Abstract
Purpose
Although SABR can improve oncologic outcomes for patients with oligometastatic disease, treatment of metastases near critical organs remains challenging. The purpose of this study is to determine the dosimetric feasibility of delivering magnetic resonance imaging (MRI)-guided adaptive SABR in a single fraction for abdominal and thoracic metastases.
Methods and Materials
Previously delivered MRI-guided radiation therapy plans for 20 patients with oligometastatic disease in the thorax or abdomen, with 70% (14/20) of the lesions within 8 mm from dose-limiting organs at risk (OARs), were used to simulate the delivery of 24 Gy in a single fraction. Planning objectives included planning target volume (PTV) V95% >90%, optimized PTV (PTVopt) V95% >90%, and PTVopt D99% >20 Gy with no OAR dose violations, where PTVopt removed overlap with nearby planning organ at risk volume (PRV). Single-fraction plans were simulated on the first 5 daily setup breath-hold MRI scans, and the plans were reoptimized to consider variations in setup position and anatomy.
Results
The mean PTV V95% for single-fraction SABR plans was lower compared with multifraction plans (mean 85.4% vs 92.6%, P = .02), but mean PTVopt V95% was not different (95.3% vs 98.2%, P = .62). After reoptimization of the single-fraction plan to the treatment day MRI, there was an increase in mean PTV V95% (85.0% vs 88.1%, P = .05), increase in mean PTVopt V95% (92.7% vs 96.3%, P = .02), increase in mean PTVopt D99% (19.7 Gy vs 23.8 Gy, P < .01), increase in mean frequency of meeting PTV D99% >20 Gy (52% vs 87%, P < .01), and increase in mean gross tumor volume minimum dose (17.5 Gy vs 19.3 Gy, P < .01). Reoptimization decreased mean frequency of OAR dose constraint violation (48% vs 0%, P < .01).
Conclusions
Single-fraction MRI-guided SABR is a dosimetrically feasible treatment for oligometastases that allows for on-table adaptation to avoid OAR dose constraint violations, but this method requires clinical validation.
Introduction
In select patients with oligometastatic disease, SABR can improve local control and overall survival at the risk of greater radiation toxicities.1, 2, 3 Studies have shown a higher risk of toxicities after SABR for metastases near serial organs, such as the trachea or the gastrointestinal tract, and these studies recommend strict adherence to dosimetric constraints.4, 5, 6 Despite awareness of the increased risks in treating lesions close to serial organs at risk (OARs) and careful planning, in the SABR-COMET phase 2 trial, which randomized patients with 1 to 5 metastases of various histologic types to standard palliative care versus standard palliative care plus SABR to all metastatic sites, 3 (4.5%) patients in the SABR arm had treatment-related death. This included 1 patient with a central lung metastasis who developed dyspnea and hemoptysis and 1 patient with an adrenal metastasis who developed a gastric perforation.3 Similarly, in the RTOG 0813 dose-escalation study for centrally located early stage non-small cell lung cancer, 8 (12.1%) of patients treated to 57.5 to 60 Gy in 5 fractions experienced grade ≥3 toxicity 1 year after treatment, with 4 (5.7%) deaths, including a possible esophageal perforation and bronchopulmonary hemorrhages.5
Randomized data support the use of single-fraction SABR for peripheral early-stage non-small cell lung cancer, and there is growing evidence that single-fraction SABR is well tolerated and can provide local control for renal cell carcinoma and oligometastases of all types of histology, in particular for those located in the liver or adrenal glands.7, 8, 9, 10, 11 Phase 1 to 2 studies report a steep dose-dependent increase in local control after single-fraction SABR between 18 and 24 Gy, plateauing at 24 Gy with 90% local control at 1 to 3 years.9 Although a single fraction of 18 Gy has been used to treat central lung lesions, higher minimum doses to the gross tumor volume (GTV) may improve the durability of local control.8,12 Subdiaphragmatic oligometastases appear to have worse local control owing to the challenges of daily setup, including respiratory motion, peristalsis, and poor soft tissue contrast on cone beam computed tomography (CBCT).9
Integrated magnetic resonance (MR)-linear accelerator and treatment planning systems (MR-linacs) may allow for safer delivery of single-fraction SABR to oligometastases as these systems are equipped with superior soft-tissue contrast visualization, on-table adaptive planning, and real-time tumor tracking with respiratory motion triggered beam gating.13, 14, 15 Phase 1 trials have demonstrated the safety of MR-guided online adaptive SABR delivered in 5 fractions for oligometastases and unresectable primaries of the abdomen and central thorax.16,17 The VU Medical Center demonstrated that MR-guided online adaptive, single-fraction SABR can be performed on patients with early-stage, peripherally located lung cancer.
The purpose of this study is to determine the dosimetric feasibility of delivering MR-guided adaptive SABR in a single fraction for abdominal and thoracic metastases, including centrally located lung lesions. We conducted a single institution retrospective analysis of previously delivered 0.35T MR-guided radiation therapy plans for oligometastatic disease and simulated the delivery of 24 Gy in a single fraction. We hypothesize that online-adaptive MR-guidance will allow for single-fraction SABR by avoiding OAR dose violations while allowing for individualized target dose escalation as permitted by surrounding OAR anatomy.
Methods and Materials
Patient selection
Patients (n = 20) with oligometastatic disease to the abdomen or lungs treated with hypofractionated radiation or SABR using maximum inhale breath hold respiratory gating on an integrated 0.35T MR-guided radiation therapy unit at our institution between 2016 and 2019 were retrospectively analyzed on an institutional review board–approved study. Ten patients with abdominal metastases and ten patients with lung metastases were selected as potentially appropriate for single-fraction online-adaptive SABR simulated treatment on a 0.35T MR-linac. These oligometastatic cases were selected because they were close to critical OARs or because they were located in areas with high respiratory motion and were representative of the patient population treated with multifraction SABR on the MR-linac at our institution. This study was approved by the institutional review board at the University of Wisconsin with approval number UW17115.
Radiation therapy planning
Selected patients underwent MR simulation on ViewRay (Oakwood Village, OH) and computed tomography (CT) simulation on Siemens Somatom Definition Edge (Siemens Healthcare, Erlangen, Germany). For both MR and CT simulation, patients were positioned and immobilized in head-first supine position on a thin mattress with an alpha cradle with both arms up using a wing board and a triangular knee support. Gadoxetate disodium (Bayer, Leverkusen, Germany) was used for liver metastases cases, and 3 hours of fasting before simulation scans and radiation delivery was required for any patients with metastases near gastrointestinal luminal organs. Planning MR field of view was either 40 × 40 × 43 cm or 54 × 47 × 43 cm with resolution of 0.15 × 0.15 × 0.3 cm with torso coils placed on patients. CT scans were acquired without dummy coils. Both MR and CT scans were acquired in maximum inhale breathe hold phase. All scans were exported to MIM (MIM Software Inc, version 6.6.11, Cleveland, OH) for segmentation of target and critical structures. All contours were completed by an experienced radiation oncologist. CT and MR scans with contours were exported to the ViewRay treatment planning system for planning. Routine commissioning involved exporting the contours between MIM and ViewRay to confirm consistency in contour location, shape, and volume. In addition, contours were visually inspected for accuracy in the ViewRay treatment planning system before planning. Plans were generated with MR as the primary image using a step-and-shoot intensity modulated radiation therapy technique and Monte Carlo dose calculation algorithm. The planning MR was registered to CT for electron density information.
In addition to the target and organs at risk, planning structures such as a ring (4 cm expansion of planning target volume [PTV] but excluding PTV), normal (everything outside the ring), and planning risk volumes with an expansion of 5 mm were contoured for better target coverage and limiting dose to OARs. An air and soft tissue override contour was created to accommodate changes in air volume in the bowel or stomach. All plans were generated with 6 MV flattening free filter photon beam. Plans used 10 to 18 beams with beams spaced 15 to 20 degrees; total beam segments varied from 24 to 104, depending on the shape of the PTV and surrounding OAR anatomy. A single isocenter was used for all plans. Total treatment time was 30 to 45 minutes and included beam-on time, which ranged from 2 to 9 minutes at a dose rate of 600 MU/min, and multileaf collimator and gantry motion, which ranged from 5 to 12 minutes. The beam-on duty cycle depended on each patient’s breath hold duration, which varied between 5 and 20 seconds. Dose optimization and final dose calculation were performed with a 2 mm dose grid and the magnetic field on to account for the Lorentz force.
Single-fraction SABR plans were created on the MR imaging (MRI) simulation with the clinically approved target and OAR contours from the original multifraction plan. Optimized PTVs (PTVopt) were created by a Boolean subtraction of the 5 mm expansion around luminal organs from the 3 mm expansion around the GTV, which is standard for breath-hold respiratory gated MR-linac treatments.18 Using a prescription of 24 Gy in 1 fraction, planning goals included PTV V95% >90%, PTVopt V95% >90%, and PTVopt D99% >20 Gy with no OAR dose violations. Dose constraints were based on the AAPM Task Group 101 and the Folkert and Timmerman recommendations.19,20 To further improve the safety of the plans, maximum voxel dose of the single-fraction plans was kept below the D0.035cc specified in the recommended dose constraints.
Adaptive reoptimization on daily MRI
To simulate the dosimetry on the day of treatment delivery, the single-fraction plans were retrospectively tested on the first 5 daily setup breath-hold MRI scans that were acquired during the clinical multifraction gated radiation treatment delivery. The daily setup MRI scans were registered to the planning MRI by manual rigid alignment of the GTVs. OAR contours were deformably registered, and contours within a 4 cm radius around the PTV were manually corrected as necessary.21 The PTVopt was updated if the anatomy of the surrounding OARs changed. The original single-fraction SABR plan was applied to the daily setup MRI scans to predict the dose distribution. The plan was then reoptimized to incorporate setup variation and new anatomy using original plan planning parameters. In cases in which the original single-fraction plan caused an OAR constraint violation, the plan was reoptimized to respect the OAR constraint. In cases with an OAR close to the PTV that did not violate any OAR dose constraints, the dose to the target was escalated through normalization until the dose constraint to the proximal OAR was met in an isotoxic approach. In lung oligometastatic cases without any proximal OARs, plans were reoptimized with a prescription dose to the target increased from 24 Gy to 26 Gy.
Evaluation parameters and statistical analysis
From the clinical multifraction plans, the volume of the GTV, PTV, and PTVopt and the frequency of clinical plan reoptimization were recorded. Dosimetric data comparing the multifraction and single-fraction plans include PTV V95%, and PTVopt V95%. Dosimetric data comparing the original single-fraction plan on the daily setup MRI (“predicted plan”) and the single-fraction plan adapted to the new anatomy (“reoptimized plan”) include PTV V95%, PTVopt V95%, PTVopt D99%, and the frequency and extent of OAR dose violations. Analysis of the predicted plans and the reoptimized plans was conducted by taking the mean of the metric of interest over the 5 setup MRI scans and then comparing the mean of these values over the 20 oligometastatic cases. The Wilcoxon signed-rank test was used to compare dosimetric data and the frequency of meeting dosimetric goals between the multifraction and single-fraction baseline plans and between the predicted and reoptimized fractional plans. All statistical analyses were done using MATLAB and Statistics Toolbox (Release 2018a, The MathWorks Inc, Natick, MA).
Results
Patient and tumor characteristics
Twenty patients with a median age of 64.5 (range, 42-85) years with oligometastatic disease of various histologic types in the thorax or abdomen were included in this study. Patients underwent radiation therapy on an MR-guided radiation therapy unit with a median dose of 50 Gy (range, 30-67.5 Gy), with 18 cases delivered in 5 fractions and 2 cases in 15 fractions. Seven of the 20 (35%) clinical multifraction plans required plan reoptimization. Overall, a mean of 17% of all clinically delivered fractions required adaptation. Six lung site metastases did not have any proximal OARs that limited the dose to the PTV, and the remaining 14 patients had dose-limiting OARs within 8 mm of the GTV (Table 1). The mean PTVopt to PTV ratio was 0.87 (range, 0.54-1.00), denoting the degree of proximity or overlap between the PTV and OARs.
Table 1.
Tumor characteristics of the 20 oligometastatic lesions included for analysis
| Site | Histology | GTV (cm3) | PTV (cm3) | PTV-opt (cm3) | PTV-opt /PTV | Constraining OAR | Max dose constraint (Gy) |
|---|---|---|---|---|---|---|---|
| Lung | Pancreas adenocarcinoma | 1.51 | 6.31 | 3.39 | 0.54 | Bronchus | 20.2 |
| Lung | Lung adenocarcinoma | 13.11 | 26.40 | 26.10 | 0.99 | Stomach | 12.4 |
| Lung | Lung adenocarcinoma | 8.07 | 25.43 | 22.00 | 0.87 | Esophagus | 15.4 |
| Lung | Esophagus adenocarcinoma | 10.85 | 24.59 | 17.75 | 0.72 | Rib | 22∗ |
| Lung | Cholangiocarcinoma | 3.35 | 13.57 | 10.02 | 0.74 | Rib | 22∗ |
| Lung | Breast invasive ductal carcinoma | 3.77 | 16.23 | 16.23 | 1.00 | Rib | 22∗ |
| Lung | Leiomyosarcoma | 1.15 | 7.77 | 7.77 | 1.00 | NA | NA |
| Lung | Melanoma | 0.79 | 7.16 | 7.16 | 1.00 | NA | NA |
| Lung | Renal cell carcinoma | 4.18 | 12.55 | 12.55 | 1.00 | NA | NA |
| Lung | Colorectal adenocarcinoma | 1.02 | 4.02 | 4.02 | 1.00 | NA | NA |
| Kidney | Colorectal adenocarcinoma | 8.57 | 19.63 | 16.00 | 0.82 | Small bowel | 15.4 |
| Kidney | Esophagus adenocarcinoma | 6.82 | 17.46 | 14.95 | 0.86 | Small bowel | 15.4 |
| Adrenal | Lung adenocarcinoma | 49.27 | 87.22 | 79.74 | 0.91 | Stomach | 12.4 |
| Portal lymph node | Hepatocellular carcinoma | 65.66 | 112.06 | 108.70 | 0.97 | Duodenum | 12.4 |
| Portacaval lymph node | Gall bladder adenocarcinoma | 7.17 | 60.74 | 53.56 | 0.88 | Stomach | 12.4 |
| Gastrohepatic lymph node | Esophagus adenocarcinoma | 12.50 | 21.61 | 16.57 | 0.77 | Stomach | 12.4 |
| Liver | Breast invasive ductal carcinoma | 1.64 | 9.51 | 8.87 | 0.93 | Stomach | 12.4 |
| Liver | Gall bladder adenocarcinoma | 84.18 | 144.98 | 133.62 | 0.92 | Duodenum | 12.4 |
| Liver | Lung adenocarcinoma | 5.61 | 13.78 | 9.60 | 0.70 | Colon | 18.4 |
| Liver | Pancreas adenocarcinoma | 33.34 | 58.12 | 49.79 | 0.86 | Colon | 18.4 |
| Mean (range) | 16.12 (0.79-84.18) | 34.46 (4.02-144.98) | 30.92 (3.39-133.62) | 0.87 (0.54-1.00) | |||
Each lesion with a corresponding dose-limiting OAR and the maximum dose constraint of that OAR is indicated. PTVopt created by a Boolean subtraction of the 3 to 5 mm expansion around luminal organs from the PTV.
Abbreviations: GTV = gross tumor volume; NA = not applicable; OAR = organ at risk; PTV = planning target volume.
D1cc (minimum dose to the 1 cm3 receiving the highest dose).
Multifraction versus single-fraction plans based on simulation images
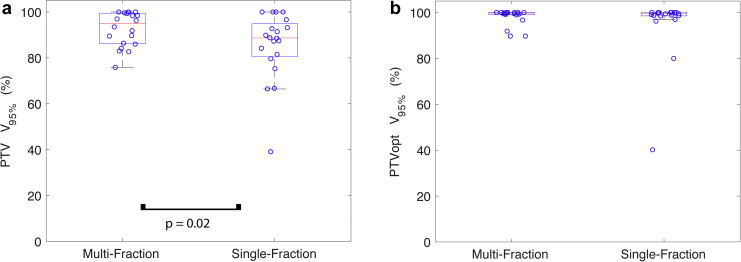
Single-fraction SABR plans were created that respected OAR dose constraints while maximizing PTV coverage. Total treatment time is estimated to be 50 to 60 minutes. This would include beam-on time of 5 to 20 minutes at a dose rate of 600 MU/min and multileaf collimator and gantry motion of 10 to 25 minutes. The beam-on duty cycle would depend on each patient’s breath hold duration, which varies between 5 and 20 seconds. Although the PTV V95% was significantly higher in the original multifraction plan compared with the single-fraction plan (mean 92.6% vs 85.4%, P = .02), the PTVopt V95% was not statistically different (mean 98.2% vs 95.3%, P = .62; Fig 1). For the 14 cases with a proximal OAR that limited dose delivery, the ratio of the limiting maximum dose constraint to the prescription dose was not statistically different between the multifraction and single-fraction plans (mean 62% vs 68%, P = .47).
Figure 1.
Comparison of target dose coverage between multifraction and single-fraction magnetic resonance-guided SABR. (a) Planning target volume (PTV) V95% is significantly greater for the multifraction plan but (b) optimized PTV (PTVopt) V95% is not significantly different. Red line denotes the median, and the edges of the box are the 25th and 75th percentiles; the whiskers extend over mean ± 2.7 × standard deviation, and open circles denote each of the 20 oligometastases cases analyzed.
Predicted versus reoptimized plan target dose delivery
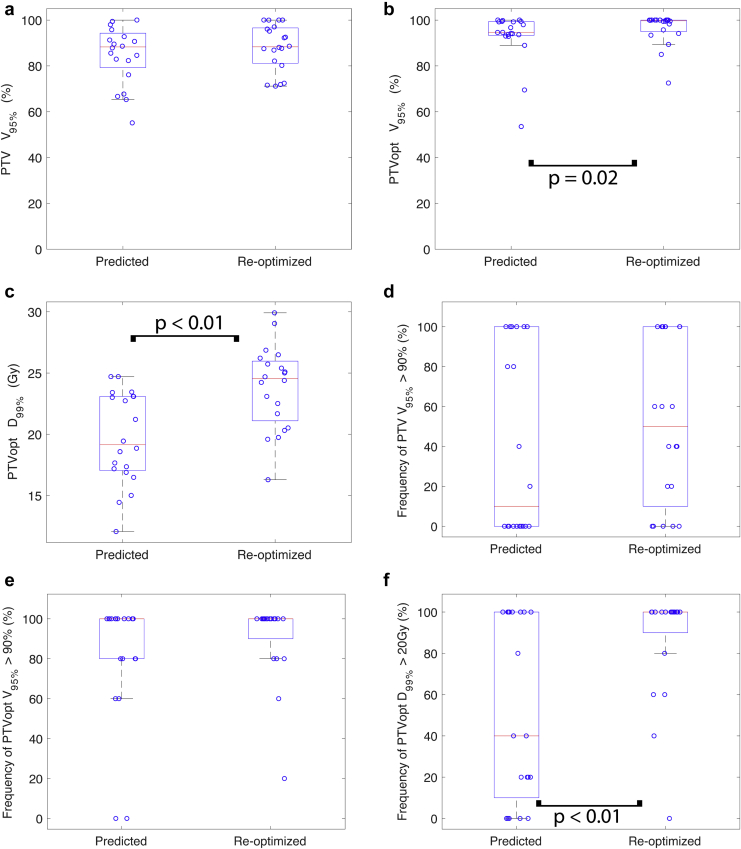
In comparing the dosimetry of the single-fraction SABR predicted plans to the reoptimized plans on the first 5 setup MRI scans, there was a borderline significant 4.7% increase in the mean PTV V95% when the predicted plan was reoptimized (85.0% vs 88.1%, P = .05; Fig 2). Similarly, there was a significant 5.1% increase in the mean PTVopt V95% (92.7% vs 96.3%, P = .02) and a significant 24.8% increase in the mean PTVopt D99% (19.7 Gy vs 23.8 Gy, P < .01). Although the mean GTV maximum dose was not different (31.5 Gy vs 32.6 Gy, P = .20), there was a 15.8% increase in the mean GTV minimum dose (17.5 Gy vs 19.3 Gy, P < .01). The mean frequency of meeting the dosimetric goals of PTV V95% >90% and PTVopt V95% >90% was similar after the predicted plan was reoptimized (41% vs 52%, P = .24; 82% vs 91%, P = .18, respectively). The mean frequency of meeting the dosimetric goal of PTVopt D99% >20 Gy increased by 35% (52% vs 87%, P < .01). An example of the dosimetric changes is shown in Figure 3.
Figure 2.
Comparison of mean target dose coverage between the original plan on 5 daily fraction magnetic resonance imaging scans (predicted) and after reoptimization (reoptimized). (a) Planning target volume (PTV) V95%, (b) optimized PTV (PTVopt) V95%, (c) PTVopt D99%, (d) frequency of PTV V95% >90%, (e) frequency of PTVopt V95% >90%, and (f) frequency of PTVopt D99% >20 Gy. Red line denotes the median, and the edges of the box are the 25th and 75th percentiles; the whiskers extend over mean ± 2.7 × standard deviation, and open circles denote each of the 20 oligometastases cases analyzed.
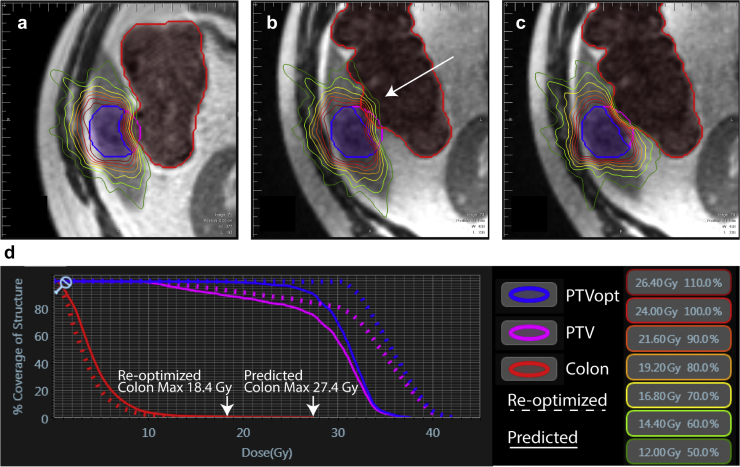
Figure 3.
Dosimetry for single-fraction magnetic resonance-guided SABR at (a) baseline planning, (b) day of treatment, and (c) day of treatment after reoptimization of an inferior liver site metastasis near the colon. (d) Dose-volume histogram curves for the planning target volume and the colon before (Planned) and after reoptimization. Note the avoidance of maximum dose violations to the colon with reoptimization (arrows).
OAR dose constraint metrics
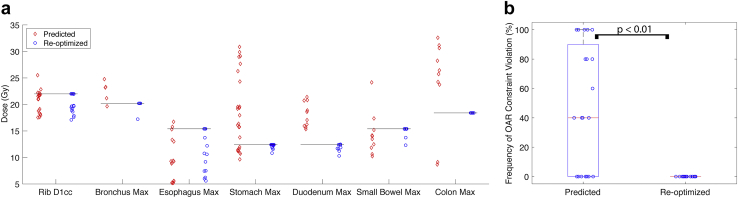
The mean frequency of violating OAR dose constraints was significantly decreased after the predicted plan was reoptimized (48% vs 0%, P < .01; Fig 4). In the remaining 52% of cases in which there were no OAR dose constraint violations, the dose to the PTV was escalated. Due to the combination of ensuring dose constraints are respected and dose escalation, the distribution of maximum doses to the proximal OARs were concentrated at or just below the OAR dose constraint after isotoxic reoptimization.
Figure 4.
(a) Comparison of the maximum doses to the constraining organs at risk (OARs) from the original plan on 5 daily fraction magnetic resonance imaging scans (predicted) and after reoptimization (reoptimized). The black lines denote the respective OAR maximum dose constraint. (b) Mean frequency of dose OAR constraint violation from the original plan on 5 daily fraction magnetic resonance imaging scans (predicted) and after reoptimization (reoptimized). Red line denotes the median, and the edges of the box are the 25th and 75th percentiles; the whiskers extend over mean ± 2.7 × standard deviation, and open circles denote each of the 20 oligometastases cases analyzed.
Discussion
In the 1950s, the stereotactic frame enabled Lars Leksell to deliver single ablative doses of radiation to brain tumors while limiting dose to healthy brain tissue.22 Similarly, the integrated MR-linac system is a technological advancement that can visualize soft tissue anatomy and allow on-table adaptation and respiratory-gated radiation therapy. The MR-linac may enable the safe delivery of single-fraction SABR to tumors close to critical serial OARs in the central lung and in the abdomen and pelvis.
In this study of 20 oligometastatic cases, with 70% (14/20) of cases with a PTV within 8 mm of a serial OAR, we show that single-fraction MR-guided SABR treatment is dosimetrically feasible, with comparable coverage of the PTVopt to multifraction clinical plans. By simulating the delivery of the single-fraction SABR plans on the first 5 daily fraction MRI scans, we show that on-table adaptation can increase the target coverage and dose and can avoid OAR dose constraint violations.
A few institutions have investigated the use of real-time MR-guided SABR for the treatment of oligometastases, most commonly in 5-fraction schedules. Using online adaptive reoptimization, these studies show an improvement in target coverage and OAR sparing, although different metrics were used as planning objectives. The VU Medical Center reported the treatment of 17 patients with adrenal metastases treated to a range of dose and fractionations (24 Gy in 3 fractions, 50 Gy in 5 fractions, and 60 Gy in 8 fractions). On-table adaptation led to an improvement in meeting the objective of PTV V95% >95% from 20% to 51%. The same institution reported the treatment of 50 patients with lung site metastases or primaries, of which 57% of cases had central lesions, treated with a range of doses in 3 to 12 fractions. On-table adaptation resulted in a median PTV V100% of 95%. Of all the delivered fractions, reoptimization was used for OAR sparing in 5%, for improvement in PTV coverage in 61% and for both in 10%. At 12 months, there was an 8% rate of grade 3 toxicity and no grade 4 or 5 toxicity.23 More recently, the VU Medical Center reported the treatment of 10 patients with early stage non-small cell lung cancer or lung metastases measuring ≤5 cm and located ≥2 cm from the proximal bronchial tree with MR-guided single-fraction SABR to a dose of 34 Gy. On-table adaptation improved PTV mean V34Gy from 89.8% to 95.0% with no OAR dose violations on any predicted or reoptimized plans. At a median follow-up of 5 months, no grade ≥3 or local recurrences were observed.24 Washington University in St Louis reported the treatment of 20 patients with abdominal oligometastatic or unresectable primary malignancies with 50 Gy in 5 fractions. After on-table adaptation, PTV V95% was improved from a mean of 76% to 79%. In addition, 63% of fractions required reoptimization due to OAR dose violations and 21% of fractions were dose escalated. At 6 months of follow-up, there were no grade ≥3 toxicities.16 The University Medical Center in Utrecht reported the treatment of 20 patients with lymph node oligometastatic disease treated to 35 Gy in 5 fractions without respiratory gating. Target coverage PTV V100 improved from a range of 47% to 100% to a range of 90% to 100% with on-table adaptation, and the median coverage was unchanged at 100% in the single lymph node plans. Target and OAR objectives were met in 19% of the predicted fractions and 84% of the reoptimized fractions.25
Other than the fractionation scheme, the abdominal SABR study by Henke et al from Washington University in St Louis was the most similar to our current study in terms of selection of patients with tumors close to serial OARs and in dosimetric outcomes.16 The 3% PTV V95% improvement in our study from 85% to 88% with reoptimization is similar the 3% improvement from 76% to 79% that reported by Henke et al, although the absolute numbers are higher in our study. However, our study had OAR dose violations in a mean of 48% of predicted plan fractions, compared with the 63% of fractions by Henke et al. This difference may be due to the inclusion of 6 patients in our study with peripheral lung metastases without any close OARs.
In comparison to the MR-guided multifraction SABR for oligometastases, there is more experience with single-fraction CBCT-based SABR. However, due to the inherently inferior soft-tissue contrast of CBCT and target position uncertainty with free-breathing, these treatments have largely been limited to bone, central nervous system, and peripheral lung metastases, away from the proximal bronchial tree and the gastrointestinal tract. The risks of treating central lung lesions with SABR is well described and can result in a 46% rate of grade ≥3 toxicity at 2 years. Therefore, the RTOG 0915 randomized control trial of 34 Gy single-fraction SABR versus multifraction SABR for early stage lung cancer excluded centrally located tumors.4,26 The SAFRON II randomized control trial of 28 Gy single-fraction SABR versus multifraction SABR for oligometastases also excluded centrally located lung metastases.27 Although Osti et al treated 49 central tumors with single-fraction SABR to 23 Gy and reported only 2 cases of grade ≥3 toxicity at 2 years, such results have not been replicated at other institutions.28
Similarly, prior studies on single-fraction SABR to the abdomen underscores the need to avoid serial OARs and most of these studies have excluded patients with lesions close to serial OARs. A retrospective review of nonmetastatic pancreatic cancer patients treated with either a single-fraction or 5-fraction SABR schedule by Pollom et al showed a significantly higher rate of grade ≥3 gastrointestinal toxicity at 12 months in the single-fraction group (12.3% vs 5.6%), highlighting the risks of delivering single-fraction SABR to tumors close to OARs in the abdomen without sufficient image guidance and the importance of tumor selection with respect to distance from OARs.29 In patients with liver metastases, single-fraction SABR can result in duodenal ulcers and biliary obstruction if the lesion is close to the porta hepatis region.30 A phase 1 trial at the UT Southwestern Medical Center on single-fraction SABR for liver metastases excluded lesions within a 2 cm expansion around the portal vein to its bifurcation.31
Other studies that have investigated the use of single-fraction SABR for oligometastases have either lowered the dose for lesions close to serial OARs or resorted to multiple-fraction regimens. In a study by Greco et al, 175 patients with 566 oligometastatic lesions at various body sites were treated with either 24 Gy in 1 fraction or 27 Gy in 3 fractions if the lesion was close to dose-limiting OARs. In contrast to most single-fraction SABR studies that have targeted relatively low-risk lesions in the bone or peripheral lung, 49% of the lesions in the Greco et al study were in lymph nodes, the liver, or soft tissue, although the majority of the lymph nodes were treated with 3 fractions. Overall, 24% of patients were treated with the 3-fraction regimen. At 5 years, local control was 92% for the single-dose regimen and 38% for the 3-fraction regimen, which underscores the need for better treatments to lesions close to OARs.9 In a study by Gandhidasan et al, 132 patients with oligometastatic lesions mainly to the lung (51%) and bones (40%) were treated with single-fraction SABR with doses determined by consensus at chart rounds ranging from 18 to 28 Gy. A dose of 18 Gy was mainly used for central lung lesions. Local control was 84% at 2 years.7 A review of single-fraction SABR studies by Ng et al showed promising data for the treatment of oligometastatic cancers. However, it concluded that multifraction SABR should be the preferred regimen given the lack of randomized control trials and lack of long-term efficacy and safety data. Future studies using MR-guided single-fraction SABR was recommended for the treatment of central lung cancers and liver metastases close to the portal vein and gastrointestinal tract.11
There are several advantages of using single-fraction MR-guided SABR. In a patient with several oligoprogressive metastatic lesions, a single-fraction treatment could substantially decrease time off systemic therapy, reduce the number of hospital visits, and improve the overall quality of life. MR-linacs are more expensive than CBCT-based devices, and the delivery of each on-table adaptive fraction takes longer and requires more supervision and expertise from therapists, physicians, and physicists.14 Single-fraction MR-guided SABR can significantly reduce costs in a resource-constrained or bundled payment environments.32 With the current uncertainty on the duration of the COVID-19 pandemic, several guidelines have been published encouraging the use of hypofractionation as a risk mitigation strategy.33, 34, 35, 36, 37 Single-fraction MR-guided SABR may provide an effective means of managing oligometastatic lesions while safely avoiding OARs with the fewest possible hospital visits for patients. Careful selection of appropriate patients through standardized classification of the extent of disease and the location and proximity to OARs may determine whether an MR-guided approach would be the most suitable.38 The best candidates for single-fraction MR-guided SABR are likely tumors within 1 to 2 cm of at least 1 critical organ without abutment or invasion. Tumors farther than 1 to 2 cm from critical organs could likely be safely treated with single fraction CBCT-based radiation therapy whereas tumors abutting or invading critical organs should be treated with multiple-fraction SABR.
Limitations of this study include the lack of clinical data to support the use of single-fraction MR-guided SABR for lesions close to serial OARs. Because this was a proof-of-concept in silico study, clinical efficacy in terms of long-term local control and rates of radiation toxicity has yet to be determined. Because tumor location with respect to proximal OARs dictates the need for plan adaptation, patient selection may have biased the results that show improvement in target coverage and OAR avoidance with MR-guided reoptimization. Six lung site metastatic tumors located greater than 1 cm from the critical OARs were included in this study to reflect the representative patient population with oligometastatic disease treated with multifraction SABR on the MR-linac at our institution. For example, oligometastatic disease to the brain was not included in this study because brain metastases are not treated with stereotactic radiosurgery on the MR-linac at our institution due to the lack of a suitable head coil and the increased positional uncertainty compared with CBCT-based treatments with the fusion of a diagnostic brain MRI. Use of the MR-linac for the treatment of oligometastatic disease in the lungs has had limited adoption not only because of the limited availability of MR-linacs but also because of the technical challenges of treating lung tumors on the MR-linac. The spatial resolution of the real-time sagittal cine imaging is low (3.5 mm × 3.5 mm × 5 mm) compared with CBCT (0.25 mm × 0.25 mm × 0.25 mm), and the breath-hold gating based on tumor tracking on the MR-linac can be difficult for tumors with a diameter less than 1.1 cm or for tumors that move outside of the sagittal plane.18,24,39 This study demonstrates the dosimetric feasibility of single-fraction SABR with MR-linac for treating oligometastatic disease in the thorax and abdomen by minimizing the effect of respiratory motion. Another limitation of this study and the general use of single-fraction SABR is the paucity of data regarding OAR dose limits and corresponding clinically measured toxicities. The AAPM Task Group 101 guidelines are largely based on the experience from the University of Texas Southwestern and the University of Virginia.19 These dose limits are not viewed as absolute constraints but serve as guidelines, which are based on toxicity observation, mathematical models, and educated approximations that are constantly updated.20,22 Given the lack of clinical validation for these dose limits, we have kept the maximum voxel dose of the single-fraction plan below the D0.035cc specified in AAPM dose constraints. Because there is no need for dose accumulation or deformable registration with single-fraction respiratory-gated SABR, experience with this form of treatment may help establish the most clinically accurate OAR dose limits.
Conclusions
Single-fraction MR-guided SABR appears to be a dosimetrically feasible alternative treatment option to multifraction SABR for oligometastases. Single-fraction MR-guided SABR allows for an efficient means of performing real-time isotoxic adaptation by avoiding OAR dose constraint violations and by escalating the dose and coverage of the target. This approach will require prospective clinical validation with long-term follow-up to establish safety and efficacy.
Footnotes
Sources of support: Dr Lee was supported by the Eric Wolfe Fellowship.
Disclosures: Dr Lee reports meeting travel reimbursement support from ViewRay. outside the submitted work, Dr Bayouth reports personal fees from ViewRay and is the cofounder of consulting company MR Guidance, LLC. Dr Bassetti reports meeting travel reimbursement support from ViewRay and clinical trial support from Merck, AstraZeneca, and EMD Serono.
Data Sharing Statement: Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
References
- 1.Gomez D.R., Tang C., Zhang J. Local consolidative therapy vs maintenance therapy or observation for patients with oligometastatic non-small cell lung cancer: Long-term results of a multi-institutional, phase II, randomized study. J Clin Oncol. 2019;37:1558–1565. doi: 10.1200/JCO.19.00201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Iyengar P., Wardak Z., Gerber D.E. Consolidative radiotherapy for limited metastatic non-small cell lung cancer: A phase 2 randomized clinical trial. JAMA Oncol. 2018;4 doi: 10.1001/jamaoncol.2017.3501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Palma D.A., Olson R., Harrow S. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): A randomised, phase 2, open-label trial. Lancet. 2019;393:2051–2058. doi: 10.1016/S0140-6736(18)32487-5. [DOI] [PubMed] [Google Scholar]
- 4.Timmerman R., McGarry R., Yiannoutsos C. Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J Clin Oncol. 2006;24:4833–4839. doi: 10.1200/JCO.2006.07.5937. [DOI] [PubMed] [Google Scholar]
- 5.Bezjak A., Paulus R., Gaspar L.E. Safety and efficacy of a five-fraction stereotactic body radiotherapy schedule for centrally located non-small cell lung cancer: NRG Oncology/RTOG 0813 Trial. J Clin Oncol. 2019;371:1316–1325. doi: 10.1200/JCO.18.00622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rusthoven K.E., Kavanagh B.D., Cardenes H. Multi-institutional phase I/II trial of stereotactic body radiation therapy for liver metastases. J Clin Oncol. 2009;27:1572–1578. doi: 10.1200/JCO.2008.19.6329. [DOI] [PubMed] [Google Scholar]
- 7.Gandhidasan S., Ball D., Kron T. Single fraction stereotactic ablative body radiotherapy for oligometastasis: Outcomes from 132 consecutive patients. Clin Oncol. 2018;30:178–184. doi: 10.1016/j.clon.2017.11.010. [DOI] [PubMed] [Google Scholar]
- 8.Siva S., Kirby K., Caine H. Comparison of single-fraction and multi-fraction stereotactic radiotherapy for patients with 18F-fluorodeoxyglucose positron emission tomography-staged pulmonary oligometastases. Clin Oncol. 2015;27:353–361. doi: 10.1016/j.clon.2015.01.004. [DOI] [PubMed] [Google Scholar]
- 9.Greco C., Pares O., Pimentel N. Phenotype-oriented ablation of oligometastatic cancer with single dose radiation therapy. Int J Radiat Oncol. 2019;104:593–603. doi: 10.1016/j.ijrobp.2019.02.033. [DOI] [PubMed] [Google Scholar]
- 10.Greco C., Zelefsky M.J., Lovelock M. Predictors of local control after single-dose stereotactic image-guided intensity-modulated radiotherapy for extracranial metastases. Int J Radiat Oncol. 2011;79:1151–1157. doi: 10.1016/j.ijrobp.2009.12.038. [DOI] [PubMed] [Google Scholar]
- 11.Ng S.S.W., Ning M.S., Lee P. Single-fraction stereotactic body radiation therapy: A paradigm during the coronavirus disease 2019 (COVID-19) pandemic and beyond? Adv Radiat Oncol. 2020;5:761–773. doi: 10.1016/j.adro.2020.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yamada Y., Katsoulakis E., Laufer I. The impact of histology and delivered dose on local control of spinal metastases treated with stereotactic radiosurgery. Neurosurg Focus. 2017;42:E6. doi: 10.3171/2016.9.FOCUS16369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Klüter S. Technical design and concept of a 0.35 T MR-Linac. Clin Transl Radiat Oncol. 2019;18:98–101. doi: 10.1016/j.ctro.2019.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van Herk M., McWilliam A., Dubec M. Magnetic resonance imaging-guided radiation therapy: A short strengths, weaknesses, opportunities, and threats analysis. Int J Radiat Oncol Biol Phys. 2018;101:1057–1060. doi: 10.1016/j.ijrobp.2017.11.009. [DOI] [PubMed] [Google Scholar]
- 15.Mutic S., Dempsey J.F. The ViewRay system: Magnetic resonance-guided and controlled radiotherapy. Semin Radiat Oncol. 2014;24:196–199. doi: 10.1016/j.semradonc.2014.02.008. [DOI] [PubMed] [Google Scholar]
- 16.Henke L., Kashani R., Robinson C. Phase I trial of stereotactic MR-guided online adaptive radiation therapy (SMART) for the treatment of oligometastatic or unresectable primary malignancies of the abdomen. Radiother Oncol. 2018;126:519–526. doi: 10.1016/j.radonc.2017.11.032. [DOI] [PubMed] [Google Scholar]
- 17.Henke L.E., Olsen J.R., Contreras J.A. Stereotactic MR-guided online adaptive radiation therapy (SMART) for ultracentral thorax malignancies: Results of a phase 1 trial. Adv Radiat Oncol. 2019;4:201–209. doi: 10.1016/j.adro.2018.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van Sornsen de Koste J.R., Palacios M.A., Bruynzeel A.M.E. MR-guided gated stereotactic radiation therapy delivery for lung, adrenal, and pancreatic tumors: A geometric analysis. Int J Radiat Oncol Biol Phys. 2018;102:858–866. doi: 10.1016/j.ijrobp.2018.05.048. [DOI] [PubMed] [Google Scholar]
- 19.Benedict S.H., Yenice K.M., Followill D. Stereotactic body radiation therapy: The report of AAPM Task Group 101. Med Phys. 2010;37:4078–4101. doi: 10.1118/1.3438081. [DOI] [PubMed] [Google Scholar]
- 20.Folkert M.R., Timmerman R.D. Stereotactic ablative body radiosurgery (SABR) or stereotactic body radiation therapy (SBRT) Adv Drug Deliv Rev. 2017;109:3–14. doi: 10.1016/j.addr.2016.11.005. [DOI] [PubMed] [Google Scholar]
- 21.Bohoudi O., Bruynzeel A.M.E., Senan S. Fast and robust online adaptive planning in stereotactic MR-guided adaptive radiation therapy (SMART) for pancreatic cancer. Radiother Oncol. 2017;125:439–444. doi: 10.1016/j.radonc.2017.07.028. [DOI] [PubMed] [Google Scholar]
- 22.Timmerman R.D. An overview of hypofractionation and introduction to this issue of seminars in radiation oncology. Semin Radiat Oncol. 2008;18:215–222. doi: 10.1016/j.semradonc.2008.04.001. [DOI] [PubMed] [Google Scholar]
- 23.Finazzi T., Haasbeek C.J.A., Spoelstra F.O.B. Clinical outcomes of stereotactic MR-guided adaptive radiation therapy for high-risk lung tumors. Int J Radiat Oncol. 2020;107:270–278. doi: 10.1016/j.ijrobp.2020.02.025. [DOI] [PubMed] [Google Scholar]
- 24.Finazzi T., van Sörnsen de Koste J.R., Palacios M.A. Delivery of magnetic resonance-guided single-fraction stereotactic lung radiotherapy. Phys Imaging Radiat Oncol. 2020;14:17–23. doi: 10.1016/j.phro.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Winkel D., Bol G.H., Werensteijn-Honingh A.M. Target coverage and dose criteria-based evaluation of the first clinical 1.5T MR-linac SBRT treatments of lymph node oligometastases compared with conventional CBCT-linac treatment. Radiother Oncol. 2020;146:118–125. doi: 10.1016/j.radonc.2020.02.011. [DOI] [PubMed] [Google Scholar]
- 26.Videtic G.M., Paulus R., Singh A.K. Long-term follow-up on NRG Oncology RTOG 0915 (NCCTG N0927): A randomized phase 2 study comparing 2 stereotactic body radiation therapy schedules for medically inoperable patients with stage I peripheral non-small cell lung cancer. Int J Radiat Oncol. 2019;103:1077–1084. doi: 10.1016/j.ijrobp.2018.11.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Siva S., Kron T., Bressel M. A randomised phase II trial of stereotactic ablative fractionated radiotherapy versus radiosurgery for oligometastatic neoplasia to the lung (TROG 13.01 SAFRON II) BMC Cancer. 2016;16:183. doi: 10.1186/s12885-016-2227-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Osti M.F., Carnevale A., Valeriani M. Clinical outcomes of single dose stereotactic radiotherapy for lung metastases. Clin Lung Cancer. 2013;14:699–703. doi: 10.1016/j.cllc.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 29.Pollom E.L., Alagappan M., von Eyben R. Single- versus multifraction stereotactic body radiation therapy for pancreatic adenocarcinoma: Outcomes and toxicity. Int J Radiat Oncol. 2014;90:918–925. doi: 10.1016/j.ijrobp.2014.06.066. [DOI] [PubMed] [Google Scholar]
- 30.Goodman K.A., Wiegner E.A., Maturen K.E. Dose-escalation study of single-fraction stereotactic body radiotherapy for liver malignancies. Int J Radiat Oncol. 2010;78:486–493. doi: 10.1016/j.ijrobp.2009.08.020. [DOI] [PubMed] [Google Scholar]
- 31.Folkert M.R., Meyer J.J., Aguilera T.A. Long-term results of a phase I dose-escalation trial and subsequent institutional experience of single-fraction stereotactic radiation therapy for liver metastases. Int J Radiat Oncol Biol Phys. 2020;108:S149. doi: 10.1016/j.ijrobp.2020.12.012. [DOI] [PubMed] [Google Scholar]
- 32.Parikh N.R., Lee P.P., Raman S.S. Time-driven activity-based costing comparison of CT-guided versus MR-guided SBRT. JCO Oncol Pract. 2020;16:e1378–e1385. doi: 10.1200/JOP.19.00605. [DOI] [PubMed] [Google Scholar]
- 33.Tchelebi L.T., Haustermans K., Scorsetti M. Recommendations on the use of radiation therapy in managing patients with gastrointestinal malignancies in the era of COVID-19. Radiother Oncol. 2020;148:194–200. doi: 10.1016/j.radonc.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Braunstein L.Z., Gillespie E.F., Hong L. Breast radiation therapy under COVID-19 pandemic resource constraints: Approaches to defer or shorten treatment from a comprehensive cancer center in the United States. Adv Radiat Oncol. 2020;5:582–588. doi: 10.1016/j.adro.2020.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zaorsky N.G., Yu J.B., McBride S.M. Prostate cancer radiation therapy recommendations in response to COVID-19. Adv Radiat Oncol. 2020;5:659–665. doi: 10.1016/j.adro.2020.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Guckenberger M., Belka C., Bezjak A. Practice recommendations for lung cancer radiotherapy during the COVID-19 pandemic: An ESTRO-ASTRO consensus statement. Radiother Oncol. 2020;146:223–229. doi: 10.1016/j.radonc.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yahalom J., Dabaja B.S., Ricardi U. ILROG emergency guidelines for radiation therapy of hematological malignancies during the COVID-19 pandemic. Blood. 2020;135:1829–1832. doi: 10.1182/blood.2020006028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Guckenberger M., Lievens Y., Bouma A.B. Characterisation and classification of oligometastatic disease: A European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus recommendation. Lancet Oncol. 2020;21:e18–e28. doi: 10.1016/S1470-2045(19)30718-1. [DOI] [PubMed] [Google Scholar]
- 39.Jaffray D.A., Siewerdsen J.H., Wong J.W. Flat-panel cone-beam computed tomography for image guided radiation therapy. Int J Radiat Oncol. 2002;53:1337–1349. doi: 10.1016/s0360-3016(02)02884-5. [DOI] [PubMed] [Google Scholar]