Introduction
Reversible splenial lesion syndrome (RESLES) is a spectrum of disorders radiologically characterized by reversible lesions mainly involving the splenium of the corpus callosum [1]. RESLES most commonly occurs in patients with seizures, particularly following antiepileptic drugs withdrawal. RESLES can also cause by infections, high-altitude cerebral edema, hypoglycemia, hyper/hyponatremia, cerebral venous thrombosis, several non-epileptic pharmacological agents, and miscellaneous conditions including anorexia nervosa, malnutrition, vitamin B12 deficiency, Charco–Marie–Tooth disease, systemic lupus erythematosus, and eclampsia [1–3]. Autoimmune encephalitis as a rare condition associated with RESLES was reported in only one anti-NMDAR encephalitis, one anti-VGKC encephalitis, and one anti-Yo encephalitis case, respectively [3–5].
Autoimmune glial fibrillary acidic protein (GFAP) astrocytopathy is a novel immunotherapy-responsive autoimmune inflammatory central nervous system (CNS) disorder characterized by anti-GFAP antibodies positive encephalitis, meningoencephalitis, or meningoencephalomyelitis [6]. Brain MRI abnormalities are commonly observed. Lesions involve the subcortical white matter, basal ganglia, hypothalamus, brainstem, cerebellum, meninges, and ventricle [6]. Approximately 50% of patients demonstrated a radial linear periventricular enhancement pattern, considered as a radiological hallmark of autoimmune GFAP astrocytopathy [6].
Herein, we report a rare case of an adult confirmed with autoimmune GFAP astrocytopathy presenting brain MRI imaging that indicates RESLES. To the best of our knowledge, this is the first case to provide direct evidence for correlations between anti-GFAP antibodies and RESLES.
Case report
A previously healthy 23-year-old male presented with a 5-day history of headache and psychosis. Neurologic examination revealed decreased cognition and consciousness level, stiff neck, and weakness (grade 4/5) in both lower extremities. The deep tendon reflexes of his upper and lower extremities were increased. The Babinski sign was present bilaterally and meningeal signs were positive.
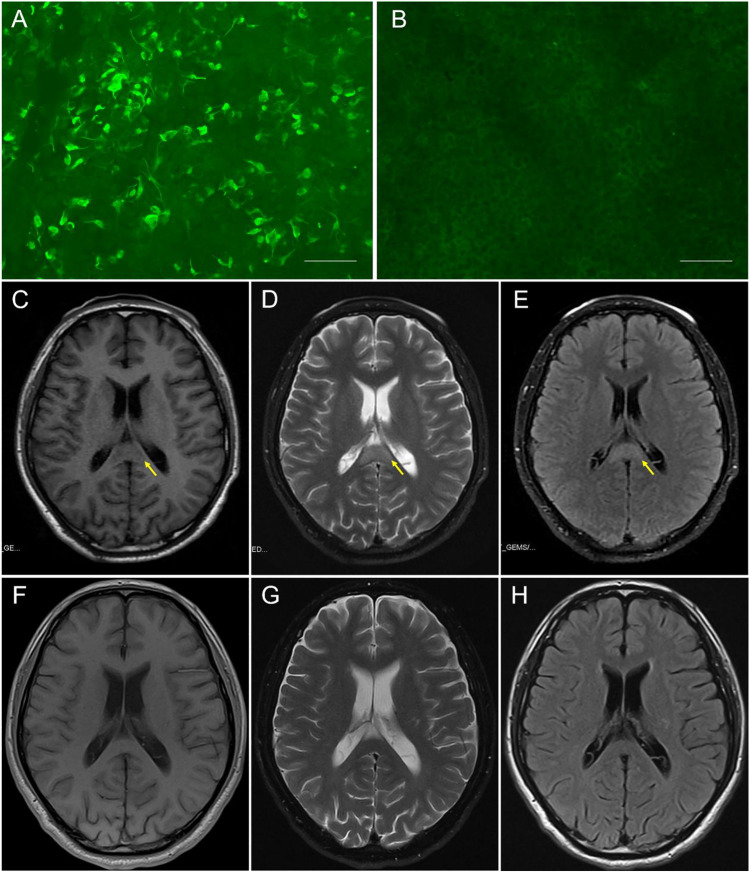
CSF studies revealed lymphocyte-predominant pleocytosis of 80/µL white blood cells (90% lymphocyte, normal range: 0–5/µL) and elevated protein (1.24 g/L, normal range: 0.15–0.45 g/L), with remarkable elevated opening pressure of 285 cm H2O. Microbiological exams in serum and CSF including serological tests, PCR methods, and culture were negative (Table 1). No infectious agent was found in CSF using metagenomics next-generation sequencing. GFAP antibodies (CSF, 1:10) were detected within patient CSF using indirect immunofluorescence (cell-based assay) on HEK293 cells (Euroimmun, Lübeck, Germany) (Fig. 1A). There were no other detectable onconeuronal or neuronal cell surface antibodies, anti-AQP4 antibodies, or anti-MOG antibodies in CSF or serum. Oligoclonal band test in serum and CSF were negative. Other hematologic test results including routine blood test, biochemical test, vitamin B12 level, C-reactive protein, microbiological test, tumor markers, immunological test, and thyroid profiles were unremarkable (Table 1). Electroencephalogram exhibited diffuse slow activities. Initial brain magnetic resonance imaging (MRI) revealed a single lesion in the splenium of the corpus callosum (SCC) hypointense on T1-weighted, hyperintense on T2-weighted, and FLAIR sequences (Fig. 1C–E). Brain MRA and spinal MRI were negative.
Table 1.
Additional auxiliary examination results
| Cerebrospinal fluid | |
| WBC | 80/µL (90% lymphocyte) |
| Glucose | (-) |
| Protein | 1.24 g/L |
| Microbiological exams | |
| Bacteria (culture) | (-) |
| Mycobacterium tuberculosis (culture) | (-) |
| Cryptococcus (culture) | (-) |
| Herpes simplex virus 1 and 2 (PCR) | (-) |
| Cytomegalovirus (PCR) | (-) |
| Epstein-Barr virus (PCR) | (-) |
| Japanese encephalitis virus (PCR) | (-) |
| Metagenomics next-generation sequencing | (-) |
| aOnconeuronal antibodies | (CSF/serum) |
| Anti-Hu | (-/-) |
| Anti-Yo | (-/-) |
| Anti-Ri | (-/-) |
| Anti-CV2 | (-/-) |
| Anti-amphiphysin | (-/-) |
| Anti-Ma2 | (-/-) |
| Anti-SOX1 | (-/-) |
| Anti-Tr | (-/-) |
| Anti-GAD65 | (-/-) |
| Anti-Zic4 | (-/-) |
| Anti-titin | (-/-) |
| Anti-recoverin | (-/-) |
| aNeuronal cell surface antibodies | (CSF/serum) |
| Anti-N-methyl-D-aspartate receptor | (-/-) |
| Anti-alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor 1 | (-/-) |
| Anti-alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor 2 | (-/-) |
| Anti-gamma-aminobutyric acid type A receptor | (-/-) |
| Anti-gamma-aminobutyric acid type B receptor | (-/-) |
| Anti-contactin-2-associated protein receptor | (-/-) |
| Anti-leucine-rich glioma inactivated-1 protein receptor | (-/-) |
| aAnti-glial fibrillary acidic protein antibodies | (1:10/-) |
| aAnti-aquaporin 4 antibodies | (-/-) |
| aAnti-myelin oligodendrocyte glycoprotein antibodies | (-/-) |
| Oligoclonal band (CSF/serum) | (-/-) |
| Hematologic tests | |
| Routine blood test | (-) |
| Biochemical test | (-) |
| C-reactive protein | (-) |
| Vitamin B12 | (-) |
| Microbiological tests | |
| Human immunodeficiency virus | (-) |
| TORCH | (-) |
| Epstein-Barr virus | (-) |
| Parasites | (-) |
| Mycobacterium tuberculosis | (-) |
| Tumor markers | |
| Carcino-embryonic antigen | (-) |
| Cancer antigen 15–3 | (-) |
| Cancer antigen 19–9 | (-) |
| Cancer antigen 125 | (-) |
| Cancer antigen 72–4 | (-) |
| Total prostate-specific antigen | (-) |
| Free prostate specific antigen | (-) |
| Neuron-specific enolase | (-) |
| Cytokeratin 19 fragment antigen 21–1 | (-) |
| Immunological test | |
| IgG4 | (-) |
| Rheumatoid factor | (-) |
| AKA | (-) |
| ANA | (-) |
| Anti-dsDNA | (-) |
| c-ANCA-IgG | (-) |
| p-ANCA-IgG | (-) |
| MPO-ANCA-IgG | (-) |
| PR3-ANCA-IgG | (-) |
| Anti-SS-A | (-) |
| Anti-SS-B | (-) |
| Anti-Scl-70 | (-) |
| Anti-Jo-1 | (-) |
| Anti-RNP | (-) |
| Anti-ACA | (-) |
| Thyroid antibodies | |
| Thyroid globulin antibodies | (-) |
| Thyroid peroxidase antibodies | (-) |
| Thyroid stimulating hormone receptor antibodies | (-) |
aCell-based assay (Euroimmun, Lübeck, Germany)
WBC, white blood cell; CSF, cerebrospinal fluid; PCR, polymerase chain reaction; TORCH, toxoplasma, treponema pallidum, rubella virus, cytomegalovirus, and herpes simplex virus 1 and 2; AKA, anti-keratin antibodies; ANA, anti-nuclear antibody; dsDNA, double-stranded deoxyribonucleic acid; ANCA, anti-neutrophil cytoplasmic antibodies; anti-SM, anti-Smith antibodies; RNP, ribonuclear protein; ACA, anticardiolipin
Fig. 1.
Auxiliary examinations of the case with autoimmune GFAP astrocytopathy and RESLES. Antibody test in serum and CSF through indirect immunofluorescence on HEK293 cells (Euroimmun, Lübeck, Germany): A Patient CSF demonstrated binding to the surface of cells expressing GFAP proteins (1:10). B Regions with no specific fluorescence to other proteins on the same slide, analyzed simultaneously, were regarded as negative controls (scale bar: 75 µm). Brain MRI: C–E Initial brain MRI on admission (5 days after onset) exhibited an isolated splenium of the corpus callosum (SCC) lesion (arrows) with hypointensity on T1WI, hyperintensity on T2WI, and on FLAIR. F–H On day 50 after onset (10 days after the second course of immunotherapy), repeat MRI highlighted complete resolution of the SCC lesion
Although 5 days of intravenous corticosteroid pulses (intravenous methylprednisolone 1000 mg/day) and IVIg (0.4 g/kg/day) were administered, the illness still progressed rapidly for the further 14 days. The patient developed coma, seizures, blurred vision, and hypoventilation and was intubated for acute respiratory failure. Following transfer to the neurological intensive care unit and treatment with 5-day additional intravenous corticosteroid pulses (intravenous methylprednisolone 1000 mg/day) and IVIg (0.4 g/kg/day) therapy, the patient gradually regained consciousness and strength after 2 weeks and was finally extubated on day 50 after onset. Repeat brain MRI exhibited complete resolution of the SCC lesion (Fig. 1F–H).
Anti-GFAP antibodies were negative both in sera and CSF after 4 months. The patient made a full recovery after a 10-month follow-up. Autoimmune GFAP astrocytopathy (anti-GFAP meningoencephalitis) and RESLES were the presumed diagnoses.
Discussion
Other known causes of RESLES were extensively excluded during diagnosis in this case. Seizures and pharmacological agents were considered as possible etiologies for RESLES. However, no patient history of seizures was present, with the SCC lesion being identified prior to seizure development and consequent treatment. Viral or other pathogenic infections seemed unlikely since no fever or other sign of infection was present, with all microbiological exams including metagenomics next-generation CSF sequencing were negative. Besides, the subsequent complete reversion of the lesion makes ischemic causes seem impossible. Moreover, biochemical, vitamin B12 level, rheumatologic, and thyroid-antibody tests exhibited normal metabolic function with no sign of other autoimmune conditions.
Reports on correlations between RESLES and neuronal or glial antibodies are extremely rare, only previously described in only one anti-NMDAR encephalitis, one anti-VGKC encephalitis, and one anti-Yo encephalitis case, respectively [3–5]. Recently, autoimmune GFAP astrocytopathy in an adult with viral infection and a child with hyponatremia was reported, showing SCC lesions [7, 8]. However, viral infections and hyponatremia itself can also cause RESLES, consequently not clarifying the role of anti-GFAP antibodies in RESLES. Therefore, with exclusion of other etiologies, this is the first case revealing direct links between anti-GFAP antibodies and RESLES.
Possible mechanism of RESLES in autoimmune GFAP astrocytopathy could be inflammatory infiltrations. Pathological studies have showed extensive inflammation, particularly around small vessels in all autoimmune GFAP astrocytopathy cases [6]. The rich vascular supply of the splenium mainly by the vertebrobasilar system could have led to intense inflammatory infiltrations in this area, leading to splenial lesion [4].
In most cases, encephalitis/encephalopathy with reversible splenial lesion showed relatively mild CNS manifestation and completely recovered within 1 month, sometimes also termed as mild encephalitis/encephalopathy with a reversible splenial lesion (MERS) [9]. However, recent studies have indicated that encephalitis/encephalopathy with reversible splenial lesion could not be simply considered as a mild clinical disorder, especially in patients who required ventilator supporting in acute phase of the disease [10]. Although there had been no established treatment guidelines, immunotherapy seems to be effective for neuronal/glial antibody-related RESLES based on limited evidences [3–5].
In conclusion, anti-GFAP autoimmunity should be added to the list of potential causes of RESLES. Autoimmune GFAP astrocytopathy should be considered when brain MRI confirms RESLES. Timely immunotherapy including corticosteroid and IVIg may improve the symptoms and benefit the long-term outcomes. Further study of future cases can promote a better understanding of the specific mechanism of autoimmune neurological antibody associated RESLES.
Acknowledgements
The authors thank the study participant for the participation in our study.
Author contribution
K. Guo and X. Lai collected data and drafted the manuscript. Y. Liu revised the figure. D. Zhou revised the manuscript. Z. Hong conceptualized and revised the manuscript.
Funding
Supported by the National Natural Science Foundation of China (grants 81671291 and 81971213).
Data statement
Further anonymized data can be made available to qualified investigators upon request to the corresponding author.
Declarations
Ethical approval
None.
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Kundian Guo and Xiaohui Lai have contributed equally to the manuscript.
Contributor Information
Kundian Guo, Email: 93502563@qq.com.
Xiaohui Lai, Email: 441607140@qq.com.
Yue Liu, Email: 979642317@qq.com.
Dong Zhou, Email: zhoudong66@yahoo.de.
Zhen Hong, Email: hongzhengoog@aliyun.com.
References
- 1.Garcia-Monco JC, Cortina IE, Ferreira E, et al. Reversible splenial lesion syndrome (RESLES): what’s in a name? J Neuroimaging. 2011;21(2):e1–14. doi: 10.1111/j.1552-6569.2008.00279.x. [DOI] [PubMed] [Google Scholar]
- 2.Liu J, Liu D, Yang B, et al. Reversible splenial lesion syndrome (RESLES) coinciding with cerebral venous thrombosis: a report of two cases. Ther Adv Neurol Disord. 2017;10(12):375–379. doi: 10.1177/1756285617727978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kaminski JA, Pruss H. N-methyl-d-aspartate receptor encephalitis with a reversible splenial lesion. Eur J Neurol. 2019;26(6):e68–e69. doi: 10.1111/ene.13900. [DOI] [PubMed] [Google Scholar]
- 4.Gilder TR, Hawley JS, Theeler BJ. Association of reversible splenial lesion syndrome (RESLES) with Anti-VGKC autoantibody syndrome: a case report. Neurol Sci. 2016;37(5):817–819. doi: 10.1007/s10072-015-2464-y. [DOI] [PubMed] [Google Scholar]
- 5.Renard D, Taieb G, Briere C, et al. Mild encephalitis/encephalopathy with a reversible splenial, white matter, putaminal, and thalamic lesions following anti-Yo rhombencephalitis. Acta Neurol Belg. 2012;112(4):405–407. doi: 10.1007/s13760-012-0080-7. [DOI] [PubMed] [Google Scholar]
- 6.Shan F, Long Y, Qiu W. Autoimmune glial fibrillary acidic protein astrocytopathy: a review of the literature. Front Immunol. 2018;9:2802. doi: 10.3389/fimmu.2018.02802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oger V, Bost C, Salah L, et al. Mild encephalitis/encephalopathy with reversible splenial lesion syndrome: an unusual presentation of anti-GFAP astrocytopathy. Eur J Paediatr Neurol. 2020;26:89–91. doi: 10.1016/j.ejpn.2020.03.002. [DOI] [PubMed] [Google Scholar]
- 8.Issa N, Martin C, Dulau C, et al. Severe anti-GFAP meningo-encephalomyelitis following viral infection. Mult Scler Relat Disord. 2020;45:102448. doi: 10.1016/j.msard.2020.102448. [DOI] [PubMed] [Google Scholar]
- 9.Tada H, Takanashi J, Barkovich AJ, et al. Clinically mild encephalitis/encephalopathy with a reversible splenial lesion. Neurology. 2004;63(10):1854–1858. doi: 10.1212/01.wnl.0000144274.12174.cb. [DOI] [PubMed] [Google Scholar]
- 10.Zhu Y, Zheng J, Zhang L, et al. Reversible splenial lesion syndrome associated with encephalitis/encephalopathy presenting with great clinical heterogeneity. BMC Neurol. 2016;16:49. doi: 10.1186/s12883-016-0572-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Further anonymized data can be made available to qualified investigators upon request to the corresponding author.