Highlights
-
•
High Choline-PET uptake is predictive of a poor outcome at 3-year follow-up.
-
•
Primary tumor should be boosted for high semiquantitative data Choline PET.
-
•
PET-guided dose to the dominant lesion can prolong the free-recurrence time.
Keywords: Prostate cancer, Radiotherapy, Dominant intra-prostatic lesion, 18F-choline, PET/CT, Prognosis
Abstract
Purpose
This study aims to establish whether metabolic parameters obtainable from FCH PET/CT can predict long-term response to radical radiotherapy (rRT) in patients with localized prostate cancer (PCa).
Methods
Drawing on a single-center database, we retrospectively reviewed the pre-treatment FCH PET/CT scans of 50 patients who underwent rRT between 2012 and 2017. Patients were enrolled if they had a follow-up of at least 3 years after rRT. Various metabolic parameters were considered for each PET/CT, including FCH multifocality. rRT was administered to all patients for a total equivalent dose of 76–80 Gy, using a standard or hypofractionated schedule. Patients were classified as disease-free (DF) if their PSA levels after rRT rose by <2 ng/mL vis-à-vis their PSA nadir, or as not disease free (NDF) if their PSA levels rose by more than 2 ng/ml.
Results
A multifocal FCH uptake in the prostate gland was identified in 27 patients (54%). At 3-year follow-up, 37 patients (74%) were judged DF, and 13 (26%) were NDF. The SUVmax and SUVmean, and the sum of the two values in all FCH foci in the prostate gland were significantly higher for NDF patients than for DF patients (all p < 0.005). The sum of the TLCKA levels in all FCH foci was likewise significantly higher in patients who were NDF than in those found DF (median 54.5 vs. 29.4; p < 0.05). At univariate analysis, the most of PET-metrics and Gleason Score were predictors of biochemical relapse after 3-year follow-up (all p < 0.05).
Conclusion
Higher SUVs seems predict a worse outcome for patients with multifocal intraprostatic lesions who are candidates for rRT.
Introduction
Prostate cancer (PCa) is the most prevalent malignancy among men in Europe. Primary treatments involve surgery or radiation therapy (RT) to the prostate gland. RT can be delivered with or without androgen deprivation therapy (ADT), depending on a patient’s risk class [1]. For now, PSA is the only biochemical marker capable of indicating the presence of recurrent disease during a patient’s follow-up. After RT, an increase in PSA levels two points above the nadir is used as a cutoff for identifying biochemical disease recurrences. Unlike the surgical approach, RT does not allow for a definitive histological assessment, so a tumor’s aggressiveness is judged on imaging findings alone, which may be misinterpreted. Methods such as magnetic resonance imaging (MRI) and positron emission tomography-computed tomography (PET/CT) with 18F-choline (FCH) play an important part in the assessment of widespread disease inside and outside the prostate gland [2]. Some studies on the accuracy of FCH PET/CT in revealing intraprostatic uptakes showed that choline can identify tumor foci inside the prostate gland [3] with a pooled sensitivity and specificity of 62.6% and 76.3%, respectively [4]. FCH PET/CT also has a prognostic role in patients with recurrent PCa, when combined with certain metabolic variables, such as the mean standardized uptake value (SUVmean), and other surrogate data [5].
A meta-analysis on the outcome of RT for PCa demonstrated that dose escalation (up to 78 Gy) improves biochemical response and progression-free survival in patients with intermediate- and high-risk PCa [6]. With this in mind, there is an increasing role for molecular imaging in the planning of RT for PCa. Pinkawa et al. demonstrated that a boost volume decided on the strength of specific metabolic variables obtainable from FCH PET/CT can be used for RT without adding to its toxicity [7].
The endpoint of the present study was to assess the prognostic value of metabolic parameters obtainable from FCH PET/CT in patients with localized PCa treated with radical-intent RT (rRT), with a view to establishing whether a potential boost (based on tumor volume ascertained from the pathological choline uptake) could improve the prognosis.
Material and methods
Patient population
Fifty patients collected in a single-center database from 2012 to 2017 were considered, identifying those meeting the following inclusion criteria: 1) biopsy-proven PCa (any D’Amico risk class); 2) FCH PET/CT prior to treatment; 2) rRT according to international guidelines; 4) a follow-up of at least 3 years after completing rRT; and 5) testosterone levels within normal range after stopping ADT (in the case of high-risk patients who were given 2-year ADT in association with rRT [8]).
Patients meeting the following criteria were excluded: 1) surgery as primary treatment; 2) metastatic disease detected on FCH PET/CT; 3) a total equivalent dose of rRT to the prostatic lesions of <76 Gy; 4) the presence or coexistence of other primary tumors; and 5) a follow-up of <2 years, or persistently suppressed testosterone levels after 2-year adjuvant ADT.
Age, Gleason score, and PSA levels at diagnosis (PSA preRT) were obtained for all patients, and follow-up data were retrieved on a medical chart by a radiation oncologist (M.S.). All patients gave their consent to the use of their PET/CT scans for research purposes in accordance with local ethical committee recommendations.
PET/CT acquisition and interpretation
Before patients underwent PET/CT examination with FCH, they fasted for at least 6 h with no restrictions on water intake. FCH (18F-fluoromethylcholine; IASON, Graz, Austria) with a scheduled activity of 3 MBq/kg body mass was administered intravenously. To prevent incomplete administration or subcutaneous accumulation, and to ensure good hydration, the radiopharmaceutical was injected through an infusion line connected to saline solution. Images were acquired with the Biograph 16 (Siemens Healthcare, Erlangen, Germany) PET/CT scanner, with low-dose CT acquired first. PET/CT whole-body acquisition (3 min/bed) was started 60 min after tracer injection, covering a skull-to-knees field of view. No dynamic PET was used. The PET data were reconstructed over a 128 × 128 matrix with a pixel size of 4.75 mm and a slice thickness of 2 mm. The processed images were displayed in coronal, transverse and sagittal planes.
From the whole-body PET/CT scans, choline uptake by the tumor was measured using the following five parameters:
-
1.
SUVmax, obtained by drawing a boundary around the whole lesion.
-
2.
SUVmean and metabolic tumor volume (MTV) were obtained by segmenting the tumor, adopting a threshold of 50%.
-
3.
total lesion choline kinase activity (TLCKA): a parameter equivalent to total lesion glycolysis (TLG) for Fluorodeoxyglucose (FDG), and calculated with the equation: TLCKA = SUVmean50% × MTV50%;
-
4.
tissue-to-background (T/B) ratio: T was calculated as the SUVmean in the isovolumetric volume of interest (VOI), and B was obtained from the SUVmean in 2 cm3 of the VOI. In cases of multiple FCH uptake foci, the VOI was positioned in the contralateral healthy prostate lobe or in the same lobe, on the basis of the uptake in the entire prostate gland.
-
5.
multifocality: the number of foci of FCH PET/CT uptake in the prostate gland.
-
6.
the sums of the SUVmax, SUVmean, MTV and TLCKA (called: SUM_SUVmax, SUM_SUVmean, SUM_MTV, and SUM_TLCKA, respectively) were calculated from the values for each metabolic parameter in all foci of FCH uptake inside the prostate gland.
Radiation therapy planning and delivery
All patients underwent simulation CT with the GE Optima CT580 RT. During the simulation, the isocenter was located in the middle of the prostate gland. The target volume and organ at risk were delineated on the CT scan. External beam radiation therapy (EBRT) with the conventional (3D-CRT) or modulated techniques [Intensity-modulated radiotherapy (IMRT) or Volumetric Modulated Arc Therapy (VMAT)] was planned with the Eclipse v11 (Varian Medical System, USA) and Oncentra v 4.2 (Elekta, UK). RapidArc VMAT plans were delivered with a Unique or TrueBeam Stx linear accelerator (Varian Medical System, USA), equipped with Millennium MLC, while 3D-CRT and Static IMRT plans were delivered with an Oncor (Siemens Healthcare, DE) equipped with 160 MLC. All patients were instructed to follow a bowel and bladder preparation protocol before each session of radiotherapy. Different fractionation schedules were used (2, 2.5 or 3 Gy per fraction) for a total equivalent dose (EQD2) of 76–80 Gy, considering an alpha/beta ratio of 1.5 [9].
Statistical analysis
A Shapiro-Wilk test was used to test the normal distribution of SUV max, SUVmean, MTV, TLCKA, T/B, Gleason score, PSA preRT, SUM_SUVmax, SUM_SUVmean, SUM_MTV, SUM_TLCKA.
Non-parametric data were analyzed using the Mann Whitney test, comparing patients with and without recurrent disease. How multifocality correlated with outcome was tested using Fisher's exact test. The specificity and sensitivity of the parameters considered were obtained using receiver operating curve (ROC) analysis. A Spearman’s rank correlation test was used for testing the relationships among clinical data, PET metrics and outcome. A univariate analysis was performed. All tests were performed with R, version 4.0.3 (2010–10-10) (The R Foundation for Statistical Computing) [10]. All p-values reported are two-sided, and a p < 0.05 was considered significant.
Results
Table 1 shows the characteristics of the 50 patients enrolled in the study. All patients were at intermediate to high risk of recurrence based on the D’Amico classification. Thirteen patients (26%) were on ADT at the time of their FCH PET/CT.
Table 1.
Characteristics of patients.
| Variables | |
|---|---|
| N of patients | 50 |
| Median age (range), years | 78 (68–89) |
| Median PSA at diagnosis (range), ng/mL | 14 (2.2–88.7) |
| Gleason score, n (%) | |
| • ≤ 6 | 3 (6%) |
| • =7 | 22 (44%) |
| • ≥ 8 | 25 (50%) |
| Grade Group, n (%) | |
| • 1 (GS 6) | = 3 (1%) |
| • 2 (GS 7: 3 + 4) | = 6 (12%) |
| • 3 (GS 7: 4 + 3) | = 6 (12%) |
| • 4 (GS 8) | = 19 (38%) |
| • 5 (GS 9–10) | = 6 (12%) |
| • GS 7 unspecified | = 10 (20%) |
| D’Amico risk classification, n (%) | |
| • Low | 0 |
| • Intermediate | 18 (36%) |
| • High | 32 (64%) |
| Multifocality at biopsy, n (%) | |
| • No | 23 (46%) |
| • Yes | 27 (54%) |
| ADT before FCH PET/CT, n (%) | |
| • No | 37 (74%) |
| • Yes | 13 (26%) |
| RT technique, n (%) | |
| • 3D-CRT | 22 (44%) |
| • IMRT/VMAT | 28 (56%) |
| RT dose (EQD2), n (%) | |
| • =76 Gy | 5 (10%) |
| • =77 Gy | 1 (2%) |
| • =78 Gy | 25 (50%) |
| • =80 Gy | 19 (38%) |
PSA: prostate specific antigen; ADT: androgen deprivation therapy; RT: radiation therapy; CRT: conformational RT; IMRT: intensity modulated RT; VMAT: volumetric modulated arc therapy; EQD2: equivalent dose
All patients only showed a significant FCH uptake in the prostate gland, with no evidence of disease outside the prostate. There were 2 foci of uptake in 10 patients (20%), 3 in 10 (20%), 4 in 4 (8%), 5 in 2 (4%), and 7 in 1 (2%), for a total of 27 patients (54%) with multifocal PCa.
A Spearman's rank correlation test showed that SUVmax, SUVmean, SUM_SUVmax, SUM_SUVmean were significantly correlated with Gleason Score (all p < 0.05), while only SUVmean and SUM_TLCKA were associated with the PSA level (both p < 0.05). However, we found also that each PET-metrics was strongly correlated with the others.
After a median follow-up of 47 months (range: 36–104 months), 13 patients (26%) had a biochemical disease recurrence while 37 (74%) were disease-free. Seven of the 13 patients (26%) whose PCa recurred subsequently died of their disease. Eleven of the 13 patients with recurrent disease had a high-risk PCa with a Gleason Score of 8 or more. Indeed, Gleason Score and Risk group based on D’Amico classification were correlated with outcome (p = 0.013 and p = 0.022, respectively).
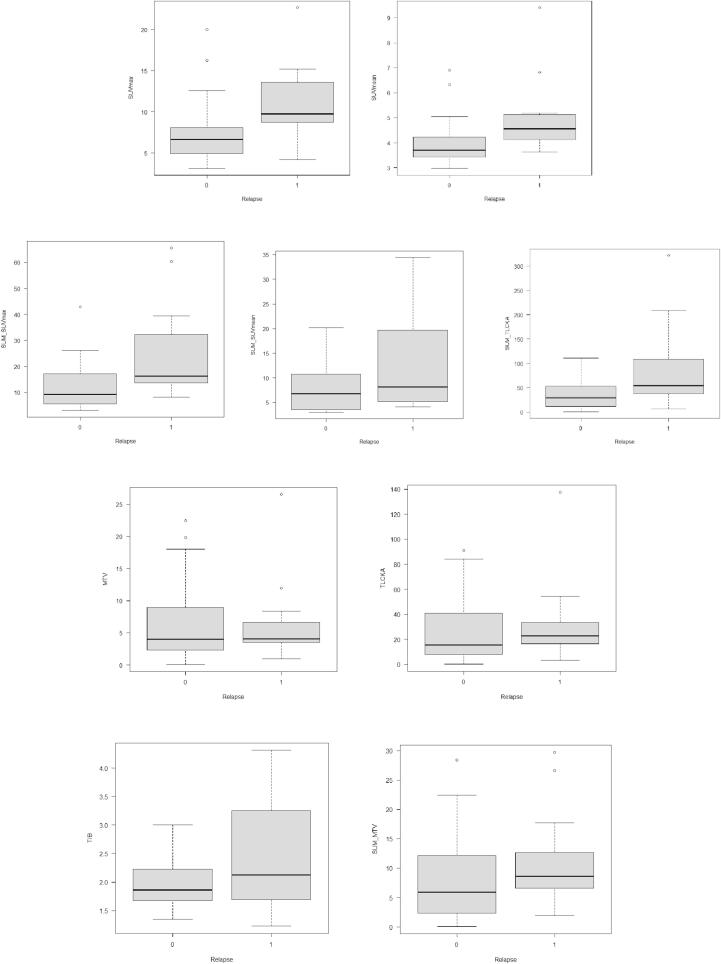
Table 2 shows the semiquantitative data obtained from the FCH PET/CT in all patients, with and without recurrent disease. Only the SUVs differed significantly between patients with and without biochemical evidence of recurrent PCa after rRT. The highest statistical significance was found for the SUVmax and SUVmean of the index lesion, and for the SUM_SUVmax in cases with multifocal disease. One in three patients with multifocal disease on FCH PET/CT had a recurrence (9 out of 27; 33%), although the difference with patients affected by non-multifocal disease was not statistically significant (p = 0.332). Similarly, patients with a single focus of FCH uptake who experienced a biochemical recurrence during the 3-year follow-up had high SUVs. The SUM_TLCKA was also significantly higher in patients with biochemical recurrences than in those remaining disease-free: this underscores the potential role of an increased choline kinase activity as a prognostic indicator of recurrent disease. Fig. 1 shows the box plots for all the metabolic variables.
Table 2.
Distribution of metabolic data based on the recurrence of disease in all 50 patients.
| Variables |
All patients (n = 50 patients) |
Relapse (n = 13 patients) |
Not relapse (n = 27 patients) |
p-value |
|---|---|---|---|---|
| SUVmax | 7.5 (3.1–22.7) |
9.8 (4.2–22.7) |
6.7 (3.1–20.0) |
0.00054* |
| SUVmean | 4.0 (3.0–9.4) |
4.6 (3.6–9.4) |
3.7 (3.0–6.9) |
0.00069* |
| MTV | 4.0 (0.1–26.6) |
4.1 (0.9–26.6) |
4.0 (0.1–22.5) |
0.5430 |
| TLCKA | 16.5 (0.2–137.6) |
22.7 (3.4–137.6) |
15.7 (0.2–90.9) |
0.1840 |
| T/B | 1.9 (1.2–4.3) |
2.1 (1.2–4.3) |
1.9 (1.4–3.0) |
0.3200 |
| SUM_SUVmax | 12.7 (3.1–65.7) |
16.3 (8.2–65.7) |
9.3 (3.1–42.9) |
0.0063* |
| SUM_SUVmean | 7.2 (3.0–34.5) |
8.1 (4.1–34.5) |
6.8 (3.0–20.2) |
0.0208* |
| SUM_MTV | 6.7 (0.1–29.7) |
8.6 (1.9–29.7) |
5.9 (0.1–28.4) |
0.1630 |
| SUM_TLCKA | 39.7 (0.2–322.2) |
54.5 (6.8–322.2) |
29.4 (0.2–111.1) |
0.0255* |
Shown here are medians (range) of each considered parameter; the reported p-values are obtained with Mann-Whitney test between patients who did and did not relapse. All marked values are statistically significant. MTV: metabolic tumor volume; TLCKA: total lesion choline kinase activity; T/B: tumor/background;
significant value.
Fig. 1.
Blox plots for all available parameters.
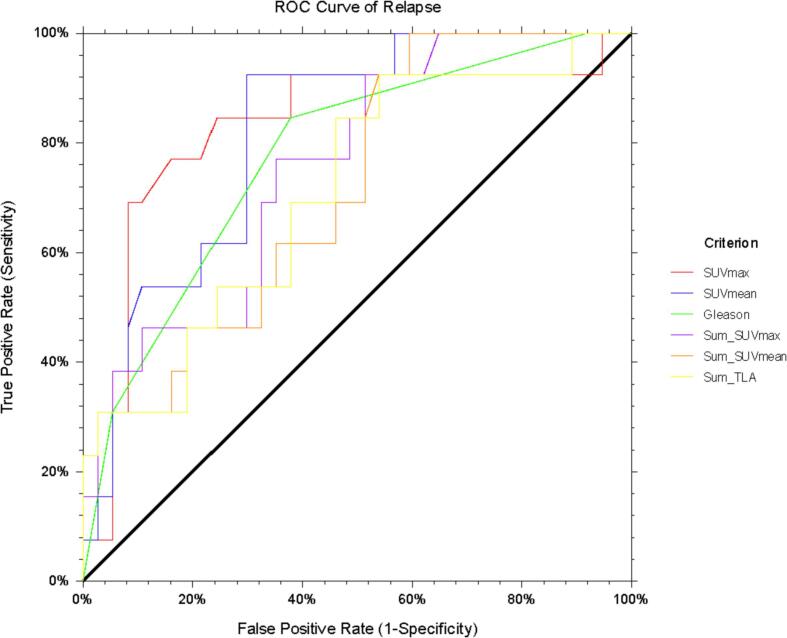
MTV, TLCKA, T/B, PSA preRT and SUM_MTV did not differ significantly between patients with and without a biochemical recurrence. T/B was significantly higher in patients who were on ADT at the time of their PET/CT than in those who were not (median value: 2.54 vs 1.94; p-value < 0.05 for the Mann Whitney test). In particular, background levels of healthy prostate gland were lower for patients on ADT (median value: 1.83 vs 2.14; p-value < 0.05). ROC curves plotted to identify patients who would develop a biochemical recurrence of PCa showed cutoffs of 8.74 for SUVmax (sensitivity 83%, specificity 77%); 4.14 for SUVmean (sensitivity 83%, specificity 77%); 11.94 for MTV (sensitivity 83%, specificity 60%); and 49.95 for TLCKA (sensitivity 83%, specificity 67%) (Fig. 2).
Fig. 2.
ROC curves for the identification of cut-off values.
At univariate analysis, SUVmax, SUVmean, SUM_SUVmax, SUM_SUVmean, SUM_TLCKA and Gleason Score were predictors of biochemical relapse after 3-year follow-up (all p < 0.05) (Table 3).
Table 3.
Univariate analysis.
| Univariate | |
|---|---|
| p-value | |
| SUVmax | 0.011 |
| SUVmean | 0.018 |
| MTV | NS |
| TLCKA | NS |
| SUM_SUVmax | 0.016 |
| SUM_SUVmean | 0.020 |
| SUM_MTV | NS |
| SUM_TLCKA | 0.026 |
| Gleason Score | 0.004 |
| PSA | NS |
| Multifocality | NS |
| N° of focalities | NS |
NS = non-significant.
Discussion
No data are currently available on the utility of radiolabeled choline PET in predicting the outcome of patients treated with rRT. Most studies have focused on the prognostic value of PET/CT with radiolabeled choline in patients with recurrent PCa [11], [12], [13], demonstrating that a positive choline PET/CT result can predict PCa-specific survival if performed during a biochemical recurrence in either hormone-naïve patients or those treated with ADT. To our knowledge, ours is the first study to correlate the use of rRT with semiquantitative variables obtainable from FCH PET/CT, after a follow-up of at least 3 years after completing rRT. Our data suggest that SUVmax, SUVmean, SUM_SUVmax, and SUM_SUVmean are correlated with the recurrence of disease. In particular, SUVmax and SUVmean are surrogates of a cancer’s aggressiveness, such as the Gleason score. In short, high SUVs correlate with a poor prognosis. SUM_TLCKA (the sum of the total lesion of choline kinase activity for all lesions) is significantly associated with biochemical PCa recurrence, underscoring the importance of a tumor’s invasiveness (MTV) and aggressiveness (SUVs). SUVmax, SUVmean and SUM_TLCKA represents PET-metrics that are correlated with the differentiation of the prostate lesions: a tumor able to produce PSA has a high activation of the choline metabolism (it means of Choline kinase activity).
As already stated in literature, radiolabeled choline PET/CT has some limitations for the purposes of identifying primary tumor, reportedly achieving a pooled sensitivity and specificity of 62.6% and 76.3%, respectively [4]. The amount of radiotracer uptake by malignant tissue overlaps significantly with that of high-grade prostatic intraepithelial neoplasia, prostatitis, benign prostatic hypertrophy, and normal tissue [14]. Focal uptake should nonetheless be viewed with a strong degree of suspicion [2]. Monofocal uptake is commonly reported in PCa patients undergoing FCH PET/CT before receiving any treatment [15]. One study reported that multifocal uptake was often associated with chronic prostatitis (in 5 of 20 patients) [16]. That said, it is impossible to rule out malignancy in patients with an inhomogeneous or multifocal pattern of choline uptake because PCa is often multifocal and the average patient has at least two distinct prostatic foci, which may have different Gleason scores [17]. In the present study, 27 out of 50 patients had a multifocal choline uptake on FCH PET/CT, and all of them had biopsy-proven multifocal lesions. We also found multifocal uptake of FCH in the prostate gland (in terms of multifocality per se and SUM_SUVmax) associated with a worse prognosis, and evidence of a significant correlation between more than one choline uptake site and the risk of recurrence after rRT.
Different semiquantitative variables were used in our study, such as SUVmax, SUVmean, T/B, MTV and TLCKA. SUVmax and SUVmean were significantly higher in patients with a poor outcome as compared to the counterpart, while MTV, TLCKA of the index lesion and T/B did not. We considered T/B as a continuous variable but as shown in Table 2, the ratio was similar for patients with and without biochemical evidence of recurrent disease (only slightly higher in the former). Neoadjuvant hormone treatment prior to any RT can reduce choline uptake in PCa [18], reducing PCa metabolism and the absolute SUVs in both primary tumor and benign prostate tissue as a result [19], [20]. In our series, 13 of the 50 patients were on ADT at the time of their PET/CT time, before undergoing rRT. In our opinion, T/B is not the best choline-PET parameter for the purposes of RT planning, particularly in the case of concurrent ADT. Other studies have tried to examine the effect of ADT on choline tracer uptake. Comparing PET/CT scans of patients with and without ADT, Chan et al. [21] found that ADT was associated with a significantly (17%) smaller overall prostate volume. Prostate SUVmax was also lower in patients on ADT than in those taking no hormone therapy, although the difference did not reach statistical significance. Similarly, visually defined isodose lines (IDLs) were significantly smaller than the corresponding 60% maximum uptake volume in patients on ADT, meaning that the treatment reduces choline tracer uptake by malignant lesions on PET imaging. These data suggest that, as PET would provide the values for IDL segmentation, it would be done before starting ADT. In a recent study, 10 patients with histologically confirmed PCa underwent three sequential choline-PET/CT scans. The first was performed at the baseline, the second after neoadjuvant ADT, and the third 4 months after RT and concomitant ADT. The combined effect of the ADT and RT-ADT was to significantly reduce tumor choline uptake, and the extent of this effect could predict long-term biochemical control or relapse. Choline PET/CT can therefore be useful for assessing rates of response to RT dose escalation and new hormone therapies for PCa [22].
In patients who underwent salvage prostatectomy and had a biochemical recurrence after RT, clinically significant recurrences always occurred at the site of primary tumor [23]. Cellini et al. [24] also found a 100% rate of local control outside the primary tumor in patients with biochemical recurrences. Many trials have shown that a dose escalation approach improves biochemical recurrence-free survival [25], [26], but the question of how to define the IDL remains to be answered. Recent studies have attempted to correlate different segmentation methods with pathology. Park et al. [27] tried to validate various segmentation methods by accurately recording tumor volume in vivo, as outlined on the image of choline uptake, with the true histological extent of the tumor. A segmentation threshold of 60% of SUVmax emerged as the best parameter in correlation with pathology. In their interesting paper, Chang et al. [3] compared several IDL contouring methods. The one that showed the strongest correlation between PET and pathology was SUV60%, although this correlation was not statistically more significant than the other options tested. Dose escalation to facilitate cell death in neoplastic clones resistant to lower doses is the most rational radiobiological principle for combating PCa cell growth, but the total dose that can be delivered to the tumor is limited by the tolerance of organs at risk (OAR). The goal of dose escalation can therefore only be achieved by reducing the exposure of OAR in order to minimize treatment-related toxicity. The best way to achieve a dose escalation is to use conformational delivery techniques such as 3D-CRT, and particularly IMRT and VMAT, combined with image-guided radiotherapy (IGRT) techniques [8]. Many retrospective series have explored the feasibility of boosting the primary tumor with a lower RT dose, reporting no differences in acute toxicities [28], [29]. A recent prospective randomized controlled trial, the FLAME study, explored the value of adding a boost dose based on MRI findings to whole prostate irradiation. Without exacerbating acute and late toxicities [30], the boost doses had a beneficial effect on biochemical disease-free survival, reducing the risk of biochemically-evidence treatment failure from 15% to 8% at 5 years [31]. Many ongoing trials are also assessing the benefit of a boost dose to the tumor during prostate gland RT, using either a moderately hypofractionated regimen or ultra-hypofractionated RT.
The feasibility of identifying patients at high risk of local recurrence, and consequently higher risk of disease progression, is crucial to the selection of those who could benefit from a boost dose to the primary tumor. Our results did not indicate any significant correlation between MTV (defined using a 50% threshold of the maximum uptake) and outcome, a finding in disagreement with other reports [3], [32], [33]. The novelty of our study is that our results allow us to be more confident in using information obtained from radiolabeled choline PET/CT for the purposes of IDL segmentation with a view to boosting the radiation dose to the primary tumor. However, these are only exploratory results, without a clear evidence that FCH PET/CT can be used as a biomarker for predict or overcome the negative prognostic features.
Various other radiopharmaceutical agents have been used in patients with PCa, as well as choline-based PET tracers [34]. Prostate-specific antigen membrane (PSMA) has recently been adopted in clinical practice, both to study recurrent PCa and in the detection of primary disease [35], [36]. Some papers are now available on the role of radiolabeled PSMA PET/CT in guiding the planning of RT for patients with primary PCa. Thomas et al. [36] studied 21 patients who underwent 68 Ga-PSMA PET/CT for the purpose of defining prostate target volumes. They found that the mean dose to the tumor tissue – defined as the pathological volume established from the 68 Ga-PSMA positive volumes – can be increased significantly without any significant increase in the mean dose to rectum and bladder. In 6 patients who underwent 18F-DCFPyL PET/CT, Goodman et al. [37] likewise found that PSMA PET can be used as a novel approach to defining the gross tumor volumes for the purposes of dominant intraprostatic lesion simultaneous in-field boost (DIL-SIB) dose escalation. No data on the effect of PSMA PET on patient prognosis in this setting have become available as yet, however.
The present paper has some limitations. The small sample size limits significantly the translation of our results in clinical practice and thus limiting the power of statistical analysis. Here in, it was assumed that a boosting dose in the primary tumor, guided by 18F-Choline PET/CT before rRT would be useful for reducing the rate of biochemical recurrence in 3-year follow-up. Moreover, due to a limited number of events, the multivariate analysis was not performed, thus further limiting the final conclusions. Therefore, the findings of the present manuscript would be considered as preliminary results that required a confirmation by a large prospective and well-designed trial.
Conclusions
A high choline uptake on PET/CT is predictive of a poor outcome at 3-year follow-up for patients with multifocal intraprostatic lesions who undergo rRT. The present study findings seem to suggest that the dose to the primary tumor would be boosted in patients with intraprostatic high SUVs, bearing in mind the radiobiology of PCa (which benefits from hypofractionation and higher doses). Further work needs to be done to assess a risk-adapted prescription protocol for patients with multifocal lesions and high SUVs, who could benefit from an increased dose to the dominant intraprostatic lesion, preferably using the latest radiopharmaceutical agents, such as radiolabeled PSMA.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.D'Amico A.V. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280(11):969–974. doi: 10.1001/jama.280.11.969. [DOI] [PubMed] [Google Scholar]
- 2.Piert M. Detection of aggressive primary prostate cancer with 11C-choline PET/CT using multimodality fusion techniques. J Nucl Med. 2009:1585–1593. doi: 10.2967/jnumed.109.063396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chang J.H., Joon D.L., Lee S.T., Gong S.J., Scott A.M., Davis I.D. Histopathological correlation of 11C-choline PET scans for target volume definition in radical prostate radiotherapy. Radiother Oncol. 2011;99(2):187–192. doi: 10.1016/j.radonc.2011.03.012. [DOI] [PubMed] [Google Scholar]
- 4.Evangelista L., Cervino A.R., Burei M., Gregianin M., Saladini G., Marzola M.C. «Comparative studies of radiolabeled choline positron emission tomography, histology of primary tumor and other imaging modalities in prostate cancer: a systematic review and meta-analysis. Clin Transl Imaging. 2013;1(2):99–109. [Google Scholar]
- 5.Colombiè M. Prognostic value of metabolic parameters and clinical impact of 18F-fluorocholine PET/CT in biochemical recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2015:1784–1793. doi: 10.1007/s00259-015-3123-5. [DOI] [PubMed] [Google Scholar]
- 6.Viani G. Higher-than-conventional radiation doses in localized prostate cancer treatment: a meta-analysis of randomized, controlled trials. Int J Radiat Oncol, Biol Phys. 2009:1405–1418. doi: 10.1016/j.ijrobp.2008.10.091. [DOI] [PubMed] [Google Scholar]
- 7.Pinkawa M. Dose-escalation using intensity-modulated radiotherapy for prostate cancer - Evaluation of the dose distribution with and without 18F-choline PET-CT detected simultaneous integrated boost. Radiother Oncol. 2009:213–219. doi: 10.1016/j.radonc.2009.07.014. [DOI] [PubMed] [Google Scholar]
- 8.NCCN . 2017. Clinical Practice Guidelines in Oncology - Prostate Cancer. [DOI] [PubMed] [Google Scholar]
- 9.Miralbell R., Roberts S.A., Zubizarreta E., Hendry J.H. Dose-Fractionation Sensitivity of Prostate Cancer Deduced From Radiotherapy Outcomes of 5,969 Patients in Seven International Institutional Datasets: α/β = 1.4 (0.9–2.2) Gy. Int J Radiat, Biol Phys. 2012;82(1):e17–e24. doi: 10.1016/j.ijrobp.2010.10.075. [DOI] [PubMed] [Google Scholar]
- 10.R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL <https://www.R-project.org/>; 2020.
- 11.Giovacchini G., Picchio M., Garcia-Parra R., Briganti A., Abdollah F., Gianolli L. 11C-choline PET/CT predicts prostate cancer-specific survival in patients with biochemical failure during androgen deprivation therapy. J Nucl Med. 2014;55(2):233–241. doi: 10.2967/jnumed.113.123380. [DOI] [PubMed] [Google Scholar]
- 12.Giovacchini G., Incerti E., Mapelli P., Kirienko M., Briganti A., Gandaglia G. [11C]choline PET/CT predicts survival in hormone-naive prostate cancer patients with biochemical failure after radical prostatectomy. Eur J Nucl Med Mol Imaging. 2015;42(6):877–884. doi: 10.1007/s00259-015-3015-8. [DOI] [PubMed] [Google Scholar]
- 13.Kwee S.A., Lim J., Watanabe A., Kromer-Baker K., Coel M.N. Prognosis related to metastatic burden measured by 18F-fluorocholine PET/CT in castration-resistant prostate cancer. J Nucl Med. 2014;55(6):905–910. doi: 10.2967/jnumed.113.135194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reske S. Imaging prostate cancer with 11C-choline PET/CT. J Nucl Med. 2006:1249–1254. [PubMed] [Google Scholar]
- 15.Jiménez Londoňo G. Revista española de medicina nuclear e imagen molecular. 2017. Role of 18F-Choline PET/CT in guiding biopsy in patients with raised PSA levels and previous negative biopsy for prostate cancer; pp. 241–246. [DOI] [PubMed] [Google Scholar]
- 16.Igerc I., Kohlfürst S., Gallowitsch H.J., Matschnig S., Kresnik E., Gomez-Segovia I. «The value of 18F-Choline PET/CT in patients with elevated PSA-level and negative prostate needle biopsy for localisation of prostate cancer. Eur J Nucl Mol Imaging. 2008;35(5):976–983. doi: 10.1007/s00259-007-0686-9. [DOI] [PubMed] [Google Scholar]
- 17.Crawford E. Prostate cancer pathology, screening, and epidemiology. Rev Urol. 2001:2–10. [PMC free article] [PubMed] [Google Scholar]
- 18.Giovacchini G. (11)C-choline uptake with PET/CT for the initial diagnosis of prostate cancer: relation to PSA levels, tumour stage and anti-androgenic therapy. Eur J Nucl Med Mol Imaging. 2008:1065–1073. doi: 10.1007/s00259-008-0716-2. [DOI] [PubMed] [Google Scholar]
- 19.Sutinen E. Kinetics of (11)C-choline uptake in prostate cancer: a PET study. Eur J Nucl Med Mol Imaging. 2004:317–324. doi: 10.1007/s00259-003-1377-9. [DOI] [PubMed] [Google Scholar]
- 20.Evangelista L. The effects of androgen deprivation therapy on the 18F-choline uptake in prostate cancer patients undergoing neoadjuvant treatment. Q J Nucl Med Mol Imaging. 2016 doi: 10.23736/S1824-4785.16.02877-6. [DOI] [PubMed] [Google Scholar]
- 21.Chan J., Carver A., Brunt J.N.H., Vinjamuri S., Syndikus I. Effect of androgen deprivation therapy on intraprostatic tumour volume identified on 18F choline PET/CT for prostate dose painting radiotherapy. Br J Radiol. 2017;90(1071):16–18. doi: 10.1259/bjr.20160818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Challapalli Exploring the potential of [11C]choline-PET/CT as a novel imaging biomarker for predicting early treatment response in prostate cancer. Nucl Med Commun. 2014:20–29. doi: 10.1097/MNM.0000000000000014. [DOI] [PubMed] [Google Scholar]
- 23.Pucar D., Hricak H., Shukla-Dave A., Kuroiwa K., Drobnjak M., Eastham J. Clinically significant prostate cancer local recurrence after radiation therapy occurs at the site of primary tumor: magnetic resonance imaging and step-section pathology evidence. Int J Radiat Oncol, Biol Phys. 2007;69(1):62–69. doi: 10.1016/j.ijrobp.2007.03.065. [DOI] [PubMed] [Google Scholar]
- 24.Cellini N., Morganti A.G., Mattiucci G.C., Valentini V., Leone M., Luzi S. Analysis of intraprostatic failures in patients treated with hormonal therapy and radiotherapy: implications for conformal therapy planning. Int J Radiat Oncol, Biol Phys. 2002;53(3):595–599. doi: 10.1016/s0360-3016(02)02795-5. [DOI] [PubMed] [Google Scholar]
- 25.Zelefsky M., Leibel S., Gaudin P., Kutcher G., Fleshner N., Venkatramen E.S. Dose escalation with three-dimensional conformal radiation therapy affects the outcome in prostate cancer. Int J Radiat Oncol, Biol Phys. 1998;41(3):491–500. doi: 10.1016/s0360-3016(98)00091-1. [DOI] [PubMed] [Google Scholar]
- 26.Pollack A., Smith L.G., von Eschenbach A.C. External beam radiotherapy dose response characteristics of 1127 men with prostate cancer treated in the PSA era. Int J Radiat Oncol, Biol Phys. 2000;48(2):507–512. doi: 10.1016/s0360-3016(00)00620-9. [DOI] [PubMed] [Google Scholar]
- 27.Ippolito E., Mantini G., Morganti A.G., Mazzeo E., Padula G.D., Digesu C. Intensity-modulated radiotherapy with simultaneous integrated boost to dominant intraprostatic lesion: preliminary report on toxicity. Am J Clin Oncol. 2012;35:158–162. doi: 10.1097/COC.0b013e318209cd8f. [DOI] [PubMed] [Google Scholar]
- 28.Sundahl N., De Meerleer G., Villeirs G., Ost P., De Neve W., Lumen N. Combining high dose external beam radiotherapy with a simultaneous integrated boost to the dominant intraprostatic lesion: analysis of genito-urinary and rectal toxicity. Radiother Oncol. 2016;119(3):398–404. doi: 10.1016/j.radonc.2016.04.031. [DOI] [PubMed] [Google Scholar]
- 29.Monninkhof Evelyn M., van Loon Juliette W.L., van Vulpen Marco, Kerkmeijer Linda G.W., Pos Floris J., Haustermans Karin, van den Bergh Laura, Isebaert Sofie, McColl Gill M., Smeenk Robert Jan, Noteboom Juus, Walraven Iris, Peeters Petra H.M., van der Heide Uulke A. Standard whole prostate gland radiotherapy with and without lesion boost in prostate cancer: Toxicity in the FLAME randomized controlled trial. Radiother Oncol. 2018;127:74–80. doi: 10.1016/j.radonc.2017.12.022. [DOI] [PubMed] [Google Scholar]
- 30.Kerkmeijer L.G.W., Groen V.H., Pos F.J., Haustermans K., Monninkhof E.M., Smeenk R.J., Kunze-Busch M., de Boer J.C.J., van der Voort van Zijp J., van Vulpen M., Draulans C., van den Bergh L., Isebaert S., van der Heide U.A. Focal boost to the intraprostatic tumor in external beam radiotherapy for patients with localized prostate cancer: results from the FLAME randomized phase III trial. J Clin Oncol. 2021 doi: 10.1200/JCO.20.02873. JCO2002873, Epub ahead of print. PMID: 33471548. [DOI] [PubMed] [Google Scholar]
- 31.Park H., Meyer C.R., Wood D., Khan A., Shah R., Hussain H. Validation of automatic target volume definition as demonstrated for 11C-Choline PET/CT of human prostate cancer using multi-modality fusion techniques. Acad Radiol. 2010;17(5):614–623. doi: 10.1016/j.acra.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chang J.H., Lim Joon D., Lee S.T., Gong S.J., Anderson N.J., Scott A.M. Intensity modulated radiation therapy dose painting for localized prostate cancer using 11C-choline positron emission tomography scans. Int J Radiat Oncol, Biol Phys. 2012;83(5):e691–e696. doi: 10.1016/j.ijrobp.2012.01.087. [DOI] [PubMed] [Google Scholar]
- 33.Wibmer A.G., Burger I.A., Sala E., Hricak H., Weber W.A., Vargas H.A. Molecular imaging of prostate cancer. Radiographics. 2016;36(1):142–159. doi: 10.1148/rg.2016150059. Epub 2015 Nov 20. PMID: 26587888; PMCID: PMC5410962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hofman M.S., Lawrentschuk N., Francis R.J., Tang C., Vela I., Thomas P. proPSMA Study Group Collaborators. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395(10231):1208–1216. doi: 10.1016/S0140-6736(20)30314-7. Epub 2020 Mar 22. PMID: 32209449. [DOI] [PubMed] [Google Scholar]
- 35.Fendler W.P., Weber M., Iravani A., Hofman M.S., Calais J., Czernin J. Prostate-specific membrane antigen ligand positron emission tomography in men with nonmetastatic castration-resistant prostate cancer. Clin Cancer Res. 2019;25(24):7448–7454. doi: 10.1158/1078-0432.CCR-19-1050. Epub 2019 Sep 11. PMID: 31511295. [DOI] [PubMed] [Google Scholar]
- 36.Thomas L., Kantz S., Hung A., Monaco D., Gaertner F.C., Essler M. 68Ga-PSMA-PET/CT imaging of localized primary prostate cancer patients for intensity modulated radiation therapy treatment planning with integrated boost. Eur J Nucl Med Mol Imaging. 2018;45(7):1170–1178. doi: 10.1007/s00259-018-3954-y. Epub 2018 Feb 21. PMID: 29468311. [DOI] [PubMed] [Google Scholar]
- 37.Goodman C.D., Fakir H., Pautler S., Chin J., Bauman G.S. Dosimetric evaluation of PSMA PET-delineated dominant intraprostatic lesion simultaneous infield boosts. Adv Radiat Oncol. 2019;5(2):212–220. doi: 10.1016/j.adro.2019.09.004. PMID: 32280821; PMCID: PMC7136625. [DOI] [PMC free article] [PubMed] [Google Scholar]