Abstract
Soil-transmitted helminths infect billions of people globally, particularly those residing in low- and middle-income regions with poor environmental sanitation and high levels of air and water pollution. Helminths display potent immunomodulatory activity by activating T helper type 2 (Th2) anti-inflammatory and Th3 regulatory immune responses. The Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), the virus that causes Coronavirus disease 2019 (COVID-19), can exacerbate Th1/Th17 pro-inflammatory cytokine production in humans, leading to a cytokine storm. Air pollutants (particulate matter, oxygen radicals, hydrocarbons and volatile organic compounds) and water pollutants (metals and organic chemicals) can also intensify Th1/Th17 immune response and could exacerbate SARS-CoV-2 related respiratory distress and failure. The present review focused on the epidemiology of SARS-CoV-2, helminths and fine particulate matter 2.5 µm or less in diameter (PM2.5) air pollution exposure in helminth endemic regions, the possible immunomodulatory activity of helminths against SARS-CoV-2 hyper-inflammatory immune response, and whether air and water pollutants can further exacerbate SARS-CoV-2 related cytokine storm and in the process hinder helminths immunomodulatory functionality. Helminth Th2/Th3 immune response is associated with reductions in lung inflammation and damage, and decreased expression levels of angiotensin-converting enzyme 2 (ACE2) receptors (SARS-CoV-2 uses the ACE2 receptors to infect cells and associated with extensive lung damage). However, air pollutants are associated with overexpression of ACE2 receptors in the epithelial cell surface of the respiratory tract and exhaustion of Th2 immune response. Helminth-induced immunosuppression activity reduces vaccination efficacy, and diminishes vital Th1 cytokine production immune responses that are crucial for combating early stage infections. This could be reversed by continuous air pollution exposure which is known to intensify Th1 pro-inflammatory cytokine production to a point where the immunosuppressive activities of helminths could be hindered. Again, suppressed activities of helminths can also be disadvantageous against SARS-CoV-2 inflammatory response. This “yin and yang” approach seems complex and requires more understanding. Further studies are warranted in a cohort of SARS-CoV-2 infected individuals residing in helminths and air pollution endemic regions to offer more insights, and to impact mass periodic deworming programmes and environmental health policies.
Keywords: SARS-CoV-2/ COVID-19, Environmental pollution, Th1/Th17-induced pro-inflammatory immune response, Cytokine storm, Helminth-induced Th2/Th3 anti-inflammatory immune response, Immunomodulatory activity of helminths
Abbreviations: ACE2, angiotensin-converting enzyme 2; ADCC, antibody-dependent cellular cytotoxicity; AREG, amphiregulin; AT1/AT2, alveolar type 1/2; BCG, bacillus Calmette–Guérin; CCL, chemokine (C-C motif) ligand; CO, carbon monoxide; COVID-19, coronavirus disease 2019; CXCL, chemokine (C-X-C motif) ligand; EGF, epidermal growth factor; G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; IFN-γ, interferon gamma; IgE, immunoglobulin E; IgG, immunoglobulin G; IL, interleukin; MCP, monocyte chemotactic protein; MIP, macrophage inflammatory protein; NO2, nitrogen dioxide; O3, ozone; PDGF, platelet-derived growth factor; PM, particulate matter; RANTES, regulated upon activation, normal T cell expressed and presumably secreted; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; sCD40L, soluble CD40 ligand; SDF1, stromal cell-derived factor 1; sICAM-1, soluble intercellular adhesion molecule-1; SO2, sulphur dioxide; sVCAM-1, soluble Vascular Cell Adhesion Molecule-1; TGF-β, transforming growth factor beta; Th1/Th17, T helper cell 1/17; TMPRSS2, transmembrane protease serine 2; TNF-α, tumour necrosis factor alpha; Tregs, regulatory T cells; TSLP, thymic stromal lymphopoietin; VEGF, vascular endothelial growth factor
1. Introduction
Soil-transmitted helminths (Ascaris lumbricoides, Trichuris trichiura, Strongyloides stercoralis, Necator americanus and Ancylostoma duodenale) and Enterobius vermicularis are parasitic intestinal worms vastly affecting people residing in underdeveloped areas with poor environmental sanitation (Hawdon 2014). According to the World Health Organization (WHO), over 1.5 billion people, which equates to 24% of the world’s population, are infected with soil-transmitted helminths. Additionally, over 267 million and 568 million preschool-age and school-age children respectively, reside in areas were helminths are intensively transmitted (World Health Organization, 2020). Infections are most prevalent in tropical and subtropical areas, with the highest numbers occurring in the Americas, East Asia, China and Sub-Saharan Africa (World Health Organization, 2020). It is estimated that around 300 million people with heavy helminth infections display severe morbidity, resulting in over 150,000 deaths annually (Hotez et al., 2006).
Lately, the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), and the associated Coronavirus Disease 2019 (COVID-19) have become a major global threat to human health. By April 24, 2021, the ever-escalating global infection rate, death rate and recovery rate is estimated to be 146,520,027, 3,103,334 and 124,259,424, respectively (Worldometer, 2021).
Patients with COVID-19 display abnormally high plasma concentrations of pro-inflammatory T lymphocyte (T cell) helper type 1 (Th1) immune cells and cytokines including TNF-α, IFN-γ, IL-2, IL-6, IL-7, IL-8, IL-17, G-CSF, GM-CSF, MCP-1 (CCL2), MIP-1α (CCL3), MIP-1β (CCL4) and IP-10 (CXCL10) which proportionally increase with disease severity (Huang et al., 2020, Schett et al., 2020).
Helminths are known to have potent immunomodulatory effects in the body through suppression of Th1 pro-inflammatory immune cells and activation of anti-inflammatory Th2 immune responses which are characterised by the production of IL-4, IL-5, IL-9, IL-10, IL-13, IgE, Tregs, mast cells, M2 macrophages, eosinophils and TGF-β (Abdoli and Ardakani, 2020). However, not all helminths induce the aforementioned immunomodulatory effects in the body (McKay, 2015). Hence, helminths can either be beneficial or disadvantageous in ameliorating or exacerbating the severity of inflammatory diseases, respectively (Abdoli and Ardakani, 2020, McKay, 2015).
Recently, the therapeutic role of helminths in the regulation and/or dysregulation of immune responses in patients with COVID-19 have become a debatable issue. Bradbury and colleagues first hypothesized that helminths could insidiously trigger the onset of severe COVID-19 infections in humans based on the theory that helminths could suppress vital Th1 immune response that is crucial in combating early stage bacterial and viral infections (Bradbury et al., 2020). Similar findings were reported elsewhere which further claimed that helminths could intensify COVID-19 morbidity and mortality (Abdoli, 2020). Conversely, Hays and colleagues hypothesized that helminth-induced Th2 anti-inflammatory responses could alleviate exacerbated inflammation in COVID-19 patients with co-morbidities such as obesity, metabolic syndrome and type 2 diabetes (Hays et al., 2020). Additionally, studies have associated helminth endemic regions with reduced COVID-19 severity and mortality rates (Gebrecherkos et al., 2021, Rodriguez, 2020).
Environmental pollution, an activator of pro-inflammatory Th1 immune responses, is proposed to also exacerbate COVID-19 severity and insidiously dampen the immunomodulatory activity of helminths in COVID-19 patients (Dvorožňáková et al., 2016, Frontera et al., 2020, Saraswathy et al., 2018). Globally, around 92% of the world population breathes unhealthy levels of air pollution (World Health Organization, 2016). The WHO reported that air pollution accounts for over 6.5 million deaths worldwide annually. This is equivalent to 11.6% (1 in every 9 deaths) of all global deaths annually, and over 90% of deaths occurred in low and middle-income countries. Over 6.1 million deaths (94%) occurred due to air pollution-induced non-communicable diseases (chronic obstructive pulmonary disease, cardiovascular disease, lung cancer and stroke) (World Health Organization, 2016). Studies have associated air pollution, a potent oxidative stress and inflammation inducing agent, with adverse alterations in host immune system and cytokine production (Gao et al., 2020, Pope et al., 2016, Saraswathy et al., 2018) and COVID-19 severity (Frontera et al., 2020) in humans. Drinking water pollutants (plastic components, plasticizers, metals, phthalates, bisphenol A and per- and polyfluoroalkyl substances) have also been reported to compromise the immune system which results in exacerbation of COVID-19 respiratory symptoms in humans (Quinete and Hauser-Davis, 2020).
Studies focusing on helminth and SARS-CoV-2 co-infections in areas affected with environmental pollution are scarce. The present review highlights the: (i) prevalence of SARS-CoV-2, helminths and fine particulate matter 2.5 µm or less in diameter (PM2.5) air pollution exposure in helminth endemic regions, (ii) regulatory role of helminths, (iii) dysregulatory role of environmental pollution, and (iv) regulatory/dysregulatory role of helminths and environmental pollution co-exposure in response to SARS-CoV-2 immune responses and severity.
2. Severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2)
2.1. Overview on SARS-CoV-2
Coronaviruses are large, positive-stranded, enveloped RNA viruses. The viral envelope is associated with at least four structural proteins namely nucleocapsid nucleoprotein, membrane, envelope and spike proteins (Malik, 2020). To date, seven human coronaviruses have been identified which are known to cause mild [HKU1, NL63 and OC43 (Betacoronaviruses) and 229E (Alphacoronavirus)] and severe [Middle East Respiratory Syndrome Coronavirus (MERS-CoV), SARS-CoV and SARS-CoV-2] respiratory illness and disease (Liu et al., 2020).
Patients with co-infections or co-morbidities such as obesity, human immunodeficiency virus (HIV) and tuberculosis, hypertension, metabolic syndrome, diabetes, cardiovascular disorders, kidney disease and respiratory disorders are most at risk of developing severe COVID-19 (Zhou et al., 2020). Patients with COVID-19 in Wuhan, China displayed symptoms of dyspnoea and lymphopenia and developed acute respiratory distress syndrome, acute cardiac injury, RNAaemia and secondary infection complications (Huang et al., 2020).
SARS-CoV-2 utilizes host cell factors, angiotensin-converting enzyme 2 (ACE2) and transmembrane protease serine 2 (TMPRSS2), to enter cells (Hoffmann et al., 2020). The SARS-CoV-2 spike protein interacts with the ACE2 receptor to promote cell entry while the TMPRSS2 plays a role in SARS-CoV-2 spike protein priming and cleavage, allowing viral and cellular membrane fusion and entry of the virus into cells (Hoffmann et al., 2020).
ACE2, an 805-amino acids captopril-insensitive carboxypeptidase, is expressed in lung alveolar epithelial cells, nasal epithelial cells, arterial and venous endothelial cells, enterocytes and arterial smooth muscle cells. ACE2 is an important regulator in lung diseases, diabetes, hypertension and cardiovascular diseases (Alifano et al., 2020). TMPRSS2, a 70-kDa serine protease, is associated with several physiological and pathological processes namely digestion, blood coagulation, tissue remodelling, tumour cell invasion, inflammatory responses and apoptosis (Antalis et al., 2011).
ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells and shown to be most vulnerable to SARS-CoV-2 infection (Lukassen et al., 2020). Studies have speculated that binding of SARS-CoV-2 to ACE2 could negatively affect the overall functionality of ACE2, leading to the overproduction of reactive oxygen species and the pathophysiology of cardiovascular and respiratory failure (Alifano et al., 2020).
Studies have found that children have lower SARS-CoV-2 infection rate, display milder symptoms and have better clinical outcomes compared to adults (Ludvigsson, 2020). Researchers attributed this to lower ACE2 gene expression levels in nasal passage epithelial cells taken from children compared to adults. It was further concluded that ACE2 gene expression was age-dependent and increases with age into adulthood (Bunyavanich et al., 2020). Researchers also investigated whether lung ACE2 and TMPRSS2 expression levels in children and adults vary using single-cell RNA sequencing (scRNA-seq) and immunohistochemistry. They also explored whether gene expression levels might influence milder SARS-CoV-2 symptoms in children. No significant changes in lung ACE2 and TMPRSS2 expression levels were observed between children and adults, even though the percentage of cells expressing TMPRSS2 were higher in adults. Alveolar type 2 (AT2) cells had the highest ACE2 expression while alveolar type 1 (AT1) cells in children and club cells in adults also showed noticeable expression. In both adults and children, AT1, AT2 and club cells were the main cell types with relatively high TMPRSS2 expression. It was concluded that lung ACE2 and TMPRSS2 expression levels was unlikely to be a major reasoning behind lower virus intrusion rate and milder symptoms in children, and unique features in children’s underdeveloped immunity systems may play a far more pivotal role (Tao et al., 2020). A possible explanation is that children have decreased mononuclear and polymorphonuclear chemotaxis, and enhanced and trained innate and adaptive immune systems if they are frequently exposed to a broad spectrum of pathogens compared to adults (Tao et al., 2020).
2.2. SARS-CoV-2 variants
In comparison to the SARS-CoV outbreak in 2002 in Guangdong, China and MERS-CoV outbreak in 2012 in Jeddah, Saudi Arabia, the current SARS-CoV-2 which originated from Wuhan, China in 2019 is treacherously and potently more infectious in humans due to mutations in the SARS-CoV-2 spike glycoprotein (mediates viral entry into cells through cell receptor binding and membrane fusion) and nucleocapsid nucleoprotein (regulates viral replication and thus influences transcription and assembly) in addition to its ability to infect and reproduce in the upper respiratory tract (Lippi and Plebani, 2020).
Several mutations in the SARS-CoV-2 spike protein can greatly enhance viral infectivity and pathogenicity, and the Center for Disease Control and Prevention (CDC) classified these mutations as variants of concern and of high consequence. This includes the B.1.1.7 (501Y.V1), B.1.351 (501Y.V2), CAL.20C (B.1.427/ B.1.429) and B.1.1.28.1 (P.1) SARS-CoV-2 lineages that originated in the United Kingdom, South Africa, California and Brazil, respectively (McCallum et al., 2021, Singh et al., 2021). In April 2021, India report on the emergence of a more infectious SARS-CoV-2 variant, B.1.617, that has the ability to enhance viral replication and evade antibodies. The B.1.617 variant carries two mutations known as E484Q and L452R. The E484Q mutation mirrors E484K, a mutation found in the South African (B.1.351) and United Kingdom (B.1.1.7) variants, while the L452R mutation is also found in the highly transmissible Californian (B.1.427/ B.1.429) variants (The Indian Express, 2021). With the ongoing emergence and discovery of highly infectious SARS-CoV-2 variant strains, this could lead to changes in pathogenicity patterns in children, and trigger higher morbidity and mortality rates in adults.
2.3. SARS-CoV-2 and the gut microbiota
SARS-CoV-2 also infects the mature human intestinal cells, the ACE2 receptors-expressing enterocytes of the small intestines in particular, which may lead to gastrointestinal symptoms such as abdominal pain, vomiting and diarrhoea during the early phases of COVID-19 (Dhar and Mohanty, 2020, Kaźmierczak-Siedlecka et al., 2020). Intestinal ACE2 receptors act as a chaperone for membrane trafficking of the sodium-dependent neutral amino acid transporter B0AT1. In the intestinal epithelium, the B0AT1/ACE2 complex regulates the composition and function of the gut microbiota, leading to ambiguous changes in host immune responses against pathogens (Viana et al., 2020).
Adverse changes in the gut microbiota composition in patients with active and severe SARS-CoV-2 gastrointestinal infections were reported. This included enrichment of opportunistic pathogens (Streptococcus infantis, Morganella morganii, Collinsella aerofaciens, Collinsella tanakaei, Coprobacillus, Clostridium ramosum, and Clostridium hathewayi), loss of short-chain fatty acid producing salutary bacteria (Alistipes onderdonkii, Lachnospiraceae bacterium, Parabacteroides merdae and Bacteroides stercoris) and enhanced functional capacity for carbohydrate metabolism and amino acid and nucleotide biosynthesis (Yeoh et al., 2021, Zuo et al., 2021, Zuo et al., 2020). In addition, as the severity of SARS-CoV-2 increased the abundance of the (i) anti-inflammatory/ immunomodulatory bacteria, Faecalibacterium prausnitzii, Eubacterium rectale and several bifidobacterial species, and (ii) ACE2 downregulating bacteria, Bacteroides massiliensis, Bacteroides ovatus, Bacteroides dorei and Bacteroides thetaiotaomicron significantly decreased (Yeoh et al., 2021, Zuo et al., 2020). As a consequence, the adverse changes in the gut microbiota, induced by SARS-CoV-2, may ultimately lead to the progression of gut dysbiosis-related diseases even after clearance of the virus, especially in elderly patients since immunity and gut microbiota diversity decreases with age (Dhar and Mohanty, 2020, Kaźmierczak-Siedlecka et al., 2020, Yeoh et al., 2021).
2.4. SARS-CoV-2, environmental pollution and cigarette smoking
Studies examining the effects of environmental pollution and cigarette smoking on COVID-19 severity are summarised in Table 1 . Chronic exposure to air pollution [particulate matter (PM), nitrogen dioxide (NO2), sulphur dioxide (SO2), ozone (O3), carbon monoxide (CO), polycyclic aromatic hydrocarbons and volatile organic compounds] can exacerbate SARS-CoV-2 related adverse health outcomes (respiratory distress and failure) and mortality rates (Domingo and Rovira, 2020, Konstantinoudis et al., 2021, Wu et al., 2020, Zhu et al., 2020, Bashir et al., 2020). Studies have postulated that the SARS-CoV-2 might have the ability to attach to airborne atmospheric particulates (PM2.5-10) which could be a contributing factor towards the high prevalence of indirect transmission rates in humans (Sanità di Toppiet al., 2020). Frontera and colleagues (2020) proposed a “double-hit hypothesis”. They found chronic exposure to PM2.5 and NO2 intensified SARS-CoV-2 related mortality rates. Chronic PM2.5 exposure led to the overexpression of alveolar ACE2 receptor which was associated with high viral load in patients. This then led to the exhaustion of ACE2 receptors and impaired host defences. Chronic NO2 exposure provided a second hit which caused a more severe form of SARS-CoV-2 in the ACE2 depleted lungs and was responsible for extensive lung damage (Frontera et al., 2020). Cigarette smoking and tobacco smoke exposure was also associated with increased ACE2 and TMPRSS2 expression which led to severe SARS-CoV-2 infections (Cai, 2020, Gülsen et al., 2020, Zhao et al., 2020).
Table 1.
Summary of studies focusing on the effects of environmental pollution and cigarette smoking on COVID-19 severity.
| References | Aim of study | Methodology | Major findings |
|---|---|---|---|
| Environmental pollution | |||
| Konstantinoudis et al., 2021 | - Temporal association between long-term air pollution (NO2 and PM2.5) and COVID-19 mortality in England. | - Cross-sectional study. - 38,573 COVID-19 deaths were reported up to 30 June 2021. - Air pollution concentrations were retrieved between 2014 and 2018. - Bayesian hierarchical models were used for data analysis. |
- A 1 µg/m3 increase in NO2 and PM2.5 was associated with a 0.5% (95% CrI: 0.2% − 1.2%) and 1.4% (95% CrI: 2.1%, − 5.1%) increase in COVID-19 mortality rate, respectively. - Posterior probability of a positive effect: NO2 (0.93) and PM2.5 (0.78). |
| Wu et al., 2020 | - Temporal association between long-term PM2.5 exposure and COVID-19 mortality in the U.S.A. | - Cross-sectional study. - COVID-19 death counts were collected from 3,000 counties in the U.S.A up to 22 April 2020. - Zero-inflated negative binomial models were used for data analysis. |
- A 1 µg/m3 increase in PM2.5 was associated with an 8% (95% CI: 2% − 15%) increase in COVID-19 mortality rate. |
| Zhu et al., 2020 | - Temporal association between air pollution (PM2.5, PM10, NO2 and O3) and daily confirmed COVID-19 cases in China. | - Retrospective study. - Air pollution concentration, meteorological variables and daily confirmed COVID-19 cases were retrieved between 23 January – 29 February 2020. - Generalized Additive Models (GAM) were used for data analysis. |
- A 10 μg/m3 increase (lag0–14) in O3, NO2, PM2.5 and PM10 was associated with a 4.76% (95% CI: 1.99–7.52), 6.94% (95% CI: 2.38–11.51), 2.24% (95% CI: 1.02–3.46), and 1.76% (95% CI: 0.89 to 2.63) increase in daily confirmed COVID-19 cases, respectively. |
| Bashir et al., 2020 | - Correlation between environmental pollution indicators (PM2.5, PM10, NO2, SO2, CO, VOC and Pb) and total confirmed cases and mortality of COVID-19 in California. | - Retrospective study. - Environmental pollution concentration and total confirmed cases and mortality of COVID-19 were retrieved between 4 March – 24 April 2020. - Kendall (K) and Spearman (S) correlation (r) tests were used for data analysis. |
Total confirmed cases: - PM10 (K r: −0.287; S r: −0.375) - PM2.5 (K r: −0.359; S r: −0.453) - SO2 (K r: −0.333; S r: −0.426) - CO (Kr: 0.045; S r: 0.083) - VOC (K r: 0.044; S r: 0.054) - Pb (K r: 0.109; S r: 0.178) - NO2 (K r: −0.514; S r: −0.736) Total confirmed mortality: - PM10 (K r: −0.267; S r: −0.350) - PM2.5 (K r: −0.339; S r: −0.429) - SO2 (K r: −0.309; S r: −0.397) - CO (Kr: 0.079; S r: 0.123) - VOC (K r: 0.032; S r: 0.038) - Pb (K r: 0.109; S r: 0.174) - NO2 (K r: −0.485; S r: −0.731) |
| Cigarette smoking | |||
| Gülsen et al., 2020 | - Association between smoking and clinical COVID-19 severity | - Systematic meta-analysis approach. − 16 articles published between December 2019 – 15 April 2020, and detailing 11,322 COVID-19 patients. |
- History of smoking and severe COVID-19 (OR: 2.17; 95% CI: 1.37–3.46; p < 0.001) (I2: 71.4%; p < 0.001). - Current smoking status and severe COVID-19 (fixed-effects model; OR: 1.51; 95% CI: 1.12–2.05; p < 0.008) (I2: 48.9%; p = 0.40. − 10.7% (978/9067) of non-smokers and 21.2% (65/305) of active smokers had severe COVID-19. |
| Zhao et al., 2020 | - Effect of pre-existing chronic obstructive pulmonary disease (COPD) and ongoing smoking history on COVID-19 severity. | - Systematic meta-analysis approach. − 11 articles published between December 2019 – 22 March 2020, and detailing 2002 COVID-19 patients. |
- Pre-existing COPD and development of severe COVID-19 (fixed-effects model; OR: 4.38; 95% CI: 2.34–8.20) (I2 = 41%; p = 0.08). - Ongoing smoking and severe COVID-19 (fixed‐effects model; OR: 1.98; 95% CI: 1.29‐3.05) (I2 = 44%; p = 0.10). - COPD and ongoing smoking collectively worsen COVID-19 severity and outcomes. |
| Cai, 2020 | - To investigate the disparity related to tobacco use in lung ACE2 gene expression and its distribution among cell types in order to predict COVID-19 susceptibility. | - Cross-sectional study - Large-scale bulk transcriptomic datasets of normal lung tissue were retrieved to investigate the disparity related to smoking status in ACE2 gene expression. - Single-cell transcriptomic datasets were retrieved to investigate the distribution of ACE2 gene expression among cell types. |
- Former smoker’s lung had higher ACE2 gene expression compared to non-smoker’s lung. - Bronchial epithelium: ACE2 is highly expressed in goblet cells and club cells of current smokers and non-smokers, respectively. - Alveoli: former smokers had high ACE2 expression in remodelled AT2 cells. - Collectively, smokers, particularly former smokers, were more susceptible to COVID-19. |
Woodby and colleagues (2021) hypothesized that air pollutants have the ability to promote SARS-CoV-2 entry and life cycle in the body through several mechanisms, including preventing antiviral activity of antimicrobial peptides and surfactant protein D, inhibition of mucociliary clearance, promoting TLR2 and TLR4 signalling that are specific for bacterial sensing, increasing ACE2 receptor expression levels, promoting TMPRSS2 cleavage, altering viral replication and assembly mediated by autophagy, modulating interferon production, preventing uptake by macrophages, and increasing epithelial permeability to promote viral spread (Woodby et al., 2021).
3. Intestinal helminths
Helminths are parasitic worms affecting billions of people globally. The main groups of parasitic helminths include: (i) Platyhelminthes (Flatworms), (ii) Nematodes (Roundworms), (iii) Acanthocephalins (Thorny-headed worms), (iv) Cestodes (Tapeworms), and (v) Trematodes (Flukes) (Lindquist and Cross, 2017, Parija et al., 2017). Majority of people worldwide are infected with soil-transmitted intestinal nematodes (Ascaris lumbricoides, Trichuris trichiura, Strongyloides stercoralis, Necator americanus and Ancylostoma duodenale) and Enterobius vermicularis. The morphological characteristics of soil-transmitted helminths are presented in Table 2 (Lindquist and Cross, 2017, Parija et al., 2017).
Table 2.
Global prevalence and morphological characteristics of intestinal helminths (Lindquist and Cross, 2017, Parija et al., 2017).
| Intestinal helminths | Global prevalence | Disease and morphology |
|---|---|---|
| Ascaris lumbricoides | 1.2 billion | - Disease in humans: Ascariasis - Adult male: 15–31 cm by 2–4 mm. - Adult female: 20–35 cm by 3–6 mm, produce 240 000 eggs/day. |
| Trichuris trichiura | 795 million | - Disease in humans: Trichuriasis - Adult male: 30–45 mm by 0.1–0.5 mm. - Adult female: 35–55 mm by 0.1–0.5 mm, produce 3000–10000 eggs/day. |
| Strongyloides stercoralis | 600 million | - Disease in humans: Strongyloidiasis - Adult males (free-living cycle only): 0.9 mm by 40–50 µm. - Adult females (free-living and parasitic cycles): 2.0–2.5 mm by 40–50 µm, produce 30–40 partially embryonated eggs/ day. |
| Necator americanus and Ancylostoma duodenale | 740 million | - Disease in humans: Necatoriasis and Ancylostomiasis - Adult male: 6–11 mm by 0.4–0.6 mm. - Adult female: 10–13 mm by 0.4–0.6 mm, produce 10 000–20 000 (A. duodenale) and 5000–10 000 (N. americanus) eggs/day. |
| Enterobius vermicularis | 200 million | - Disease in humans: Enterobiasis - Adult male: 2.5 mm by 0.1–0.2 mm. - Adult female: 8–13 mm long by 0.3–0.5 mm, produce 16 000 eggs/day. |
Helminthic infections (the most prevalent neglected tropical diseases) predominantly occur in the intestines as well as in the blood, lungs, liver and brain (Hotez et al., 2006). Risk factors associated with soil-transmitted intestinal helminth infection are low socioeconomic status, poor availability of clean water, poor sanitation and poor hygiene (Hotez et al., 2006). Mebendazole (most favoured), praziquantel, albendazole and diethylcarbamazine are some of the anthelmintic drugs used in the mass control and prevention of helminth infections (Dayan, 2003). Lately, ivermectin, an anti-parasitic and anthelmintic drug, was reported to inhibit the replication of SARS-CoV-2 in vitro (Caly et al., 2020).
Soil-transmitted helminth infections often tend to be mild or asymptomatic in humans however severe infections do occur leading to nutritional deficiencies, several allergy-related atopic diseases, mental delays, growth and development delays, organ failure and death (Cepon-Robins and Gildner, 2020).
Helminth infections can modify the gut microbiota composition in the intestines and exacerbate bacterial colitis (Su et al., 2018). They secrete excretory-secretory products that have antimicrobial activity (Abner et al., 2001, Brosschot and Reynolds, 2018), and also modify human production of anti-microbial peptides (Brosschot and Reynolds, 2018, Fricke et al., 2015). Helminths and Lactobacilli species have coevolved and display a mutualistic relationship with the aim of regulating mucosal immune responses in order to thrive in the intestinal environment; Lactobacilli species increases in abundance during chronic helminth infection and promotes the pathogenesis of persistent helminth infection (Brosschot and Reynolds, 2018). Nutritional deficiencies in the intestines and diminished epithelial glucose absorption are also associated with helminth infections (Brosschot and Reynolds, 2018, Shea-Donohue et al., 2001).
In a Malaysian cohort, helminth parasites (Ascaris species and Trichuris species) was associated with increased faecal abundance of Paraprevotellaceae (strongly associated with the Trichuris species), Alphaproteobacteria, Bacteroidales and Mollicutes, and enriched metabolic pathways involving amino acids and nucleotides biosynthesis in the microbiota. In helminth-negative patients, a high abundance of Bifidobacterium, and enriched xenobiotic and carbohydrate metabolism pathways was observed in the microbiota. Trichuris worms alone was associated with highly enriched pathways involved in nucleotide metabolism, and cell growth and death while Ascaris worms alone was correlated with reduced activity of carbohydrate metabolic pathways in the microbiota (Lee et al., 2014). In Zimbabwe, children infected with Schistosoma hematobium displayed increased faecal abundance of bacteria within the genus Prevotella (Kay et al., 2015).
4. Epidemiology of SARS-CoV-2, helminths and PM2.5 air pollution in helminth endemic regions
The prevalence of SARS-CoV-2, helminths and PM2.5 air pollution levels in helminth endemic WHO regions worldwide are summarized in Table 3, Table 4, Table 5 . Data for helminths (World Health Organization, 2021a) and PM2.5 air pollution (World Health Organization, 2021b) prevalence were retrieved from the WHO online website. Data for the prevalence of SARS-CoV-2 (total cases, total deaths and total recovered) was retrieved from the Worldometer online website that gives real time world statistics (Worldometer, 2021). Helminth endemic countries or territories are grouped into 6 WHO regions: Africa, The Americas, Eastern Mediterranean, European region, South East Asia and Western Pacific.
Table 3.
Prevalence of helminths, COVID-19 and PM2.5 air pollution in Sub-Saharan African countries.
| Countries ranked based on helminth prevalence | Total population, n | Helminths prevalence, n | COVID-19 prevalence up to 19 April 2021, n |
Air quality index (AQI) | ||
|---|---|---|---|---|---|---|
| Total cases, n | Total deaths, n | Total recovered, n | PM2.5 (µg/m3), mean [95% CI] | |||
| Nigeria | 210,162,742 | 45,205,565 | 164,207 | 2,061 | 154,325 | 48.73 [41.08–125.8] |
| Democratic Republic of Congo | 91,700,032 | 29,643,689 | 28,894 | 745 | 25,841 | 37.62 [22.75–74.02] |
| Ethiopia | 117,205,948 | 27,758,240 | 240,236 | 3,328 | 178,705 | 34.36 [22.68–58.24] |
| United Republic of Tanzania | 61,071,876 | 18,788,363 | 509 | 21 | 183 | 25.59 [18.57–47.77] |
| Uganda | 46,875,190 | 17,938,970 | 41,378 | 338 | 40,898 | 48.41 [27.91–79.63] |
| South Africa | 59,900,468 | 15,827,969 | 1,565,680 | 53,711 | 1,489,457 | 23.58 [17.99–33.35] |
| Angola | 33,669,143 | 12,194,554 | 24,300 | 561 | 22,576 | 27.95 [16.01–53.83] |
| Ghana | 31,584,523 | 10,904,105 | 91,663 | 771 | 89,530 | 31.95 [18.53–66.8] |
| Mozambique | 31,944,020 | 10,862,269 | 69,134 | 798 | 61,207 | 19.44 [12.45–33.08] |
| Niger | 24,893,250 | 10,391,142 | 5,116 | 190 | 4,771 | 70.8 [35.94–250.7] |
| Cameroon | 27,066,166 | 10,100,056 | 64,809 | 939 | 57,821 | 65.26 [43.03–113.4] |
| Madagascar | 28,251,355 | 8,124,525 | 32,205 | 542 | 25,798 | 21.44 [13.66–36.05] |
| Malawi | 19,519,025 | 7,414,608 | 33,919 | 1,136 | 31,717 | 22.14 [12.09–40.26] |
| Kenya | 54,705,045 | 5,937,691 | 151,287 | 2,463 | 101,362 | 25.85 [17.78–44.07] |
| Zimbabwe | 15,034,019 | 5,849,280 | 37,699 | 1,552 | 35,005 | 19.35 [10.38–35.2] |
| Zambia | 18,788,816 | 4,733,164 | 90,918 | 1,235 | 88,718 | 24.7 [13.41–45.28] |
| Rwanda | 13,205,039 | 3,987,005 | 23,866 | 322 | 22,082 | 40.75 [16.13–96.97] |
| Sierra Leone | 8,104,959 | 3,420,897 | 4,020 | 79 | 2,848 | 20.63 [11.09–42.47] |
| Togo | 8,431,597 | 2,393,570 | 12,496 | 119 | 10,163 | 32.71 [19.72–65.19] |
| Guinea | 13,413,017 | 2,383,636 | 21,460 | 138 | 18,882 | 22.43 [12.3–47.42] |
| Cote d’Ivoire | 26,920,447 | 2,280,850 | 45,519 | 274 | 45,064 | 23.72 [14.53–49.57] |
| Burundi | 12,169,053 | 2,242,532 | 3,477 | 6 | 773 | 35.61 [15.11–91.6] |
| Benin | 12,373,092 | 2,208,676 | 7,611 | 95 | 6,728 | 33.11 [20.65–68.88] |
| Senegal | 17,091,143 | 1,077,768 | 39,782 | 1,091 | 38,511 | 37.52 [24.54–67.85] |
| Central African Republic | 4,896,095 | 996,622 | 5,728 | 75 | 5,112 | 49.5 [24.96–120.6] |
| Liberia | 5,151,337 | 903,257 | 2,042 | 85 | 1,899 | 17.19 [11.2–28.18] |
| Namibia | 2,577,241 | 810,983 | 46,515 | 602 | 44,437 | 22.59 [12.16–46.31] |
| Congo | 5,625,046 | 809,022 | 10,084 | 139 | 8,208 | 38.67 [18.16–94.74] |
| South Sudan | 11,297,620 | 669,759 | 10,432 | 114 | 10,148 | 41.12 [21.73–87.92] |
| Chad | 16,796,394 | 544,879 | 4,691 | 168 | 4,335 | 53.01 [23.25–132.4] |
| Gabon | 2,267,109 | 483,207 | 21,858 | 133 | 18,507 | 38.51 [19.88–85.08] |
| Lesotho | 2,155,713 | 382,336 | 10,709 | 315 | 5,028 | 27.78 [16.82–43.44] |
| Comoros | 884,209 | 231,998 | 3,815 | 146 | 3,589 | 18.60 [9.22–38.73] |
| Equatorial Guinea | 1,439,155 | 144,713 | 7,259 | 106 | 6,885 | 45.9 [21.86–108] |
| Cabo Verde | 560,769 | 137,073 | 19,975 | 189 | 17,870 | 31.99 [16.01–61.12] |
| Guinea-Bissau | 2,004,645 | 129,110 | 3,710 | 66 | 3,117 | 27.12 [14.26–62.11] |
| Botswana | 2,389,088 | 116,050 | 44,075 | 671 | 39,493 | 21.24 [13.38–35.22] |
| Sao Tome and Principe | 222,372 | 77,429 | 2,275 | 35 | 2,210 | 25.66 [14.26–52.11] |
| Gambia | 2,470,054 | 70,776 | 5,720 | 170 | 5,190 | 32.2 [17.46–61.81] |
| Eswatini | 1,169,679 | 16,659 | 18,415 | 671 | 17,682 | 16.26 [10.32–25.61] |
| Overall prevalence, n (%) | 1,045,986,491 (13,31)a | 268,192,997 (25,64)b | 3,017,488 (0,29)c | 76,200 (2.53)d | 2,746,675 (91.03)e | Average PM2.5:32.52 [18.58–68.77] |
% Overall prevalence (Total population) = Total population in Sub-Saharan Africa / World population as of 21 April 2021 (7,860,798,912) × 100.
% Overall prevalence (Helminths prevalence) = Total helminths prevalence in Sub-Saharan Africa / Total population in Sub-Saharan Africa × 100.
% Overall prevalence (Total COVID-19 cases) = Total COVID-19 cases in Sub-Saharan Africa / Total population in Sub-Saharan Africa × 100.
% Overall prevalence (Total COVID-19 deaths) = Total COVID-19 deaths in Sub-Saharan Africa / Total COVID-19 cases in Sub-Saharan Africa × 100.
% Overall prevalence (Total COVID-19 recovery rate) = Total COVID-19 recovery rate in Sub-Saharan Africa / Total COVID-19 cases in Sub-Saharan Africa × 100.
Table 4.
Prevalence of helminths, COVID-19 and PM2.5 air pollution in the Americas, Eastern Mediterranean and European region.
| Countries ranked based on helminth prevalence | Total population, n | Helminths prevalence, n |
COVID-19 prevalence up to 19 April 2021, n |
Air quality index (AQI) | ||
|---|---|---|---|---|---|---|
| Total cases, n | Total deaths, n | Total recovered, n | PM2.5 (µg/m3), mean [95% CI] | |||
| The Americas | ||||||
| Mexico | 130,009,363 | 19,900,177 | 2,311,172 | 213,048 | 1,836,377 | 20.08 [16.66–27.19] |
| Brazil | 213,761,802 | 9,547,212 | 13,943,071 | 373,442 | 12,391,599 | 11.49 [10.92–14.92] |
| Venezuela | 28,371,608 | 8,049,594 | 183,190 | 1,905 | 165,858 | 15.82 [10.85–25.87] |
| Guatemala | 18,178,124 | 4,957,871 | 212,734 | 7,21 | 190,944 | 23.59 [17.8–30.87] |
| Colombia | 51,313,902 | 3,186,736 | 2,652,947 | 68,328 | 2,471,498 | 15.24 [11.62–23.37] |
| Dominican Republic | 10,934,383 | 2,749,409 | 261,129 | 3,418 | 218,724 | 12.95 [6.38–26.69] |
| Honduras | 10,029,541 | 2,214,843 | 200,935 | 4,957 | 76,339 | 20.12 [14.64–28.86] |
| Paraguay | 7,202,123 | 1,974,836 | 250,165 | 5,313 | 205,883 | 11.16 [7.56–16.46] |
| Nicaragua | 6,687,161 | 1,611,101 | 6,778 | 180 | 4,225 | 16.87 [11.29–25.23] |
| El Salvador | 6,512,268 | 1,443,743 | 67,557 | 2,078 | 63,787 | 23.42 [16.14–36.25] |
| Haiti | 11,512,932 | 920,867 | 12,918 | 251 | 11,791 | 14.63 [7.6–31.21] |
| Peru | 33,336,408 | 342,185 | 1,704,757 | 57,230 | 1,627,969 | 24.27 [20.32–32.81] |
| Guyana | 789,575 | 204,303 | 11,863 | 271 | 10,180 | 20.46 [9.29–43.63] |
| Bolivia | 11,799,417 | 190,910 | 289,066 | 12,648 | 238,195 | 20.24 [14.83–32.18] |
| Argentina | 45,525,551 | 160,777 | 2,694,014 | 59,228 | 2,364,171 | 11.83 [9.82–18.99] |
| Panama | 4,368,639 | 51,175 | 360,841 | 6,188 | 350,610 | 11.18 [8.62–15.18] |
| Antigua and Barbuda | 98,574 | 1,201 | 1,216 | 31 | 986 | 17.92 [10.4–34.1] |
| Saint Vincent and Grenadines | 111,223 | 702 | 1819 | 10 | 1677 | 21.2 [11.39–43.17] |
| Dominica | 72,129 | 551 | 165 | 0 | 159 | 18.17 [10.84–34.39] |
| Overall prevalence, n (%) | 590,614,723 (7,51)a | 57,508,193 (9,74)b | 25,160,767 (4,26)c | 808,538 (3,22)d | 45,355,837 (88,34)e | Average PM2.5: 17.40 [11.95–28.49] |
| Eastern Mediterranean | ||||||
| Pakistan | 224,289,117 | 25,234,450 | 761,437 | 16,316 | 662,845 | 55.21 [46.7–75.74] |
| Afghanistan | 39,621,758 | 16,222,053 | 58,038 | 2,550 | 52,244 | 53.17 [40.08–75.76] |
| Yemen | 30,345,627 | 9,833,727 | 5,858 | 1,132 | 2,261 | 44.96 [20.93–111.1] |
| Syrian Arab Republic | 17,837,821 | 2,440,286 | 21,279 | 1,456 | 14,958 | 39.43 [32.43–58.91] |
| Iraq | 40,936,402 | 2,170,486 | 984,950 | 15,026 | 864,039 | 57.73 [42.24–87.6] |
| Somalia | 16,243,130 | 1,972,987 | 13,079 | 670 | 5,413 | 29.51 [17–56.06] |
| Sudan | 44,658,737 | 1,217,659 | 31,790 | 2,208 | 25,539 | 47.92 [25.05–113.4] |
| Djibouti | 999,401 | 110,561 | 10,460 | 116 | 9,172 | 40.38 [16.09–111.5] |
| Overall prevalence, n (%) | 414,931,993 (5,28)a | 59,202,209 (14,27)b | 1,886,891 (0,45)c | 39,474 (2,09)d | 1,636,471 (86,73)e | Average PM2.5: 46.04 [20.07–86.26] |
| European region | ||||||
| Tajikistan | 9,707,083 | 3,161,746 | 13,308 | 90 | 13,218 | 40.05 [19.93–84.69] |
| Kyrgyzstan | 6,609,541 | 2,169,854 | 92,095 | 1,555 | 87,056 | 18.12 [12.77–35.24] |
| Azerbaijan | 10,211,851 | 686,000 | 301,661 | 4,169 | 265,539 | 18.2 [10.8–35.39] |
| Uzbekistan | 33,854,114 | 405,951 | 86,982 | 638 | 84,307 | 25.29 [13.15–51.5] |
| Overall prevalence, n (%) | 60,382,589 (0,77)a | 6,423,551 (10,64)b | 494,046 (0,82)c | 6,452 (1,31)d | 450,120 (91,11)e | Average PM2.5: 25.42 [14.16–51.71] |
% Overall prevalence (Total population) = Total population in The Americas or Eastern Mediterranean or European region / World population as of 21 April 2021 (7,860,798,912) × 100.
% Overall prevalence (Helminths prevalence) = Total helminths prevalence in The Americas or Eastern Mediterranean or European region / Total population in The Americas or Eastern Mediterranean or European region × 100.
% Overall prevalence (Total COVID-19 cases) = Total COVID-19 cases in The Americas or Eastern Mediterranean or European region / Total population in The Americas or Eastern Mediterranean or European region × 100.
% Overall prevalence (Total COVID-19 deaths) = Total COVID-19 deaths in The Americas or Eastern Mediterranean or European region / Total COVID-19 cases in The Americas or Eastern Mediterranean or European region × 100.
% Overall prevalence (Total COVID-19 recovery rate) = Total COVID-19 recovery rate in The Americas or Eastern Mediterranean or European region / Total COVID-19 cases in The Americas or Eastern Mediterranean or European region × 100.
Table 5.
Prevalence of helminths, COVID-19 and PM2.5 air pollution in South East Asia and Western Pacific.
| Countries ranked based on helminth prevalence | Total population, n | Helminths prevalence, n |
COVID-19 prevalence up to 19 April 2021, n |
Air quality index (AQI) |
||
|---|---|---|---|---|---|---|
| Total cases, n | Total deaths, n | Total recovered, n | PM2.5 (µg/m3), mean [95% CI] | |||
| South East Asia | ||||||
| India | 1,390,790,274 | 435,638,057 | 15,295,118 | 180,322 | 13,088,427 | 65.2 [78.61–96.56] |
| Indonesia | 275,822,293 | 70,642,364 | 1,609,300 | 43,567 | 1,461,414 | 15.58 [13.54–20.16] |
| Bangladesh | 165,993,675 | 56,339,394 | 723,221 | 10,497 | 621,300 | 58.33 [51.02–68.72] |
| Myanmar | 54,700,104 | 12,575,540 | 142,644 | 3,206 | 131,903 | 34.69 [31.44–41.24] |
| Nepal | 29,552,077 | 8,573,482 | 285,900 | 3,091 | 275,555 | 94.33 [70.99–139.8] |
| Democratic People’s Republic of Korea | 51,304,493 | 5,418,928 | 115,195 | 1,802 | 105,227 | 30.4 [23.64–43.35] |
| Timor-Leste | 1,338,335 | 467,672 | 1,368 | 2 | 695 | 17.88 [8.25–39.37] |
| Bhutan | 778,359 | 229,846 | 961 | 1 | 881 | 35.32 [32.75–44.99] |
| Overall prevalence, n (%) | 1,970,279,610 (25,06)a | 589,885,283 (29,94)b | 18,173,707 (0,92)c | 242,488(1,33)d | 15,685,402 (86,31)e | Average PM2.5: 43.97 [38.78–61.77] |
| Western Pacific | ||||||
| Philippines | 110,738,989 | 45,437,063 | 945,745 | 16,048 | 788,322 | 18.38 [15.07–21.35] |
| China | 1,439,323,776 | 26,222,138 | 90,510 | 4,636 | 85,559 | 49.16 [49.66–53.77] |
| Vietnam | 98,035,043 | 7,368,702 | 2,791 | 35 | 2,475 | 29.66 [24.57–37.08] |
| Cambodia | 16,902,622 | 5,000,734 | 7,031 | 45 | 2,524 | 23.98 [14.44–37.96] |
| Papua New Guinea | 9,081,270 | 3,511,004 | 9,738 | 89 | 846 | 10.91 [4.38–30.76] |
| Laos People’s Democratic Republic | 7,359,625 | 2,173,051 | 58 | 0 | 49 | 24.49 [19.08–34.95] |
| Fiji | 901,614 | 332,494 | 72 | 2 | 65 | 10.19 [4.67–22.1] |
| Solomon Islands | 700,195 | 251,562 | 20 | 0 | 18 | 10.67 [4.58–27.84] |
| Vanuatu | 312,820 | 115,109 | 3 | 0 | 1 | 10.31 [4.38–26.32] |
| Tonga | 106,672 | 37,131 | 0 | 0 | 0 | 10.08 [5.03–21.9] |
| Micronesia | 115,981 | 36,871 | 1 | 0 | 1 | 10.23 [4.5–24.93] |
| Kiribati | 120,955 | 33,294 | 0 | 0 | 0 | 10.45 [4.69–24.1] |
| Marshall Islands | 59,508 | 19,594 | 4 | 0 | 4 | 9.43 [4.52–21.78] |
| Tuvalu | 11,911 | 3,120 | 0 | 0 | 0 | 11.42 [7.26–16.56] |
| Nauru | 10,879 | 2,844 | 0 | 0 | 0 | 12.53 [7.99–18.41] |
| Overall prevalence, n (%) | 1,683,781,860 (21,42)a | 90,544,711 (5,38)b | 1,048,942 (0.06)c | 20,855 (1,97)d | 879,864 (83,32)e | Average PM2.5:16.79 [11.65–27.99] |
% Overall prevalence (Total population) = Total population in South East Asia or Western Pacific / World population as of 21 April 2021 (7,860,798,912) × 100.
% Overall prevalence (Helminths prevalence) = Total helminths prevalence in South East Asia or Western Pacific / Total population in South East Asia or Western Pacific × 100.
% Overall prevalence (Total COVID-19 cases) = Total COVID-19 cases in South East Asia or Western Pacific / Total population in South East Asia or Western Pacific × 100.
% Overall prevalence (Total COVID-19 deaths) = Total COVID-19 deaths in South East Asia or Western Pacific / Total COVID-19 cases in South East Asia or Western Pacific × 100.
% Overall prevalence (Total COVID-19 recovery rate) = Total COVID-19 recovery rate in South East Asia or Western Pacific / Total COVID-19 cases in South East Asia or Western Pacific × 100.
Helminth infections were most prevalent in South East Asia (29,94%) followed by Sub-Saharan Africa (25,64%), Eastern Mediterranean (14,27%), European region (10,64%), The Americas (9,74%) and Western Pacific (5,38%). For PM2.5 air pollution, the Eastern Mediterranean region had the highest mean PM2.5 levels (46,04 µg/m3) followed by South East Asia (43,97 µg/m3), Sub-Saharan Africa (32,52 µg/m3), European region (25,42 µg/m3), The Americas (17,40 µg/m3) and Western Pacific region (16,79 µg/m3). Both Sub-Saharan Africa and European region (91%), and Eastern Mediterranean region and South East Asia (86%) had similar SARS-CoV-2 recovery rates, followed by The Americas (88%) and Western Pacific region (83%) (Table 3, Table 4, Table 5).
The Americas had the highest SARS-CoV-2 mortality rate (3,22%) followed by Sub-Saharan Africa (2,52%), Eastern Mediterranean (2,09%), Western Pacific (1,97%), South East Asia (1,33%) and European region (1,31%) (Table 3, Table 4, Table 5). COVID-19 lethality rate deviate between continents, regions and countries, with notable higher rates in Europe and the United States of America (non-helminth endemic regions as specified by the WHO), regions that are economically developed with optimal health systems. On the contrary, helminth endemic regions with poor economies and inadequate health care facilities tend to have low COVID-19 lethality (Fonte et al., 2020). It was hypothesized that the immunomodulatory role of helminths could be a reason behind the low lethality rates. However, several other factors could also influence these outcomes such as age, population genetics, unavailability of diagnostic testing, SARS-CoV-2 mutations, and environmental humidity and temperature that oppose viral replication (Fonte et al., 2020). Both HIV and tuberculosis infections are independent risk factors for increasing COVID-19 mortality rates (WCDH and NICD, 2020); Sub-Saharan Africa has the highest prevalence of both infections in the world, and had the second highest COVID-19 mortality rate (Table 3). The harmful impacts of air pollution in adversely affecting host immune response and triggering the manifestation of severe SARS-CoV-2 infections will be discussed further later on.
5. Immunological features of soil-transmitted helminths, SARS-CoV-2 and environmental pollution
Cytokines are synthesized by a wide variety of immune cells, predominantly, by T cells, macrophages and neutrophils, which play an important role in promoting and regulating immune responses towards a broad spectrum of parasites, bacteria and viruses (Lucas et al., 2020). Cytokines can elicit anti-inflammatory (IL-10, TGF-β), pro-inflammatory (IL-1, IL-6, IL-8, TNF-α) and anti-infectious (TNF-α, IFN-α, IFN-β, IFN-γ) activities in the body. They also play a role in activating T cells (IL-2, IL-4, IL-10, IL-13, IL-15), B cells (IL-3, IL-4, IL-5, IL-6, IL-21), mast cells (IL-3, IL-4, IL-5, IL-13) and haematopoietic cells (IL-3, IL-5, IL-7, G-CSF, M-CSF, GM-CSF) (Saraswathy et al., 2018).
5.1. Soil-transmitted helminths immune responses
The host defence mechanism against helminths is dependent on Th2 immune responses, anti-inflammatory cytokines (IL-4, IL-5, IL-9, IL-13), antibody isotypes (IgE, IgG1, IgG4), and high levels of mast cells, basophils and eosinophils (Abdoli and Ardakani, 2020, Anthony et al., 2007). The host immune system can also be ineffective against helminths since antibody-dependent cellular cytotoxicity (ADCC) via eosinophils and IgE antibody can usually fail in destroying the impenetrable surface layer of adult helminths (Moreau and Chauvin, 2010). Additionally, helminths can also display “molecular mimicry” by mimicking a host structure and function (Abdoli and Ardakani, 2020, Moreau and Chauvin, 2010).
Chronic helminth infections induce regulatory dendritic cells, T cells and B cells, and IL-10 and TGF-β (Abdoli and Ardakani, 2020). Human cytokine expression levels in the presence of soil-transmitted helminths are as follows: (i) Ascaris lumbricoides: increased IL-4, IL-5 and IL-10 and decreased IL-6, IL-12, TNF-α and IFN-γ expression levels (Blish et al., 2010, Cepon-Robins and Gildner, 2020, Cooper et al., 2000, Geiger et al., 2002, Santos et al., 2019); (ii) Trichuris trichiura: increased IL-4, IL-9, IL-10, IL-13 and decreased TNF-α and IFN-γ expression levels (Cepon-Robins and Gildner, 2020, Faulkner et al., 2002, Geiger et al., 2002, MacDonald et al., 1994); (iii) Necator americanus and Ancylostoma duodenale: increased IL-4, IL-5, IL-9, IL-10, IL-13 and TGF-β and decreased IL-6, IL-17, TNF-α and IFN-γ expression levels (Cepon-Robins and Gildner, 2020, Gaze et al., 2012, Geiger et al., 2011, McSorley et al., 2011); (iv) Strongyloides stercoralis: increased IL-4, IL-5, IL-9, IL-10, IL-13, IL-27, IL-37 and TGF-β and decreased IL-1β, TNF-α and IFN-γ expression levels (Anuradha et al., 2016).
Helminths display potent immunomodulatory activities via suppression of Th1 pro-inflammatory immune responses and activation of Th2 anti-inflammatory immune responses (Fonte et al., 2020). Helminths controls Th1 and Th2 immune responses by enhancing the activities of alternatively activated macrophages (AAMs), FOXP3+ Treg cells and regulatory B cells, leading to the release of anti-inflammatory cytokines (IL-10, TGF-β) (Fonte et al., 2020). Helminth-induced tissue injury activates the production of cytokine alarmins (ILC2, IL-25, IL-33), leading to the activation of Th2 cytokines production by Th2 cells, thymic stromal lymphopoietin (TSLP) cells, eosinophils and basophils (Abdoli and Ardakani, 2020, Salgame et al., 2013). The production of cytokines IL-4, IL-5, IL-9 and IL-13, the primary mediators of Th2 responses, is triggered by IL-25 and IL-33. IL-12 production by dendritic cells, a major promoter of Th1 responses, is limited by TSLP (Fonte et al., 2020). Additionally, macrophages exposure to the Th2 cytokines IL-4 or IL-13 can result in the suppression and stimulation of the classically activated macrophages (M1) and alternatively activated macrophages (M2), respectively (Abdoli and Ardakani, 2020, Salgame et al., 2013). Helminth-produced excretory-secretory antigens stimulate dendritic cells to produce immunoregulatory cytokines (IL-10 and TGF-β) and also inhibit dendritic cell synthesis of pro-inflammatory costimulatory molecules, cytokines and chemokines (Abdoli and Ardakani, 2020, Salgame et al., 2013). Helminth infection can also stimulate regulatory dendritic cells to promote the synthesis and differentiation of Tregs, leading to the down-regulation of pro-inflammatory Th1 and Th17 responses (and its related cytokines IFN-γ, TNF-α, IL-12, IL-17, IL-23) by Th2 cytokines (Abdoli and Ardakani, 2020, Fonte et al., 2020, Salgame et al., 2013). Th17 cells secrete IL-17A and IL-17F and produces IL-21 and IL-22, maintain the mucosal barrier, manages host defence against pathogens and also play a role in the induction of tissue inflammation and destruction (Guglani and Khader, 2010).
5.2. SARS-CoV-2 immune responses
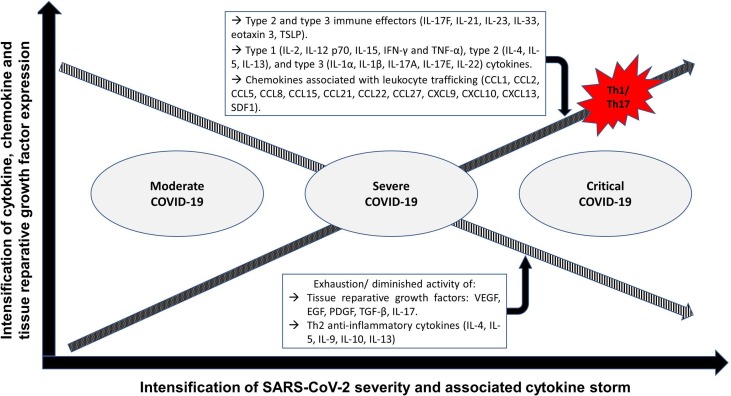
Patients with COVID-19 initially display respiratory disease symptoms which can further lead to pneumonia and disseminated inflammation, and eventually resulting in the occurrence of a “cytokine storm” and multiple complications including sepsis-like syndrome, disseminated intravascular coagulation, acute respiratory distress syndrome and failure of multiple organs (Azkur et al., 2020). The “cytokine storm”, an indicator of disease severity and fatal outcomes, is stimulated by the activation of several white blood cells (monocytes, neutrophils, dendritic cells, macrophages, NK cells, B cells and T cells), tissue cells (endothelial and epithelial cells) and bacteria and viruses (Azkur et al., 2020, Behrens and Koretzky, 2017). Adverse changes in the gut microbiota, induced by SARS-CoV-2, stimulates COVID-19 disease severity and dysfunctional immune responses by intensifying the magnitude of several inflammatory chemokines, cytokines and tissue damage blood markers (Yeoh et al., 2021). The expression of cytokines and tissue reparative growth factors in patients with moderate, severe and critical COVID-19 is summarized in Fig. 1 .
Fig. 1.
Cytokine, chemokine and tissue reparative growth factor expression profiles in patients with moderate, severe and critical COVID-19. As the severity of COVID-19 and associated cytokine storm intensifies the expression profiles of pro-inflammatory Th1/ Th17 associated cytokines and chemokines increases while the expression of anti-inflammatory Th2 cytokines and tissue reparative growth factors decreases. Fig. 1 footnote: CCL: C-C motif chemokine ligand; CXCL: chemokine (C-X-C motif) ligand; EGF: Epidermal growth factor; INF-γ: Interferon gamma; IL: Interleukin; PDGF: Platelet-derived growth factor; SDF1: stromal cell-derived factor 1; TGF-β: Transforming growth factor beta; Th1: T helper type 1 cells; Th2: T helper type 2 cells; Th17: T helper type 17 cells; TNF-α: Tumour necrosis factor alpha; TSLP: Thymic stromal lymphopoietin; VEGF: Vascular endothelial growth factor.
Patients infected with SARS-CoV-2 display a similar “cytokine storm” as was observed during the SARS-CoV outbreak in 2002–2003 and MERS-CoV outbreak in 2012. Patients that were infected with SARS-CoV displayed elevated serum concentrations of pro-inflammatory cytokines (IFN-γ, IL-1β, IL-6, IL-12, MCP-1, CXCL10) (Channappanavar and Perlman, 2017, Huang et al., 2020, Wong et al., 2004). Increased amounts of pro-inflammatory cytokines (TNF-α, IFN-γ, IL-6, IL-15, IL-17, CXCL10) was also observed in patients with MERS-CoV (Channappanavar and Perlman, 2017, Huang et al., 2020, Mahallawi et al., 2018).
Studies in Wuhan, China found that patients with severe COVID-19 infections in ICU had increased levels of Th1 and Th17 cells, and elevated plasma concentrations of pro-inflammatory cytokines (TNF-α, IFN-γ, IL-2, IL-6, IL-7, IL-8, IL-17), anti-inflammatory cytokines (IL-4, IL-10), chemokines (MCP-1, MIP-1α, MIP-1β, IP-10) and haematopoietic progenitor cells growth factors (G-CSF, M-CSF, GM-CSF) compared to non-ICU patients (Huang et al., 2020).
Several components of the innate immune system, including IL-1, IL-6, IL-18, TNF-α and type I/III IFN are present in the upper respiratory tract to control the infection of SARS-CoV-2. However, in some cases SARS-CoV-2 bypasses the first step of control and spreads rapidly along the airway to the alveoli, leading to pneumonia, acute respiratory distress syndrome, acute lung damage and in some cases death (Fonte et al., 2020, Qian et al., 2013, Vabret et al., 2020). As seen with COVID-19, a similar “cytokine storm” was also observed in patients with severe SARS-CoV-2 however anti-inflammatory cytokines (IL-4, IL-10) secretion was not initiated (Huang et al., 2020, Wong et al., 2004).
Lucas and colleagues (2020) identified specific immunological markers that occur early in patients with COVID-19 which strongly correlated with poor health outcomes and death. They associated increased levels of IFN-α before the first 12 days from symptom onset with lengthier hospital stays and death in patients with COVID-19. In addition, patients who eventually died of COVID-19 displayed significantly higher levels of IL-1Ra, IFN-α and IFN-λ, as well as chemokines (CCL1, MCP-1, CCL21, M-CSF, IL-2, IL-16) associated with the recruitment and survival of monocytes and T cells within the first 12 days from symptom onset (Lucas et al., 2020).
5.3. Environmental pollution-induced immune responses
Similar to SARS-CoV-2 and COVID-19, environmental pollution (air and water pollution) can also trigger the activation of several inflammation associated pathways and diseases, and adversely affects the host immune system (Quinete and Hauser-Davis, 2020, Saraswathy et al., 2018). Prolonged and chronic exposure to environmental pollution can lead to the exhaustion and diminished activity of Th2 immune response (Quinete and Hauser-Davis, 2020, Saraswathy et al., 2018). Air pollution is associated with the pathogenesis of several complications and diseases in the lungs (chronic obstructive pulmonary disease and asthma), heart (myocardial ischemia, atherosclerosis and hypertension), blood (increased coagulability, reduced oxygen saturation and peripheral thrombosis) and brain (cerebrovascular ischemia, neuro-degeneration and impaired neurotransmitter signalling) (Quinete and Hauser-Davis, 2020, Saraswathy et al., 2018).
Air pollutants (NO2 and PM2.5) promotes the overexpression of ACE2 receptors in the epithelial cell surface of the respiratory tract (SARS-CoV-2 gain entry and infect cells using the ACE2 receptor) (Paital and Agrawal, 2020). Acute air pollution exposure (SO2, PM2.5, NO2, and CO) was associated with increased serum levels of Th1 and Th17 cytokines (IFN-γ, IL-2, IL-12, IL-17A, MCP-1 and soluble CD40 ligand (sCD40L)), and decreased Th2 cytokine levels (IL-4, IL-13 and eotaxin) and forced vital capacity % predicted (FVC % pred) in patients with chronic obstructive pulmonary disease (Gao et al., 2020). Similar findings were reported elsewhere (Gu et al., 2017). Acute exposure to PM2.5 alone was associated with cardiovascular injury which was evident by the suppressed circulating levels of pro-angiogenic growth factors (PDGF, RANTES, GROα, EGF and VEGF), and increased levels pro-inflammatory (IL-1β, IL-6, MCP-1 and MIP-1α/β) and anti-angiogenic (IP-10 and TNF-α) cytokines and endothelial adhesion markers (sICAM-1 and sVCAM-1). PM2.5 also triggered inflammatory responses as evident by the increased circulating levels of CD4+, CD8+, CD14+ and CD16+ cells (Pope et al., 2016).
Water pollutants such as metals (copper, cadmium, mercury, lead, chromium and arsenic) and organic chemicals (polyfluoroalkyl substances, bisphenol A and phthalates) can also be immunotoxic and compromise the immune response of humans by dysregulating Th1 and Th2 cytokine production (Quinete and Hauser-Davis, 2020). They display endocrine disrupting properties and are associated with autoimmune diseases, and reproductive, developmental and neurological disorders (Quinete and Hauser-Davis, 2020).
In humans, high arsenic levels were correlated with increased incidences of allergies, asthma and parasitic infections, and significantly inhibited the secretion of IL-2, resulting in immunosuppression and favouring opportunistic infections (Quinete and Hauser-Davis, 2020, Soto-Peña et al., 2006). Increased IL-1β (chromium and mercury) and TNF-α (low mercury concentration), and decreased IL-6, IL-8, IL-10 and IFN-γ (chromium) and TNF-α and IL-1β (high mercury concentration) expression levels were noted in human PMBCs (Maria et al., 2000). BALB/c mice consuming water containing lead and copper displayed an upregulation of IFN-γ and IL-4 expression downregulated in the lead-treated group only (Radbin et al., 2014).
Human exposure to polyfluoroalkyl substances is associated with increased IL-4 and IL-5 levels, limited seroprotection from the influenza A virus subtype H3N2 and asthma (Dong et al., 2013, Looker et al., 2014, Quinete and Hauser-Davis, 2020, Zhu et al., 2016). Mice consuming water containing perfluorooctane sulfonate displayed significantly increased IL-6 levels (Fair et al., 2011, Quinete and Hauser-Davis, 2020), while another study noted a reduction in IL-2 and IFN-γ secretion in favour of IL-4 and IL-10 which indicated that perfluorooctane sulfonate exposure directly suppress cellular responses and enhances the activation of humoral responses (Quinete and Hauser-Davis, 2020, Zheng et al., 2011).
Both bisphenol A (an environmental estrogen) and phthalates are also implicated in airway inflammation, allergic sensitization and asthma (Nakajima et al., 2012, Quinete and Hauser-Davis, 2020). Mice drinking water containing butyl benzyl phthalate displayed increased susceptibility for Th2-mediated immune responses and no significant changes in Th1 immune responses was noted, leading to increased vulnerability in developing allergic asthma and immune-related diseases (Jahreis et al., 2018, Quinete and Hauser-Davis, 2020). Bisphenol A exposure was associated with highly intensified Th1 cellular immune response (IFN-γ) and suppression of IL-4 in mice. It was proposed that the estrogenic activity of bisphenol A stimulated the production of prolactin which affected cytokine expression profiles, leading to dysregulated Th1 cellular immune response (Quinete and Hauser-Davis, 2020, Youn et al., 2002).
6. Immunomodulatory role of helminths against SARS-CoV-2/ COVID-19: the pros and cons
6.1. Advantages
Helminths are potent activators of immunosuppressive anti-inflammatory Th2 immune response cytokines and Tregs (TGF-β, IL-4, IL-10, IL-13, IL-33) which could be beneficial in modulating heightened pro-inflammatory Th1 and Th17 immune response in humans (Cepon-Robins and Gildner, 2020, Maizels and McSorley, 2016). A study in Ethiopia associated intestinal parasites with reduced COVID-19 severity and fatality rates, and it was suggested that parasite-induced immunomodulatory responses may dampen hyperinflammation in patients with COVID-19 (Gebrecherkos et al., 2021). Studies found helminth-induced IL-4 signaling in CD8+ T cells expands bystander or virtual memory CD8+ T cells for early control of viral infection (Rolot et al., 2018). Hence, this could be one of the reasons behind milder COVID-19 symptoms and diminished cytokine storm in patients co-infected with helminths and COVID-19 (Cepon-Robins and Gildner, 2020, Munjita et al., 2020).
Helminths or helminth-derived antigens display therapeutic potential for the resolution of inflammation and cytokine storm syndrome in patients with COVID-19 (Abdoli, 2019, Abdoli and Mirzaian Ardakani, 2020). Excretory-secretory molecules from Trichuris trichiura reduces intestinal inflammation by activating Th2 and Treg immune responses (Cepon-Robins and Gildner, 2020; Cepon‐Robins et al., 2019). Helminth-induced expression of IL-9, IL-10, TGF-β and amphiregulin (AREG; associated with wound repair and tissue formation in the lung and gut) play a role in reducing lung inflammation and enhancing lung repair (Schwartz et al., 2018).
Helminths play a crucial role in modifying the intestinal microbiota by promoting the rapid growth and development of several immune-regulating bacterial species. The anti-inflammatory Th2 cytokines produced by the intestinal microbiota was reported to minimize viral infection-induced lung tissue damage (Cepon-Robins and Gildner, 2020, McFarlane et al., 2017).
Patients presenting with coexisting helminth infections and metabolic syndrome and type 2 diabetes, chronic inflammatory conditions that are risk factors for COVID-19 morbidity and mortality, displayed decreased levels of Th1 pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, IL-12, IL-18, IL-23, IL-27, G-CSF, GM-CSF) versus those without helminth infections (Hays et al., 2020, Rajamanickam et al., 2020). This highlight the putative role of helminth induced Th2 immune response in balancing Th1 and Th17 hyper-inflammatory responses in individuals infected with chronic inflammatory conditions and/or SARS-CoV-2.
As mentioned, the SARS-CoV-2 bind directly to the ACE2 receptors in order to infect cells (Hoffmann et al., 2020). Studies associated heightened inflammation and activated Th2 immune response with increased and decreased levels of host ACE2 receptors, respectively (Cepon-Robins and Gildner, 2020, Jackson et al., 2020, Wrapp et al., 2020). Inflammatory conditions such as smoking, diabetes, respiratory disease and cardiovascular disease are associated with increased levels of ACE2 receptors (Butler et al., 2020, Cepon-Robins and Gildner, 2020, Sajuthi et al., 2020, Smith et al., 2020). Unlike non-allergic asthma, allergic asthma, which is initiated by activated Th2 immune response and linked with decreased ACE2 expression, was proposed to offer protection from severe COVID-19 symptoms and outcomes in humans (Cepon-Robins and Gildner, 2020, Jackson et al., 2020). Considering that helminths trigger Th2 immune response in the lung (Cepon-Robins and Gildner, 2020, Schwartz et al., 2018), this could also lead to decreased ACE2 levels in respiratory cells, diminished ability of SARS-CoV-2 to infect host cells and decreased viral load (Cepon-Robins and Gildner, 2020).
Patients with severe COVID-19 have very low levels of eosinophils (Cepon-Robins and Gildner, 2020, Tanni et al., 2020). Helminth infections activates the production of IL-5 which is directly associated with high eosinophils levels (Cepon-Robins and Gildner, 2020, Huang and Appleton, 2016, Sanderson, 1992). Eosinophils display immunoregulatory and antiviral effects in the body in response to combating helminth larvae in the lungs (Cepon-Robins and Gildner, 2020, Huang and Appleton, 2016, Lindsley et al., 2020). A strong Th2 immune response and high levels of eosinophils could be beneficial in fighting off early stages of SARS-CoV-2 infection, resulting in less tissue damage and reductions in viral loads (Cepon-Robins and Gildner, 2020, Lindsley et al., 2020, Rodriguez, 2020).
6.2. Disadvantages
Helminths are linked with nutritional deficiencies and anaemia which leads to compromised and dampened immune response against pathogens in humans (Abdoli, 2020, Bethony et al., 2006, Cepon-Robins and Gildner, 2020, Le et al., 2017). As mentioned previously, SARS-CoV-2 and species-specific helminths can adversely alter the gut microbiota composition by favouring and retarding the growth of opportunistic pathogens and short-chain fatty acid producing salutary bacteria, respectively which could lead manifestation of gastrointestinal infections, bacterial colitis and gut dysbiosis-related diseases (Lee et al., 2014, Su et al., 2018, Yeoh et al., 2021, Zuo et al., 2021, Zuo et al., 2020). These outcomes may be further aggravated if individuals are co-infected.
Chronic helminth infections can skew the host immune response toward anti-inflammatory Th2 immunity, leading to the suppression of vital pro-inflammatory Th1 immune response that is crucial for attacking intracellular pathogens as seen in cases with co-infections between helminths and tuberculosis (Babu and Nutman, 2016). Helminth-induced immunosuppression could also lead to increased viral loads and disease severity (Abdoli, 2020, Cepon-Robins and Gildner, 2020). This is in accordance with human studies associating helminth infections in endemic regions with increased susceptibility and severity to malaria, HIV/AIDS and tuberculosis (Abdoli, 2020, Salgame et al., 2013).
Helminth-induced immunosuppression was reported to dampen Bacillus Calmette–Guérin (BCG), a vaccine for tuberculosis, vaccination efficacy (Abdoli, 2020, Elias et al., 2008). Lately, the BCG vaccine was proposed to be a candidate in protecting against severe COVID-19 (Abdoli, 2020, Redelman-Sidi, 2020). Studies using mice found helminth infections drastically reduced vaccination efficacy against the H1N1 influenza A virus due to the systemic and sustained expansion of IL-10-producing CD4+CD49+LAG-3+ type 1 regulatory T cells. The above observations remained the same even after termination of helminth infection, and blockade of the IL-10 receptor only partially restored anti-influenza vaccination efficacy (Abdoli, 2020, Hartmann et al., 2019). These findings suggest that helminth infections could inhibit the advancement of long-term immunity against SARS-CoV-2.
7. Concluding remarks
In conclusion, there is scarcity of studies focusing on: (i) the immunomodulatory role of helminths against SARS-CoV-2 pro-inflammatory Th1/Th17 immune response, and (ii) the insidious role of prolonged and chronic air and water pollution exposure on the functionality and immunomodulatory activity of helminths in the host, and in exacerbating pro-inflammatory Th1/Th17 cytokine production in patients with SARS-CoV-2.
Nonetheless, considering that helminths are potent activators of anti-inflammatory Th2 immune response, studies have associated elevated Th2 related cytokines and tissue reparative growth factors with reductions in lung inflammation and damage, and decreased expression levels of ACE2 receptors (SARS-CoV-2 uses the ACE2 receptors to infect cells and associated with extensive lung damage), which could lead to reduced SARS-CoV-2 infection rate and low viral load in the host. However, this can be reversed if the individual is continuously being exposed to air pollution because studies have associated overexpression of ACE2 receptors in the epithelial cell surface of the respiratory tract with air pollution and exhaustion of Th2 immune response which could promote severe SARS-CoV-2 infection in high air pollution endemic regions.
Conversely, helminth-induced immunosuppression activity could also be disadvantageous by reducing vaccination efficacy, and diminishing vital Th1/Th17 immune response cytokine production that are crucial for combating early stage infections. However, this could be reversed by: (i) continuous air pollution exposure which is associated with exhaustion of Th2 immune response, or (ii) hyper-inflammatory disorders (obesity, diabetes, hypertension and metabolic disorders) which are known to intensify Th1/Th17 pro-inflammatory cytokine production to a point where the immunosuppressive activities of helminths could be hindered. Again, suppressed activities of helminths can also be disadvantageous against SARS-CoV-2 inflammatory response. This “yin and yang” approach seems complex and requires more understanding.
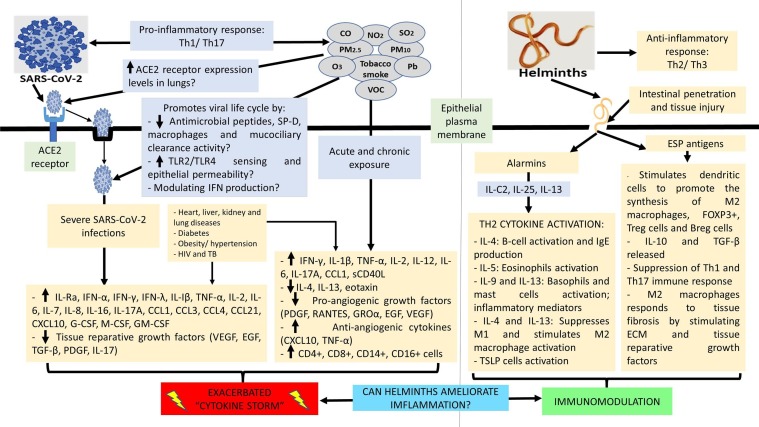
All the above suggest that environmental pollution could hinder the activity of helminths which could be both harmful and beneficial in patients with SARS-CoV-2. It is also known that chronic air pollution exposure exacerbates SARS-CoV-2 related adverse health outcomes (respiratory distress and failure). Further studies are warranted in a cohort of SARS-CoV-2 infected individuals residing in helminth and air pollution endemic regions to offer more clarification. Data from these studies could influence mass periodic deworming programmes and environmental health policies in order to minimise SARS-CoV-2 severity in affected regions. Fig. 2 highlights the hypothesized mechanisms on how air pollutants can promote SARS-CoV-2 entry and life cycle, the role of SARS-CoV-2 and air pollutants in stimulating Th1/ Th17 pro-inflammatory cytokine production and associated cytokine storm, the role of helminth infections in stimulating Th2 anti-inflammatory cytokine production and tissue reparative growth factors, and collectively on how helminths may modulate inflammation induced by SARS-CoV-2 and air pollutants.
Fig. 2.
Immune response against SARS-CoV-2 and interaction with environmental pollution and helminths. Environmental pollutants may promote SARS-CoV-2 entry and replication by increasing ACE2 receptor expression, TLR2/ TLR4 sensing and epithelial permeability, decreasing the activity of antimicrobial peptides, surfactant protein D, macrophages and mucociliary clearance, and modulating the production of interferon. Both SARS-CoV-2 and environmental pollution can trigger the activation of pro-inflammatory Th1/ Th17 immune response, leading to the intensified production of inflammatory cytokines and chemokines and the manifestation of a cytokine storm. This scenario may be worsened if individuals have co-morbidities. Exhaustion/ diminished activity of tissue reparative/ pro-angiogenic growth factors occurs as disease severity and inflammation is aggravated. Helminths are potent stimulators of anti-inflammatory Th2/ Th3 immune response. Helminth induced intestinal penetration and tissue injury activates the production of alarmins, leading to the activation of Th2 associated cytokines (IL-4, IL-5, IL-9, IL13), B-cells, eosinophils, basophils, masts cells, M2 macrophages (responds to tissue fibrosis by stimulating the extracellular matrix and tissue reparative growth factors) and TSLP cells. Helminths associated excretory-secretory antigen products stimulates dendritic cells to promote the activation and production of M2 macrophages, FOXP3+, Treg cells, Breg cells, IL-10 (regulates Th1/Th17-cell immune responses) and TGF-β (regulator of haematopoiesis and tissue repair). Collectively, helminths are important immunomodulators and may have the potential to ameliorate SARS-CoV-2 and environmental pollution induced inflammation. Fig. 2 footnote: ACE2: Angiotensin-converting enzyme 2; Breg: Regulatory B cells; CCL: C-C motif chemokine ligand; CO: Carbon monoxide; CXCL: chemokine (C-X-C motif) ligand; ECM: Extracellular matrix; EGF: Epidermal growth factor; ESP: Excretory-secretory products; FOXP3+: forkhead box P3; G-CSF: Granulocyte colony-stimulating factor; GM-CSF: Granulocyte-macrophage colony-stimulating factor; GROα: Growth-regulated alpha protein; IFN: Interferon; IL: Interleukin; M1: classically activated macrophages; M2: alternatively activated macrophages; M-CSF: Macrophage colony-stimulating factor; NO2: Nitrogen dioxide; O3: Ozone; Pb: Lead; PDGF: Platelet-derived growth factor; PM: Particulate matter; RANTES: Regulated on Activation, Normal T Cell Expressed and Secreted; SARS-CoV-2: Severe Acute Respiratory Syndrome Coronavirus 2; sCD40L: Soluble CD40 ligand; SO2: Sulphur dioxide; SP-D: Surfactant protein D; TGF-β: Transforming growth factor beta; Th1: T helper type 1 cells; Th2: T helper type 2 cells; Th3: T helper type 3 cells; Th17: T helper type 17 cells; TLR: Toll-like receptor; TNF-α: Tumour necrosis factor alpha; Treg: Regulatory T cells; VEGF: Vascular endothelial growth factor; VOC: Volatile organic compounds.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgments
We would like to acknowledge the South African Medical Research Council (SAMRC) for funding.
Funding
Professor ZL Mkhize-Kwitshana was partially supported as a private investigator (PI) (and researchers PN, MNMM, KNB, NN and RP) by funding from the South African Medical Research Council (SAMRC) Mid-Career Scientist Programme (MCSP) Grant (no award/grant number), through its Division of Research Capacity Development under the RCDI programme from funding received from the South African National Treasury. The content hereof is the sole responsibility of the authors and do not necessarily represent the official views of the SAMRC or the funders.
Handling Editor: Frederic Coulon
References
- Abdoli A. Therapeutic potential of helminths and helminth-derived antigens for resolution of inflammation in inflammatory bowel disease. Arch. Med. Res. 2019;50:58–59. doi: 10.1016/j.arcmed.2019.03.001. [DOI] [PubMed] [Google Scholar]
- Abdoli A. Helminths and COVID-19 Co-infections: a neglected critical challenge. ACS Pharmacol. Transl. Sci. 2020;3:1039–1041. doi: 10.1021/acsptsci.0c00141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdoli A., Ardakani H.M. Helminth infections and immunosenescence: the friend of my enemy. Exp. Gerontol. 2020;133 doi: 10.1016/j.exger.2020.110852. [DOI] [PubMed] [Google Scholar]
- Abdoli A., Mirzaian Ardakani H. Potential application of helminth therapy for resolution of neuroinflammation in neuropsychiatric disorders. Metab. Brain Dis. 2020;35:95–110. doi: 10.1007/s11011-019-00466-5. [DOI] [PubMed] [Google Scholar]
- Abner S.R., Parthasarathy G., Hill D.E., Mansfield L.S. Trichuris suis: detection of antibacterial activity in excretory-secretory products from adults. Exp. Parasitol. 2001;99:26–36. doi: 10.1006/expr.2001.4643. [DOI] [PubMed] [Google Scholar]
- Alifano M., Alifano P., Forgez P., Iannelli A. Renin-angiotensin system at the heart of COVID-19 pandemic. Biochimie. 2020;174:30–33. doi: 10.1016/j.biochi.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antalis T.M., Bugge T.H., Wu Q. Membrane-anchored serine proteases in health and disease. Prog. Mol. Biol. Transl. Sci. 2011;99:1–50. doi: 10.1016/b978-0-12-385504-6.00001-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anthony R.M., Rutitzky L.I., Urban J.F., Stadecker M.J., Gause W.C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 2007;7:975–987. doi: 10.1038/nri2199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anuradha R., Munisankar S., Bhootra Y., Jagannathan J., Dolla C., Kumaran P., Shen K., Nutman T.B., Babu S. Systemic cytokine profiles in strongyloides stercoralis infection and alterations following treatment. Infect. Immun. 2016;84:425–431. doi: 10.1128/iai.01354-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azkur A.K., Akdis M., Azkur D., Sokolowska M., van de Veen W., Brüggen M.C., O'Mahony L., Gao Y., Nadeau K., Akdis C.A. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in COVID-19. Allergy. 2020;75:1564–1581. doi: 10.1128/iai.01354-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babu S., Nutman T.B. Helminth-tuberculosis co-infection: an immunologic perspective. Trends Immunol. 2016;37:597–607. doi: 10.1016/j.it.2016.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bashir M.F., Ma B.J., Bilal, Komal B., Bashir M.A., Farooq T.H., Iqbal N., Bashir M. Correlation between environmental pollution indicators and COVID-19 pandemic: a brief study in Californian context. Environ. Res. 2020;187:109652. doi: 10.1016/j.envres.2020.109652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrens E.M., Koretzky G.A. Review: cytokine storm syndrome: looking toward the precision medicine era. Arthritis Rheumatol. (Hoboken, NJ) 2017;69:1135–1143. doi: 10.1002/art.40071. [DOI] [PubMed] [Google Scholar]
- Bethony J., Brooker S., Albonico M., Geiger S.M., Loukas A., Diemert D., Hotez P.J. Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. The Lancet. 2006;367:1521–1532. doi: 10.1016/S0140-6736(06)68653-4. [DOI] [PubMed] [Google Scholar]
- Blish C.A., Sangaré L., Herrin B.R., Richardson B.A., John-Stewart G., Walson J.L. Changes in plasma cytokines after treatment of ascaris lumbricoides infection in individuals with HIV-1 infection. J. Infect. Dis. 2010;201:1816–1821. doi: 10.1086/652784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradbury R.S., Piedrafita D., Greenhill A., Mahanty S. Will helminth co-infection modulate COVID-19 severity in endemic regions? Nat. Rev. Immunol. 2020;20:342. doi: 10.1038/s41577-020-0330-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brosschot T.P., Reynolds L.A. The impact of a helminth-modified microbiome on host immunity. Mucosal Immunol. 2018;11:1039–1046. doi: 10.1038/s41385-018-0008-5. [DOI] [PubMed] [Google Scholar]
- Bunyavanich S., Do A., Vicencio A. Nasal gene expression of angiotensin-converting enzyme 2 in children and adults. JAMA. 2020;323:2427–2429. doi: 10.1001/jama.2020.8707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler M.W., O’Reilly A., Dunican E.M., Mallon P., Feeney E.R., Keane M.P., McCarthy C. Prevalence of comorbid asthma in COVID-19 patients. J. Allergy Clin. Immunol. 2020;146:334–335. doi: 10.1016/j.jaci.2020.04.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai H. Sex difference and smoking predisposition in patients with COVID-19. Lancet Respir. Med. 2020;8 doi: 10.1016/S2213-2600(20)30117-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caly L., Druce J.D., Catton M.G., Jans D.A., Wagstaff K.M. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res. 2020;178 doi: 10.1016/j.antiviral.2020.104787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cepon-Robins T.J., Gildner T.E. Old friends meet a new foe: A potential role for immune-priming parasites in mitigating COVID-19 morbidity and mortality. Evolut., Med., Public Health. 2020;2020:234–248. doi: 10.1093/emph/eoaa037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Channappanavar R., Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017;39:529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper P.J., Chico M.E., Sandoval C., Espinel I., Guevara A., Kennedy M.W., Urban J.F., Jr., Griffin G.E., Nutman T.B. Human infection with Ascaris lumbricoides is associated with a polarized cytokine response. J. Infect. Dis. 2000;182:1207–1213. doi: 10.1086/315830. [DOI] [PubMed] [Google Scholar]
- Dayan A.D. Albendazole, mebendazole and praziquantel. Review of non-clinical toxicity and pharmacokinetics. Acta Trop. 2003;86:141–159. doi: 10.1016/S0001-706X(03)00031-7. [DOI] [PubMed] [Google Scholar]
- Dhar D., Mohanty A. Gut microbiota and Covid-19- possible link and implications. Virus Res. 2020;285 doi: 10.1016/j.virusres.2020.198018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domingo J.L., Rovira J. Effects of air pollutants on the transmission and severity of respiratory viral infections. Environ. Res. 2020;187 doi: 10.1016/j.envres.2020.109650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong G.-H., Tung K.-Y., Tsai C.-H., Liu M.-M., Wang D., Liu W., Jin Y.-H., Hsieh W.-S., Lee Y.L., Chen P.-C. Serum polyfluoroalkyl concentrations, asthma outcomes, and immunological markers in a case–control study of Taiwanese children. Environ. Health Perspect. 2013;121:507–513. doi: 10.1289/ehp.1205351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dvorožňáková E., Dvorožňáková M., Šoltys J. Heavy metal intoxication compromises the host cytokine response in Ascaris Suum model infection. Helminthologia. 2016;53:14–23. doi: 10.1515/helmin-2015-0063. [DOI] [Google Scholar]
- Elias D., Britton S., Aseffa A., Engers H., Akuffo H. Poor immunogenicity of BCG in helminth infected population is associated with increased in vitro TGF-β production. Vaccine. 2008;26:3897–3902. doi: 10.1016/j.vaccine.2008.04.083. [DOI] [PubMed] [Google Scholar]
- Fair P.A., Driscoll E., Mollenhauer M.A., Bradshaw S.G., Yun S.H., Kannan K., Bossart G.D., Keil D.E., Peden-Adams M.M. Effects of environmentally-relevant levels of perfluorooctane sulfonate on clinical parameters and immunological functions in B6C3F1 mice. J. Immunotoxicol. 2011;8:17–29. doi: 10.3109/1547691X.2010.527868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faulkner H., Turner J., Kamgno J., Pion S.D., Boussinesq M., Bradley J.E. Age- and infection intensity-dependent cytokine and antibody production in human trichuriasis: the importance of IgE. J. Infect. Dis. 2002;185:665–672. doi: 10.1086/339005. [DOI] [PubMed] [Google Scholar]
- Fonte L., Acosta A., Sarmiento M.E., Ginori M., García G., Norazmi M.N. COVID-19 lethality in sub-Saharan Africa and helminth immune modulation. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.574910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fricke W.F., Song Y., Wang A.-J., Smith A., Grinchuk V., Pei C., Ma B., Lu N., Urban J.F., Shea-Donohue T., Zhao A. Type 2 immunity-dependent reduction of segmented filamentous bacteria in mice infected with the helminthic parasite Nippostrongylus brasiliensis. Microbiome. 2015;3:40. doi: 10.1186/s40168-015-0103-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frontera A., Cianfanelli L., Vlachos K., Landoni G., Cremona G. Severe air pollution links to higher mortality in COVID-19 patients: the “double-hit” hypothesis. J. Infect. 2020;81:255–259. doi: 10.1016/j.jinf.2020.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao N., Xu W., Ji J., Yang Y., Wang S.-T., Wang J., Chen X., Meng S., Tian X., Xu K.-F. Lung function and systemic inflammation associated with short-term air pollution exposure in chronic obstructive pulmonary disease patients in Beijing, China. Environ. Health. 2020;19:1–10. doi: 10.1186/s12940-020-0568-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaze S., McSorley H.J., Daveson J., Jones D., Bethony J.M., Oliveira L.M., Speare R., McCarthy J.S., Engwerda C.R., Croese J., Loukas A. Characterising the mucosal and systemic immune responses to experimental human hookworm infection. PLoS Pathog. 2012;8 doi: 10.1371/journal.ppat.1002520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebrecherkos, T., Gessesse, Z., Kebede, Y., Gebreegzabher, A., Tassew, G., Abdulkader, M., Ebuy, H., Desta, A., Hailu, A., Harris, V., 2021. Effect of co-infection with parasites on severity of COVID-19. medRxiv. 10.1101/2021.02.02.21250995. [DOI]
- Geiger S.M., Fujiwara R.T., Freitas P.A., Massara C.L., Dos Santos Carvalho O., Corrêa-Oliveira R., Bethony J.M. Excretory-secretory products from hookworm l(3) and adult worms suppress proinflammatory cytokines in infected individuals. J. Parasitol. Res. 2011;2011 doi: 10.1155/2011/512154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geiger S.M., Massara C.L., Bethony J., Soboslay P.T., Carvalho O.S., Corrêa-Oliveira R. Cellular responses and cytokine profiles in Ascaris lumbricoides and Trichuris trichiura infected patients. Parasite Immunol. 2002;24:499–509. doi: 10.1046/j.1365-3024.2002.00600.x. [DOI] [PubMed] [Google Scholar]
- Gu, X.Y., Chu, X., Zeng, X.L., Bao, H.R., Liu, X.J., 2017. Effects of PM2.5 exposure on the Notch signaling pathway and immune imbalance in chronic obstructive pulmonary disease. Environ. Pollut. (Barking, Essex: 1987) 226, 163–173. 10.1016/j.envpol.2017.03.070. [DOI] [PubMed]
- Guglani L., Khader S.A. Th17 cytokines in mucosal immunity and inflammation. Curr. Opin. HIV and AIDS. 2010;5:120–127. doi: 10.1097/COH.0b013e328335c2f6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gülsen A., Yigitbas B.A., Uslu B., Drömann D., Kilinc O. The effect of smoking on COVID-19 symptom severity: systematic review and meta-analysis. Pulmonary Med. 2020;2020:7590207. doi: 10.1155/2020/7590207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartmann W., Brunn M.-L., Stetter N., Gagliani N., Muscate F., Stanelle-Bertram S., Gabriel G., Breloer M. Helminth infections suppress the efficacy of vaccination against seasonal influenza. Cell Rep. 2019;29:2243–2256. doi: 10.1016/j.celrep.2019.10.051. [DOI] [PubMed] [Google Scholar]
- Hawdon J.M. Controlling soil-transmitted helminths: time to think inside the box? J. Parasitol. 2014;100:166–188. doi: 10.1645/13-412.1. [DOI] [PubMed] [Google Scholar]
- Hays R., Pierce D., Giacomin P., Loukas A., Bourke P., McDermott R. Helminth coinfection and COVID-19: An alternate hypothesis. PLoS Negl.Trop. Dis. 2020;14 doi: 10.1371/journal.pntd.0008628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hotez P.J., Bundy D.A., Beegle K., Brooker S., Drake L., de Silva N., Montresor A., et al. In: Disease Control Priorities in Developing Countries 2nd edition: The International Bank for Reconstruction and Development/The World Bank. Jamison D.T., editor. Oxford University Press; New York: 2006. Chapter 24: Helminth infections: soil-transmitted helminth infections and schistosomiasis; pp. 467–482. [Google Scholar]
- Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L., Appleton J.A. Eosinophils in helminth infection: defenders and dupes. Trends Parasitol. 2016;32:798–807. doi: 10.1016/j.pt.2016.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson D.J., Busse W.W., Bacharier L.B., Kattan M., O’Connor G.T., Wood R.A., Visness C.M., Durham S.R., Larson D., Esnault S., et al. Association of respiratory allergy, asthma and expression of the SARS-CoV-2 receptor, ACE2. J. Allergy Clin. Immunol. 2020;146:203–206. doi: 10.1016/j.jaci.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jahreis S., Trump S., Bauer M., Bauer T., Thürmann L., Feltens R., Wang Q., Gu L., Grützmann K., Röder S. Maternal phthalate exposure promotes allergic airway inflammation over 2 generations through epigenetic modifications. J. Allergy Clin. Immunol. 2018;141:741–753. doi: 10.1016/j.jaci.2017.03.017. [DOI] [PubMed] [Google Scholar]
- Kay G.L., Millard A., Sergeant M.J., Midzi N., Gwisai R., Mduluza T., Ivens A., et al. Differences in the Faecal Microbiome in Schistosoma haematobium Infected Children vs. Uninfected Children. PLoS Negl. Trop. Dis. 2015;9 doi: 10.1371/journal.pntd.0003861. https://dx.doi.org/10.1371%2Fjournal.pntd.0003861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaźmierczak-Siedlecka K., Vitale E., Makarewicz W. COVID-19 - gastrointestinal and gut microbiota-related aspects. Eur. Rev. Med. Pharmacol. Sci. 2020;24:10853–10859. doi: 10.26355/eurrev_202010_23448. https://doi.org/10.26355/eurrev_202010_23448. [DOI] [PubMed] [Google Scholar]
- Konstantinoudis G., Padellini T., Bennett J., Davies B., Ezzati M., Blangiardo M. Long-term exposure to air-pollution and COVID-19 mortality in England: a hierarchical spatial analysis. Environ. Int. 2021;146 doi: 10.1016/j.envint.2020.106316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le H.T., Brouwer I.D., Verhoef H., Nguyen K.C., Kok F.J. Anemia and intestinal parasite infection in school children in rural Vietnam. Asia Pacific J. Clin. Nutrit. 2017;16:716–723. [PubMed] [Google Scholar]
- Lee S.C., Tang M.S., Lim Y.A., Choy S.H., Kurtz Z.D., Cox L.M., Gundra U.M., Cho I., et al. Helminth colonization is associated with increased diversity of the gut microbiota. PLoS Negl. Trop. Dis. 2014;8 doi: 10.1371/journal.pntd.0002880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindquist H.A., Cross J.H. In: Infectious Diseases. 4th ed. Cohen J., Powderly W.G., Opal S.M., editors. Elsevier; 2017. Section 8 Clinical Microbiology: Parasites, Chapter 195: Helminths; pp. 1763–1779. [DOI] [Google Scholar]
- Lindsley A.W., Schwartz J.T., Rothenberg M.E. Eosinophil responses during COVID-19 infections and coronavirus vaccination. J. Allergy Clin. Immunol. 2020;146:1–7. doi: 10.1016/j.jaci.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lippi G., Plebani M. The critical role of laboratory medicine during coronavirus disease 2019 (COVID-19) and other viral outbreaks. Clin. Chem. Lab. Med. (CCLM) 2020;58:1063–1069. doi: 10.1515/cclm-2020-0240. [DOI] [PubMed] [Google Scholar]
- Liu D.X., Liang J.Q., Fung T.S. Human Coronavirus-229E, -OC43, -NL63, and -HKU1. Ref. Mod. Life Sci. 2020;2020:1–13. doi: 10.1016/B978-0-12-809633-8.21501-X. [DOI] [Google Scholar]
- Looker C., Luster M.I., Calafat A.M., Johnson V.J., Burleson G.R., Burleson F.G., Fletcher T. Influenza vaccine response in adults exposed to perfluorooctanoate and perfluorooctanesulfonate. Toxicol. Sci. 2014;138:76–88. doi: 10.1093/toxsci/kft269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucas C., Wong P., Klein J., Castro T.B.R., Silva J., Sundaram M., Ellingson M.K., et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020;584:463–469. doi: 10.1038/s41586-020-2588-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludvigsson, J.F., 2020. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatrica (Oslo, Norway: 1992) 109, 1088–1095. 10.1111/apa.15270. [DOI] [PMC free article] [PubMed]
- Lukassen S., Chua R.L., Trefzer T., Kahn N.C., Schneider M.A., Muley T., Winter H., Meister M., Veith C., Boots A.W., Hennig B.P., Kreuter M., Conrad C., Eils R. SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells. EMBO J. 2020;39 doi: 10.15252/embj.20105114. https://doi.org/10.15252/embj.20105114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDonald T.T., Spencer J., Murch S.H., Choy M.Y., Venugopal S., Bundy D.A., Cooper E.S. Immunoepidemiology of intestinal helminthic infections. 3. Mucosal macrophages and cytokine production in the colon of children with Trichuris trichiura dysentery. Trans. R. Soc. Trop. Med. Hyg. 1994;88:265–268. doi: 10.1016/0035-9203(94)90072-8. [DOI] [PubMed] [Google Scholar]
- McCallum, M., Bassi, J., Marco, A., Chen, A., Walls, A.C., Iulio, J.D., et al., 2021. SARS-CoV-2 immune evasion by variant B.1.427/B.1.429. BioRxiv. 10.1101/2021.03.31.437925. [DOI]
- McKay D.M. Not all parasites are protective. Parasite Immunol. 2015;37:324–332. doi: 10.1111/pim.12160. [DOI] [PubMed] [Google Scholar]
- Mahallawi W.H., Khabour O.F., Zhang Q., Makhdoum H.M., Suliman B.A. MERS-CoV infection in humans is associated with a pro-inflammatory Th1 and Th17 cytokine profile. Cytokine. 2018;104:8–13. doi: 10.1016/j.cyto.2018.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maizels R.M., McSorley H.J. Regulation of the host immune system by helminth parasites. J. Allergy Clin. Immunol. 2016;138:666–675. doi: 10.1016/j.jaci.2016.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik Y.A. Properties of Coronavirus and SARS-CoV-2. Malaysian J. Pathol. 2020;42:3–11. [PubMed] [Google Scholar]
- Maria B., Villanueva G., Koizumi S., Jonai H. Cytokine production by human peripheral blood mononuclear cells after exposure to heavy metals. J. Health Sci. 2000;46:6–21. doi: 10.1248/jhs.46.358. [DOI] [Google Scholar]
- McFarlane A.J., McSorley H.J., Davidson D.J., Fitch P.M., Errington C., Mackenzie K.J., Gollwitzer E.S., Johnston C.J., MacDonald A.S., Edwards M.R. Enteric helminth-induced type I interferon signaling protects against pulmonary virus infection through interaction with the microbiota. J. Allergy Clin. Immunol. 2017;140:1068–1078. doi: 10.1016/j.jaci.2017.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McSorley H.J., Gaze S., Daveson J., Jones D., Anderson R.P., Clouston A., Ruyssers N.E., Speare R., McCarthy J.S., Engwerda C.R., Croese J., Loukas A. Suppression of inflammatory immune responses in celiac disease by experimental hookworm infection. PLoS ONE. 2011;6 doi: 10.1371/journal.pone.0024092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreau E., Chauvin A. Immunity against helminths: interactions with the host and the intercurrent infections. J. Biomed. Biotechnol. 2010;2010 doi: 10.1155/2010/428593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munjita S.M., Samutela M., Ndashe K., Munsaka S.M. Immunity, parasites, genetics and sex hormones: contributors to mild inflammatory responses in COVID-19? Pan African Med. J. 2020;35:36. doi: 10.11604/pamj.supp.2020.35.2.23267. https://doi.org/10.11604/pamj.supp.2020.35.2.23267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima Y., Goldblum R.M., Midoro-Horiuti T. Fetal exposure to bisphenol A as a risk factor for the development of childhood asthma: an animal model study. Environ. Health. 2012;11:1–7. doi: 10.1186/1476-069X-11-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paital B., Agrawal P.K. Air pollution by NO(2) and PM(2.5) explains COVID-19 infection severity by overexpression of angiotensin-converting enzyme 2 in respiratory cells: a review. Environ. Chem. Lett. 2020:1–18. doi: 10.1007/s10311-020-01091-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parija S.C., Chidambaram M., Mandal J. Epidemiology and clinical features of soil-transmitted helminths. Trop. Parasitol. 2017;7:81–85. doi: 10.4103/tp.TP_27_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pope C.A., Bhatnagar A., McCracken J.P., Abplanalp W., Conklin D.J., O’Toole T. Exposure to fine particulate air pollution is associated with endothelial injury and systemic inflammation. Circ. Res. 2016;119:1204–1214. doi: 10.1161/CIRCRESAHA.116.309279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qian Z., Travanty E.A., Oko L., Edeen K., Berglund A., Wang J., Ito Y., Holmes K.V., Mason R.J. Innate immune response of human alveolar type II cells infected with severe acute respiratory syndrome-coronavirus. Am. J. Respir. Cell Mol. Biol. 2013;48:742–748. doi: 10.1165/rcmb.2012-0339OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinete N., Hauser-Davis R.A. Drinking water pollutants may affect the immune system: concerns regarding COVID-19 health effects. Environ. Sci. Pollut. Res. 2020;28:1235–1246. doi: 10.1007/s11356-020-11487-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radbin R., Vahedi F., Chamani J. The influence of drinking-water pollution with heavy metal on the expression of IL-4 and IFN-γ in mice by real-time polymerase chain reaction. Cytotechnology. 2014;66:769–777. doi: 10.1007/s10616-013-9626-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajamanickam A., Munisankar S., Dolla C., Menon P.A., Thiruvengadam K., Nutman T.B., Babu S. Helminth infection modulates systemic pro-inflammatory cytokines and chemokines implicated in type 2 diabetes mellitus pathogenesis. PLoS Negl. Trop. Dis. 2020;14 doi: 10.1371/journal.pntd.0008101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Redelman-Sidi G. Could BCG be used to protect against COVID-19? Nat. Rev. Urol. 2020;17:316–317. doi: 10.1038/s41585-020-0325-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez C. The global helminth belt and Covid-19: the new eosinophilic link. Qeios. 2020 doi: 10.32388/IWKQH9.2. [DOI] [Google Scholar]
- Rolot M., Dougall A.M., Chetty A., Javaux J., Chen T., Xiao X., Machiels B., Selkirk M.E., Maizels R.M., Hokke C. Helminth-induced IL-4 expands bystander memory CD8+ T cells for early control of viral infection. Nat. Commun. 2018;9:1–16. doi: 10.1038/s41467-018-06978-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sajuthi S.P., DeFord P., Li Y., Jackson N.D., Montgomery M.T., Everman J.L., Rios C.L., Pruesse E., Nolin J.D., Plender E.G., et al. Type 2 and interferon inflammation regulate SARS-CoV-2 entry factor expression in the airway epithelium. Nat. Commun. 2020;11:1–18. doi: 10.1038/s41467-020-18781-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salgame P., Yap G.S., Gause W.C. Effect of helminth-induced immunity on infections with microbial pathogens. Nat. Immunol. 2013;14:1118–1126. doi: 10.1038/ni.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanderson C.J. Interleukin-5, eosinophils, and disease. Blood. 1992;79:3101–3109. [PubMed] [Google Scholar]
- Sanità di Toppi L., Sanità di Toppi L., Bellini E. Novel coronavirus: How atmospheric particulate affects our environment and health. Challenges. 2020;11:1–12. doi: 10.3390/challe11010006. [DOI] [Google Scholar]
- Santos J.H.A., Bührer-Sékula S., Melo G.C., Cordeiro-Santos M., Pimentel J.P.D., Gomes-Silva A., Costa A.G., Saraceni V., Da-Cruz A.M., Lacerda M.V.G. Ascaris lumbricoides coinfection reduces tissue damage by decreasing IL-6 levels without altering clinical evolution of pulmonary tuberculosis or Th1/Th2/Th17 cytokine profile. Rev. Soc. Bras. Med. Trop. 2019;52 doi: 10.1590/0037-8682-0315-2019. [DOI] [PubMed] [Google Scholar]
- Saraswathy S., Dixit R., Agrawal P., Krishnan M. Air pollution and cytokines. J. Med. Acad. 2018;1:43–49. doi: 10.5005/jp-journals-10070-0008. [DOI] [Google Scholar]
- Schett G., Sticherling M., Neurath M.F. COVID-19: risk for cytokine targeting in chronic inflammatory diseases? Nat. Rev. Immunol. 2020;20:271–272. doi: 10.1038/s41577-020-0312-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz C., Hams E., Fallon P.G. Helminth modulation of lung inflammation. Trends Parasitol. 2018;34:388–403. doi: 10.1016/j.pt.2017.12.007. [DOI] [PubMed] [Google Scholar]
- Shea-Donohue, T., Sullivan, C., Finkelman, F.D., Madden, K.B., Morris, S.C., Goldhill, J., Piñeiro-Carrero, V., Urban, J.F., 2001. The role of IL-4 in Heligmosomoides polygyrus-induced alterations in murine intestinal epithelial cell function. J. Immunol. (Baltimore, Md: 1950) 167, 2234–2239. 10.4049/jimmunol.167.4.2234. [DOI] [PubMed]
- Singh J., Samal J., Kumar V., Sharma J., Agrawal U., Ehtesham N.Z., et al. Structure-function analyses of new SARS-CoV-2 variants B.1.1.7, B.1.351 and B.1.1.28.1: clinical, diagnostic, therapeutic and public health implications. Viruses. 2021;3:439. doi: 10.3390/v13030439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith J.C., Sausville E.L., Girish V., Yuan M.L., Vasudevan A., John K.M., Sheltzer J.M. Cigarette smoke exposure and inflammatory signaling increase the expression of the SARS-CoV-2 receptor ACE2 in the respiratory tract. Dev. Cell. 2020;53:514–529. doi: 10.1016/j.devcel.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto-Peña G.A., Luna A.L., Acosta-Saavedra L., Conde-Moo P., López-Carrillo L., Cebrián M.E., Bastida M., Calderón-Aranda E.S., Vega L. Assessment of lymphocyte subpopulations and cytokine secretion in children exposed to arsenic. FASEB J. 2006;20:779–781. doi: 10.1096/fj.05-4860fjen. [DOI] [PubMed] [Google Scholar]
- Su C., Su L., Li Y., Long S.R., Chang J., Zhang W., Walker W.A., Xavier R.J. Helminth-induced alterations of the gut microbiota exacerbate bacterial colitis. Mucosal Immunol. 2018;11:144–157. doi: 10.1038/mi.2017.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanni F., Akker E., Zaman M.M., Figueroa N., Tharian B., Hupart K.H. Eosinopenia and COVID-19. J. Am. Osteopathic Assoc. 2020;120:504–508. doi: 10.7556/jaoa.2020.091. [DOI] [PubMed] [Google Scholar]
- Tao, Y., Yang, R., Wen, C., Fan, J., Ma, J., He, Q., Zhao, Z., Song, X., Chen, H., Shi, G., 2020. SARS-CoV-2 entry related genes are comparably expressed in children's lung as adults. medRxiv 2020.05.25.20110890. 10.1101/2020.05.25.20110890. [DOI]
- The Indian Express, 2021. Explained: B.1.617 variant and the Covid-19 surge in India. https://indianexpress.com/article/explained/maharashtra-double-mutant-found-b-1-617-variant-and-the-surge-7274080/ (accessed 27 April 2021).
- Vabret N., Britton G.J., Gruber C., Hegde S., Kim J., Kuksin M., Levantovsky R., et al. Immunology of COVID-19: current state of the science. Immunity. 2020;52:910–941. doi: 10.1016/j.immuni.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viana S.D., Nunes S., Reis F. ACE2 imbalance as a key player for the poor outcomes in COVID-19 patients with age-related comorbidities - Role of gut microbiota dysbiosis. Ageing Res. Rev. 2020;62 doi: 10.1016/j.arr.2020.101123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Western Cape Department of Health (WCDH) in collaboration with the National Institute for Communicable Diseases (NICD) in South Africa, 2020. Risk Factors for Coronavirus Disease 2019 (COVID-19) Death in a Population Cohort Study from the Western Cape Province, South Africa. Clinical Infectious Diseases ciaa1198, 10.1093/cid/ciaa1198. [DOI]
- Wong C.K., Lam C.W., Wu A.K., Ip W.K., Lee N.L., Chan I.H., Lit L.C., Hui D.S., Chan M.H., Chung S.S., Sung J.J. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin. Exp. Immunol. 2004;136:95–103. doi: 10.1111/j.1365-2249.2004.02415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodby B., Arnold M.M., Valacchi G. SARS-CoV-2 infection, COVID-19 pathogenesis, and exposure to air pollution: What is the connection? Ann. N. Y. Acad. Sci. 2021;1486:15–38. doi: 10.1111/nyas.14512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization (WHO), 2016. WHO releases country estimates on air pollution exposure and health impact. http://www.who.int/medicentre/news/releases/2016/air-pollution-estimates/en/ (accessed 20 January 2021).
- World Health Organization (WHO), 2020. Soil-transmitted helminth infections. https://www.who.int/news-room/fact-sheets/detail/soil-transmitted-helminth-infections (accessed 20 January 2021).
- World Health Organization (WHO), 2021a. Soil-transmitted helminthiases. https://apps.who.int/neglected_diseases/ntddata/sth/sth.html (accessed 20 April 2021).
- World Health Organization (WHO), 2021b. SDG Indicator 11.6.2 Concentrations of fine particulate matter (PM2.5). https://www.who.int/data/gho/data/indicators/indicator-details/GHO/concentrations-of-fine-particulate-matter-(pm2-5) (accessed 22 April 2021).
- Worldometer, 2021. COVID-19 coronavirus pandemic live updates. https://www.worldometers.info/coronavirus/ (accessed 24 April 2021).
- Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.-L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, X., Nethery, R.C., Sabath, B.M., Braun, D., Dominici, F., 2020. Exposure to air pollution and COVID-19 mortality in the United States: A nationwide cross-sectional study. MedRxiv 2020.04.05.20054502. 10.1101/2020.04.05.20054502. [DOI]
- Yeoh Y.K., Zuo T., Lui G.C., Zhang F., Liu Q., Li A.Y., Chung A.C., Cheung C.P., et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021;70:698–706. doi: 10.1136/gutjnl-2020-323020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youn J.-Y., Park H.-Y., Lee J.-W., Jung I.-O., Choi K.-H., Kim K., Cho K.-H. Evaluation of the immune response following exposure of mice to bisphenol A: induction of Th1 cytokine and prolactin by BPA exposure in the mouse spleen cells. Arch. Pharmacal Res. 2002;25:946–953. doi: 10.1007/BF02977018. [DOI] [PubMed] [Google Scholar]
- Zhao Q., Meng M., Kumar R., Wu Y., Huang J., Lian N., Deng Y., Lin S. The impact of COPD and smoking history on the severity of COVID-19: A systemic review and meta-analysis. J. Med. Virol. 2020;92:1915–1921. doi: 10.1002/jmv.25889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng L., Dong G.-H., Zhang Y.-H., Liang Z.-F., Jin Y.-H., He Q.-C. Type 1 and Type 2 cytokines imbalance in adult male C57BL/6 mice following a 7-day oral exposure to perfluorooctanesulfonate (PFOS) J. Immunotoxicol. 2011;8:30–38. doi: 10.3109/1547691X.2010.537287. [DOI] [PubMed] [Google Scholar]
- Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet (London, England) 2020;395:1054–1062. doi: 10.1016/s0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y., Qin X.-D., Zeng X.-W., Paul G., Morawska L., Su M.-W., Tsai C.-H., Wang S.-Q., Lee Y.L., Dong G.-H. Associations of serum perfluoroalkyl acid levels with T-helper cell-specific cytokines in children: By gender and asthma status. Sci. Total Environ. 2016;559:166–173. doi: 10.1016/j.scitotenv.2016.03.187. [DOI] [PubMed] [Google Scholar]
- Zhu Y., Xie J., Huang F., Cao L. Association between short-term exposure to air pollution and COVID-19 infection: evidence from China. Sci. Total Environ. 2020;727 doi: 10.1016/j.scitotenv.2020.138704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo T., Liu Q., Zhang F., Lui G.C., Tso E.Y., Yeoh Y.K., et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut. 2021;70:276–284. doi: 10.1136/gutjnl-2020-322294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo T., Zhang F., Lui G.C.Y., Yeoh Y.K., Li A.Y.L., Zhan H., Wan Y., et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020;159:944–955. doi: 10.1053/j.gastro.2020.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]