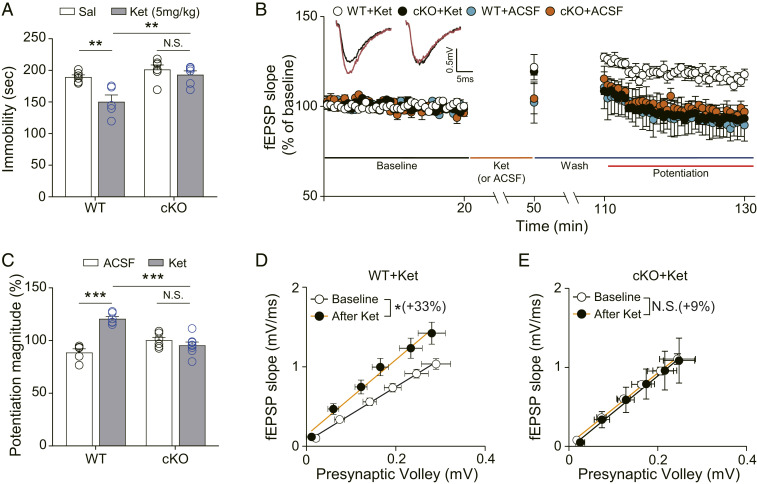
Fig. 1.
Ketamine does not produce antidepressant-like behavioral changes and synaptic potentiation in Reelin cKO mice. (A) Accumulated time mice spent immobile during the FST performed 2 h after ketamine injection. Ketamine significantly decreased immobility in WT mice but not in cKO mice [two-way ANOVA with Tukey’s multiple comparisons, Genotype × Drug: F(1, 19) = 4.815, P = 0.0408, Genotype: F(1, 19) = 15.68, P = 0.0008, Drug: F(1, 19) = 11.47, P = 0.0031, n = 5 to 6 per group]. (B) Ketamine potentiation was measured after 20 min of baseline measurement, 30 min of ketamine perfusion, and 1 h of washout period in the hippocampal CA1 region. All responses were normalized to respective baselines. (Inset) Representative waveforms from baseline (black) or ketamine potentiation (red) measurement. (C) A bar graph that summarizes the magnitude of ketamine potentiation of respective groups measured in B. Significantly increased potentiation magnitude by ketamine was observed in WT mice but not in cKO mice [two-way ANOVA with Tukey’s multiple comparisons, Genotype × Drug: F(1, 21) = 37.82, P < 0.0001, Genotype: F(1, 21) = 5.226, P = 0.0327, Drug: F(1, 21) = 20.50, P = 0.0002, n = 5 to 8 per group]. (D and E) I–O curves measured during baseline and potentiation measurement in B. Initial slopes of fEPSPs versus presynaptic volley values are plotted at 4, 8, 12, 16, 20, and 24 μA stimulation intensity. The slope of the I–O curve is significantly increased after ketamine treatment in slices from WT mice but not in slices from cKO mice [paired t test, WT+Ket (D): t(5) = 3.671, P = 0.0144, n = 6; cKO+Ket (E): t(7) = 1.129, P = 0.2961, n = 8]. All data represented as mean ± SEM; *P < 0.05, **P < 0.01, and ***P < 0.001.