Abstract
We report mean severe acute respiratory syndrome coronavirus 2 serial intervals for Montana, USA, from 583 transmission pairs; infectors’ symptom onset dates occurred during March 1–July 31, 2020. Our estimate was 5.68 (95% CI 5.27–6.08) days, SD 4.77 (95% CI 4.33–5.19) days. Subperiod estimates varied temporally by nonpharmaceutical intervention type and fluctuating incidence.
Keywords: SARS-CoV-2, COVID-19, coronavirus, 2019 novel coronavirus disease, severe acute respiratory syndrome coronavirus 2, zoonoses, coronavirus disease, viruses, epidemics, infectious disease transmission, epidemiology, mathematical model, communicable disease control, rural population, coronavirus, basic reproduction number, Montana
In support of efforts in response to the emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the pathogen causing novel coronavirus disease (COVID-19), the scientific community has attempted to predict its transmission trends, often through disease modeling. However, disease-specific parameter estimates for SARS-CoV-2 vary greatly. These parameters include the serial interval (SI), or the duration between onset of symptoms in connected primary and secondary cases, which is crucial in estimating epidemic reproduction numbers (R0) and assessing the effects of nonpharmaceutical interventions (NPIs) on transmission (1). Recent studies report SARS-CoV-2 SIs ranging from 2.97 to 7.5 days, with estimates representing primarily densely populated and urban settings (Table 1; Figure 1). The rural United States was relatively untouched in early epidemic waves, but major outbreaks followed in subsequent waves, so it is unknown whether rural- and urban-based transmission differ. Our objective was to report and compare SARS-CoV-2 SI values for Montana, USA, a primarily rural population, with other global and urban estimates. The study was defined as a public health surveillance activity by the University of Montana Institutional Review Board.
Table 1. Published mean serial interval estimates for severe acute respiratory syndrome coronavirus 2*.
| Publication† | Study location, dates (all in 2020 except as indicated) | No. cases (pairs) | SI mean (95% CI) | SI SD (95% CI) | SI estimate method |
|---|---|---|---|---|---|
| This study | Montana, USA, Mar 1–Jul 31 | 4,793 (583) | 5.68 (5.27–6.08) | 4.77 (4.33–5.19) | Forward |
| Prete et al., 2020 (13)‡ | Brazil, Feb 25–Mar 19 | NA (65) | 2.97 | 3.29 | Other |
| Talmoudi et al., 2020 (14)‡ | Tunisia, Feb 29–May 5 | NA (491) | 5.30 (4.66–5.95) | 0.26 (0.23–0.30) | Other |
| Lavezzo et al., 2020 (15) | Vo’, Italy, Feb 21–Mar 7 | 81 (41) | 7.2 (5.9–9.6) | NA | Other |
| Aghaali et al., 2020 (16) | Qom, Iran, Feb 20–Mar 8 | 88 (37) | 4.55 | 3.30 | Forward |
| You et al., 2020 (17)‡ | China (OHP), as of Mar 31 | 14,828 (198) | 4.60 | 5.55 | Intrinsic |
| Ali et al., 2020 (1)‡ | China (OHP), Jan 9–Feb 13 | 9,120 (677) | 5.1 (4.7–5.5) | 5.3 (5.0–5.6) | Forward |
| Zhang et al., 2020 (18) | China (OHP), Jan 19–Feb 17 | 8,579 (35) | 5.1 (1.3–11.6) | NA | Forward |
| Du et al., 2020 (10)‡ | China (OHP), Jan 21–Feb 8 | 752 (468) | 3.96 (3.53–4.39) | 4.75 (4.46–5.07) | Backward |
| Liao et al., 2020 (19) | China (CTGCH), Jan 7–Mar 20 | 46 (12) | 6.50 (2.45–17.38) | NA | Forward |
| Zhao et al., 2020 (20) | Hong Kong, Jan 16–Feb 15 | 56 (21) | 4.9 (3.6–6.2) | 4.4 (2.9–8.3) | Other |
| Chan et al., 2020 (21) | Hong Kong, Jan 23–Apr 6 | 915 (47) | 6.5 (0–18) | 4.7 | Unknown |
| Bi et al., 2020 (22) | Shenzhen, China, Jan 14–Feb 9 | 391 (48) | 6.3 (5.2–7.6) | 4.2 (3.1–5.3) | Other |
| Wang et al., 2020 (23) | Shenzhen, China, Jan 19–Feb 22 | 417 (27) | 5.9 (3.9–9.6) | 4.8 (3.1–10.1) | Other |
| Ganyani et al., 2020 (24)‡ | Tianjin, China, Jan 14–Feb 27 | 135 (NA) | 3.95 (–4.47 to 12.51) | 4.24 (4.03–4.95) | Other |
| Tindale et al., 2020 (25) | Tianjin, China, Jan 21–Feb 22 | 135 (72) | 4.31 (2.91–5.72) | 0.716 | Forward |
| Li et al., 2020 (26) | Wuhan, China, as of Jan 22 | 425 (6) | 7.5 (5.3–19.0) | 3.4 | Other |
| Ganyani et al., 2020 (24)‡ | Singapore, Jan 21–Feb 26 | 91 (NA) | 5.21 (–3.35 to 13.94) | 4.32 (4.06–5.58) | Other |
| Tindale et al., 2020 (25) | Singapore, Jan 23–Feb 26 | 93 (56) | 4.17 (2.44–5.89) | 0.882 | Forward |
| Ki et al., 2020 (27) | South Korea, Jan 10–Feb 10 | 28 (12) | 6.6 (3–15) | NA | Unknown |
| Mettler et al., 2020 (12)‡ | South Korea, Jan 20–Jun 30 | 5,201 (102) | 3.43 (2.62–4.24) | NA | Forward |
| Chun et al., 2020 (28)‡ | South Korea, Jan 23–Mar 31 | 9,887 (69) | 3.18 (2.22–4.24) | 0.75 (0.47–1.03) | Forward |
| Son et al., 2020 (29) | Busan, South Korea, Feb 21–Mar 24 | 108 (28) | 5.54 (4.08–7.01) | 3.90 (2.47–5.32) | Other |
| Nishiura et al., 2020 (30) | Meta-analysis, 2019 Dec 21–2020 Feb 12 | NA (28) | 4.7 (3.7–6.0) | 2.9 (1.9–4.9) | Other |
| He et al., 2020 (11)‡ | Meta-analysis, Jan 21–Feb 12 | NA (77) | 5.8 (4.8–6.8) | NA | Other |
*All articles published during 2020 except this study. CTGCH, Chongqing Three Gorges Central Hospital; NA, not available; OHP, outside Hubei Province; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SI, serial interval. †See References and Appendix for full publication information. ‡Study included negative-valued serial interval pairs in the estimate.
Figure 1.
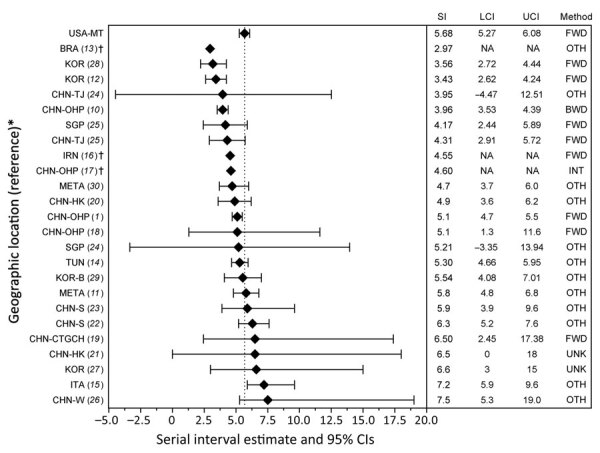
Published mean serial interval estimates for severe acute respiratory syndrome coronavirus 2. *See References and Appendix for full study information. †These studies did not report CIs. Only point estimates are given. BRA, Brazil; BWD, backward; CHN-CTGCH, China–Chongqing Three Gorges Central Hospital; CHN-HK, China–Hong Kong; CHN-OHP, China–outside Hubei Province; CHN-S, China–Shenzhen; CHN-TJ, China–Tianjin; CHN-W, China–Wuhan; FWD, forward; INT, intrinsic; IRN, Iran; ITA, Italy; KOR, South Korea; KOR-B, South Korea–Busan; LCI, lower confidence interval; META, meta-analysis; NA, data not available; OTH, other; SGP, Singapore; TUN, Tunisia; UCI, upper confidence interval; UNK, unknown; USA-MT, United States–Montana.
The Study
We acquired COVID-19 data, reported by local health jurisdictions, from the Montana Department of Public Health and Human Services; we obtained 45,102 case records as of November 15, 2020. We examined a subset of cases with symptom onset dates during March 1–July 31, 2020 (n = 4,793), as well as secondary cases resulting from primary infections during that period, regardless of onset date. We selected this period because all reported cases were PCR positive, all NPI stages were represented (pre–shelter-in-place [pre-SIP], shelter-in-place [SIP], and reopening phase 1 and phase 2), and the proportion of identifiable transmission chains among cases was relatively high (March–June 39%–44%; July 11%) compared with later periods (August–November 0%–2%).
We assessed the records to identify all epidemiologic links. We defined links as cases having contact with another reported case, when viral infection through accepted modes of transmission was plausible. Linked records (n = 1,005) were organized into pairs and designated as primary or secondary cases. When appropriate, cases were listed as primaries for multiple secondary cases; however, cases were limited to 1 secondary designation. For some secondary cases, 1 specific primary was not clearly defined. To estimate a serial range in these situations, we assigned upper and lower bounds using the shortest and longest SIs from all possible primaries. We excluded records when we could not determine an epidemiologic link or transmission direction. We identified 583 pairs, with 466 primary and 583 secondary cases.
We gave temporal markers to pairs on the basis of the primary case’s symptom onset date, consistent with forward-looking SIs (2), and grouped them by the corresponding statewide NPI: pre-SIP, March 1–27; SIP, March 28–April 25; phase 1, April 26–May 31; phase 2 (June), June 1–30; and phase 2 (July), July 1–31. We divided phase 2 into 2 subperiods to account for changing incidence trends.
We analyzed data using R version 3.6.2 and the EpiEstim package (3,4). Complying with EpiEstim functional requirements, we assigned pairs with a zero-valued SI an upper bound of 1 day, with lower bounds unchanged (n = 52 pairs). No negative-valued SIs were identified. We excluded pairs with a SI >2 incubation periods (>28 days). We determined that a gamma distribution was most appropriate using the R0 package est.GT function (5). Next, we used EpiEstim estimate_R, with case-pair and daily incidence data, to perform a Bayesian estimation of the SI gamma distribution using Markov chain Monte Carlo specified for the joint posterior sample of possible SI values (6,7).
Montana’s overall mean SI estimate was 5.68 (95% CI 5.27–6.08) days (SD 4.77 [95% CI 4.33–5.19] days) (Figure 2). Pre-SIP provided the longest subperiod estimate, 6.84 (95% CI 5.84–7.87) days. The SI shortened during SIP, to 5.54 (95% CI 3.34–8.26) days, and again during phase 1, to 5.26 (95% CI 3.64–7.21) days. However, the SI lengthened during phase 2 (June) to 6.23 (95% CI 5.59–6.85) days, almost reaching pre-SIP levels. Phase 2 (July) demonstrated a sharp reduction to the shortest SI observed, 4.42 (95% CI 3.92–4.93) days. Sensitivity analyses of NPI impact delays resulted in altered subperiod estimates, especially for phase 1 relative to other subperiods (Table 2). Additional sensitivity analyses, comparing forward- and backward-looking SIs, produced vastly dissimilar point estimates and trends.
Figure 2.
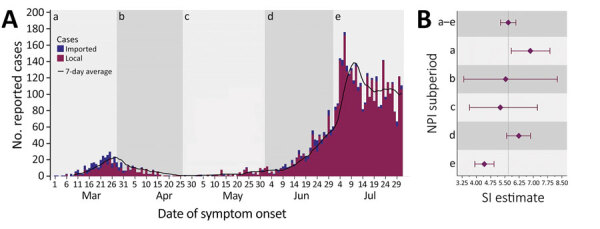
Reported COVID-19 cases and SARS-CoV-2 SI estimates by NPI subperiod, Montana, USA, March 1–July 31, 2020. A) COVID-19 cases, by date of symptom onset. Total cases, 4,793; total pairs, 583. For subperiod pair totals, see the Forward section of Table 2. B) SI estimates and 95% CIs (error bars). Overall mean SI was 5.68 (95% CI 5.27–6.08) days, overall SD 4.77 (95% CI 4.33–5.19) days. For subperiod SI and SD estimates, see the Forward section of Table 2. SI estimates are forward-looking and are based on the symptom onset date of the primary case in the infector–infectee pair. NPI subperiods: a) Pre-SIP, March 1–27, no NPIs in place; no. cases, 285. b) SIP, March 28–April 25, statewide stay-at-home order instituted and all nonessential businesses closed; no. cases, 168. c) Phase 1, April 26–May 31, statewide stay-at-home order lifted and limited business types allowed to open with reduced capacity; no. cases, 99. d) Phase 2 (June), June 1–30, all business types allowed to open under less restrictive capacity regulations; no. cases, 824. e) Phase 2 (July), July 1–31, all business types allowed to open under less restrictive capacity regulations; no. cases 3,417. Black line is the average number of cases for the preceding 7 days. Imported case: COVID-19 case linked to out-of-state OR out-of-county transmission; local case: nonimported COVID-19 case linked to in-state AND in-county transmission. COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; SI, serial interval; SIP, shelter-in-place.
Table 2. Sensitivity analyses: forward and backward severe acute respiratory syndrome coronavirus 2 serial interval estimates by nonpharmaceutical intervention subperiod and length of intervention effects delay*.
| SI estimate method | NPI subperiod | Measure | Sensitivity analysis scenarios† |
||
|---|---|---|---|---|---|
| No delay | 1-week delay | 2-week delay | |||
| Forward: onset of
primary case |
Pre–shelter-in-place, Mar 1–27 |
No. pairs | 95 | 105 | 113 |
| Mean SI (95%CI) | 6.84 (5.84–7.87) | 6.83 (5.67–8.07) | 6.66 (5.61–7.80) | ||
| SD (95% CI) |
5.56 (4.45–6.80) |
5.78 (4.48–7.24) |
5.61 (4.50–6.84) |
||
| Shelter-in-place, Mar 28–Apr 25 |
No. pairs | 20 | 10 | 3 | |
| Mean SI (95% CI) | 5.54 (3.34–8.26) | 4.08 (2.61–5.85) | 2.46 (1.24–4.10) | ||
| SD (95% CI) |
5.30 (2.69–8.76) |
2.83 (1.47–4.66) |
1.52 (0.38–3.38) |
||
| Reopening, phase 1, Apr 26–May 31 |
No. pairs | 25 | 64 | 114 | |
| Mean SI (95% CI) | 5.26 (3.64–7.21) | 7.45 (6.02–9.02) | 7.10 (6.08–8.16) | ||
| SD (95% CI) |
4.74 (2.86–7.09) |
6.24 (4.70–8.03) |
5.82 (4.77–6.99) |
||
| Reopening, phase 2, Jun 1–30 |
No. pairs | 248 | 296 | 289 | |
| Mean SI (95% CI) | 6.23 (5.59–6.85) | 5.39 (4.88–5.94) | 5.08 (4.56–5.59) | ||
| SD (95% CI) |
5.32 (4.61–6.05) |
4.59 (4.01–5.21) |
4.32 (3.75–4.94) |
||
| Reopening, phase 2, Jul 1–31 |
No. pairs | 195 | 117 | 76 | |
| Mean SI (95% CI) | 4.42 (3.92–4.93) | 4.20 (3.65–4.78) | 3.98 (3.36–4.67) | ||
| SD (95% CI) |
3.51 (2.97–4.06) |
3.20 (2.65–3.80) |
2.90 (2.29–3.60) |
||
| Backward: onset of secondary case | Pre–shelter-in-place, Mar 1–27 |
No. pairs | 61 | 89 | 105 |
| Mean SI (95% CI) | 4.82 (3.88–5.84) | 5.83 (4.86–6.82) | 6.48 (5.55–7.51) | ||
| SD (95% CI) |
3.84 (2.88–4.93) |
4.91 (3.86–6.08) |
5.50 (4.44–6.63) |
||
| Shelter-in-place, Mar 28–Apr 25 |
No. pairs | 54 | 26 | 11 | |
| Mean SI (95% CI) | 8.57 (6.77–10.58) | 9.03 (6.73–11.66) | 7.58 (4.29–11.83) | ||
| SD (95% CI) |
6.95 (5.10–8.99) |
6.52 (4.28–9.22) |
6.21 (2.91–10.73) |
||
| Reopening, phase 1, Apr 26–May 31 |
No. pairs | 19 | 30 | 62 | |
| Mean SI (95% CI) | 3.79 (2.46–5.37) | 4.95 (3.53–6.60) | 4.57 (3.64–5.60) | ||
| SD (95% CI) |
3.10 (1.70–4.90) |
4.41 (2.78–6.43) |
3.73 (2.72–4.90) |
||
| Reopening, phase 2, Jun 1–30 |
No. pairs | 202 | 280 | 310 | |
| Mean SI (95% CI) | 5.38 (4.72–6.08) | 5.14 (4.64–5.67) | 5.22 (4.73–5.77) | ||
| SD (95% CI) |
4.59 (3.86–5.41) |
4.31 (3.77–4.90) |
4.38 (3.85–4.97) |
||
| Reopening, phase 2, Jul 1–31 | No. pairs | 233 | 161 | 106 | |
| Mean SI (95% CI) | 5.43 (4.85–6.05) | 5.82 (5.12–6.56) | 6.45 (5.37–7.57) | ||
| SD (95% CI) | 4.52 (3.90–5.17) | 4.88 (4.14–5.70) | 5.41 (4.35–6.64) | ||
*NPI, nonpharmaceutical intervention; SI, serial interval. †Serial interval estimation methods and delay scenarios contain dissimilar pair totals because of their temporal differences (forward pairs, n) no delay: 583; 1-week delay: 592; 2-week delay: 595; (backward pairs, n) no delay: 569; 1-week delay: 586; 2-week delay: 594.
Conclusions
Analysis of SARS-CoV-2 transmission in Montana during March 1–July 31, 2020, identified a mean SI of 5.68 (95% CI 5.27–6.08) days, falling within the bounds of 16 of 24 published estimates from more urbanized settings across the globe (Table 1; Figure 1). However, an aggregate estimate derived from data spanning multiple outbreak stages may not accurately describe Montana-based transmission because changing contact patterns and environmental influences may cause variation (1,2). Temporal analyses suggest that NPIs influenced transmission patterns, as demonstrated by Montana’s epidemic curve and fluctuating SI values (Figure 2). Ali found that SIs shorten as stricter NPIs are applied (1,8), which our subperiod estimates mostly support. However, phase 2 (July) contradicts the premise, with the shortest subperiod SI and a less restrictive NPI (Table 2). Furthermore, when accounting for NPI impact delays, the alignment falters during phase 2. This difference may occur because Ali did not assess additional epidemic waves, which complicates direct NPI comparisons (1). Park agreed with Ali, while also offering a mathematical proof for the relationship between epidemic growth rates, calculated from incidence data, and forward-looking SIs (2,9). Park showed that as growth rates increase, forward SIs lengthen, and that when incidence decreases (either over time or because of external factors) forward SIs shorten (2). This better describes Montana’s incidence and our subperiod estimates, with NPIs providing context (Figure 2). Increased incidence and longer SIs during pre-SIP and phase 2 (June) stem from nonexistent and relaxed NPIs, whereas decreased incidence and shorter SIs during SIP and phase 2 (July) likely result from stricter NPIs and increased compliance with public health recommendations (e.g., mask wearing and social distancing). Additional data describing social compliance would benefit this interpretation.
The first limitation of this study is that the proportion of cases with identifiable transmission chains was lower during July than in previous periods. Despite this limitation, we felt it was necessary to report an SI for a period experiencing sizable incidence fluctuations. In addition, whereas others have reported negative-valued SIs among 1.2%–14.46% of infector–infectee pairs (10–14), we failed to identify any within our data. This difference could be caused by multiple factors, including incorrectly reported symptom onset dates, misidentified transmission direction between pairs, or both. However, the absence of negative SIs was not unique to our study; 14 of 24 published SI estimates did not include negative-valued pairs (Table 1).
Furthermore, to include pairs with a zero-valued SI, our study required changing their upper range. A sensitivity analysis of the adjustment showed minimal impact to the resulting estimate, whereas another sensitivity analysis, examining zero-valued pairs’ exclusion, returned a substantially elevated estimate. These analyses indicate that nontraditional SIs play key roles in generation time, SI, and R0 studies, especially for SARS-CoV-2, and that inclusive methods should be used when possible.
Our study offers evidence that rural-based SARS-CoV-2 SI estimates are consistent with those describing transmission occurring in urban settings. Furthermore, temporal variations in incidence, which can be caused by NPIs, must be considered when assessing SI distributions and other transmission measures. More period-based analyses of varying NPIs and their effects on transmission dynamics would help corroborate these findings.
Additional references for study of SARS-CoV-2 serial infection variation, Montana.
Acknowledgments
We thank Montana’s local county and tribal health jurisdictions for their dedication and commitment in responding to the COVID-19 pandemic and for collecting the data used in this study. We also thank the Montana Department of Public Health and Human Services, Communicable Disease Epidemiology Section, for allowing us access to the state’s COVID-19 case investigation data; the authors of the serial interval publications referenced in this paper who kindly responded to our inquiries concerning their studies; as well as Todd Harwell, Curtis Noonan, Erin Semmens, Jeffrey Shaman, Scott Whittenburg, Laura Williamson, and the anonymous reviewers for offering feedback on the manuscript.
This research was supported by the National Institute of General Medical Sciences of the National Institutes of Health (NIH), United States (award no. P20GM130418) and Montana state special revenue funds.
Biography
Mr. Reed is an epidemiologist with the Office of Epidemiology and Scientific Support at the Montana Department of Public Health and Human Services, where he manages the state’s hospital discharge data and public health data visualization systems. His primary research interests include infectious disease modeling, vectorborne disease, and global health.
Footnotes
Suggested citation for this article: Reed IG, Walker ES, Landguth EL. Severe acute respiratory syndrome coronavirus 2 serial interval variation, Montana, USA, March 1–July 31, 2020. Emerg Infect Dis. 2021 May [date cited]. https://doi.org/10.3201/eid2705.204663
References
- 1.Ali ST, Wang L, Lau EHY, Xu XK, Du Z, Wu Y, et al. Serial interval of SARS-CoV-2 was shortened over time by nonpharmaceutical interventions. Science. 2020;369:1106–9. 10.1126/science.abc9004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Park SW, Sun K, Champredon D, Li M, Bolker BM, Earn DJD, et al. Forward-looking serial intervals correctly link epidemic growth to reproduction numbers. Proc Natl Acad Sci U S A. 2021;118:e2011548118. 10.1073/pnas.2011548118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.R Core Team. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2019. [cited 2021 Feb 11]. https://www.R-project.org
- 4.Cori A. EpiEstim: estimate time varying reproduction numbers from epidemic curves. R package version 2.2–1; 2019. [cited 2021 Feb 11]. https://CRAN.R-project.org/package=EpiEstim.
- 5.Boelle P, Obadia T. R0: estimation of R0 and real-time reproduction number from epidemics. R package version 1.2–6; 2015. [cited 2021 Feb 11]. https://CRAN.R-project.org/package=R0.
- 6.Reich NG, Lessler J, Cummings DAT, Brookmeyer R. Estimating incubation period distributions with coarse data. Stat Med. 2009;28:2769–84. 10.1002/sim.3659 [DOI] [PubMed] [Google Scholar]
- 7.Thompson RN, Stockwin JE, van Gaalen RD, Polonsky JA, Kamvar ZN, Demarsh PA, et al. Improved inference of time-varying reproduction numbers during infectious disease outbreaks. Epidemics. 2019;29:100356. 10.1016/j.epidem.2019.100356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Griffin J, Casey M, Collins Á, Hunt K, McEvoy D, Byrne A, et al. Rapid review of available evidence on the serial interval and generation time of COVID-19. BMJ Open. 2020;10:e040263. 10.1136/bmjopen-2020-040263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ma J, Dushoff J, Bolker BM, Earn DJD. Estimating initial epidemic growth rates. Bull Math Biol. 2014;76:245–60. 10.1007/s11538-013-9918-2 [DOI] [PubMed] [Google Scholar]
- 10.Du Z, Xu X, Wu Y, Wang L, Cowling BJ, Meyers LA. Serial interval of COVID-19 among publicly reported confirmed cases. Emerg Infect Dis. 2020;26:1341–3. 10.3201/eid2606.200357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.He X, Lau EHY, Wu P, Deng X, Wang J, Hao X, et al. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat Med. 2020;26:672–5. 10.1038/s41591-020-0869-5 [DOI] [PubMed] [Google Scholar]
- 12.Mettler SK, Kim J, Maathuis MH. Diagnostic serial interval as a novel indicator for contact tracing effectiveness exemplified with the SARS-CoV-2/COVID-19 outbreak in South Korea. Int J Infect Dis. 2020;99:346–51. 10.1016/j.ijid.2020.07.068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Prete CA, Buss L, Dighe A, Porto VB, da Silva Candido D, Ghilardi F, et al. Serial interval distribution of SARS-CoV-2 infection in Brazil. J Travel Med. 2021;28:taaa115. 10.1093/jtm/taaa115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Talmoudi K, Safer M, Letaief H, Hchaichi A, Harizi C, Dhaouadi S, et al. Estimating transmission dynamics and serial interval of the first wave of COVID-19 infections under different control measures: a statistical analysis in Tunisia from February 29 to May 5, 2020. BMC Infect Dis. 2020;20:914. 10.1186/s12879-020-05577-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional references for study of SARS-CoV-2 serial infection variation, Montana.


