Fig. 4. Evolution of the PDLLA-Zn-nitrided Fe BRS in a rabbit abdominal aorta.
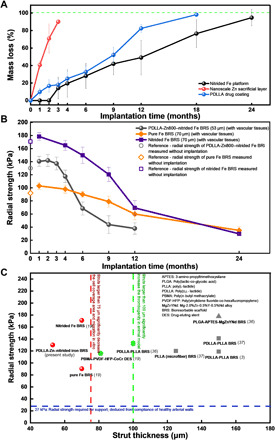
(A) Mass loss of each layer after careful separation of the layers. The black line represents the mass loss of the nitrided Fe platform (obtained by weighing the remaining struts after removing the tissues and biodegradation products; n = 11), the red line represents the mass loss of the nanoscale Zn sacrificial layer (Zn in its elemental state; obtained using AAS; n = 11), and the blue line represents the mass loss of the PDLLA drug coating (obtained using gel permeation chromatography; n = 11) as a function of time point after BRS implantation. (B) Radial strength evolution of the PDLLA-Zn800-nitrided Fe BRS in the present study and the pure and nitrided Fe BRSs reported in our previous study (19) as a function of time point after implantation. The PDLLA-Zn800-nitrided Fe BRS’s metallic strut was 53 μm thick, while the pure and nitrided Fe BRSs’ metallic struts were 70 μm thick. The radial strength was measured with vascular tissues, except for the initial values (n = 8). (C) Summary of radial strength–strut thickness features of the scaffolds/stents. Simon et al. (33) has reported 75 μm (dashed line in red) as a critical strut thickness, above which the strut will affect the cell coverage and migration of endothelial cells in vitro. Kolandaivelu et al. (22), on the other hand, has reported 100 μm (dashed line in green) as a critical strut thickness value in animal models, with the risk of thrombogenicity increasing when the strut thickness is larger than such value. Twenty-seven kilopascal is considered a threshold of radial strength (which is required for support) deduced from the compliance of healthy arterial walls (34).
