Fig. 3.
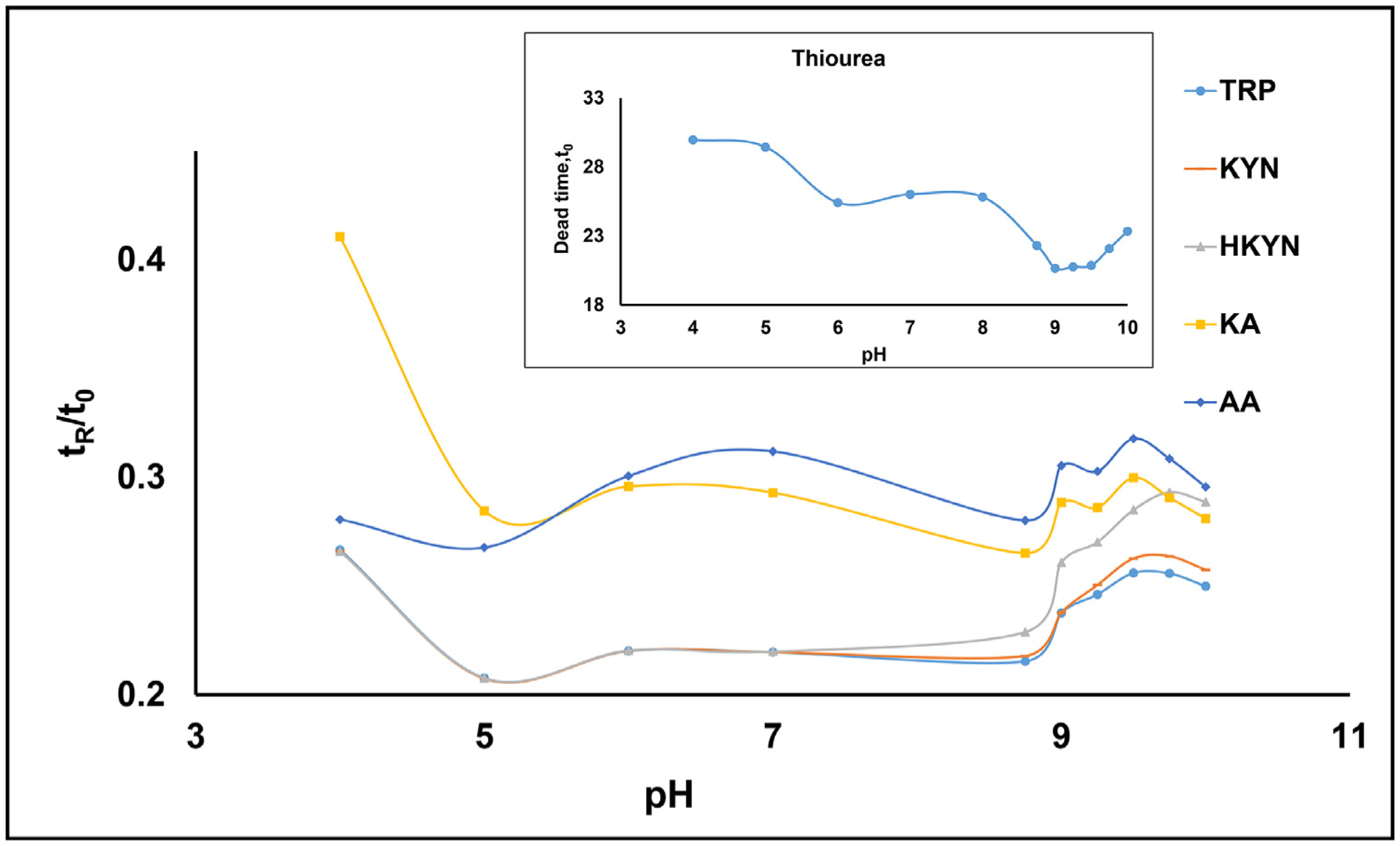
Effect of pH variation on the relative retention time (tr/t0) of five upstream KP metabolites. The AMPS covalent bonded column (65 cm × 50 um i.d.) was used to perform CE-ESI-MS/MS. Mobile phase: 15 mmol/L ammonium carbonate [(NH4)2CO3], prepared at pH 4.0–9.0 using 17.4 M acetic acid and above pH 9 using 14.5 M NH4OH. The applied voltage was +15 kV during separation generating a current range of 8.5–14.0 μA. Capillary temperature, 20 °C. Injection: 5 mbar for 100 s. A 100 mbar of internal pressure was applied to the AMPS column during CEC-MS runs. Spray chamber parameters: sheath liquid [80/20 (%v/v) MeOH/water, 40 mM HOAc], sheath liquid flow rate, 5 μL/min; capillary voltage, +3500 V; drying gas flow rate, 5.0 L/min; drying gas temperature, 200 °C; nebulizer pressure, 7 psi; collision cell gas (N2) pressure, 30 psi. The multiple reaction monitoring (MRM) in the positive ion mode was used (see Table 1 for MRM conditions). The inset plot shows the trend line for the dead time of thiourea over the pH range of 4.0–10.0. Curve identification for individual metabolites is listed in the legend.
