulin IV, a marker for cilia following viral infection.45,46 In another study, A549 cells were infected with human bocavirus VP1 unique region (HBoV-VP1u), which resulted in a significant decrease in TEER and increase in permeability in these cells.47 The same study found an increase in claudin-1 and a decrease in occludin. Secretory phospholipases A2 (sPLA2) are a family of enzymes that play an important role in lung inflammation and tissue damage, and it was shown that VP1u exhibits sPLA2-like activity, which could explain the impact of the virus on epithelial barrier injury.47
Vaccinia virus (VV)
VV is a member of the Poxviridae family and is an enveloped, double-stranded DNA virus.115 VV is known to cause smallpox, but there is limited knowledge of how it is spread.116 Infection of AECs showed that this virus primarily infects through the basolateral rather than the apical surface, which is accessible near the edges of the cell monolayers. In AECs, VV had no effect on TEER or ZO-1 localization. This is in contrast to studies done on HeLa and Vero cultured cell lines that have shown rapid cell death, decrease TEER, and changes in ZO-1 expression.48 More research is needed to delineate VV’s impact on the airway epithelial TJ.
Immune response following viral infection and tight junction restoration
Following viral infection, two simultaneous mechanisms need to occur: repairing the tight junction barrier in order to prevent further viral infectivity and inciting an immune response to combat the virus. The restoration of tight junction morphology following a viral infection is crucial to establish a barrier between our internal system and the outside world. Virus-induced tight junction disassembly and dysfunction contribute to producing an immune response. The immune system has generated multiple mechanisms in response to viral infection to reestablish the AJC and fight the viral infection. Viruses induce the upregulations of various cytokines and chemokines. While some cytokines, such as interleukin (IL) −4 and IL-13, have been shown to provoke barrier disruption in 16HBE cells and air-liquid interface cultivated paranasal sinus mucosa cells, others play a vital role in protecting injured cells.
In human bronchial epithelial cells, IL-4 and IL-13 act via the Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway. Studies have shown that these cytokines lead to the disassembly of ZO-1, occludin, E-cadherin, and β-catenin.117 There were no significant changes in claudin-1, −2, or −3. In a mouse model, lung IL-13 reduced claudin-18, while it increased claudin-4 expression.7
IL-22, a member of the IL-10 cytokine family, provides stability to injured cells and is produced by natural killer (NK) and natural killer T (NKT) cells following many viral infections, in particular influenza infection.118 No studies have looked at IL-22 activation following former coronaviruses such as SARS-CoV or MERS.119 However, recent research suggests that IL-22 may be activated by COVID-19 infection, which signifies the need for further investigation to elucidate a causal relationship between this virus and IL-22 signaling.119 In vivo studies in wild-type C57BI/6 mice and in vitro studies in AECs suggest that increased IL-22 protects the epithelial barrier by promoting increased levels of claudin-4, ZO-1, and ZO-2.118 As mentioned in the previous section, influenza viruses cause dysfunction of claudin-4, and influenza H1N1, specifically, has also been shown to decrease levels of ZO-1.29,98 Therefore, IL-22 repair of these tight junctions following viral infection allows for proper functioning.
IL-22 helps restore the tight junction barrier, which protects against secondary bacterial infection. Continuous dysfunction of the AJC can lead to bacterial infection superimposed on the viral infection. Streptococcus pneumoniae and Staphylococcus aureus are the leading causes of secondary bacterial pneumonia following influenza infection, and both have been shown to activate IL-22.119 IL-22 has also been shown to be stimulated following Klebsiella pneumonia and Pseudomonas aeruginosa bacterial infections. Increases in IL-22 expression following bacterial superinfection allows for continued repair of the viral-induced disassembled TJ proteins.119 IL-22BP is a soluble binding protein that acts as a competitive inhibitor of IL-22. In one study, both wild-type C57BL/6 and IL-22BP-/ – mice (i.e., mice without IL-22BP) were infected with influenza A for 6 days prior to S. aureus infection.120 IL-22BP-/ – mice were protected against S. aureus infection in comparison to the wild-type mice. The researchers then conducted a similar experiment with S. pneumoniae infection following influenza A disease, and similar results were observed.120 After 48 hours, there was complete clearance of S. pneumoniae in IL-22BP-/ – mice, while the bacterial load in wild-type mice remained high. Further studies showed that claudin-1, claudin-4, claudin-5, and claudin-8 gene expression was increased in IL-22BP-/ – mice following S. aureus superinfection, despite similar expression during influenza infection alone.120 These results suggest that IL-22 may exert its effect on tight junction function similar to what occurred in IL-22BP-/ – mice. TJ impairment can lead to an increased likelihood of bacterial superinfection and disease severity, and IL-22 helps mitigate these effects through increased expression of ZO-1, ZO-2, claudin-1, claudin-4, claudin-5, and claudin-8.118,120
HRV may be responsible for up to half of all viral-associated COPD and asthma exacerbations.22,121 Betamethasone, a steroid typically used to treat skin diseases, allergic conditions, and some rheumatic disorders, has shown promising outcomes in restoring the AJC barrier following HRV infection. Inhaled corticosteroids, such as fluticasone propionate (FP) and budesonide (BUD) are used in managing of COPD. They may also aid in protecting against epithelial barrier dysfunction in response to viral infection and environmental insults, like cigarette smoking.
Cigarette smoke leads to TJ disorganization, directly through the cleavage of occludin and claudin and indirectly through oxidative stress, which leads to the production of cytokines, most notably interleukins IL-6 and IL-8, which results in airway inflammation. There is a decrease in TEER following cigarette smoke extract (CSE) exposure in 16HBE cells.122 In one study, primary nasal epithelial cells and 16HBE cells were exposed to both CSE and HRV infection, and this resulted in an exaggerated decrease in barrier function in both systems.123 Interestingly, a single dose of betamethasone, the same amount used in commercially-available nose drops, showed significant protection of the barrier.123 CSE and poly(I:C) led to the secretion of IL-8, which was efficiently suppressed by both fluticasone propionate (FP) and budesonide (BUD).122 BUD and FP also attenuated ZO-1 disruption following CSE exposure alone. It is hypothesized that BUD interferes with epidermal growth factor (EGF) receptor activation, thus helping maintain the AJC barrier. Following poly(I:C) infection, there was a significant reduction in TEER, which was attenuated following BUD and FP exposure.122 Further research is warranted to determine the role of betamethasone, BUD, and FP in the treatment of HRV-induced asthma and COPD.
As mentioned above, HRV is a significant cause of asthma exacerbations.121 Azithromycin, a macrolide antibiotic used to treat many pulmonary bacterial infections, has been shown to reduce the severity of an acute asthma exacerbation by inhibiting Intercellular Adhesion Molecule 1 (ICAM-1), a receptor for HRV.124 It may help in reestablishing the AJC barrier following infection. In primary AECs, azithromycin induced intracellular localization of claudin-1 and −4, occludin, and JAM-A.125 There were no effects on E-cadherin. Of note, improved TEER following azithromycin administration suggests that this processing of TJ proteins helps decrease permeability and increase cell integrity, thereby potentially preventing entry of viral infections.124,125 Further studies are warranted to elucidate its mechanism in therapeutic strategies and its effect on the tight junction barrier.
The coinfection of viruses and bacteria have been documented in multiple respiratory conditions, like pneumonia, cystic fibrosis, and COPD. H. influenzae is an opportunistic pathogen among patients with COPD, and coinfection with HRV is common. In 16HBE monolayers, HRV promotes paracellular migration of non-typeable H. influenzae from the apical to basolateral surface strictly in the cells with disrupted ZO-1, thereby suggesting that it is the ZO-1 disassembly that allows H. influenzae superinfection.22 This phenomenon was not observed in either RSV or influenza A.
In addition to repairing the tight junction barrier to prevent further viral or bacterial infection, the immune system must fight against the virus at hand. Tight junction disassembly helps trigger the immune system response. A limited number of studies have investigated this, and further research is essential to help understand the role of tight junctions in immune responses. In 16HBE cells, downregulation of E-cadherin leads to epidermal growth factor receptor-dependent increases in the thymus – and activation-regulated chemokine (TARC), a chemokine that plays an important role in activating Th2 immune responses.126 Multiple viruses, like RSV, SARS-CoV, COVID-19, Enterovirus, and Coxsackievirus, decrease E-cadherin expression, thereby potentiating a Th2 immune response.
Conclusion
Airway epithelial barrier integrity is crucial for internal homeostasis and has been recognized to play an essential role in health and disease. In addition to homeostasis maintenance, epithelial cells constitute the first line of defense from the environment. Epithelial cells possess AJCs comprised of multiple TJ and AJ proteins that regulate the passage of allergens, pollutants, viruses, and other pathogens into and out of the airways. TJ proteins are involved in key regulatory cell functions such as differentiation, proliferation, and migration by recruiting signaling proteins in response to extracellular stimuli, while AJ proteins are necessary for regulating the actin cytoskeleton and stabilizing cell-cell connections.
Extensive research has shown that many respiratory viruses enter the body through the airway epithelia and disrupt this barrier through dysregulation of the AJC. Recent studies highlighted in this review discuss the disruption mechanisms in which viruses enter these cells, causing infection and initiation of their cytopathic actions. ZO-1 is the most highly implicated protein in respiratory viral targeting. Some viruses lead to decreased levels of ZO-1, while others caused ZO-1 disassembly and internalization without reducing levels of ZO-1. Other TJ proteins involved in viral entry into the epithelia include occludin, E-cadherin, β-catenin, and numerous proteins in the claudin family. Understanding the interaction between the airway epithelial barrier and viral pathogens is of great significance. Identifying mechanisms of viral-induced epithelial pathology will help in understanding the role of the airway epithelial barrier in diseases such as ARDS, asthma, COPD, and cystic fibrosis, as well as in the design of anti-viral strategies.
GRANTS
This work was supported by the Mark Lauer Pediatric Research Grant, Cleveland Clinic Children’s (FR), as well as National Heart, Lung, and Blood Institute Grants R01-HL-148057 (F.R.).
Funding Statement
This work was supported by the National Heart, Lung, and Blood Institute [R01-HL-148057]; Mark Lauer Pediatric Research Grant, Cleveland Clinic Children’s .
Abbreviations
| 16HBE | 16HBE14o- human bronchial epithelial |
| AdV | Human adenovirus |
| AECs | Airway epithelial cells |
| AJ | Adherens junction |
| AJC | Apical junctional complex |
| Camp | Cyclic adenosine monophosphate |
| CAR | Coxsackie and Adenovirus Receptor |
| COPD | Chronic obstructive pulmonary disease |
| COVID-19 | Coronavirus disease 2019 |
| CV | Coxsackievirus |
| EV | Enterovirus |
| H1N1 | 2009 pandemic Influenza A/H1N1pdm09 strain |
| HBoV | Human bocavirus |
| HIV | Human immunodeficiency virus |
| HPIV | Human parainfluenza virus |
| HRV | Human rhinovirus |
| IAV | Influenza A Virus |
| Madin | Darby Canine Kidney II (MDCKII) |
| MERS-CoV | Middle East respiratory syndrome |
| MeV | Measles virus |
| PALS1 | Proteins Associated with Lin Seven 1 |
| PATJ | PALS1-Associated Tight Junction protein |
| PVRL1 | Poliovirus receptor related protein 1 |
| PKD | Protein kinase D |
| RSV | Respiratory syncytial virus |
| SARS-CoV & SARS-CoV-2 | Severe acute respiratory syndrome coronavirus |
| TEER | Trans-epithelial electrical resistance |
| TJ | Tight junction |
| VV | Vaccinia virus |
Disclosures
No conflicts of interest, financial or otherwise, are declared by the authors. The authors have no financial relationship with a biotechnology and/or pharmaceutical manufacturer that has an interest in the subject matter or materials discussed in the submitted manuscript.
References
- 1.Rezaee F, Georas SN.. Breaking barriers. New insights into airway epithelial barrier function in health and disease. Am J Respir Cell Mol Biol. 2014;50(5):1–19. doi: 10.1165/rcmb.2013-0541RT. PubMed PMID: 24467704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Georas SN, Rezaee F. Epithelial barrier function: at the front line of asthma immunology and allergic airway inflammation. J Allergy Clin Immunol. 2014;134(3):509–520. doi: 10.1016/j.jaci.2014.05.049. PubMed PMID: 25085341; PMCID: PMC4170838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dong D, Xie W, Liu M. Alteration of cell junctions during viral infection. Thorac Cancer. 2020;11(3):519–525. Epub 2020/02/06. doi: 10.1111/1759-7714.13344. PubMed PMID: 32017415; PMCID: PMC7049484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mineta K, Yamamoto Y, Yamazaki Y, Tanaka H, Tada Y, Saito K, Tamura A, Igarashi M, Endo T, Takeuchi K, et al. Predicted expansion of the claudin multigene family. FEBS Lett. 2011;585(4):606–612. Epub 2011/02/01. doi: 10.1016/j.febslet.2011.01.028. PubMed PMID: 21276448. [DOI] [PubMed] [Google Scholar]
- 5.Schneeberger EE, Lynch RD. The tight junction: a multifunctional complex. Am J Physiol Cell Physiol. 2004;286(6):C1213–28. Epub 2004/05/21. doi: 10.1152/ajpcell.00558.2003. PubMed PMID: 15151915. [DOI] [PubMed] [Google Scholar]
- 6.Furuse M. Molecular basis of the core structure of tight junctions. Cold Spring Harb Perspect Biol. 2010;2(1):a002907. Epub 2010/02/26. doi: 10.1101/cshperspect.a002907. PubMed PMID: 20182608; PMCID: PMC2827901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wittekindt OH. Tight junctions in pulmonary epithelia during lung inflammation. Pflugers Arch. 2017;469(1):135–147. Epub 2016/12/07. doi: 10.1007/s00424-016-1917-3. PubMed PMID: 27921210; PMCID: PMC5203840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Koval M. Differential pathways of claudin oligomerization and integration into tight junctions. Tissue Barriers. 2013;1(3):e24518. Epub 2014/03/26. doi: 10.4161/tisb.24518. PubMed PMID: 24665398; PMCID: PMC3867512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Turksen K. All about claudins. Tissue Barriers. 2013;1(3):e26750. Epub 2014/03/26. doi: 10.4161/tisb.26750. PubMed PMID: 24665405; PMCID: PMC3866198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jang AS. The apical junctional complex in respiratory diseases. Chonnam Med J. 2014;50(1):1–5. Epub 2014/05/24. doi: 10.4068/cmj.2014.50.1.1. PubMed PMID: 24855600; PMCID: PMC4022791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Eisenreich W, Rudel T, Heesemann J, Goebel W. How viral and intracellular bacterial pathogens reprogram the metabolism of host cells to allow their intracellular replication. Front Cell Infect Microbiol. 2019;9:42. Epub 2019/03/20. doi: 10.3389/fcimb.2019.00042. PubMed PMID: 30886834; PMCID: PMC6409310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guttman JA, Finlay BB. Tight junctions as targets of infectious agents. Biochim Biophys Acta. 2009;1788(4):832–841. Epub 2008/12/09. doi: 10.1016/j.bbamem.2008.10.028. PubMed PMID: 19059200. [DOI] [PubMed] [Google Scholar]
- 13.Maginnis MS. Virus-receptor interactions: the key to cellular invasion. J Mol Biol. 2018;430(17):2590–2611. Epub 2018/06/21. doi: 10.1016/j.jmb.2018.06.024. PubMed PMID: 29924965; PMCID: PMC6083867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Torres-Flores JM, Arias CF. Tight junctions go viral! Viruses. 2015;7(9):5145–5154. Epub 2015/09/26. doi: 10.3390/v7092865. PubMed PMID: 26404354; PMCID: PMC4584309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rezaee F, Harford TJ, Linfield DT, Altawallbeh G, Midura RJ, Ivanov AI, Piedimonte G. cAMP-dependent activation of protein kinase A attenuates respiratory syncytial virus-induced human airway epithelial barrier disruption. PLoS One. 2017;12(7):e0181876. Epub 2017/08/02. doi: 10.1371/journal.pone.0181876. PubMed PMID: 28759570; PMCID: PMC5536269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Singh D, McCann KL, Imani F. MAPK and heat shock protein 27 activation are associated with respiratory syncytial virus induction of human bronchial epithelial monolayer disruption. Am J Physiol Lung Cell Mol Physiol. 2007;293(2):L436–45. Epub 2007/06/15. doi: 10.1152/ajplung.00097.2007. PubMed PMID: 17557802; PMCID: PMC2231338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Masaki T, Kojima T, Okabayashi T, Ogasawara N, Ohkuni T, Obata K, Takasawa A, Murata M, Tanaka S, Hirakawa S, et al. A nuclear factor-kappa B signaling pathway via protein kinase C delta regulates replication of respiratory syncytial virus in polarized normal human nasal epithelial cells. Mol Biol Cell. 2011;22(13):2144–2156. doi: 10.1091/mbc.E10-11-0875. PubMed PMID: WOS:000292293000002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Smallcombe CC, Linfield DT, Harford TJ, Bokun V, Ivanov AI, Piedimonte G, Rezaee F. Disruption of the airway epithelial barrier in a murine model of respiratory syncytial virus infection. Am J Physiol-Lung C. 2019;316(2):L358–L68. doi: 10.1152/ajplung.00345.2018. PubMed PMID: WOS:000458506200005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim S, Kim GH. Roles of claudin-2, ZO-1 and occludin in leaky HK-2 cells. PLoS One. 2017;12(12):e0189221. Epub 2017/12/19. doi: 10.1371/journal.pone.0189221. PubMed PMID: 29252987; PMCID: PMC5734727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Odenwald MA, Choi W, Buckley A, Shashikanth N, Joseph NE, Wang Y, Warren MH, Buschmann MM, Pavlyuk R, Hildebrand J, et al. ZO-1 interactions with F-actin and occludin direct epithelial polarization and single lumen specification in 3D culture. J Cell Sci. 2017;130(1):243–259. Epub 2016/11/02. doi: 10.1242/jcs.188185. PubMed PMID: 27802160; PMCID: PMC5394778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Colpitts CC, Baumert TF. Claudins in viral infection: from entry to spread. Pflugers Arch. 2017;469(1):27–34. Epub 2016/11/26. doi: 10.1007/s00424-016-1908-4. PubMed PMID: 27885488; PMCID: PMC5299967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sajjan U, Wang Q, Zhao Y, Gruenert DC, Hershenson MB. Rhinovirus disrupts the barrier function of polarized airway epithelial cells. Am J Respir Crit Care Med. 2008;178(12):1271–1281. Epub 2008/09/13. doi: 10.1164/rccm.200801-136OC. PubMed PMID: 18787220; PMCID: PMC2599868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Looi K, Buckley AG, Rigby PJ, Garratt LW, Iosifidis T, Zosky GR, Larcombe AN, Lannigan FJ, Ling KM, Martinovich KM, et al. Effects of human rhinovirus on epithelial barrier integrity and function in children with asthma. Clin Exp Allergy. 2018;48(5):513–524. Epub 2018/01/20. 10.1111/cea.13097. PubMed PMID: 29350877. [DOI] [PubMed] [Google Scholar]
- 24.Yeo NK, Jang YJ. Rhinovirus infection-induced alteration of tight junction and adherens junction components in human nasal epithelial cells. Laryngoscope. 2010;120(2):346–352. doi: 10.1002/lary.20764. PubMed PMID: WOS:000274605000024. [DOI] [PubMed] [Google Scholar]
- 25.Hu Y, Song J, Liu L, Zhang Y, Wang L, Li Q. microRNA-4516 contributes to different functions of epithelial permeability barrier by targeting poliovirus receptor related protein 1 in enterovirus 71 and coxsackievirus A16 infections. Front Cell Infect Microbiol. 2018;8:110. Epub 2018/04/25. doi: 10.3389/fcimb.2018.00110. PubMed PMID: 29686973; PMCID: PMC5900049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cohen CJ, Shieh JT, Pickles RJ, Okegawa T, Hsieh JT, Bergelson JM. The coxsackievirus and adenovirus receptor is a transmembrane component of the tight junction. Proc Natl Acad Sci U S A. 2001;98(26):15191–15196. Epub 2001/12/06. doi: 10.1073/pnas.261452898. PubMed PMID: 11734628; PMCID: PMC65005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Coyne CB, Bergelson JM. CAR: a virus receptor within the tight junction. Adv Drug Deliv Rev. 2005;57(6):869–882. Epub 2005/04/12. doi: 10.1016/j.addr.2005.01.007. PubMed PMID: 15820557. [DOI] [PubMed] [Google Scholar]
- 28.Raschperger E, Thyberg J, Pettersson S, Philipson L, Fuxe J, Pettersson RF. The coxsackie- and adenovirus receptor (CAR) is an in vivo marker for epithelial tight junctions, with a potential role in regulating permeability and tissue homeostasis. Exp Cell Res. 2006;312(9):1566–1580. Epub 2006/03/18. doi: 10.1016/j.yexcr.2006.01.025. PubMed PMID: 16542650. [DOI] [PubMed] [Google Scholar]
- 29.LeMessurier KS, Tiwary M, Morin NP, Samarasinghe AE. Respiratory barrier as a safeguard and regulator of defense against influenza a virus and streptococcus pneumoniae. Front Immunol. 2020;11:3. Epub 2020/03/03. doi: 10.3389/fimmu.2020.00003. PubMed PMID: 32117216; PMCID: PMC7011736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Short KR, Kasper J, van der Aa S, Andeweg AC, Zaaraoui-Boutahar F, Goeijenbier M, Richard M, Herold S, Becker C, Scott DP, et al. Influenza virus damages the alveolar barrier by disrupting epithelial cell tight junctions. Eur Respir J. 2016;47(3):954–966. Epub 2016/01/09. doi: 10.1183/13993003.01282-2015. PubMed PMID: 26743480. [DOI] [PubMed] [Google Scholar]
- 31.Pharo EA, Williams SM, Boyd V, Sundaramoorthy V, Durr PA, Baker ML. Host-pathogen responses to pandemic influenza h1n1pdm09 in a human respiratory airway model. Viruses. 2020;12(6):679. Epub 2020/07/01. 10.3390/v12060679. PubMed PMID: 32599823; PMCID: PMC7354428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Travanty E, Zhou B, Zhang H, Di YP, Alcorn JF, Wentworth DE, Mason R, Wang J. Differential susceptibilities of human lung primary cells to H1N1 influenza viruses. J Virol. 2015;89(23):11935–11944. Epub 2015/09/18. 10.1128/JVI.01792-15. PubMed PMID: 26378172; PMCID: PMC4645340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Brune KA, Ferreira F, Mandke P, Chau E, Aggarwal NR, D’Alessio FR, Lambert AA, Kirk G, Blankson J, Drummond MB, et al. HIV impairs lung epithelial integrity and enters the epithelium to promote chronic lung inflammation. PLoS One. 2016;11(3):e0149679. Epub 2016/03/02. 10.1371/journal.pone.0149679. PubMed PMID: 26930653; PMCID: PMC4773117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34..Fan X, Staitieh BS, Jensen JS, Mould KJ, Greenberg JA, Joshi PC, Koval M, Guidot DM. Activating the Nrf2-mediated antioxidant response element restores barrier function in the alveolar epithelium of HIV-1 transgenic rats. Am J Physiol Lung Cell Mol Physiol. 2013;305(3):L267–77. Epub 2013/06/12. 10.1152/ajplung.00288.2012. PubMed PMID: 23748533; PMCID: PMC3743010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fan X, Joshi PC, Koval M, Guidot DM. Chronic alcohol ingestion exacerbates lung epithelial barrier dysfunction in HIV-1 transgenic rats. Alcohol Clin Exp Res. 2011;35(10):1866–1875. Epub 2011/05/17. 10.1111/j.1530-0277.2011.01531.x. PubMed PMID: 21569054; PMCID: PMC3157600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lassiter C, Fan X, Joshi PC, Jacob BA, Sutliff RL, Jones DP, Koval M, Guidot DM. HIV-1 transgene expression in rats causes oxidant stress and alveolar epithelial barrier dysfunction. AIDS Res Ther. 2009;6(1):1. Epub 2009/02/06. 10.1186/1742-6405-6-1. PubMed PMID: 19193217; PMCID: PMC2644707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Teoh KT, Siu YL, Chan WL, Schluter MA, Liu CJ, Peiris JS, Bruzzone R, Margolis B, Nal B. The SARS coronavirus E protein interacts with PALS1 and alters tight junction formation and epithelial morphogenesis. Mol Biol Cell. 2010;21(22):3838–3852. Epub 2010/09/24. 10.1091/mbc.E10-04-0338. PubMed PMID: 20861307; PMCID: PMC2982091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.De Maio F, Lo Cascio E, Babini G, Sali M, Della Longa S, Tilocca B, Roncada P, Arcovito A, Sanguinetti M, Scambia G, et al. Improved binding of SARS-CoV-2 Envelope protein to tight junction-associated PALS1 could play a key role in COVID-19 pathogenesis. Microbes Infect. 2020;22(10):592–597. Epub 2020/09/07. 10.1016/j.micinf.2020.08.006. PubMed PMID: 32891874; PMCID: PMC7473260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Robinot R, Hubert M, GDd M, Lazarini F, Bruel T, Smith N, Levallois S, Larrous F, Fernandes J, Gellenoncourt S, et al. SARS-CoV-2 infection damages airway motile cilia and impairs mucociliary clearance. bioRxiv. 2020. doi: 10.1101/2020.10.06.328369. [DOI] [Google Scholar]
- 40.Singh BK, Pfaller CK, Cattaneo R, Sinn PL. Measles virus ribonucleoprotein complexes rapidly spread across well-differentiated primary human airway epithelial cells along F-actin rings. mBio. 2019;10(6). Epub 2019/11/28. doi: 10.1128/mBio.02434-19. PubMed PMID: 31772054; PMCID: PMC6879720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Muhlebach MD, Mateo M, Sinn PL, Prufer S, Uhlig KM, Leonard VH, Navaratnarajah CK, Frenzke M, Wong XX, Sawatsky B, et al. Adherens junction protein nectin-4 is the epithelial receptor for measles virus. Nature. 2011;480(7378):530–533. Epub 2011/11/04. 10.1038/nature10639. PubMed PMID: 22048310; PMCID: PMC3245798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhang L, Collins PL, Lamb RA, Pickles RJ. Comparison of differing cytopathic effects in human airway epithelium of parainfluenza virus 5 (W3A), parainfluenza virus type 3, and respiratory syncytial virus. Virology. 2011;421(1):67–77. Epub 2011/10/12. 10.1016/j.virol.2011.08.020. PubMed PMID: 21986028; PMCID: PMC3208758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yumine N, Matsumoto Y, Ohta K, Fukasawa M, Nishio M. Claudin-1 inhibits human parainfluenza virus type 2 dissemination. Virology. 2019;531:93–99. Epub 2019/03/12. 10.1016/j.virol.2019.01.031. PubMed PMID: 30856486. [DOI] [PubMed] [Google Scholar]
- 44.Henrickson KJ. Parainfluenza viruses. Clin Microbiol Rev. 2003;16(2):242–264. Epub 2003/04/15. 10.1128/cmr.16.2.242-264.2003. PubMed PMID: 12692097; PMCID: PMC153148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Deng X, Li Y, Qiu J. Human bocavirus 1 infects commercially available primary human airway epithelium cultures productively. J Virol Methods. 2014;195:112–119. Epub 2013/10/19. 10.1016/j.jviromet.2013.10.012. PubMed PMID: 24134939; PMCID: PMC3855471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Huang Q, Deng X, Yan Z, Cheng F, Luo Y, Shen W, Lei-Butters DC, Chen AY, Li Y, Tang L, et al. Establishment of a reverse genetics system for studying human bocavirus in human airway epithelia. PLoS Pathog. 2012;8(8):e1002899. Epub 2012/09/08. 10.1371/journal.ppat.1002899. PubMed PMID: 22956907; PMCID: PMC3431310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chiu CC, Shi YF, Yang JJ, Hsiao YC, Tzang BS, Hsu TC. Effects of human Parvovirus B19 and Bocavirus VP1 unique region on tight junction of human airway epithelial A549 cells. PLoS One. 2014;9(9):e107970. Epub 2014/10/01. doi: 10.1371/journal.pone.0107970. PubMed PMID: 25268969; PMCID: PMC4182432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vermeer PD, McHugh J, Rokhlina T, Vermeer DW, Zabner J, Welsh MJ. Vaccinia virus entry, exit, and interaction with differentiated human airway epithelia. J Virol. 2007;81(18):9891–9899. Epub 2007/06/22. 10.1128/JVI.00601-07. PubMed PMID: 17581984; PMCID: PMC2045410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rima B, Collins P, Easton A, Fouchier R, Kurath G, Lamb RA, Lee B, Maisner A, Rota P, Wang L, et al. ICTV virus taxonomy profile: pneumoviridae. J Gen Virol. 2017;98(12):2912–2913. Epub 2017/11/01. 10.1099/jgv.0.000959. PubMed PMID: 29087278; PMCID: PMC5775899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Geoghegan S, Erviti A, Caballero MT, Vallone F, Zanone SM, Losada JV, Bianchi A, Acosta PL, Talarico LB, Ferretti A, et al. Mortality due to Respiratory Syncytial Virus. Burden and Risk Factors. Am J Respir Crit Care Med. 2017;195(1):96–103. doi: 10.1164/rccm.201603-0658OC. PubMed PMID: 27331632 [DOI] [PubMed] [Google Scholar]
- 51.Hall CB, Weinberg GA, Iwane MK, Blumkin AK, Edwards KM, Staat MA, Auinger P, Griffin MR, Poehling KA, Erdman D, et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med. 2009;360(6):588–598. Epub 2009/02/07. doi: 10.1056/NEJMoa0804877. PubMed PMID: 19196675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Falsey AR, Hennessey PA, Formica MA, Cox C, Walsh EE. Respiratory syncytial virus infection in elderly and high-risk adults. N Engl J Med. 2005;352(17):1749–1759. Epub 2005/04/29. 10.1056/NEJMoa043951. PubMed PMID: 15858184. [DOI] [PubMed] [Google Scholar]
- 53.Rezaee F, Linfield DT, Harford TJ, Piedimonte G. Ongoing developments in RSV prophylaxis: a clinician’s analysis. Curr Opin Virol. 2017;24:70–78. PubMed PMID: 28500974; PMCID: PMC5541395. doi: 10.1016/j.coviro.2017.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Linfield DR, F. RSV: Available prophylactic options and vaccines in clinical trials. In: IntechOpen, editor. The Burden of Respiratory Syncytial Virus Infection in the Young and the Old 2019.
- 55.Stein RT, Sherrill D, Morgan WJ, Holberg CJ, Halonen M, Taussig LM, Wright AL, Martinez FD. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet. 1999;354(9178):541–545. Epub 1999/09/02. 10.1016/S0140-6736(98)10321-5. PubMed PMID: 10470697. [DOI] [PubMed] [Google Scholar]
- 56.Thomsen SF, van der Sluis S, Stensballe LG, Posthuma D, Skytthe A, Kyvik KO, Duffy DL, Backer V, Bisgaard H. Exploring the association between severe respiratory syncytial virus infection and asthma: a registry-based twin study. Am J Respir Crit Care Med. 2009;179(12):1091–1097. Epub 2009/03/17. 10.1164/rccm.200809-1471OC. PubMed PMID: 19286626. [DOI] [PubMed] [Google Scholar]
- 57.Piedimonte G, Hegele RG, Auais A. Persistent airway inflammation after resolution of respiratory syncytial virus infection in rats. Pediatr Res. 2004;55(4):657–665. Epub 2004/01/09. 10.1203/01.PDR.0000112244.72924.26. PubMed PMID: 14711892. [DOI] [PubMed] [Google Scholar]
- 58.Smallcombe CC, Harford TJ, Linfield DT, Lechuga S, Bokun V, Piedimonte G, Rezaee F. Titanium dioxide nanoparticles exaggerate respiratory syncytial virus-induced airway epithelial barrier dysfunction. Am J Physiol Lung Cell Mol Physiol. 2020;319(3):L481–L496. Epub 2020/07/10. 10.1152/ajplung.00104.2020. PubMed PMID: 32640839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Rezaee F, Meednu N, Emo JA, Saatian B, Chapman TJ, Naydenov NG, De Benedetto A, Beck LA, Ivanov AI, Georas SN. Polyinosinic:polycytidylic acid induces protein kinase D-dependent disassembly of apical junctions and barrier dysfunction in airway epithelial cells. J Allergy Clin Immunol. 2011;128(6):1216–24 e11. Epub 2011/10/15. doi: 10.1016/j.jaci.2011.08.035. PubMed PMID: 21996340; PMCID: 3273326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rezaee F, DeSando SA, Ivanov AI, Chapman TJ, Knowlden SA, Beck LA, Georas SN. Sustained protein kinase D activation mediates respiratory syncytial virus-induced airway barrier disruption. J Virol. 2013;87(20):11088–11095. doi: 10.1128/Jvi.01573-13. PubMed PMID: WOS:000325275800014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bougneres L, Girardin SE, Weed SA, Karginov AV, Olivo-Marin JC, Parsons JT, Sansonetti PJ, Van Nhieu GT. Cortactin and Crk cooperate to trigger actin polymerization during Shigella invasion of epithelial cells. J Cell Biol. 2004;166(2):225–235. Epub 2004/07/21. 10.1083/jcb.200402073. PubMed PMID: 15263018; PMCID: PMC2172305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Weed SA, Parsons JT. Cortactin: coupling membrane dynamics to cortical actin assembly. Oncogene. 2001;20(44):6418–6434. Epub 2001/10/19. 10.1038/sj.onc.1204783. PubMed PMID: 11607842. [DOI] [PubMed] [Google Scholar]
- 63.Li Z, Zhang C, Chen L, Li G, Qu L, Balaji KC, Du C. E-cadherin facilitates protein kinase d1 activation and subcellular localization. J Cell Physiol. 2016;231(12):2741–2748. Epub 2016/03/19. 10.1002/jcp.25382. PubMed PMID: 26991955. [DOI] [PubMed] [Google Scholar]
- 64.Gan H, Wang G, Hao Q, Wang QJ, Tang H. Protein kinase D promotes airway epithelial barrier dysfunction and permeability through down-regulation of claudin-1. J Biol Chem. 2013;288(52):37343–37354. Epub 2013/11/23. 10.1074/jbc.M113.511527. PubMed PMID: 24265314; PMCID: PMC3873586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Beavo JA, Brunton LL. Cyclic nucleotide research – still expanding after half a century. Nat Rev Mol Cell Biol. 2002;3(9):710–718. Epub 2002/09/05. doi: 10.1038/nrm911. PubMed PMID: 12209131. [DOI] [PubMed] [Google Scholar]
- 66.Kast JI, McFarlane AJ, Globinska A, Sokolowska M, Wawrzyniak P, Sanak M, Schwarze J, Akdis CA, Wanke K. Respiratory syncytial virus infection influences tight junction integrity. Clin Exp Immunol. 2017;190(3):351–359. Epub 2017/09/01. doi: 10.1111/cei.13042. PubMed PMID: 28856667; PMCID: PMC5680068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Raleigh DR, Marchiando AM, Zhang Y, Shen L, Sasaki H, Wang Y, Long M, Turner JR. Tight junction-associated MARVEL proteins marveld3, tricellulin, and occludin have distinct but overlapping functions. Mol Biol Cell. 2010;21(7):1200–1213. Epub 2010/02/19. doi: 10.1091/mbc.E09-08-0734. PubMed PMID: 20164257; PMCID: PMC2847524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Muto S, Hata M, Taniguchi J, Tsuruoka S, Moriwaki K, Saitou M, Furuse K, Sasaki H, Fujimura A, Imai M, et al. Claudin-2-deficient mice are defective in the leaky and cation-selective paracellular permeability properties of renal proximal tubules. Proc Natl Acad Sci U S A. 2010;107(17):8011–8016. Epub 2010/04/14. doi: 10.1073/pnas.0912901107. PubMed PMID: 20385797; PMCID: PMC2867900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Looi K, Troy NM, Garratt LW, Iosifidis T, Bosco A, Buckley AG, Ling KM, Martinovich KM, Kicic-Starcevich E, Shaw NC, et al. Effect of human rhinovirus infection on airway epithelium tight junction protein disassembly and transepithelial permeability. Exp Lung Res. 2016;42(7):380–395. Epub 2016/10/12. doi: 10.1080/01902148.2016.1235237. PubMed PMID: 27726456. [DOI] [PubMed] [Google Scholar]
- 70.Jacobs SE, Lamson DM, St George K, Walsh TJ. Human rhinoviruses. Clin Microbiol Rev. 2013;26(1):135–162. Epub 2013/01/09. doi: 10.1128/CMR.00077-12. PubMed PMID: 23297263; PMCID: PMC3553670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kennedy JL, Turner RB, Braciale T, Heymann PW, Borish L. Pathogenesis of rhinovirus infection. Curr Opin Virol. 2012;2(3):287–293. Epub 2012/05/01. 10.1016/j.coviro.2012.03.008. PubMed PMID: 22542099; PMCID: PMC3378761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Stobart CC, Nosek JM, Moore ML. Rhinovirus Biology, Antigenic Diversity, and Advancements in the Design of a Human Rhinovirus Vaccine. Front Microbiol. 2017;8:2412. Epub 2017/12/21. doi: 10.3389/fmicb.2017.02412. PubMed PMID: 29259600; PMCID: PMC5723287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.McIntyre CL, Knowles NJ, Simmonds P. Proposals for the classification of human rhinovirus species A, B and C into genotypically assigned types. J Gen Virol. 2013;94(Pt 8):1791–1806. Epub 2013/05/17. doi: 10.1099/vir.0.053686-0. PubMed PMID: 23677786; PMCID: PMC3749525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Faris AN, Ganesan S, Chattoraj A, Chattoraj SS, Comstock AT, Unger BL, Hershenson MB, Sajjan US. Rhinovirus delays cell repolarization in a model of injured/regenerating human airway epithelium. Am J Respir Cell Mol Biol. 2016;55(4):487–499. Epub 2016/04/28. doi: 10.1165/rcmb.2015-0243OC. PubMed PMID: 27119973; PMCID: PMC5070101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Panday A, Sahoo MK, Osorio D, Batra S. NADPH oxidases: an overview from structure to innate immunity-associated pathologies. Cell Mol Immunol. 2015;12(1):5–23. Epub 2014/09/30. doi: 10.1038/cmi.2014.89. PubMed PMID: 25263488; PMCID: PMC4654378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fink K, Duval A, Martel A, Soucy-Faulkner A, Grandvaux N. Dual role of NOX2 in respiratory syncytial virus- and sendai virus-induced activation of NF-kappaB in airway epithelial cells. J Immunol. 2008;180(10):6911–6922. Epub 2008/05/06. doi: 10.4049/jimmunol.180.10.6911. PubMed PMID: 18453612. [DOI] [PubMed] [Google Scholar]
- 77.Snelgrove RJ, Edwards L, Rae AJ, Hussell T. An absence of reactive oxygen species improves the resolution of lung influenza infection. Eur J Immunol. 2006;36(6):1364–1373. Epub 2006/05/17. doi: 10.1002/eji.200635977. PubMed PMID: 16703568. [DOI] [PubMed] [Google Scholar]
- 78.Di Bona D, Cippitelli M, Fionda C, Camma C, Licata A, Santoni A, Craxi A. Oxidative stress inhibits IFN-alpha-induced antiviral gene expression by blocking the JAK-STAT pathway. J Hepatol. 2006;45(2):271–279. Epub 2006/04/06. doi: 10.1016/j.jhep.2006.01.037. PubMed PMID: 16595158. [DOI] [PubMed] [Google Scholar]
- 79.Comstock AT, Ganesan S, Chattoraj A, Faris AN, Margolis BL, Hershenson MB, Sajjan US. Rhinovirus-induced barrier dysfunction in polarized airway epithelial cells is mediated by NADPH oxidase 1. J Virol. 2011;85(13):6795–6808. Epub 2011/04/22. doi: 10.1128/JVI.02074-10. PubMed PMID: 21507984; PMCID: PMC3126521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Xiang RF, Stack D, Huston SM, Li SS, Ogbomo H, Kyei SK, Mody CH. Ras-related C3 (SFK) are proximal and essential for phosphatidylinositol 3-kinase (PI3K) activation in natural killer (NK) cell-mediated direct cytotoxicity against cryptococcus neoformans. J Biol Chem. 2016;291(13):6912–6922. Epub 2016/02/13. doi: 10.1074/jbc.M115.681544. PubMed PMID: 26867574; PMCID: PMC4807276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pons-Salort M, Parker EP, Grassly NC. The epidemiology of non-polio enteroviruses: recent advances and outstanding questions. Curr Opin Infect Dis. 2015;28(5):479–487. Epub 2015/07/24. doi: 10.1097/QCO.0000000000000187. PubMed PMID: 26203854; PMCID: PMC6624138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Tuthill TJ, Groppelli E, Hogle JM, Rowlands DJ. Picornaviruses. Curr Top Microbiol Immunol. 2010;343:43–89. Epub 2010/04/17. doi: 10.1007/82_2010_37. PubMed PMID: 20397067; PMCID: PMC3018333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Xiang Z, Gonzalez R, Wang Z, Ren L, Xiao Y, Li J, Li Y, Vernet G, Paranhos-Baccala G, Jin Q, et al. Coxsackievirus A21, enterovirus 68, and acute respiratory tract infection, China. Emerg Infect Dis. 2012;18(5):821–824. Epub 2012/04/21. doi: 10.3201/eid1805.111376. PubMed PMID: 22516379; PMCID: PMC3358056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Walters RW, Freimuth P, Moninger TO, Ganske I, Zabner J, Welsh MJ. Adenovirus fiber disrupts CAR-mediated intercellular adhesion allowing virus escape. Cell. 2002;110(6):789–799. Epub 2002/09/26. doi: 10.1016/s0092-8674(02)00912-1. PubMed PMID: 12297051. [DOI] [PubMed] [Google Scholar]
- 85.Cook J, Radke J. Mechanisms of pathogenesis of emerging adenoviruses. F1000Res. 2017;6:90. Epub 2017/02/12. 10.12688/f1000research.10152.1. PubMed PMID: 28184296; PMCID: PMC5289147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Cheng J, Qi X, Chen D, Xu X, Wang G, Dai Y, Cui D, Chen Q, Fan P, Ni L, et al. Epidemiology and transmission characteristics of human adenovirus type 7 caused acute respiratory disease outbreak in military trainees in East China. Am J Transl Res. 2016;8(5):2331–2342. Epub 2016/06/28. PubMed PMID: 27347341; PMCID: PMC4891446 [PMC free article] [PubMed] [Google Scholar]
- 87.Zeng X, Carlin CR. Host cell autophagy modulates early stages of adenovirus infections in airway epithelial cells. J Virol. 2013;87(4):2307–2319. Epub 2012/12/14. doi: 10.1128/JVI.02014-12. PubMed PMID: 23236070; PMCID: PMC3571477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kotha PL, Sharma P, Kolawole AO, Yan R, Alghamri MS, Brockman TL, Gomez-Cambronero J, Excoffon KJ. Adenovirus entry from the apical surface of polarized epithelia is facilitated by the host innate immune response. PLoS Pathog. 2015;11(3):e1004696. Epub 2015/03/15. doi: 10.1371/journal.ppat.1004696. PubMed PMID: 25768646; PMCID: PMC4358923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.McAuley JL, Gilbertson BP, Trifkovic S, Brown LE, McKimm-Breschkin JL. Influenza virus neuraminidase structure and functions. Front Microbiol. 2019;10:39. Epub 2019/02/15. doi: 10.3389/fmicb.2019.00039. PubMed PMID: 30761095; PMCID: PMC6362415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kalil AC, Thomas PG. Influenza virus-related critical illness: pathophysiology and epidemiology. Crit Care. 2019;23(1):258. Epub 2019/07/22. doi: 10.1186/s13054-019-2539-x. PubMed PMID: 31324202; PMCID: PMC6642581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hatta M, Hatta Y, Kim JH, Watanabe S, Shinya K, Nguyen T, Lien PS, Le QM, Kawaoka Y. Growth of H5N1 influenza A viruses in the upper respiratory tracts of mice. PLoS Pathog. 2007;3(10):1374–1379. Epub 2007/10/10. doi: 10.1371/journal.ppat.0030133. PubMed PMID: 17922570; PMCID: PMC2000968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Zielecki F, Semmler I, Kalthoff D, Voss D, Mauel S, Gruber AD, Beer M, Wolff T. Virulence determinants of avian H5N1 influenza A virus in mammalian and avian hosts: role of the C-terminal ESEV motif in the viral NS1 protein. J Virol. 2010;84(20):10708–10718. Epub 2010/08/06. 10.1128/JVI.00610-10. PubMed PMID: 20686040; PMCID: PMC2950580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Yu H, Gao Z, Feng Z, Shu Y, Xiang N, Zhou L, Huai Y, Feng L, Peng Z, Li Z, et al. Clinical characteristics of 26 human cases of highly pathogenic avian influenza A (H5N1) virus infection in China. PLoS One. 2008;3(8):e2985. Epub 2008/08/22. doi: 10.1371/journal.pone.0002985. PubMed PMID: 18716658; PMCID: PMC2515635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Blumel J, Burger R, Drosten C, Groner A, Gurtler L, Heiden M, Hildebrandt M, Jansen B, Klamm H, Montag-Lessing T, et al. Influenza Virus. Transfus Med Hemother. 2008;35(1):42–49. Epub 2008/01/01. doi: 10.1159/000111480. PubMed PMID: 21547110; PMCID: PMC3083280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bouvier NM, Palese P. The biology of influenza viruses. Vaccine. 2008;26(Suppl 4):D49–53. Epub 2009/02/21. doi: 10.1016/j.vaccine.2008.07.039. PubMed PMID: 19230160; PMCID: PMC3074182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Golebiewski L, Liu H, Javier RT, Rice AP. The avian influenza virus NS1 ESEV PDZ binding motif associates with Dlg1 and Scribble to disrupt cellular tight junctions. J Virol. 2011;85(20):10639–10648. Epub 2011/08/19. doi: 10.1128/JVI.05070-11. PubMed PMID: 21849460; PMCID: PMC3187509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Denney L, Ho LP. The role of respiratory epithelium in host defence against influenza virus infection. Biomed J. 2018;41(4):218–233. Epub 2018/10/24. doi: 10.1016/j.bj.2018.08.004. PubMed PMID: 30348265; PMCID: PMC6197993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Zhang L, Peeples ME, Boucher RC, Collins PL, Pickles RJ. Respiratory syncytial virus infection of human airway epithelial cells is polarized, specific to ciliated cells, and without obvious cytopathology. J Virol. 2002;76(11):5654–5666. Epub 2002/05/07. doi: 10.1128/jvi.76.11.5654-5666.2002. PubMed PMID: 11991994; PMCID: PMC137037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.UNAIDS . Global HIV & AIDS statistics - 2020 fact sheet 2020. [November/12/2020]. Available from: https://www.unaids.org/en/resources/fact-sheet.
- 100.Hladik F, McElrath MJ. Setting the stage: host invasion by HIV. Nat Rev Immunol. 2008;8(6):447–457. Epub 2008/05/13. doi: 10.1038/nri2302. PubMed PMID: 18469831; PMCID: PMC2587276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fitzpatrick ME, Kunisaki KM, Morris A. Pulmonary disease in HIV-infected adults in the era of antiretroviral therapy. AIDS. 2018;32(3):277–292. Epub 2017/12/02. doi: 10.1097/QAD.0000000000001712. PubMed PMID: 29194119; PMCID: PMC5937696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Benvenuto D, Giovanetti M, Ciccozzi A, Spoto S, Angeletti S, Ciccozzi M. The 2019-new coronavirus epidemic: evidence for virus evolution. J Med Virol. 2020;92(4):455–459. Epub 2020/01/30. doi: 10.1002/jmv.25688. PubMed PMID: 31994738; PMCID: PMC7166400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. Epub 2015/02/28. 10.1007/978-1-4939-2438-7_1. PubMed PMID: 25720466; PMCID: PMC4369385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.ECDC ECfDCaP . COVID-19 situation update worldwide, as of week 2 2021 2020 [January/26/2021]. Available from: https://www.ecdc.europa.eu/en/geographical-distribution-2019-ncov-cases.
- 105.Raghu G, Wilson KC. COVID-19 interstitial pneumonia: monitoring the clinical course in survivors. Lancet Respir Med. 2020;8(9):839–842. Epub 2020/08/08. doi: 10.1016/S2213-2600(20)30349-0. PubMed PMID: 32758440; PMCID: PMC7398671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in covid-19. N Engl J Med. 2020;383(2):120–128. Epub 2020/05/22. doi: 10.1056/NEJMoa2015432. PubMed PMID: 32437596; PMCID: PMC7412750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.CDC CfDCaP . Understanding How COVID-19 Vaccines Work 2021. [January/25/2021]. Available from: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/different-vaccines/how-they-work.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Fvaccines%2Fabout-vaccines%2Fhow-they-work.html.
- 108.Zhu N, Wang W, Liu Z, Liang C, Wang W, Ye F, Huang B, Zhao L, Wang H, Zhou W, et al. Morphogenesis and cytopathic effect of SARS-CoV-2 infection in human airway epithelial cells. Nat Commun. 2020;11(1):3910. Epub 2020/08/09. doi: 10.1038/s41467-020-17796-z. PubMed PMID: 32764693; PMCID: PMC7413383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Takeda M. Measles virus breaks through epithelial cell barriers to achieve transmission. J Clin Invest. 2008;118(7):2386–2389. Epub 2008/06/24. doi: 10.1172/JCI36251. PubMed PMID: 18568081; PMCID: PMC2430502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Leonard VH, Sinn PL, Hodge G, Miest T, Devaux P, Oezguen N, Braun W, McCray PB Jr., McChesney MB, Cattaneo R. Measles virus blind to its epithelial cell receptor remains virulent in rhesus monkeys but cannot cross the airway epithelium and is not shed. J Clin Invest. 2008;118(7):2448–2458. Epub 2008/06/24. doi: 10.1172/JCI35454. PubMed PMID: 18568079; PMCID: PMC2430500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ludlow M, Rennick LJ, Sarlang S, Skibinski G, McQuaid S, Moore T, de Swart RL, Duprex WP. Wild-type measles virus infection of primary epithelial cells occurs via the basolateral surface without syncytium formation or release of infectious virus. J Gen Virol. 2010;91(\(Pt 4)):971–979. Epub 2009/11/20. doi: 10.1099/vir.0.016428-0. PubMed PMID: 19923259. [DOI] [PubMed] [Google Scholar]
- 112.Burke CW, Bridges O, Brown S, Rahija R, Russell CJ. Mode of parainfluenza virus transmission determines the dynamics of primary infection and protection from reinfection. PLoS Pathog. 2013;9(11):e1003786. Epub 2013/11/28. doi: 10.1371/journal.ppat.1003786. PubMed PMID: 24278024; PMCID: PMC3836739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Falsey AR. Current management of parainfluenza pneumonitis in immunocompromised patients: a review. Infect Drug Resist. 2012;5:121–127. Epub 2012/08/16. doi: 10.2147/IDR.S25874. PubMed PMID: 22893749; PMCID: PMC3418768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Jartti T, Hedman K, Jartti L, Ruuskanen O, Allander T, Soderlund-Venermo M. Human bocavirus-the first 5 years. Rev Med Virol. 2012;22(1):46–64. Epub 2011/11/01. doi: 10.1002/rmv.720. PubMed PMID: 22038931. [DOI] [PubMed] [Google Scholar]
- 115.Tolonen N, Doglio L, Schleich S, Krijnse Locker J. Vaccinia virus DNA replication occurs in endoplasmic reticulum-enclosed cytoplasmic mini-nuclei. Mol Biol Cell. 2001;12(7):2031–2046. Epub 2001/07/14. doi: 10.1091/mbc.12.7.2031. PubMed PMID: 11452001; PMCID: PMC55651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Xu R, Johnson AJ, Liggitt D, Bevan MJ. Cellular and humoral immunity against vaccinia virus infection of mice. J Immunol. 2004;172(10):6265–6271. Epub 2004/05/07. doi: 10.4049/jimmunol.172.10.6265. PubMed PMID: 15128815. [DOI] [PubMed] [Google Scholar]
- 117.Saatian B, Rezaee F, Desando S, Emo J, Chapman T, Knowlden S, Georas SN. Interleukin-4 and interleukin-13 cause barrier dysfunction in human airway epithelial cells. Tissue Barriers. 2013;1(2):e24333. doi: 10.4161/tisb.24333. PubMed PMID: 24665390; PMCID: 3875607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Hebert KD, McLaughlin N, Galeas-Pena M, Zhang Z, Eddens T, Govero A, Pilewski JM, Kolls JK, Pociask DA. Targeting the IL-22/IL-22BP axis enhances tight junctions and reduces inflammation during influenza infection. Mucosal Immunol. 2020;13(1):64–74. Epub 2019/10/11. doi: 10.1038/s41385-019-0206-9. PubMed PMID: 31597930; PMCID: PMC6917921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Alcorn JF. IL-22 plays a critical role in maintaining epithelial integrity during pulmonary infection. Front Immunol. 2020;11:1160. Epub 2020/06/26. 10.3389/fimmu.2020.01160. PubMed PMID: 32582219; PMCID: PMC7296169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Abood RN, McHugh KJ, Rich HE, Ortiz MA, Tobin JM, Ramanan K, Robinson KM, Bomberger JM, Kolls JK, Manni ML, et al. IL-22-binding protein exacerbates influenza, bacterial super-infection. Mucosal Immunol. 2019;12(5):1231–1243. Epub 2019/07/13. doi: 10.1038/s41385-019-0188-7. PubMed PMID: 31296910; PMCID: PMC6717528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Friedlander SL, Busse WW. The role of rhinovirus in asthma exacerbations. J Allergy Clin Immunol. 2005;116(2):267–273. Epub 2005/08/09. doi: 10.1016/j.jaci.2005.06.003. PubMed PMID: 16083778. [DOI] [PubMed] [Google Scholar]
- 122.Heijink IH, Jonker MR, de Vries M, van Oosterhout AJ, Telenga E, Ten Hacken NH, Postma DS, van den Berge M. Budesonide and fluticasone propionate differentially affect the airway epithelial barrier. Respir Res. 2016;17(1):2. Epub 2016/01/08. doi: 10.1186/s12931-015-0318-z. PubMed PMID: 26739349; PMCID: PMC4704248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Waltl EE, Selb R, Eckl-Dorna J, Mueller CA, Cabauatan CR, Eiwegger T, Resch-Marat Y, Niespodziana K, Vrtala S, Valenta R, et al. Betamethasone prevents human rhinovirus- and cigarette smoke- induced loss of respiratory epithelial barrier function. Sci Rep. 2018;8(1):9688. Epub 2018/06/28. doi: 10.1038/s41598-018-27022-y. PubMed PMID: 29946071; PMCID: PMC6018698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Hashimoto S, Matsumoto K, Gon Y, Ichiwata T, Takahashi N, Kobayashi T. Viral infection in asthma. Allergol Int. 2008;57(1):21–31. Epub 2008/01/23. doi: 10.2332/allergolint.R-07-156. PubMed PMID: 18209504. [DOI] [PubMed] [Google Scholar]
- 125.Asgrimsson V, Gudjonsson T, Gudmundsson GH, Baldursson O. Novel effects of azithromycin on tight junction proteins in human airway epithelia. Antimicrob Agents Chemother. 2006;50(5):1805–1812. Epub 2006/04/28. doi: 10.1128/AAC.50.5.1805-1812.2006. PubMed PMID: 16641453; PMCID: PMC1472195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Heijink IH, Kies PM, Kauffman HF, Postma DS, van Oosterhout AJ, Vellenga E. Down-regulation of E-cadherin in human bronchial epithelial cells leads to epidermal growth factor receptor-dependent Th2 cell-promoting activity. J Immunol. 2007;178(12):7678–7685. Epub 2007/06/06. doi: 10.4049/jimmunol.178.12.7678. PubMed PMID: 17548604. [DOI] [PubMed] [Google Scholar]


