Abstract
Background
Pandemics greatly interfere with overall health care delivery as resources are diverted to combat the crisis. Kidney transplantation programs were closed temporarily during the COVID-19 pandemic. Given the critical shortage of organs, their short shelf life, and their overall importance to improving length and quality of life for those with kidney disease, this analysis examines the impact of discarding deceased donor organs.
Methods
The net benefit (or harm) of discarding deceased donor organs was measured in projected life years from a societal and individual perspective using a Markov model. A wide range of infection rates, pandemic durations, and case fatality rates associated with infection in wait listed and transplant recipients were examined.
Results
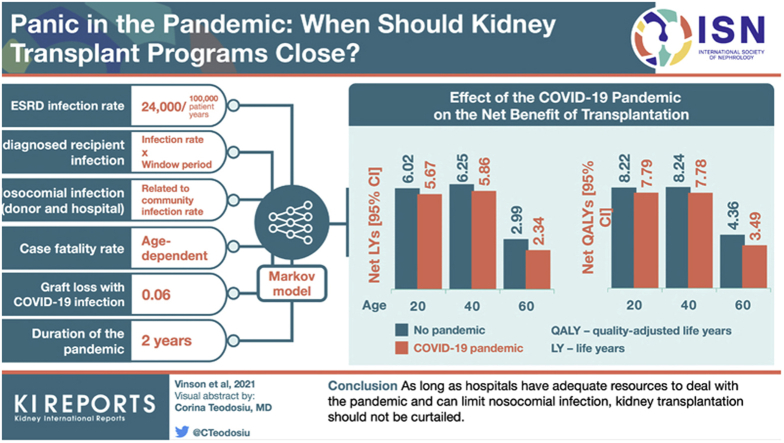
Overall, patient life expectancy fell for both wait listed and transplant recipients as the pandemic conditions became more unfavorable. However, the overall net benefit of a transplant during the pandemic was preserved. For example, prior to the pandemic, the net benefit of a kidney transplant over dialysis was calculated to be 6.25 life years (LYs) or 8.24 quality-adjusted life years (QALYs) in a 40-year old recipient. This fell to 5.86 LYs (7.78 QALYs) during the pandemic. Even assuming plausible but higher relative case fatality rates and risks of nosocomial and donor transmission in transplant recipients compared to wait listed patients, the net benefit remained >4 years for most deceased donor organs.
Conclusion
As long as hospitals have adequate resources to deal with the pandemic and can limit nosocomial infection, kidney transplantation should not be curtailed.
Keywords: COVID-19, deceased donor, decision analysis, kidney, transplantation
Graphical abstract
Early in the COVID-19 pandemic, there was a marked reduction in organ transplantation, and recent surveys suggest that many programs have rates that remain below historic rates of transplantation.1, 2, 3, 4 The lack of hospital resources that differed by region and time, uncertainty in the rate and mortality of infection in patients with end-stage kidney disease (ESKD) (case fatality rate), the initial difficulty identifying those with infection (lack of testing), the lack of antiviral therapy, and the potential need to reduce immunosuppression in transplant recipients who were infected, all were very understandable reasons to close programs.
During program closure/slow-down, many potential years of life were lost in patients with end organ disease from the lack of transplantation. Given the current state of knowledge, experts and transplant societies have made reasonable recommendations of what donors should and should not be considered for retrieval and the testing required of donors and recipients.5, 6, 7, 8 Decisions about closing transplant programs remain vague. Transplantation is not recommended if there is widespread community transmission or there is a lack of hospital resources.6 Importantly, there will always be the possibility of pandemics, and learning how to deal with transplantation during this pandemic might inform us how best to manage transplantation in future events.
Recently, a medical decision analysis was published that looked at who should accept a kidney transplant during the pandemic and who should defer (from the perspective of the patient).9 As expected, longer delays until subsequent transplant, high case fatality rates in transplant patients relative to wait list patients, recipient age, and overall infection rates in the community had a significant impact on the decision. The overall outlook was only 5 years, but long enough to surmise which option was the best. Importantly, this study did not examine the lost benefit (both in terms of life years and quality-adjusted life years) of discarded organs from a societal perspective if kidney transplant programs close during a pandemic.
The primary purpose of this study was to estimate potential life years lost from discarding a deceased donor kidney organ during the COVID-19 pandemic. Because transplant recipients and those on the wait list are at risk for COVID-19, measuring the net benefit of transplantation requires consideration of the consequences of this infection both before and after transplantation. The outcomes are likely to vary based on infection rate and associated risk of death, especially in older cohorts, in which fatality rates are quite high. Information from this analysis might help to inform programs on when to suspend activity in response to a pandemic, and whether certain populations should defer kidney transplantation to a later time.
Materials and Methods
This medical decision analysis examines the option of transplanting an available deceased donor kidney organ during a pandemic, or discarding it to avoid the increased pandemic-attributable risk. As a comparison, the study also examines the option of transplanting an available organ or discarding it in the absence of a pandemic. In a further analysis, we examined the impact of waiting for the pandemic to subside before accepting a transplant compared to accepting a transplant during the pandemic.
Model Assumptions
Rate of Infection
Rate of Infection
Daily infection rates in the United States (population 331 million) have varied widely between 20,000 and 200,000 new cases per day, and this has also varied by region.10 As a baseline, we examined the equivalent of 108,000 cases per day (about 12,000 cases per 100,000 population per year) but also examined higher rates that would be equivalent to about 325,000 cases per day (36,000 cases per 100,000 person years) in the United States.
Sources of Infection
Donor and Recipient
The overall rate of infection transmission from a donor organ is related to the incidence rate in the population, the test characteristics, and the duration of time that a patient is infected before testing positive (window period). The model assumes that potential donors are screened, that those with pre-hospital exposure or symptoms of lung inflammation are to be excluded, and that nucleic acid amplification testing (NAT) for COVID-19 in nasopharyngeal secretions is performed and negative. Viral infection in the blood has been detected in 15% of cases, and therefore transmission through the organ is possible, although to date this has not been reported.5,11,12 Despite testing by NAT, the false-negative rate is high.13,14 The tests are often negative up to 3 days after exposure, but the rate of false-negative results can also be high thereafter.13 As a very conservative estimate, we accommodated a large window period (from 7 to 21 days) along with a high false-negative NAT rate (38% with range of 18%−65%) in our determination.14 We also considered that 15% of cases would have viremia (an expected overestimate) and transmit infection. The model also included the possibility that the potential recipient had an asymptomatic infection on admission and was tested to be false negative. With immunosuppression, we assumed that the infection would become symptomatic and would be associated with increased mortality. Because infection rates might be higher in ESKD patients, this increase in risk was taken into account.15, 16, 17
In-Hospital Nosocomial
Spread from medical personnel or undiagnosed patients is a potential source of infection. This is also likely related to the general population incidence as well as hospital resources and practice. Although nosocomial infection to patients has been reported to be high, patient transmission with current infection control policies is likely to be much lower.18,19 We assumed a 4-fold higher rate of exposure than the baseline general population during hospital and early post-transplantation follow-up period (Table 1).
Table 1.
Probabilities in model
| Variable | Baseline | Range |
|---|---|---|
| Incidence rate in general population (per 100,000 patient years) | 12,000 | 6,000–36,000 |
| Dialysis/wait list patients | 2-fold | (2–4)a |
| Transplant recipients | 2-fold | (2–4) |
| Nosocomial risk (duration of exposure in hospital 21 days with a range of 14–30 days) | 4-fold | (2–10) |
| Case fatality rate - (age dependent [see text]) | ||
| Nosocomial acquired infection | Age dependent | (0.05–0.55) |
| Community acquired infection | Age dependent | (0.03–0.35) |
| In sensitivity analysis ∗ community acquired infection | 1-fold | (1–2) |
| Graft loss with COVID 19 infection | 0.06 | (0.03–0.10) |
| False negative (%) | 38 | (18–65) |
| Window period (days) | 14 | (7–21) |
| Duration of pandemic | 2 years | (1–3) |
A 4-fold increase with upper general population infection incident rate would result in >100% population. Rates were capped at 72,000 per 100,000 patient years.
Community
Infection rates have been reported to be higher among in-center dialysis patients, but the magnitude of this increase is unclear.13, 14, 15, 16,20 Whether the incidence of infection in transplant recipients is higher than the general population is also unknown. There is uncertainty as to whether the infection risk is higher in wait listed patients than in transplant recipients. As a baseline, a 2-fold higher rate was modeled in the ESKD population (24,000 cases per 100,000 ESKD population per year). Importantly, a very large range of infection rates were considered, such that if relative rates in the ESKD population were underestimated, the higher community rates would compensate. Differential infection rate increases were also explored such that wait list infection rate was greater than the transplant infection rate and the transplant rate was greater than the wait list rate.
Outcome of Infection
Case fatality rates have been reported in ESKD and CKD nondialysis patients.21, 22, 23, 24, 25, 26, 27 In one large European study, case fatality rates were approximately 3% in those aged <49 years, 10% in those aged 50 to 59 years, 25% in those aged 60 to 69 years, and 35% in those aged 70 to 79 years.21 The absolute case fatality rate for dialysis and transplant patients is nearly the same (28-day death rate about 20%). Although the absolute rates are the same in both populations, the adjusted relative risk is higher in transplant recipients (lower baseline mortality rate), likely reflecting the impact of immunosuppression. Wait listed patients are also at increased risk for death from COVID-19.22, 23, 24
Case fatality rates in patients who have recently undergone transplantation are not known, in part because many programs were suspended early on. One could assume that the risk of death is higher during the period of intense immunosuppression. The model assumed that the case fatality rate during the period of intense immunosuppression would be approximately 1.5-fold higher (5% in those < aged 49, 15% in those aged 50 to 59, 40% in those aged 60 to 69, and 55% in those aged 70 to 79 years).
Transplant recipients with acute infection are recommended to have their immunosuppression reduced.5, 6, 7 In addition, there is an increased risk of acute kidney injury with severe infection. Both could contribute to graft loss in recipients. The evidence for permanent graft loss varies considerably and may be very high if only hospitalized patients are considered but less when non-hospitalized patients are included in the denominator. We examined a range (3%−10%) of acute graft loss rates for patients developing a symptomatic COVID-19 infection.25, 26, 27
As a baseline, we assumed that the pandemic lasted 2 years but examined a 1- to 3-year duration.
The net benefit was measured in projected life years using a Markov model. To measure the net benefit of an organ, we assumed that the reference population would be wait listed patients using the rationale described by Schnitzler et al.28 As with their analysis, the time horizon was limited to 40 years, a time when most organs would have failed. The expected remaining life years (area under the survival curve) on the wait list was subtracted from the expected remaining life years with a transplant, including the possibility of graft loss and return to dialysis.
Annual mortality rates per 1000 patient years for dialysis patients on the wait list, death with a functioning transplant, and return to dialysis from a failed transplant were taken from the United States Renal Data System (USRDS) (unadjusted tables).29 Tables provide information on age groups (18−21, 22−24, 25−29, 30−34 years, etc.). Because annual rates of graft loss are not available, rates were calculated from 5-year survival tables (return to dialysis or re-transplantation) for deceased donor transplants for each of the age groups. Rates were converted to transition probabilities. The model assumed only 1 transplant, as the objective was to examine the net benefit of using or discarding a kidney organ during a pandemic. In the determination of survival on the wait list, we did not take into account that patients may be taken off the wait list from illness, as these patients would not be eligible for transplantation. In addition, we did not take into account that some patients with a failed transplant would be relisted and undergo retransplantation. Several other studies that have calculated net benefit have taken these events (wait list withdrawal and censoring at retransplantation) into account, and no method is perfect.28,30, 31, 32, 33 Our net benefit is a more conservative measure, since wait list survival is slightly overestimated and a patient’s posttransplantation graft failure (return to dialysis) survival is underestimated. In the deferred analysis, patients were assumed to be relisted for transplantation after the first year. The model examined different transplant rates (30, range 20−50 transplants per 100 patient wait-years) based on expected differences in Donor Service Areas and the likelihood that patients would be “near the top” of the list.34 The utility health state scaling factors assigned were as follows: dialysis = 0.8, functioning transplant = 1.0, and death = 0, based on previous health outcome studies.35 No utility adjustments were made for time spent in the hospital.
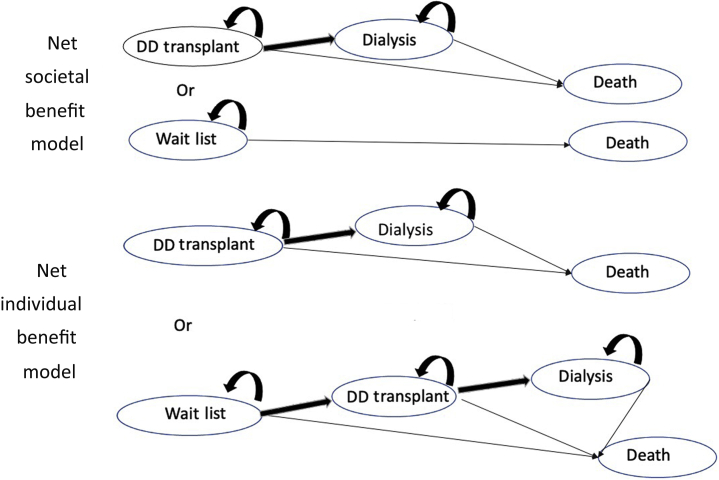
The Markov model was developed in TreeAge Pro Healthcare Version 2019 R2.1 (Tree Age Software, LLC, Williamstown, MA) and used for this medical decision analysis. The decision trees are shown in Figure 1. Patients were considered to be their own controls such that the characteristics of a patient accepting an immediate transplant was identical to the one deferring or remaining on the transplant list. Uncertainty in the conclusions was examined by 1-way sensitivity analysis for all model variables. Two- and 3-way sensitivity analyses for key variables (age, incidence of infection, case fatality rate, relative infection rates) were examined. Confidence intervals (95% CIs) for estimates of the net benefit for a transplanted organ were determined by probabilistic sensitivity analysis using Monte Carlo simulations of 100 trials. Distributions for age-related mortality rates, graft loss (donor quality as measured by the Kidney Donor Profile Index [KDPI]), and kidney transplant rates (deferred transplantation) were used in the simulation studies. The range of KDPI relative risks were calibrated to expected 5-year graft survival for deceased donor transplants.36 Differences in net benefit by sex, diabetes mellitus status, and race (Black/White) were explored using relative risks taken from the literature.9,29 Research ethics approval was waived by our institution’s research ethics board (the Nova Scotia Health Research Ethics Board).
Figure 1.
Decision trees. With COVID-19 infection, mortality rates for health states will increase accordingly. DD, deceased donor.
Results
Table 2 shows the projected life years in adults aged 25 to 70 years at the time of deceased donor kidney transplantation during and not during a pandemic. At a baseline rate of 12,000 infections per 100,000 patient years in the general population (with 2-fold higher rates in the transplant and wait listed/dialysis cohorts), receiving a deceased donor kidney transplant during a pandemic provided only modestly fewer LYs and QALYs than receiving one prior to a pandemic. For example, the net benefit of a deceased donor transplant was 5.86 compared to 6.25 LYs and 7.78 compared to 8.24 QALYs in a 40-year old patient, during and prior to the pandemic, respectively. Absolute net benefit during and prior to a pandemic were much lower in older patient groups.
Table 2.
Life years by options: baseline variables
| Age (yr) | Transplant COVID-19 LYs (QALYs) |
Transplant No COVID-19 LYs (QALYs) |
Wait List (WL) COVID-19 LYs (QALYs) |
Wait List No COVID-19 LYs (QALYs) |
Difference COVID-19 (Transplant WL) No COVID-19 (Transplant WL) LYs [95% CI] (QALYs [95% CI]) |
|---|---|---|---|---|---|
| 25 | 24.14 (22.57) |
24.84 (23.28) |
18.47 (14.78) |
18.82 (15.06) |
5.67 [5.44–5.90] 6.02 [5.78–6.26] (7.79 [7.44–8.14]) (8.22 [7.86–8.58]) |
| 40 | 19.35 (18.57) |
20.05 (19.28) |
13.49 (10.79) |
13.80 (11.04) |
5.86 [5.66–6.06] 6.25 [6.03–6.47] (7.78 [7.56–8.00]) (8.24 [8.01–8.47]) |
| 50 | 14.24 (13.80) |
15.54 (15.09) |
9.93 (7.95) |
10.67 (8.53) |
4.31 [4.20–4.43] 4.87 [4.75–4.89] (5.85 [5.69–6.01]) (6.56 [6.39–6.73]) |
| 60 | 9.27 (9.03) |
11.14 (10.88) |
6.93 (5.54) |
8.15 (6.52) |
2.34 [2.17–2.41] 2.99 [2.91–3.07] (3.49 [3.43–3.55]) (4.36 [4.26–4.46]) |
| 70 | 5.88 (5.75) |
7.53 (7.37) |
5.10 (4.08) |
6.36 (5.09) |
0.78 [0.72–0.82] 1.17 [1.12–1.22] (1.67 [1.62–1.72]) (2.28 [2.22–2.34]) |
LYs, life years; QALY, quality-adjusted life years; WL, wait list.
Table 3 shows the impact of higher and lower general population infection rates. The model predicts significant reductions in remaining life years from this pandemic for all age groups as the infection rate increases. The largest proportional reduction in net benefit is in the older age cohorts. However, in younger cohorts, 6-fold increases in infection rates were associated with a < 20% fall in the net benefit of a transplant.
Table 3.
Effect of COVID-19 general population incidence rate on transplant outcome and net benefit (over wait list) during a pandemic
| Age (yr) | 36,000 cases per 100,000 patient years LYs [95% CI] (QALYs [95% CI]) |
12,000 cases per 100,000 patient years LYs [95% CI] (QALYs [95% CI]) |
6,000 cases per 100,000 patient years LYs [95% CI] (QALYs [95% CI]) |
No COVID-19 LYs [95% CI] (QALY- [95% CI]) |
|---|---|---|---|---|
| 25 | ||||
| Transplant | ||||
| LYs | 23.17 | 24.14 | 24.46 | 24.84 |
| QALYs | 21.59 | 22.57 | 22.90 | 23.28 |
| Net benefit | ||||
| LYs | 5.18 [4.93–5.43] | 5.67 [5.44–5.90] | 5.83 [5.53–6.13] | 6.02 [5.78–6.26] |
| QALYs | 7.20 [6.91–7.49] | 7.79 [7.44–8.14] | 7.99 [6.69–8.29] | 8.22 [7.86–8.58] |
| 40 | ||||
| Transplant | ||||
| LYs | 18.39 | 19.35 | 19.68 | 20.05 |
| QALYs | 17.59 | 18.57 | 18.90 | 19.28 |
| Net benefit | ||||
| LYs | 5.33 [5.13–5.53] | 5.86 [5.66–6.06] | 6.05 [5.85–6.25] | 6.25 [6.03–6.47] |
| QALYs | 7.14 [6.94–7.34] | 7.78 [7.56–8.00] | 7.99 [7.76–8.22] | 8.24 [8.01–8.47] |
| 50 | ||||
| Transplant | ||||
| LYs | 12.55 | 14.24 | 14.84 | 15.54 |
| QALYs | 12.12 | 13.80 | 14.40 | 15.09 |
| Net benefit | ||||
| LYs | 3.58 [3.47–3.69] | 4.31 [4.20–4.43] | 4.57 [4.44–4.70] | 4.87 [4.75–4.89] |
| QALYs | 4.95 [4.83–5.07] | 5.85 [5.69–6.01] | 6.18 [6.02–6.34] | 6.56 [6.39–6.73] |
| 60 | ||||
| Transplant | ||||
| LYs | 7.01 | 7.39 | 10.12 | 11.14 |
| QALYs | 6.80 | 9.03 | 9.87 | 10.88 |
| Net benefit | ||||
| LYs | 1.58 [1.53–1.63] | 2.34 [2.17–2.41] | 2.63 [2.55–2.71] | 2.99 [2.91–3.07] |
| QALYs | 2.45 [2.39–2.51] | 3.49 [3.43–3.55] | 3.88 [3.78–3.98] | 4.36 [4.26–4.46] |
| 70 | ||||
| Transplant | ||||
| LYs | 4.01 | 5.88 | 6.62 | 7.53 |
| QALYs | 3.90 | 5.75 | 6.48 | 7.37 |
| Net benefit | ||||
| LYs | 0.38 [0.27–0.40] | 0.78 [0.72–0.82] | 0.95 [0.91–0.99] | 1.17 [1.12–1.22] |
| QALYs | 1.0 [0.96–1.04] | 1.67 [1.62–1.72] | 1.95 [1.89–2.01] | 2.28 [2.22–2.34] |
End-stage kidney disease (ESKD) infection rate was 2-fold higher than in the general population. Therefore, if infection rate is 36,000 per 100,000 patient years in community, then 72,000 cases per 100,000 ESKD patient years. LYs, life years; QALY, quality-adjusted life years; WL, wait list.
Table 4 shows the impact of different relative infection rates and case fatality rates between wait listed and transplant recipients. Even under circumstances in which both the infection rate and case fatality rate were higher in transplant recipients relative to wait list/dialysis patients, most transplants provided a net benefit, the exception being among the older recipients, where there was no net benefit and potentially was harm (fewer LYs). However, when adjusted for quality of life, even this older cohort benefited.
Table 4.
Higher/lower relative risk of infection, higher/baseline case fatality rate (CFR) in wait listed/transplant recipients on net benefit of transplant during a pandemic (over wait list)
| Age (yr) | Risk of infection with a transplant is equal to on the wait list [95% CI] |
Risk of infection 2-fold higher in transplant than wait list |
Risk of infection 2-fold higher in wait list than transplant |
Risk of Infection 1.5-fold higher CFR 1.5-fold higher in transplant that wait list |
Risk of infection 1.5-fold higher CFR 1.5-fold higher in wait list than transplant |
Risk of Infection 1.5-fold higher in wait list CFR 1.5-fold higher in transplant |
60,000 cases per 100,00 patient years in wait list; 45,000 cases per 100,000 patient years in transplant; CFR 2-fold higher in transplant than wait list |
|---|---|---|---|---|---|---|---|
| 25 | |||||||
| Transplant | |||||||
| LYs | 24.14 | 23.65 | 24.12 | 23.65 | 24.14 | 23.91 | 22.93 |
| QALYs | 22.57 | 22.04 | 22.56 | 22.00 | 22.57 | 22.35 | 21.40 |
| net benefit | |||||||
| LYs | 5.67 [5.44–5.90] | 5.18 [4.91–5.45] | 5.70 [5.45–5.95] | 5.18 [4.88–5.48] | 6.03 [5.73–6.33] | 5.58 [5.28–5.88] | 4.84 [4.55–5.13] |
| QALYs | 7.79 [7.44–8.14] | 7.26 [6.90–7.62] | 8.00 [7.60–8.40] | 7.22 [6.90–7.54] | 8.09 [7.74–8.44] | 7.69 [7.33–8.05] | 6.93 [6.59–7.27] |
| 40 | |||||||
| Transplant | |||||||
| LYs | 19.35 | 18.83 | 19.34 | 18.77 | 19.35 | 19.13 | 18.16 |
| QALYs | 18.57 | 18.04 | 18.56 | 17.99 | 18.57 | 18.35 | 17.39 |
| net benefit | |||||||
| LYs | 5.86 [5.66–6.06] | 5.34 [5.15–5.53] | 6.09 [5.89–6.29] | 5.28 [5.28–5.68] | 6.19 [5.98–6.40] | 5.77 [5.56–5.98] | 5.01 [4.79–5.23] |
| QALYs | 7.78 [7.56–8.00] | 7.25 [7.04–7.46] | 7.96 [7.73–8.19] | 7.20 [6.96–7.45] | 8.05 [7.81–8.29] | 7.67 [7.44–7.90] | 6.88 [6.67–7.09] |
| 50 | |||||||
| Transplant | |||||||
| LYs | 14.24 | 13.32 | 14.23 | 13.05 | 14.24 | 13.73 | 11.81 |
| QALYs | 13.80 | 12.88 | 13.79 | 12.63 | 13.80 | 13.31 | 11.42 |
| net benefit | |||||||
| LYs | 4.31 [4.20–4.43] | 3.39 [3.26–3.52] | 4.84 [4.72–4.96] | 3.12 [2.98–3.26] | 5.07 [4.97–5.21] | 4.09 [3.96–4.22] | 2.65 [2.54–2.76] |
| QALYs | 5.85 [5.69–6.01] | 4.93 [4.78–5.08] | 6.28 [6.13–6.43] | 4.68 [4.55–4.81] | 6.46 [5.31–6.61] | 5.59 [5.42–5.76] | 4.09 [3.97–4.22] |
| 60 | |||||||
| Transplant | |||||||
| LYs | 7.39 | 8.02 | 9.25 | 7.59 | 7.39 | 8.50 | 5.97 |
| QALYs | 9.03 | 7.80 | 9.01 | 7.38 | 9.03 | 8.28 | 5.800 |
| net benefit | |||||||
| LYs | 2.34 [2.17–2.41] | 1.09 [1.03–1.15] | 3.19 [3.07–3.26] | 0.66 [0.61–0.71] | 3.53 [3.43–3.63] | 2.05 [1.99–2.11] | 0.25 [0.19–0.31] |
| QALYs | 3.49 [3.43–3.55] | 2.25 [2.19–2.31] | 4.16 [4.09–4.23] | 1.84 [1.77–1.91] | 4.44 [4.33–4.55] | 3.12 [3.04–3.20] | 1.22 [1.17–1.27] |
| 70 | |||||||
| Transplant | |||||||
| LYs | 5.88 | 4.84 | 5.87 | 5.10 | 5.88 | 5.22 | 3.18 |
| QALYs | 5.75 | 4.72 | 5.74 | 4.36 | 5.75 | 5.10 | 3.10 |
| net benefit | |||||||
| LYs | 0.78 [0.72–0.82] | –0.26 [–0.29 to –0.23] | 1.63 [1.58–1.68] | –0.63 [0.66 to –0.60] | 1.96 [1.86–2.06] | 0.60 0.56–0.64] | –0.73 [–0.76 to –0.70] |
| QALYs | 1.67 [1.62–1.72] | 0.64 [0.61–0.67] | 2.35 [2.30–2.40] | 0.28 [0.26–0.20] | 2.61 [2.51–2.71] | 1.40 [1.34–1.46] | –0.03 [–0.05 to –0.01] |
Community infection rate is 12,000 cases per 100,000 patient years; baseline end-stage kidney disease (ESKD) population infection rate is 24,000 cases per 100,000 ESKD patient years; 1.5-fold higher ESKD rate is 36,000 cases per 100,000 ESKD patient years; 2-fold higher in ESKD population is 48,000 cases per 100,000 ESKD patient years. Last column 20,000 cases in the general population, 60,000 cases in the wait list population, 50,000 cases in transplant patients all per 100,000 cohort patient years. LYs, life years; QALYs, quality-adjusted life years.
Scenarios in which there was no net benefit (LYs) of a transplant during the pandemic occurred in patients aged ≥70 years, with a sustained infection rate of > 40,000 cases per 100,000 transplanted patient years (compared to 24,000 infections per 100,000 wait list patient years), or a case fatality rate of 1.5-fold higher compared to wait list patients along with a sustained infection of > 30,000 infections per 100,000 transplant patient years (compared to 24,000 in wait list patients). No net benefit (LYs) was also seen in patients aged 60 to 70 years, with a sustained infection rate of > 2-fold higher in transplant compared to wait list patients and a case fatality rate that was > 1.8-fold higher in transplant compared to wait list patients. When considering the net benefit in terms of QALYs rather than LYs, some of these scenarios were associated with net benefit (Table 4).
In further sensitivity analyses, at a baseline infection rate of 12,000 per 100,000 patient years in the general population (2-fold higher in the ESKD population), other factors in the analysis that affected the net benefit of a deceased donor transplant were less important. If acute graft loss with COVID-19 infection increased from 6% to 10%, there was fall in the net benefit during a pandemic from 5.86 LYs (7.78 QALYs) to 5.71 LYs (7.61 QALYs). If the duration of the pandemic lengthened from 2 to 3 years, there was a reduction in the net benefit to 5.71 LYs (7.59 QALYs). Over the range of probabilities for a nosocomial infection in-hospital, false-negative COVID-19 test rates (18%−65%) and window period (7−21 days) of infection, the net benefit for a 40-year-old to undergo transplantation changed by < 0.05 LYs.
In the overall analysis, the net benefit for a deceased donor transplant in a 40-year-old patient was reduced by 0.39 LYs (6.25−5.86 LYs) or 0.46 QALYs (8.24−7.78 QALYs) during the pandemic. The reduction was 0.40 LYs (0.46 QALYs) for male and 0.34 LYs (0.40 QALYs) for female patients. The reduction for patients with diabetes mellitus was 0.49 LYs (0.57 QALYs) and for patients without diabetes mellitus was 0.38 LYs (0.44 LYs). The reduction for Black patients was 0.38 LYs (0.48 QALYs) and for White patients was 0.45 LYs (0.54 QALYs).
In our prespecified analysis from a patient perspective, a patient was put back on the transplant list after 1 year (pandemic ongoing) with a transplant rate of 30 transplants per 100 patient wait years. Most patients benefit from immediate rather than deferred transplantation (Table 5). However, if the relative rate of infection or the case fatality rate is higher in transplant recipients, then the younger and older cohorts fare less well, and some may be harmed. Longer delays in relisting and lower subsequent transplantation rates improve the benefits of continuing with transplantation during a pandemic (data not shown).
Table 5.
Net benefit of immediate transplant during a pandemic compared to deferred transplant outside of a pandemic: relatively higher infection rates and case fatality rates (CFR)
| Age (yr) | Baseline [95% CI] |
1.5-fold higher CFR in transplant recipients relative to dialysis/wait list | 1.5 Higher infection rate in transplant relative to dialysis/wait list | 1.5 Higher infection rate in wait list; 1.5-fold higher CFR in transplant |
1.5 Higher infection rate in wait list; CFR 2-fold higher in transplant than wait list |
|---|---|---|---|---|---|
| 25 | |||||
| LYs | 0.48 [0.12–0.84] | 0.26 [–0.13 to 0.60] | 0.21 [–0.13 to 0.55] | 0.42 [0.08–0.76] | 0.20 [–0.13 to 053] |
| QALYs | 0.63 [0.23–1.03] | 0.42 [0.06–0.78] | 0.35 [–0.03 to 0.73] | 0.57 [0.17–0.97] | 0.36 [–0.04 to 0.76] |
| 40 | |||||
| LYs | 1.32 [1.06–1.58] | 1.10 [0.85–1.35] | 1.04 [0.79–1.29] | 1.25 [0.99–1.51] | 1.03 [0.76–1.20] |
| QALYs | 1.63 [1.35–1.91] | 1.42 [1.12–1.72] | 1.35 [1.05–1.65] | 1.56 [1.27–1.85] | 1.35 [1.06–1.64] |
| 50 | |||||
| LYs | 1.32 [1.17–1.47] | 0.81 [0.66–0.96] | 0.83 [0.68–0.98] | 1.15 [0.98–1.32] | 0.66 [0.51–0.81] |
| QALYs | 1.67 [1.50–1.84] | 1.18 [1.00–1.36] | 1.18 [1.00–1.36] | 1.50 [1.32–1.68] | 1.02 [0.84–1.20] |
| 60 | |||||
| LYs | 0.90 [0.81–0.98] | 0.15 [0.07–0.23] | 0.23 [0.16–0.30] | 0.65 [0.57–0.73] | –0.06 [–0.16 to 0.04] |
| QALYs | 1.23 [1.13–1.33] | 0.28 [0.18–0.38] | 0.56 [0.46–0.66] | 0.96 [0.86–1.06] | 0.27 [0.16–0.38] |
| 70 | |||||
| LYs | 0.32 [0.26–0.38] | –0.33 [–0.39 to –0.27] | –0.25 [–0.29 to –0.21] | 0.13 [0.08–0.18] | –0.47 [–0.53 to –0.41] |
| QALYs | 0.62 [0.57–0.67] | –0.01 [–0.07 to 0.05] | 0.06 [0.01–0.11] | 0.41 [0.32–0.50] | –0.18 [–0.24 to –0.12] |
Based on 12,000 cases per 100,000 patient years in general population; 24,000 cases per 100,000 (end-stage kidney disease) ESKD patient years; 1.5-fold higher in wait list patients is equal to 36,000 case per 100,000 per wait list patient years. LYs, life years; QALYs, quality adjusted life years.
Discussion
The study shows that discarding kidneys during a pandemic may result in a significant lost opportunity from a societal perspective. Although the pandemic is associated with a significant reduction in ESKD life expectancy, much of the net benefit of a transplant is preserved. At this point, how many potential transplant organs were not transplanted is unclear, but this information will become available with time as the transplant data are collected. Assuming that kidney programs remained closed to deceased donor transplantation over the last 9 months because of the pandemic, there could be 10,000 fewer deceased donor transplants, and this could mean as many as 50,000 lost LYs. The impact of potential life years lost is not easily captured when transplants are not performed. However, patients remaining on dialysis with higher mortality rates will contribute to the COVID-19−related excess mortality in the general population, even though their deaths may not be from the infection. During the early period of considerable uncertainty with respect to the rate of infection, its impact on various populations, and the lack of hospital personal protection equipment, caution was prudent and justified. Now that there is some information, this study highlights the potential gains of maintaining kidney transplant programs open, even during periods of moderately high infection risk, assuming that hospitals are adequately resourced.
Some centers have advocated for continued transplantation in the pandemic, especially for those patients who have no dialysis option equivalent (e.g., liver failure) or those who are unlikely to receive a kidney (highly sensitized ESKD wait list patients). One could argue that less sensitized wait list patients should also undergo transplantation, assuming that they are fully informed and willing to undergo transplantation during the pandemic. The risk of infection-related death and morbidity will likely be the same in sensitized and unsensitized recipients (assuming similar intensities of induction); graft outcomes will be the same or better in the less sensitized recipients; highly sensitized individuals are already prioritized by the allocation algorithms; and denying the unsensitized patient the option of a transplant effectively converts that individual to one who is sensitized (forced to wait).
The model shows that there are scenarios in which the net benefit is small and even negative. The most at risk are the older recipients (age > 65 years), for whom the net benefit of a transplant is already considerably less compared to that in younger patients. To achieve a negative net benefit (harm) in LYs, older transplant recipients would need to have sustained infection rates and case fatality rates that were considerably higher than wait listed patients. Even in these circumstances, however, the net benefit as measured by QALYs (rather than LYs) was positive. Under most reasonable scenarios, the reduction in net benefit in younger patients is < 20% compared to transplantation during a non-pandemic period. Unfortunately, much of the information needed to provide a more precise estimate of net benefit in the pandemic is still not available or is changing.
The results of this study need to be considered in the context of the existing literature. Although the benefits to society can be large, the patient perspective is also of paramount importance. This approach was taken by a recently published study.9 Both this and our study used Markov models with similar decision trees and data sources; however, our model had a longer period of follow-up to capture the full effect of the transplant, considered quality of life, and included the additional risk of graft loss associated with COVID-19 infection in transplant recipients. Their study advantages included a calculator projecting benefit (or loss) based on individual characteristics and rate of subsequent transplantation if deferred to after the pandemic passed. Knowing when the pandemic will end is an important unknown when considering the patient perspective. Since the publication of that study, infection rates have increased. Despite the differences, both studies concluded that transplantation during the pandemic was superior to deferring, except if the case fatality rate was considerably higher in transplant recipients relative to the wait list and infection rates were high. Our study also uniquely considered higher infection rates in transplant recipients relative to the wait list. Under these scenarios, both studies showed that selected patients might benefit from a delay, but only if they subsequently underwent transplantation. Delay likely benefited younger patients because of lower wait list mortality (more likely to survive the pandemic). Immediate transplantation in the elderly patients was poorly tolerated during high infection rates and with relatively high case fatality rates. The bigger reality is that most programs have re-opened even while the spread of infection has increased.
In addition to exploring the effects of age, we also explored other potential confounders and subgroups. ESKD patients with diabetes mellitus have higher rates of death.29 Nonetheless, the reductions in net benefit during a pandemic are modest and in keeping with the overall analysis. Individuals of Black ethnicity are at higher risk for COVID-19 infection and may be at higher mortality risk independent of comorbidity.37,38 Wait list mortality is lower in Black than White individuals, but Black patients experience higher rates of graft loss.29 Regardless, the absolute reductions in net benefit from COVID-19 were similar compared to the general analysis. The net benefit of proceeding with kidney transplantation would be expected to be even greater in those populations with lower infection and case fatality rates, less virulent strains of SARS-CoV-2 (the virus responsible for COVID-19 disease), or in patients who are more tolerant of infection. Likewise, given the superior outcomes observed in patients undergoing preemptive kidney transplantation and the increased morbidity and mortality observed in patients with ESKD before and after transition to maintenance dialysis, preemptive transplantation would still be expected to be of net benefit during this and future pandemics. Finally, once vaccine uptake is ubiquitous among ESKD and transplant recipients, the net benefit with transplantation will approach that in a non-pandemic period.
There are limitations to this analysis. More information is forthcoming even at the time of this analysis, and assumptions may need to be adjusted accordingly. The incident rates of infection in any region vary over time (waves) and are not predictable.10 Higher incidence rates over shorter periods of time may be experienced; however, under these circumstances, hospital resources would be stretched and transplantation would not be possible. Some of the sustained infection rates included in the analysis would likely overwhelm hospital resources. The duration of the pandemic and the potential benefit of a vaccine, especially in our kidney disease population, is unknown. If the pandemic remains forever, then comparing it to a time of “no−COVID-19” is irrelevant. Importantly, waiting for a COVID-19−free period may results in thousands of lost years of life due to discarding kidney transplant organs.
We did not address live donor kidney transplantation, as the procedure can be considered elective. Deferring the procedure seems reasonable. However, waiting might expose the patient to higher infection risks if dialysis centers are superspreading areas, in addition to a lower quality of life and a higher mortality on dialysis, and the potential live donor may no longer be available at a later date. Conversely, the donor avoids the nosocomial risk of infection by delaying to a non-pandemic period.
The assumed period of time that an infection is undetectable in a donor and recipient is still unknown; however, we used a large window period and high false-negative test result assumption in the model.14 Our estimate is likely to be very conservative. It is not clear whether a kidney organ can transmit the infection from deceased donors that are appropriately screened and tested to be negative. The actual general population infection rate is likely underreported based on current antibody testing, and more recent increases in testing have detected asymptomatic infections.20 Case fatality rates have been dropping both from better care and from detection of cases that were not previously known (from antibody testing or NAT testing in asymptomatic people).39 Together these would suggest that the negative impact of the pandemic on transplantation outcomes in this analysis may be even less.
We did not consider other risk-mitigating strategies such as having only patients undergo transplantation who have resolved infection, have generated an antibody response and are now well, or increasing organ utilization by using viable organs from infected or high-risk donors to those with immunity. At this point, the strength of the immunity and its duration are unknown. Precise probabilities on mortality and graft loss with acute infection during intense immunosuppression, and nosocomial spread from health care providers or from patient to patient, are also unknown but were considered in this analysis.
Finally, we modeled the impact of only a single transplant organ. It is uncertain what the likelihood of re-transplantation and subsequent graft survival would be for an individual who underwent transplantation during the COVID-19 pandemic, became infected, and lost their transplant but survived. It is not clear whether this would differ from the situation of a similar patient who underwent transplantation before the COVID-19 pandemic and lost their transplant from a COVID-19−related illness.
In summary, the study shows that the net benefit of a kidney organ is not greatly reduced during this pandemic compared with a non-pandemic period. Therefore, this study supports kidney transplantation at this time, with very little reason to suspend activity unless other circumstances (hospital resources, nosocomial infection) are an issue. The lost benefit of a discarded deceased donor organ cannot be retrieved. Patients would need to be fully informed, although the magnitude of the risk is uncertain. Deferred transplantation to a COVID-19−free period may be a reasonable option for some, but the pandemic may persist for a longer than hoped and a prolonged delay may be harmful. Hopefully what we have learned from this experience is that certain aspects of health care delivery such as transplantation, cancer, and cardiovascular care must go on.
Disclosure
All the authors declared no competing interests.
Data Availablity
All probabilities and rates are published at the United States Renal Data System (USRDS). A copy of the decision tree is available on request from the corresponding author.
Footnotes
Modified STROBE Statement
Supplementary Material
Modified STROBE Statement
References
- 1.Loupy A., Aubert O., Reese P.P. Organ procurement and transplantation during the COVID-19 pandemic. Lancet. 2020;395:e95–e96. doi: 10.1016/S0140-6736(20)31040-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boyarsky B.J., Po-Yu Chiang T., Werbel W.A. Early impact of COVID-19 on transplant center practices and policies in the United States. Am J Transplant. 2020;20:1809–1818. doi: 10.1111/ajt.15915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lentine K.L., Vest L.S., Schnitzler M.A. Survey of US living kidney donation and transplantation practices in the COVID-19 era. Kidney Int Rep. 2020;5:1894–1905. doi: 10.1016/j.ekir.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cholankeril G., Podboy A., Alshuwaykh O. More early impact of COVID-19 on solid organ transplantation in the United States. Transplantation. 2020;104:2221–2224. doi: 10.1097/TP.0000000000003391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kumar D., Manuel O., Natori Y. COVID-19: a global transplant perspective on successfully navigating a pandemic. Am J Transplant. 2020;20:1773–1779. doi: 10.1111/ajt.15876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Guidance on Coronavirus Disease 2019 (COVID-19) for Transplant Clinicians. https://tts.org/23-tid/tid-news/657-tid-update-and-guidance-on-2019-novel-coronavirus-2019-ncov-for-transplant-id-clinicians Updated June 2020. Available at:
- 7.Ritschl P.V., Nevermann N., Wiering L. Solid organ transplantation programs facing lack of empiric evidence in the COVID-19 pandemic: a by-proxy Society Recommendation Consensus approach. Am J Transplant. 2020;20:1826–1836. doi: 10.1111/ajt.15933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Galvan N.T.N., Moreno N.F., Garza J.E. Donor and transplant candidate selection for solid organ transplantation during the COVID-19 pandemic [e-pub ahead of print]. Am J Transplant. https://doi.org/10.1111/ajt.16138 [DOI] [PMC free article] [PubMed]
- 9.Massie A.B., Boyarsky B.J., Werbel W.A. Identifying scenarios of benefit or harm from kidney transplantation during the COVID-19 pandemic: a stochastic simulation and machine learning study [e-pub ahead of print] Am J Transplant. 2020 doi: 10.1111/ajt.16117. Accessed October 17, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.https://www.worldometers.info/coronavirus/country/us/ Worldometer. Available at:
- 11.Su H., Yang M., Wan C. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020;98:219–227. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19-issues-related-to-solid-organ-transplantation COVID-19: Issues related to solid organ transplantation. Available at:
- 13.Woloshin S., Patel N., Kesselheim A.S. False negative tests for SARS-CoV-2 infection—challenges and implications. N Engl J Med. 2020;383:e38. doi: 10.1056/NEJMp2015897. [DOI] [PubMed] [Google Scholar]
- 14.Kucirka L.M., Lauer S.A., Laeyendecker O. Variation in false-negative rate of reverse transcriptase polymerase chain reaction-based SARS-CoV-2 tests by time since exposure. Ann Intern Med. 2020;173:262–267. doi: 10.7326/M20-1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Weiss S., Bhat P., del Pilar COVID-19 infection in ESKD: findings from a prospective disease surveillance program at dialysis facilities in New York City and Long Island. J Am Soc Nephrol. 2020;31:2517–2521. doi: 10.1681/ASN.2020070932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Clarke C., Prendecki M., Dhutia A. High prevalence of asymptomatic COVID-19 infection in hemodialysis patients detected using serologic screening. J Am Soc Nephrol. 2020;31:1969–1975. doi: 10.1681/ASN.2020060827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Corbett R.W., Blakey S., Nitsch D. West London Renal and Transplant Centre. Epidemiology of COVID-19 in an urban dialysis center. J Am Soc Nephrol. 2020;31:1815–1823. doi: 10.1681/ASN.2020040534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Carter B., Collins J.T., Barlow-Pay F. Nosocomial COVID-19 infection: examining the risk of mortality. The COPE-Nosocomial Study (COVID in Older PEople) J Hosp Infect. 2020;106:376–384. doi: 10.1016/j.jhin.2020.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rhee C., Baker M., Vaidya V. Incidence of nosocomial COVID-19 in patients hospitalized at a large US academic medical center. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.20498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Anand S., Montez-Rath M., Boseman J. Prevalence of SARS-CoV-2 antibodies in a large nationwide sample of patients on dialysis in the USA: a cross-sectional study. Lancet. 2020;396:1335–1344. doi: 10.1016/S0140-6736(20)32009-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jager K.J., Kramer A., Chesnave N.C. Results from the ERA-EDTA Registry indicate a high mortality due to COVD-19 in dialysis patients and kidney transplant recipients across Europe. Kidney Int. 2020;98:1540–1548. doi: 10.1016/j.kint.2020.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Craig-Schapiro R., Salinas T., Lubetzky M. COVID-19 outcomes in patients wait listed for kidney transplantation and kidney transplant recipients [e-pub ahead of print] Am J Transplant. 2020 doi: 10.1111/ajt.16351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Thaunat O, Legeai C, Anglicheau D et al. IMPact of the COVID-19 epidemic on the moRTAlity of kidney transplant recipients and candidates in a French Nationwide registry sTudy (IMPORTANT). Kidney Int. (in press). [DOI] [PMC free article] [PubMed]
- 24.Clarke C., Lucisano G., Prendecki M. Informing the risk of kidney transplantation versus remaining on the wait list in the COVID-19 era. Kidney Int Rep. 2021;6(1) doi: 10.1016/j.ekir.2020.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Azzi Y., Parides M., Alani O. COVID-19 infection in kidney transplant recipients at the epicenter of pandemics. Kidney Int. 2020;98:1559–1567. doi: 10.1016/j.kint.2020.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Elias M., Pievani D., Randoux C. COVID-19 infection in kidney transplant recipients: disease incidence and clinical outcomes. J Am Soc Nephrol. 2020;31:2413–2423. doi: 10.1681/ASN.2020050639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lubetzky M., Aull M.J., Craig-Schapiro R. Kidney allograft recipients, immunosuppression, and coronavirus disease—2019: a report of consecutive cases from a New York City transplant center. Nephrol Dial Transplant. 2020;35:1250–1261. doi: 10.1093/ndt/gfaa154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schnitzler M.A., Whiting J.F., Brennan D.C. The life-years saved by a deceased organ donor. Am J Transplant. 2005;5:2289–2296. doi: 10.1111/j.1600-6143.2005.01021.x. [DOI] [PubMed] [Google Scholar]
- 29.United States Renal Data System . National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; Bethesda, MD: 2019. 2019 USRDS annual data report: epidemiology of kidney disease in the United States. [Google Scholar]
- 30.Wolfe R.A., Ashby V.B., Milford E.L. Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med. 1999;341:1725–1730. doi: 10.1056/NEJM199912023412303. [DOI] [PubMed] [Google Scholar]
- 31.Wong G., Howard K., Chapman J.R. Comparative survival and economic benefits of deceased donor kidney transplantation and dialysis in people with varying ages and co-morbidities. PLoS One. 2012;7 doi: 10.1371/journal.pone.0029591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Foster M.C., Varothai N., Weiner D.E. Estimation of life-years saved by solid-organ transplant. JAMA Surg. 2015;150:1015. doi: 10.1001/jamasurg.2015.1936. [DOI] [PubMed] [Google Scholar]
- 33.Cecka J.M. Comparing survival after transplantation with survival on dialysis: net benefit or a way to identify healthier patients? Transplantation. 2006;82:619–620. doi: 10.1097/01.tp.0000235435.82445.e8. [DOI] [PubMed] [Google Scholar]
- 34.Organ Procurement and Transplantation Network (OPTN) and Scientific Registry of Transplant Recipients (SRTR). OPTN/SRTR 2018 Annual Data Report. Department of Health and Human Services, Health Resources and Services Administration; Rockville, MD: 2019. [Google Scholar]
- 35.Wolfe R.A., McCullough K., Schaubel D. Calculating life years from transplant (LYFT): methods for kidney and kidney-pancreas candidates. Am J Transplant. 2008;8:997–1011. doi: 10.1111/j.1600-6143.2008.02177.x. [DOI] [PubMed] [Google Scholar]
- 36.A guide to calculating and interpreting the Kidney Donor Profle Index (KDPI). Updated March 23, 2020. https://optn.transplant.hrsa.gov/media/1512/guide_to_calculating_interpreting_kdpi.pdf Available at:
- 37.Rentsch C.T., Kidwai-Khan F., Tate J.P. COVID-19 by race and ethnicity: a national cohort study of 6 million United States veterans [preprint]. medRxiv. https://doi.org/10.1101/2020.05.12.20099135
- 38.Yehia B.R., Winegar A., Fogel R. Association of race with mortality among patients hospitalized with coronavirus disease 2019 (COVID-19) at 92 US hospitals. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.18039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ghayda R.A., Lee K.H., Han Y.J. Estimation of global case fatality rate of coronavirus disease 2019 (COVID-19) using meta-analyses: comparison between calendar date and days since the outbreak of the first confirmed case. Int J Infect Dis. 2020;100:302–308. doi: 10.1016/j.ijid.2020.08.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.