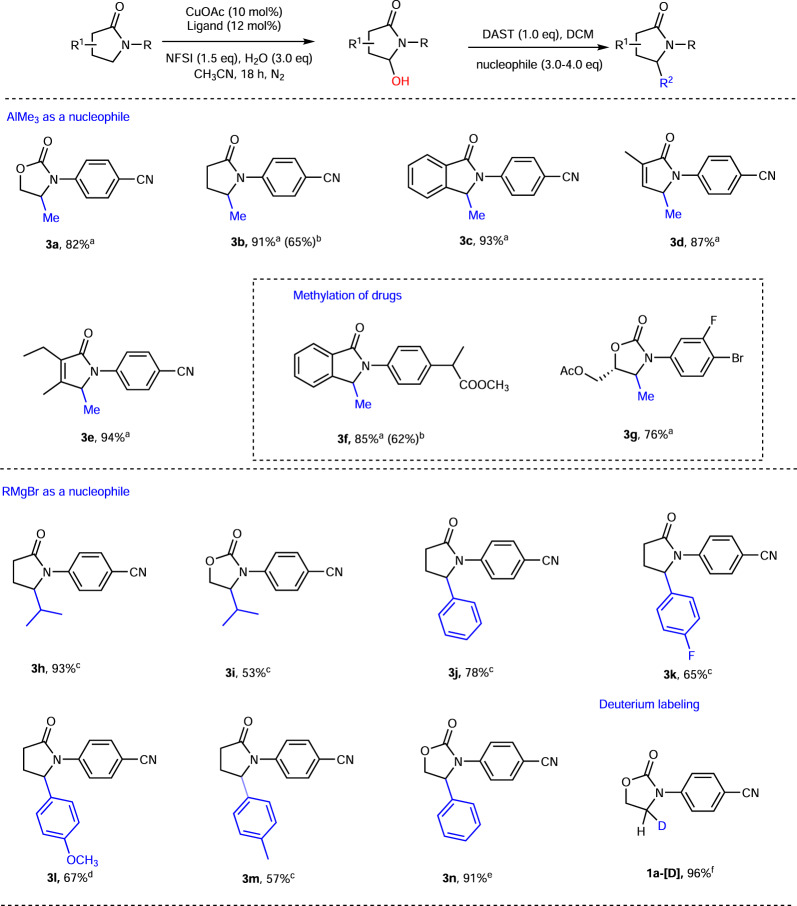
Fig. 3. Late-stage alkylations.
Reaction conditions: aHemiaminal 2 (0.1 mmol), CH2Cl2 (1.0 mL), DAST (0.1 mmol) added at −78 °C; rt for 1 h; cooled to −78 °C, AlMe3 (0.3 mmol) added, stirred 2 h; rt for 1 h, yield of the second step. b1l or 1y (0.2 mmol), H2O (0.3 mmol), CuOAc (0.02 mmol), L2 or L1 (0.022 mmol), NFSI (0.3 mmol), CH3CN (2.0 ml), under N2, followed by CH2Cl2 (1.0 mL), DAST (0.1 mmol) at −78 °C; rt for 1 h; cooled to −78 °C, AlMe3 (0.3 mmol) added, stirred 2 h; rt for 1 h, overall yield of two steps. cHemiaminal 2 (0.1 mmol), CH2Cl2 (1.0 mL), DAST (0.1 mmol) added at −78 °C; rt for 1 h; cooled to −78 °C, RMgBr (0.4 mmol) added, stirred for 5 h; rt for 1 h. d(A) bpy (0.4 mmol), THF (0.5 mL), RMgBr (0.4 mmol) added at −40 °C for 0.5 h; rt for 0.5 h; (B) 1l (0.1 mmol), CH2Cl2 (1.0 mL), DAST (0.1 mmol) added at −78 °C; rt for 1 h; (B) added to (A), stirred 5 h at −40 °C; rt for 1 h. e(A) CuBr·Me2S (0.4 mmol), THF (0.5 mL), RMgBr (0.4 mmol) added at −40 °C for 0.5 h; rt for 0.5 h; (B) 1a (0.1 mmol), CH2Cl2 (1.0 mL), DAST (0.1 mmol) added at −78 °C; rt for 1 h; (B) added to (A), stirred for 5 h at −40 °C; rt for 1 h. f1a (1.0 mmol), TFA (2.0 mL), NaBD4 (4.0 mmol) added at 0 °C; rt for 2 h.