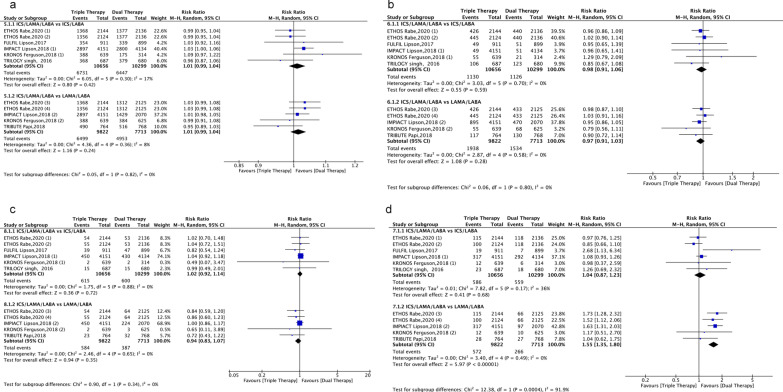
Fig. 5.
Forest plot for safety endpoints. Single-inhaler triple therapy vs. single-inhaler dual therapy (LABA/LAMA or ICS/LABA FDC) on the risk of adverse events (AEs) and serious adverse events (a, b) and the risk of cardiovascular events and pneumonia (c, d) in COPD patients. ICSs: Inhaled corticosteroids; LABA: Long-acting beta2-agonist; LAMA: Long-acting muscarinic antagonist; ETHOS Rabe, 2020 (1): BUD/GLY/FOR (320/18/9.6) vs. BUD/FOR (320/9.6); ETHOS Rabe, 2020 (2): BUD/GLY/FOR (160/18/9.6) vs. BUD/FOR (320/9.6); ETHOS Rabe, 2020 (3): BUD/GLY/FOR (320/18/9.6) vs. GLY/FOR (18/9.6); ETHOS Rabe, 2020 (4): BUD/GLY/FOR (160/18/9.6) vs. GLY/FOR (18/9.6); IMPACT Lipson, 2018 (1): FF/UMEC/VI (100/62.5/25) vs. FF/VI (100/25); IMPACT Lipson, 2018 (2): FF/UMEC/VI (100/62.5/25) vs. UMEC/VI (62.5/25); KRONOS Ferguson, 2018 (1): BUD/GLY/FOR (320/18/9.6) vs. BUD/FOR (320/9.6); KRONOS Ferguson, 2018 (2): BUD/GLY/FOR (320/18/9.6) vs. GLY/FOR (18/9.6). BUD/GLY/FOR: budesonide/glycopyrronium bromide/formoterol fumarate; FF/UMEC/VI: fluticasone furoate/umeclidinium/vilanterol; BUD/FOR: budesonide/formoterol fumarate; UMEC/VI: umeclidinium bromide/vilanterol; GLY/FOR: glycopyrronium bromide/formoterol fumarate