Abstract
Background
Coronavirus disease 2019 (COVID‐19) is associated with activation of coagulation that mainly presents as thrombosis. Sepsis is also associated with activation of coagulation that mainly presents as disseminated intravascular coagulation. Many studies have reported increased levels of plasma d‐dimer in patients with COVID‐19 that is associated with severity, thrombosis, and mortality.
Objectives
The aim of this study was to compare levels of circulating extracellular vesicle tissue factor (EVTF) activity and active plasminogen activator inhibitor 1 (PAI‐1) in plasma from patients with COVID‐19 or sepsis.
Methods
We measured levels of d‐dimer, EVTF activity, and active PAI‐1 in plasma samples from patients with COVID‐19 (intensive care unit [ICU], N = 15; and non‐ICU, N = 20) and patients with sepsis (N = 35).
Results
Patients with COVID‐19 had significantly higher levels of d‐dimer, EVTF activity, and active PAI‐1 compared with healthy controls. Patients with sepsis had significantly higher levels of d‐dimer and EVTF activity compared with healthy controls. Levels of d‐dimer were significantly lower in patients with COVID‐19 compared with patients with sepsis. Levels of EVTF activity were significantly higher in ICU patients with COVID‐19 compared with patients with sepsis. Levels of active PAI‐1 were significantly higher in patients with COVID‐19 compared with patients with sepsis.
Conclusions
High levels of both EVTF activity and active PAI‐1 may promote thrombosis in patients with COVID‐19 due to simultaneous activation of coagulation and inhibition of fibrinolysis. The high levels of active PAI‐1 in patients with COVID‐19 may limit plasmin degradation of crosslinked fibrin and the release of d‐dimer. This may explain the lower levels of D‐dimer in patients with COVID‐19 compared with patients with sepsis.
Keywords: coagulation, COVID‐19, PAI‐1, sepsis, tissue factor
Essentials.
Patients with coronavirus disease 2019 (COVID‐19) have lower levels of D‐dimer than patients with sepsis.
Patients with COVID‐19 in the intensive care unit have higher circulating tissue factor activity than patients with sepsis.
Patients with COVID‐19 have higher circulating active plasminogen activator inhibitor 1 (PAI‐1) than patients with sepsis.
The combination of extracellular vesicle tissue factor activity and active PAI‐1 may promote thrombosis in patients with COVID‐19.
1. INTRODUCTION
Infection is commonly associated with activation of the coagulation system as part of the host immune response. 1 For instance, bacterial infections can cause sepsis, which is associated with thrombosis, disseminated intravascular coagulation (DIC) and a consumptive coagulopathy. 2 , 3 Patients with DIC have elevated plasma levels of D‐dimer, prolonged activated partial thromboplastin time, prolonged prothrombin time, thrombocytopenia, and microangiopathic thrombosis in multiple organs. 2 Severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) infection leads to coronavirus disease 2019 (COVID‐19). Patients with COVID‐19 rarely present with overt DIC, apart from terminally ill patients, but thrombosis is present. Early studies reported a wide range of rates of venous thromboembolism (VTE) in patients with COVID‐19 (0.9%–69%). 4 However, a more recent study of 3334 patients in New York reported a rate of VTE of 6.2% in patients with COVID‐19. 5 At present, the mechanism(s) promoting thrombosis in patients with COVID‐19 is not known. 4 Elevated D‐dimer levels are observed in several diseases caused by viral infection, including patients infected with influenza A/H1N1, SARS‐CoV or SARS‐CoV‐2. 6 , 7 , 8 , 9 , 10 , 11 In patients with COVID‐19, D‐dimer levels are associated with disease severity, thrombosis, and mortality. 8 , 9 , 10 , 11
Tissue factor (TF) expression is induced in monocytes and endothelial cells during infection. 12 Inhibition of TF attenuates the coagulopathy in septic baboons, indicating that TF is a major driver of coagulation in sepsis. 13 We speculated that TF may also contribute to the activation of coagulation in patients with COVID‐19. 4 Extracellular vesicles (EVs) are small membrane vesicles released from a variety of cells, including monocytes and endothelial cells. 14 We and others have observed elevated levels of extracellular vesicle (EV) TF activity in various diseases, including influenza A/H1N1 infection and endotoxemia, 15 , 16 , 17 and levels of EVTF activity are associated with mortality in patients with influenza A/H1N1. 16 Recently, we demonstrated TF‐dependent shortening of the onset of thrombin generation in plasmas from patients with COVID‐19 and sepsis. 18 Moreover, we also detected increased levels of circulating EVTF activity in patients with COVID‐19, 19 which is consistent with the notion that TF + EVs may promote thrombosis.
Plasminogen activator inhibitor‐1 (PAI‐1) circulates in an active form as well as a latent inactive form. 20 Elevated levels of PAI‐1 are associated with thrombosis in a variety of diseases. 20 PAI‐1 increases the risk of thrombosis by inhibiting the plasminogen activators tissue plasminogen activator and urokinase. This results in reduced conversion of plasminogen to plasmin and plasmin‐dependent lysis of thrombi. Interestingly, a meta‐analysis of 19 studies, which included studies that measured either total or active PAI‐1, found that PAI‐1 levels are associated with severity and all‐cause mortality in sepsis. 21 Furthermore, one study found that plasma PAI‐1 levels are higher in patients with SARS compared with patients with other infectious pneumonias or healthy controls. 22
In this study, we measured levels of D‐dimer, EVTF activity, and active PAI‐1 to define the changes induced by COVID‐19 and sepsis and elucidate mechanisms that increase thrombosis risk in patients with COVID‐19.
2. MATERIALS AND METHODS
2.1. Study participants
Participants with COVID‐19 were a subset of subjects enrolled in previous studies. 18 , 23 , 24 Hospitalized patients (N = 35 total, 15 intensive care unit [ICU] and 20 non‐ICU) with SARS‐CoV‐2 infections confirmed by reverse transcriptase polymerase chain reaction were recruited from the University of Utah Health Sciences Center in Salt Lake City between March 17 and August 1, 2020. Nine of the patients with COVID‐19 were mechanically ventilated. Enrollment criteria included age >18, positive SARS‐CoV‐2 testing, respiratory symptoms or fever, and hospital admission. The majority of the ICU patients were critically ill due to acute respiratory distress syndrome. No clinically apparent thrombotic events were observed in the patients with COVID‐19. Critically ill patients with sepsis (N = 35) were recruited from the ICU at University of Utah Health in the Salt Lake City Science Center between July 2017 and March 2020. Thirty of the patients with sepsis were mechanically ventilated. Patients with sepsis were a subset of subjects enrolled in a previous study of plasma biomarkers of coagulation and fibrinolysis and function. 18 Enrollment criteria included age >18, SEPSIS‐3 criteria, and ICU admission. All patients underwent clinically directed investigations to identify the pathogen causing the infection, but as is common in patients with sepsis, only 29% of the patients had a pathogen specifically identified. Pathogens that were identified include Streptococcus (11%), Staphylococcus (9%), Escherichia coli (6%), and influenza A/H1N1 (3%). We also determined the site or organ of the primary infection. These included pneumonia (34%), skin and soft‐tissue infection (26%), urosepsis (23%), intra‐abdominal infection (9%), blood (6%), or unknown (2%). All patients were enrolled within 72 hours of ICU admission and were recruited under protocols approved by the Institutional Review Board of the University of Utah (IRB no. 00102638, 00093575). Anticoagulation was used in 94% of the patients. Age‐, sex‐, and race‐matched heathy donors (N = 10) were used as controls. All participants or their legal authorized representative gave written informed consent for study enrollment in accordance with the Declaration of Helsinki.
2.2. Anticoagulation
At the time of blood draw, the majority of patients with COVID‐19 (89%) or sepsis (94%) were receiving anticoagulants to prevent thrombosis, as ordered by the patient’ primary team in accordance with institutional guidelines. Patients with COVID‐19 were more frequently administered low‐molecular‐weight heparin (LMWH) (enoxaparin) compared with patients with sepsis. In addition to LWMH, one patient with COVID‐19 was on unfractionated heparin and one patient was on rivaroxaban. Patients with sepsis were more frequently on unfractionated heparin than were patients with COVID‐19. Most patients on anticoagulants were on weight‐based prophylactic dosing per institutional guidelines, except for one patient with sepsis who was on therapeutic heparin. Four non‐ICU patients with COVID‐19 and two patients with sepsis did not receive anticoagulants.
2.3. Thrombosis
The standard of care at the University of Utah does not include screening ultrasound for VTE. Clinically apparent, overt thrombotic events were rare in our cohort. Only two patients with sepsis had thrombotic events and none of the patients with COVID‐19 had symptomatic, clinically detected thrombotic complications.
2.4. Plasma preparation
Blood was collected in the same manner in each of the 3 groups using a 21‐g needle vacutainer butterfly into acid/citrate/dextrose anticoagulant. Blood was centrifuged (150 g, 20 minutes) to generate platelet‐rich plasma. Platelet‐rich plasma was then centrifuged (1500 g, 20 minutes) to produce platelet‐poor plasma, which was flash frozen in liquid nitrogen and stored at −80°C.
2.5. Measurement of biomarkers in plasma
EVTF activity was measured as described. 25 Briefly, EVs were isolated from plasma by centrifugation, washed, and resuspended in buffer. TF activity of the EVs was determined by adding factor VIIa and factor X and then measuring the levels of factor Xa using a chromogenic substrate. Active PAI‐1 was measured with a kit that captures active PAI‐1 with urokinase and detects PAI‐1 using an antihuman PAI‐1 antibody (Cat. no. HPAIKT; Molecular Innovations, Novi, MI, USA). D‐dimer was measured using an ELISA (ab260076; Abcam, Cambridge, MA, USA). All assays were run in a batch together at the same time except for D‐dimer levels on seven patients with COVID‐19, which were run on a separate day using the same D‐dimer ELISA kit and lot from Abcam. There was no significant difference between COVID‐19 D‐dimer levels run on separate days. All technicians were blinded to the study groups.
2.6. Statistical analysis
Since the Shapiro‐Wilk test indicated that data were not normally distributed, we used the Mann‐Whitney U test for two group comparisons and the Kruskal‐Wallis test with Dunn’s multiple comparisons for multiple group comparisons. Data are shown as median (interquartile range [IQR]). The relationship between biomarkers was evaluated using the Spearman’s rho. All statistical analyses were performed with Prism version 7.03 (GraphPad Software, La Jolla, CA, USA). P values <.05 were considered statistically significant.
3. RESULTS AND DISCUSSION
3.1. Patient characteristics
The study included 35 patients with COVID‐19 (N = 20 non‐ICU and N = 15 ICU), 35 patients with sepsis (all in the ICU), and 10 healthy controls (Table 1). The COVID‐19 and sepsis groups were well matched based on age, sex, comorbidities, and in‐hospital mortality. However, sequential organ failure assessment (SOFA) scores were significantly different between the two groups, as multiorgan failure is common in sepsis but not in COVID‐19 (Table 1). The patients with COVID‐19 in the ICU were significantly older and were more severely ill compared with the non‐ICU patients with COVID‐19 (Table 2).
TABLE 1.
Clinical characteristics of healthy donors and hospitalized patients with COVID‐19 and sepsis
| Healthy donors (N = 10) | COVID‐19 (N = 35) | Sepsis (N = 35) | P value | |
|---|---|---|---|---|
| Age, mean (±SD) | 54.2 (±11.3) | 54.3 (±17.8) | 57.9 (±19.4) | .62 |
| Male, % | 70 | 46 | 57 | .35 |
| Hispanic/Latino, % | 10 | 34 | 17 | .14 |
| BMI, mean (±SD) | 32.6 (±9.4) | 32.2 (±12.8) | .44 | |
| Diabetes, % | 0 | 42.8 | 25.7 | .21 |
| Hypertension, % | 0 | 42.9 | 51.4 | .63 |
| Chronic lung disease, % | 0 | 20.0 | 17.1 | .99 |
| SOFA score, mean (±SD) | 3.3 (±2.3) | 6.3 (±3.9) | <.001 | |
| ARDS, % | 45.7 | 31.1 | .33 | |
| Mechanical ventilation, % | 25.7 | 17.1 | .56 | |
| Survival to date, % | 82.9 | 85.7 | >.99 | |
| White blood cell count, K/µL, mean (±SD) a | 7.1 (±2.5) | 16.1 (±7.7) | <.001 | |
| Platelet count, K/µL, mean (±SD) a | 242 (±87.5) | 196 (±97.9) | .42 | |
| Anticoagulation, % | 88.6 | 94 | .67 | |
| D‐dimer, median (IQR) |
129 (50‐335) |
922 (611‐1907) |
3195 (1610‐6742) |
<.001 |
| EVTF activity, median (IQR) | 0 (0‐0) |
0.24 (0.14‐0.37) |
0.14 (0‐0.34) | <.001 |
| PAI‐1, median (IQR) |
10.5 (1.5‐24.8) |
56.2 (38.3‐105.5) |
23.5 [9.2–80.9] | <.001 |
The COVID‐19 and sepsis groups were compared using the Mann‐Whitney U test for two group comparisons or the Kruskal‐Wallis test with Dunn’s multiple comparisons for multiple group comparisons.
Abbreviations: ARDS, acute respiratory distress syndrome; BMI, body mass index; COVID‐19, coronavirus disease 2019; EVTF, extracellular vesicle tissue factor; IQR, interquartile range; PAI‐1, plasminogen activator inhibitor‐1; SD, standard deviation; SOFA, sequential organ failure assessment.
TABLE 2.
Clinical characteristics of COVID‐19 non‐ICU and ICU patients
|
COVID‐19 Non‐ICU (N = 20) |
COVID‐19 ICU (N = 15) |
P value | |
|---|---|---|---|
| Age, mean (±SD) | 48.5 (±17.2) | 62.0 (±15.3) | .02 |
| Male, % | 40 | 53 | .51 |
| Hispanic/Latino, % | 30 | 40 | .72 |
| BMI, mean (±SD) | 34.1 (±9.7) | 30.6 (±9.0) | .16 |
| Diabetes, % | 30 | 60 | .10 |
| Hypertension, % | 35 | 53 | .32 |
| Chronic lung disease, % | 15.0 | 26.7 | .67 |
| SOFA score, mean (±SD) | 1.8 (±1.1) | 5.3 (±1.8) | <.001 |
| ARDS, % | 10 | 93.8 | <.001 |
| Mechanical ventilation, % | 0 | 60.0 | <.001 |
| Survival to date, % | 100 | 60 | .003 |
| White blood cell count, mean (±SD) | 6.2 (±2.3) | 8.3 (±2.2) | .009 |
| Platelet count, K/µL, mean (±SD) | 241 (±107) | 244 (±54) | .38 |
| Hydroxychloroquine, % | 20 | 33 | .45 |
| Remdesivir, % | 27 | 25 | >0.99 |
| Anticoagulation, % | 80 | 100 | .12 |
| D‐dimer, median (IQR) | 765 (495‐1382) |
1340 (696‐4297) |
.04 |
| EVTF activity, median (IQR) |
0.24 (0.14‐0.37) |
0.47 (0.30‐0.81) |
<.001 |
| PAI−1, median (IQR) |
66.5 (37.5‐108.6) |
47.7 (38.3‐83.4) |
0.610 |
Study participants were a subset of subjects previously reported. 18 , 23 , 24 Groups were compared using the Mann‐Whitney U test.
Abbreviations: ARDS, acute respiratory distress syndrome; BMI, body mass index; COVID‐19, coronavirus disease 2019; EVTF, extracellular vesicle tissue factor; ICU, intensive care unit; IQR, interquartile range; PAI‐1, plasminogen activator inhibitor‐1; SD, standard deviation; SOFA, sequential organ failure assessment.
3.2. D‐dimer
Levels of D‐dimer were significantly higher in both patients with COVID‐19 (median, 922 [IQR, 611‐1907]; P < .01) and patients with sepsis (median, 3195 [IQR, 1610‐6742]; P < .001) compared with healthy controls (median, 129 [IQR, 50‐335]; and higher in sepsis compared with patients with COVID‐19 (Figure 1A).
FIGURE 1.
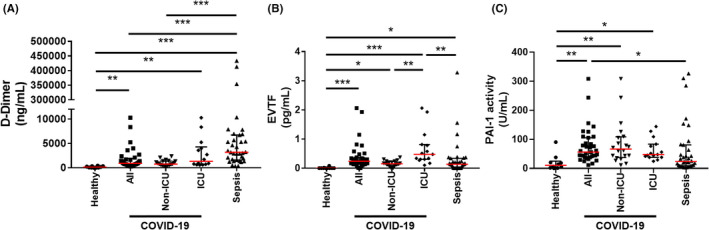
Plasma biomarkers in COVID‐19 patients and sepsis patients. Levels of D‐dimer (A), extracellular vesicle tissue factor (EVTF) activity (B), and active plasminogen activator inhibitor‐1 (PAI‐1) (C) were measured in patients with coronavirus disease 2019 (COVID‐19) (N = 35; intensive care unit [ICU], N = 15 and non‐ICU, N = 20) or sepsis (N = 35). Data is shown as median (red line) ± interquartile range. For the D‐dimer analysis, we could not measure values for one ICU patient with COVID‐19 and one non‐ICU patient with COVID‐19 because of lack of sample. Zero D‐dimer value was measured in 1 healthy control. Zero EVTF activity values were measured in 8 healthy controls, 1 non‐ICU patient with COVID‐19, and 9 patient with sepsis. Groups were compared using Kruskal‐Wallis test with Dunn’s multiple comparisons. Statistical significance is indicated above the different groups: *P < .05, **P < .01, ***P < .001
3.3. EVTF activity
Levels of EVTF activity were significantly higher in patients with COVID‐19 (median, 0.24 [IQR, 0.14‐0.37]; P < .001) and patients with sepsis (median, 0.14 [IQR, 0‐0.34];P < .05) compared with healthy controls (median, 0 [IQR, 0‐0]; (Figure 1B). The data for patients with COVID‐19 are consistent with our recent study showing that patients with COVID‐19 (N = 100) from Sweden have elevated levels of EVTF activity (median, 0.21 [IQR, 0‐0.53]) compared with healthy controls (median, 0 [IQR, 0‐0]). 19 ICU patients had higher levels of EVTF activity compared with non‐ICU patients (Figure 1B). We recently showed that patients with COVID‐19 requiring high respiratory support had higher levels of EVTF compared with patients with low respiratory support. 19 Importantly, ICU patients with COVID‐19 had significantly higher levels of EVTF activity compared with patients with sepsis (Figure 1B). A previous study with a human endotoxemia model reported relatively low levels of EVTF activity. 17 EVTF activity was significantly associated with the SOFA score of all patients with COVID‐19 (Spearman’s rho, 0.49; P = 0.004) but not patients with sepsis.
3.4. Plasminogen activator inhibitor 1
Levels of active PAI‐1 were significantly higher in patients with COVID‐19 (median, 56.2 [IQR, 38.3‐105.5];P < .01) compared with healthy controls (median, 10.5 [IQR, 1.5‐24.8]) (Figure 1C). Patients with sepsis (median, 23.5 [IQR, 9.2‐80.9]) had higher levels of active PAI‐1 compared with healthy controls, but this difference was not statistically significant. Interestingly, levels of active PAI‐1 were significantly higher in patients with COVID‐19 compared with patients with sepsis (Figure 1C). Notably, patients with sepsis were sicker than patients with COVID‐19 (Table 1). Active PAI‐1 was significantly associated with both D‐dimer (Spearman’s rho, 0.381; P = 0.02) and SOFA score (Spearman’s rho, 0.40; P = .02) in patients with sepsis, but not in patients with COVID‐19.
3.5. Differences between the coagulopathies in patients with COVID‐19 and patients with sepsis
Collectively, these data indicate that patients with COVID‐19 have higher levels of EVTF activity (an activator of coagulation) and PAI‐1 (an inhibitor of fibrinolysis), but lower levels of D‐dimer (a marker of fibrin dissolution) compared with patients with sepsis (Figure 2). We propose that these combined changes create an environment in patients with COVID‐19 that promotes thrombosis rather than DIC. In patients with sepsis, elevated circulating EVTF activity will activate the coagulation system and PAI‐1 will inhibit the fibrinolytic system but to a lesser extent than in patients with COVID‐19 resulting in higher D‐dimer levels, an environment favoring DIC over thrombosis (Figure 2).
FIGURE 2.
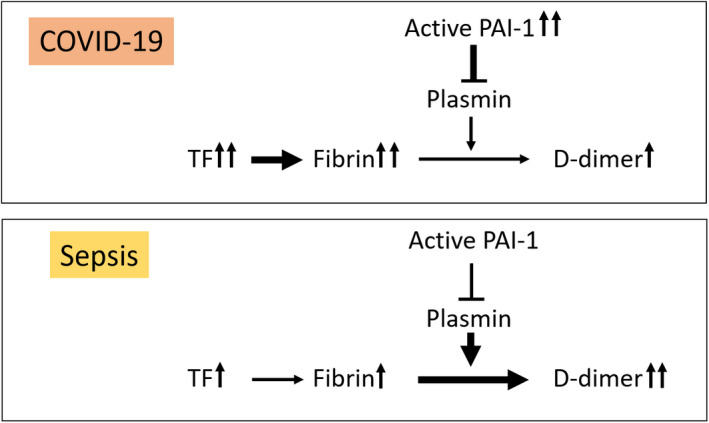
Model of the coagulopathies in coronavirus disease 2019 (COVID‐19) and sepsis. In patients with COVID‐19, increased tissue factor (TF) leads to fibrin generation but low D‐dimer due to the concurrent increase in plasminogen activator inhibitor 1 (PAI‐1) and inhibition of fibrinolysis. In sepsis, increased TF leads to fibrin generation with high D‐dimer due to a smaller increase in PAI‐1
3.6. Limitations of the study
The study has several limitations. The number of patients with COVID‐19 and patients with sepsis is small and samples were collected at a single time point. In addition, most of the patients with COVID‐19 and patients with sepsis were receiving anticoagulant therapy to prevent thrombosis. Four of the non‐ICU patients with COVID‐19 and two of the patients with sepsis did not receive anticoagulation. We did not observe any differences in the levels of EVTF and PAI‐1 in patients with COVID‐19 and patients with sepsis with or without anticoagulation. We used platelet‐poor plasma rather than platelet‐free plasma. We know that higher levels of EVTF activity are observed in platelet‐poor plasma compared with platelet‐free plasma. 15 Platelets are also a major source of PAI‐1. 26 , 27 However, all samples were collected in the same manner. There was no significant difference in age or sex between patients with COVID‐19 and patients with sepsis. A higher percentage of male donors were included in our healthy donor group, but this was not significantly different when compared with our COVID‐19 or sepsis cohorts.
RELATIONSHIP DISCLOSURE
MTR is a member of the Scientific Advisory Board for Acticor Biotech SAS. All other authors declare no conflicts of interest.
AUTHOR CONTRIBUTIONS
RAC, MTR, and NM designed the study. RAC and FD performed the experiments. RAC, YH, SPG, and NM analyzed the data. EAM collected the samples from the patients. NM wrote the manuscript. RAC, YH, SPG, ASW, EGB, and MTR edited the manuscript. All authors read and approved the final manuscript.
ACKNOWLEDGMENTS
The authors thank the patients who participated in this study.
Handling Editor: Dr Johnny Mahlangu.
Funding information
This work was supported by grants from the NIH (U24NS107228 to FD; K01AG059892 to RAC; R01HL142804, R01AG048022, R56AG059877, and R01HL130541 to MTR; and U01HL143403 and HL126974 to ASW); the University of Utah Triple I Program (EAM), and Fonds voor Wetenschappelijk Onderzoek Vlaanderen FWO 12U7818 N (FD); and NIH R01 HL119523 (NM) and a University of North Carolina at Chapel Hill School of Medicine and NCTraCS ECBR pilot grant (NM). This work was also supported in part by Merit Review Award Number I01 CX001696 to MTR from the US Department of Veterans Affairs Clinical Sciences R&D. This material is the result of work supported with resources and the use of facilities at the George E. Wahlen VA Medical Center, Salt Lake City, Utah. The contents do not represent the views of the US Department of Veterans Affairs or the US government
Contributor Information
Alisa S. Wolberg, @aswolberg.
Nigel Mackman, Email: nigel.mackman@med.unc.edu.
REFERENCES
- 1. Antoniak S. The coagulation system in host defense. Res Pract Thromb Haemost. 2018;2:549‐557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Iba T, Levy JH, Warkentin TE et al. Diagnosis and management of sepsis‐induced coagulopathy and disseminated intravascular coagulation. J Thromb Haemost. 2019;17:1989‐1994. [DOI] [PubMed] [Google Scholar]
- 3. Kaplan D, Casper TC, Elliott CG et al. VTE incidence and risk factors in patients with severe sepsis and septic shock. Chest. 2015;148:1224‐1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mackman N, Antoniak S, Wolberg AS, Kasthuri R, Key NS. Coagulation abnormalities and thrombosis in patients infected with SARS‐CoV‐2 and other pandemic viruses. Arterioscler Thromb Vasc Biol. 2020;40:2033‐2044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bilaloglu S, Aphinyanaphongs Y, Jones S, Iturrate E, Hochman J, Berger JS. Thrombosis in hospitalized patients with COVID‐19 in a New York City health system. JAMA. 2020;324:799‐801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Davey RT Jr, Lynfield R, Dwyer DE et al. The association between serum biomarkers and disease outcome in influenza A(H1N1)pdm09 virus infection: results of two international observational cohort studies. PLoS One. 2013;8:e57121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lee N, Hui D, Wu A et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1986‐1994. [DOI] [PubMed] [Google Scholar]
- 8. Chen G, Wu D, Guo W et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130:2620‐2629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Berger JS, Kunichoff D, Adhikari S et al. Prevalence and outcomes of D‐dimer elevation in hospitalized patients with COVID‐19. Arterioscler Thromb Vasc Biol. 2020;40:2539‐2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zhou F, Yu T, Du R et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054‐1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844‐847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Grover SP, Mackman N. Tissue factor: an essential mediator of hemostasis and trigger of thrombosis. Arterioscler Thromb Vasc Biol. 2018;38:709‐725. [DOI] [PubMed] [Google Scholar]
- 13. Taylor FB Jr, Chang A, Ruf W et al. Lethal E. coli septic shock is prevented by blocking tissue factor with monoclonal antibody. Circ Shock. 1991;33:127‐134. [PubMed] [Google Scholar]
- 14. Coumans FAW, Brisson AR, Buzas EI et al. Methodological guidelines to study extracellular vesicles. Circ Res. 2017;120:1632‐1648. [DOI] [PubMed] [Google Scholar]
- 15. Hisada Y, Alexander W, Kasthuri R et al. Measurement of microparticle tissue factor activity in clinical samples: a summary of two tissue factor‐dependent FXa generation assays. Thromb Res. 2016;139:90‐97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Rondina MT, Tatsumi K, Bastarache JA, Mackman N. Microvesicle tissue factor activity and interleukin‐8 levels are associated with mortality in patients with influenza A/H1N1 infection. Crit Care Med. 2016;44:e574‐e578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mooberry MJ, Bradford R, Hobl EL, Lin FC, Jilma B, Key NS. Procoagulant microparticles promote coagulation in a factor XI‐dependent manner in human endotoxemia. J Thromb Haemost. 2016;14:1031‐1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Bouck EG, Denorme F, Holle LA et al. COVID‐19 and sepsis are associated with different abnormalities in plasma procoagulant and fibrinolytic activity. Arterioscler Thromb Vasc Biol. 2021;41:401‐414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Rosell A, Havervall S, von Meijenfeldt F et al. Patients with COVID‐19 have elevated levels of circulating extracellular vesicle tissue factor activity that is associated with severity and mortality. Arterioscler Thromb Vasc Biol. 2021;41:878–882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Westrick RJ, Eitzman DT. Plasminogen activator inhibitor‐1 in vascular thrombosis. Curr Drug Targets. 2007;8:966‐1002. [DOI] [PubMed] [Google Scholar]
- 21. Tipoe TL, Wu WKK, Chung L et al. Plasminogen activator inhibitor 1 for predicting sepsis severity and mortality outcomes: a systematic review and meta‐analysis. Front Immunol. 2018;9:1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wu YP, Wei R, Liu ZH et al. Analysis of thrombotic factors in severe acute respiratory syndrome (SARS) patients. Thromb Haemost. 2006;96:100‐101. [DOI] [PubMed] [Google Scholar]
- 23. Middleton EA, He XY, Denorme F et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID‐19 acute respiratory distress syndrome. Blood. 2020;136:1169‐1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Manne BK, Denorme F, Middleton EA et al. Platelet gene expression and function in patients with COVID‐19. Blood. 2020;136:1317‐1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hisada Y, Mackman N. Measurement of tissue factor activity in extracellular vesicles from human plasma samples. Res Pract Thromb Haemost. 2019;3:44‐48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Brogren H, Karlsson L, Andersson M, Wang L, Erlinge D, Jern S. Platelets synthesize large amounts of active plasminogen activator inhibitor 1. Blood. 2004;104:3943‐3948. [DOI] [PubMed] [Google Scholar]
- 27. Brogren H, Wallmark K, Deinum J, Karlsson L, Jern S. Platelets retain high levels of active plasminogen activator inhibitor 1. PLoS ONE. 2011;6:e26762. [DOI] [PMC free article] [PubMed] [Google Scholar]


