Abstract
Sugarcane bagasse is a fibrous material containing cellulose as its main component. It is produced in large quantities across the world. It is a kind of waste material that comes from the sugar industry. It is most commonly used in paper industries, but researchers have suggested that different mechanical and chemical treatments can help to extract cellulosic fibers, pure cellulose, cellulose nanofibers, and cellulose nanocrystals. These extracted materials have diverse applications in regenerated cellulosic fiber and composite material production. This paper will discuss the extraction procedures and typical applications in composite industries of these extracted materials. And an assessment will also be done on the production process and the properties of the end products to find out some common factors which can control the properties of these extracted material reinforced composites to some extent.
Keywords: Cellulose, Linear density, Tenacity, Tensile strength, Alkali treatment
Cellulose, Linear Density, Tenacity, Tensile Strength, Alkali Treatment
1. Introduction
Sugarcane (Saccharum officinarum) is cultivated in considerable quantities in tropical countries. In 2017, about 1.84 billion tons of sugarcane were produced worldwide [1]. It is used in sugar mills and alcohol mills. But it cannot be consumed entirely by those mills as about 30% pulpy fibrous residue is produced after being utilized in those mills [2, 3,4]. These residues are called bagasse [5]. The bagasse is used in various applications, including paper industries, as feedstock, as biofuel, etc [6, 7]. Sugarcane bagasse is a lingo cellulosic material [8]. It is generally a kind of waste [2], which may have some particular uses. Since it contains quite a fair amount of cellulose, this cellulose can be extracted, and that cellulose can have different applications. The fibrous materials may also be used as fiber in the textile and civil engineering sector, too though they may need some unique treatments before being used. More specifically, this bagasse can be used to reinforce composite materials for creating a totally new type of material [7]. The main advantage of using bagasse is, it is pure waste material, and if this material can be utilized in any application even after a few simple pretreatments, the process still produces a very economical product, and also the product will surely be fully or partially biodegradable, which is quite an important factor these days. Also, the extracted fiber can show quite good mechanical properties if an appropriate technique is used [7]. The extracted cellulose can be used for producing sustainable regenerated textile fibers, too [9]. Other than that, the bagasse can also be a source for producing nanoparticles though that would not come cheaply as alternatives [10, 11]. In this paper, structural properties and applications of sugarcane bagasse in different sectors will be discussed, along with some essential pretreatments or fiber extraction procedures.
2. Physical structure and chemical composition of sugarcane bagasse fiber
Vegetable fibers that are derived from stalks or stems are called bast fibers [12]. Jute, Flax, Ramie, etc., are bast fibers, while these are also classified dicotyledons as these plants have net veined leaves [13]. Sugarcane contains parallel-veined leaves. The fiber bundles are randomly arranged throughout the stem of the fiber, but in bast fiber, the fiber bundles are arranged in a certain ring pattern, and that's why it is not classified as bast fiber [13]. The sugar cane stalk can be divided into two portions, the outside rind and an inner pith. The outside rind portion contains longer and finer bundles of fibers, while the inner portion contains the short fibers [12, 14]. Bagasse actually contains both types of fibers [2]. Cellulose covers about one-third of the plant tissues of sugarcane. Sugarcane bagasse contains about 40–50% cellulose and 25–35% hemicellulose. The rest contains lignin, wax, etc [[15], [16], [17]]. Cellulose has a crystalline structure (about 50–90% crystalline depending on the source of cellulose [18]), while hemicellulose is an amorphous structure containing xylose, glucose, etc [8, 19]. Cellulose is more like a natural linear polymer containing anhydroglucose units linked by β 1, 4 glycosidic bonds [8, 17]. It contains three hydroxyl groups of different reactivity as C-2 and C-3 got secondary –OH groups while a primary –OH can be found at C-6 position [17, 20]. These hydroxyl groups help to produce strong intermolecular and intramolecular hydrogen bonds [20]. These cellulose polymers are distributed in fibrils that are surrounded by hemicellulose and lignin. Lignin actually works as a glue between cellulose and hemicellulose and helps the material to gain rigidity [8]. It is a three-dimensional polymer containing three different phenyl-propane precursor monomer namely, p-coumaryl, coniferyl, and sinapyl alcohol, which are joined together by alkyl-aryl, aryl-aryl, and alkyl-alkyl bonds [8, 21]. The chemical composition of sugarcane bagasse is given below in Table 1 [7, 8, 22, 23, 24, 25, 26, 27, 28, 29, 30].
Table 1.
Chemical composition of sugarcane bagasse.
| Name of the Content | Percentage |
|---|---|
| Cellulose | 26–47 |
| Hemicellulose | 19–33 |
| Lignin | 14–23 |
| Ash | 1–5 |
This composition stands for untreated raw sugarcane bagasse. But different pretreatments can help to reduce the contents, such as hemicellulose, lignin, etc., so that 55–89% cellulose can be yielded from these bagasse samples [7, 27].
3. Extraction process of fiber and cellulose from bagasse
Sugarcane bagasse has been used for different purposes. Cellulosic fiber has been extracted from it. It has also been used for extracting pure cellulose, while cellulose nanoparticles have also been tried to be extracted from sugarcane bagasse. Different researchers have used various techniques, and the outcomes have also been different. Some extraction processes are described below.
3.1. Fiber extraction
Sugarcane bagasse contains two different types of fibers in the rind and the inner pith. Generally, the fibers of the rind can be useful due to their longer length and comparatively better mechanical properties. Fibers from inner pith can also be used in various applications, but those are not expected to show better mechanical properties due to having minimal fiber length [12, 14, 31]. The fiber extraction procedure varied by different researchers, but the main operations remained quite similar. A general process flow diagram is shown below in Figure 1.
Figure 1.
Process flow diagram of fiber extraction.
Collier et al. tried to extract fibers from sugarcane rind [12]. They separated the rind from the pith and then removed the nodal regions from the rind as those regions have different physical structures [14]. They pretreated the fibers at the beginning of the process. At first, 4 cm long and 1–2 mm wide rind pieces were immersed in hot water. It was done to remove the sugars. 0.1N and 1N concentrations of sodium hydroxide (NaOH) at high atmospheric pressure (2 atm) and normal atmospheric pressure for 1–2 h and 1–4 h, respectively, were used in that study. The used temperature was 121 °C. Continuous stirring was used for standard atmospheric pressure treatment. 100 ml alkaline solution was used for 1 gm of the rind. They found that higher concentration of NaOH, higher temperature, and higher atmospheric pressure removed more amount of lignin and resulted in fiber bundles of lower linear density [21.8 Tex (Tex is a unit of linear density which means mass per unit length [32]) at 2 atmospheric pressure by using 1N NaOH in 2 h of treatment, and 22.5 Tex at normal atmospheric pressure by using the same concentration of NaOH in 4 h treatment with continuous stirring]. They found the ultimate cell length of fiber about 2 mm, which was within the range of the ultimate cell length of jute fiber [33, 34]. But the fiber bundles of lower linear density (finer fiber bundles) showed comparatively inferior mechanical properties (tenacity of 28.7 cN/Tex and 15.6 cN/Tex for fiber bundles of 54.2 and 21.8 Tex, respectively).
Michel et al. also tried to extract fibers from sugarcane bagasse [2]. The process flow was similar to the given one in Figure 1, but they did have some variations in the pretreatment of bagasse. They tried two concentrations of NaOH (0.1N and 1N) for treating bagasse at 130 °C for an hour in an autoclave. They pretreated two samples with distilled water and salty water, respectively, at the same temperature in an autoclave. They obtained comparatively smaller fibers than the previously mentioned process because they took whole bagasse instead of only fibers from the rind. Treatment with salty water (with 1N NaOH) resulted in fibers of the lowest average length (29.8 mm) but finer fiber bundle (32 Tex), while pretreatment with only distilled water (with 0.1N NaOH) resulted in fibers of the highest average length (45.6 mm) but higher linear density (39 Tex). NaOH concentration did not seem to affect the mean fiber length too much as both 0.1N and 1N NaOH solution produced fibers of 37.6 and 37.7 mm, respectively. But the fiber bundle fineness was affected to a greater extent as 49 Tex, and 35 Tex were resulted by those two solutions, respectively. Again as the previously mentioned research, the finer fiber bundle showed lower strength (tenacity 7.5 cN/Tex), and the coarser fiber bundle showed higher strength (tenacity 22 cN/Tex). Salty water pretreated fibers were found to be less rigid, while fibers treated with only 0.1N NaOH were impossible to be bent at all. It means these fibers are relatively stiff and are not really suitable for textile purposes in comparison to other textile fibers such as jute, flax, etc., where the low tenacity of bagasse fibers is mainly responsible [[35], [36], [37], [38], [39]].
Meanwhile, Chiparus and Chen used a similar technique where they used NaOH solution for the delignification of sugarcane bagasse and found similar results and found fiber length varying from 50.2 mm to 70.2 mm and fiber fineness varying from 18.722 to 47.822 Tex [40]. There are not many pieces of research involving the extraction of fiber from sugarcane bagasse. Some have extracted fibers just for reinforcing composite materials. For this purpose, the fiber length does not have to be longer always as fibers of minimal length can also be used as reinforcements. Cao et al. used sugarcane bagasse fiber in their research as reinforcement [41]. They extracted fiber-containing a high percentage of cellulose by treating the bagasse with NaOH. They soaked bagasse in each 1% NaOH solution at 25 °C for 2 h, maintaining a liquor ratio of 20:1. NaOH treatment increased about 17% tensile strength and also produced finer fiber (fiber length was 0.9 cm).
3.2. Cellulose extraction
There have been some researches for extracting cellulose from sugarcane bagasse. Extracted cellulose has been used in various applications. The process of extracting cellulose from sugarcane bagasse can be summarized in the following process flow diagram in Figure 2.
Figure 2.
Process flow diagram of cellulose extraction from sugarcane bagasse.
The processes varied by different researchers in terms of the use of the chemicals and conditions. Sun et al. used three different processes to extract pure cellulose from sugarcane bagasse [17]. They ground and dewaxed sugarcane bagasse in toluene-ethanol solution before treating the samples using water at 55 °C and with or without ultrasonic radiation. Sequential treatment was done in one set of samples by using sodium hydroxide (0.5M) and hydrogen peroxide (0.5, 1, 1.5, 2, and 3%) and got 2 types of cellulose (44.7 and 45.9%). They also produced delignified samples by treating the samples sequentially with 1.3% sodium chlorite and 10% sodium hydroxide or potassium hydroxide. This process resulted in 44.7 and 44.2% cellulose yield. In the third process, they used a one-step treatment with an 80% acetic acid and 70% nitric acid mixture under controlled conditions in 20 min of treatment. This process resulted in a cellulose yield 43.0 (at 120 °C) and 43.6% (at 110 °C).
Meanwhile, Liu et al. extracted cellulose sugarcane bagasse by using quite similar processes as the previous research [42]. They dewaxed the samples by using chloroform-ethanol solution, and then they delignified the samples using chlorite and ultrasonic irradiation before treating them with 6% sodium hydroxide at 75 °C for 2 h. Then the samples were treated in two different systems. In one approach, they treated the samples sequentially by using 15 and 18% potassium hydroxide, 15 and 18% sodium hydroxide at 23 °C for 2 h, and 8 and 10% potassium hydroxide, 8 and 10% sodium hydroxide at 23 °C for 12 h. In a one-step system, they treated the samples with 10% potassium and sodium hydroxide at 23 °C for 16 h. These treatments resulted in 50.7, 49.5, 48.6, 47.8, 57.2, and 55.4% cellulose yield, respectively. They also found that crystallinity values (39.8, 40.3, 41.6, 42.7, 44.8, and 45.6%) were less than cellulose of flax, cotton, and kenaf fibers (70, 65, and 60%, respectively) [43].
Abdel-Halim extracted cellulose from sugarcane bagasse by alkaline treatment with sodium hydroxide, followed by delignification/bleaching using sodium chlorite/hexamethylenetetramine solution [44]. He used dried and ground bagasse for the process. He treated bagasse with NaOH (0.1–2.5 N) and a non-ionic wetting agent (2 gm/l) while he used 1:20 material to liquor ratio at the boiling temperature for an hour. Later for the delignification process, he treated the alkali-treated bagasse with sodium chlorite (NaClO2) (5 gm/l), hexamethylenetetramine (0–0.5 gm/l), and non-ionic wetting agent (2 gm/l) at boiling temperature with 1:30 material to liquor ratio.
Saelee et al. extracted cellulose from dried sugarcane bagasse using steam explosion and xylanase pretreatment and bleaching process [27]. They used the technique of Rocha et al., for which, first of all, the bagasse underwent a steam explosion at 13 bar pressure and 195 °C temperature for 15 min [45]. The exploded bagasse was then treated with 20 μ/g of xylanase by using fiber to liquor ratio of 1:10 for an hour at 50 °C under constant agitation. Then bleaching was done by using 0.7% sodium chlorite (NaClO2). Other than these, Shaikh et al. also used a steam explosion for cellulose extraction from bagasse [46]. While S. M. Costa et al. used a solution containing 16% sodium oxide (Na2O) and 0.15% anthraquinone, at a 12:1 liquor ratio [9]. The solution was heated at 160 °C for one and a half hours, as determined in another research [47]. They bleached the obtained fiber by dissolving it in N-methylmorpholine-N-oxide (NMMO) using the method used by Fink et al. [48] to produce pure cellulose fibers.
3.2.1. Nanocellulose extraction
Nanocellulose is cellulose in the form of fibers or crystals which have lengths in a few micrometers region and diameter within <100 nm range. Those are extracted from natural cellulose fibers. Nanocellulose materials are mainly of three types-nano fibrillated cellulose, cellulose nanocrystals, and bacterial nanocellulose. Among these, the cellulose nanocrystals are generally derived from cellulosic fibers [49]. Most recent researches have been taken place to extract nanocellulose or nanocrystals from sugarcane bagasse. Like the previous sections, this process can be summarized in the following process flow diagram in Figure 3.
Figure 3.
Process flow diagram of nanocellulose extraction from sugarcane bagasse.
The chemical treatments varied by different researchers again in terms of the chemicals, the ratios, and other conditions. Mandal and Chakrabarty extracted nanocellulose in their study [50]. The bagasse fibers were bleached with 0.7% (w/v) NaClO2 solution. Then the obtained hollow cellulose fibers were treated with NaOH (by boiling with 250 ml 17.5% (w/v) solution for 5 h) to remove any hemicellulose from the fibers. Then the fibers were dissolved in 50 ml dimethylsulfoxide solution at 80 °C, and the treatment lasted for 3 h. The obtained product was acid hydrolyzed using 60% sulphuric acid (H2SO4) at 50 °C (1:20 liquor ratio) for 5 h under vigorous agitation to produce nanocellulose. The particle sizes were found in the range of 18.17–220 nm.
Kumar et al. [51] extracted cellulose nanocrystals from bagasse by using the acid hydrolysis technique described by other researchers [52,53]. They hydrolyzed delignified and hemicellulose-free bagasse using H2SO4 solution [64% (w/w)] at 45 °C for 60 min under vigorous and constant mechanical stirring. They found that the process produced rod-shaped cellulose nanocrystals having the size in the range of 250–480 nm (length) and 20–60 nm (diameter), and the crystallinity percentage was about 72%. This process of cellulose nanocrystal preparation was again used by Slvutsky and Bertuzzi [11]. They alkali hydrolyzed bagasse by treating 10gm of sugarcane bagasse with 100 mL of NaOH (6%) at 60 °C for 4 h in a shaker before bleaching the bagasse in 200 mL of NaClO2 (30%) solution and with shaking for 24 h at room temperature. Then they acid hydrolyzed the bagasse to produce cellulose nanocrystals. The average length and diameter of the obtained particles were 247.51 (±32.34) nm and 10.11 (±3.36) nm, respectively. de Oliveira et al. used a similar technique to produce cellulose nanocrystals [54]. They bleached the bagasse pulps by using the process of Sun et al. in which they treated the bagasse with an alkaline solution containing 24% hydrogen peroxide (H2O2) and 4% NaOH at 70–80 °C [17]. Then the treated the bleached samples at 50 °C in a preheated solution of 65 wt.% H2SO4 with mechanical stirring for 40 min (5.0 gm pulp was added in 200 mL of the acid solution) [55]. They also found rod-like particles, which are similar to other results mentioned above that also include some which have not been discussed here [[56], [57], [58], [59], [60]]. Particle size varied within the ranges of 69–117 nm length and 6–7 nm diameter. Ferreira et al. [61] used the method of de Oliveira [54] for bleaching the bagasse. Then they acid hydrolyzed the bagasse by using H2SO4 where 10 gm sample was dispersed in 250 mL of H2SO4 (65% v/v) at 45 °C for 45 min.
Other than these processes, Sofia et al. used a ball milling technique [62], while Feng et al. used ultrasonic treatment without acid hydrolyzation [63]. For the first process, dewaxed bagasse fibers were dilignified by using a 1.3% sodium chlorite solution, and then the obtained holocellulose fibers were treated with 10% potassium hydroxide (KOH) and 10% NaOH at 20 °C. They used two techniques; one was used by Kumar et al. [51], which involved acid hydrolysis, while the other approach used Spex 8000M shaker mill for milling. They placed 0.25 gm of bleached bagasse fibers in a 70mL container made of polypropylene along with 50 mL of deionized water and 20gm of cerium-doped zirconia balls (ball diameter was 0.5 mm), and the process took place for an hour. Results showed that the ball milling technique did not remove the amorphous portions, whereas acid hydrolysis removed amorphous portions and increased the crystalline part of cellulose nanocrystals. In the study of Feng et al., they bleached the fibers with 30% hydrogen peroxide at 80 °C for 3 h. Then the fibers were treated with deionized water for an hour before being disintegrated in a high-speed blender at 48000 rpm for 5 min. Then the samples were treated with ultrasonication for producing nanofibers. Kathiresan and Sivaraj extracted nanocellulose from sugarcane bagasse using sodium hydroxide [64]. The fibers were collected from the outer rind at first. The fibers were chopped into small pieces and then treated with sodium hydroxide (1% sodium hydroxide solution at 80 °C for two hours) to separate the fibers easily. The collected fibers were then milled (at 300 rpm for 5 h) in a high-energy ball mill to produce nanofibers.
4. Applications
Generally, fibrous materials are most commonly used in the textile sector. But for being suitable for textile applications, the fibers need certain qualities such as fineness, crimp, tensile strength, etc. [31], which sugarcane bagasse cannot offer, as shown in Table 2.
Table 2.
| Fiber Name | Chemical Components% |
Physical Properties |
||||
|---|---|---|---|---|---|---|
| Cellulose | Lignin | Hemicellulose | Ash | Liner Density (Tex) | Tenacity (cN/Tex) | |
| Cotton | 85–90 | 0.7–1.6 | 1–3 | 0.8–2 | 0.15–0.4 | 20–40 |
| Jute | 61–72 | 12–13 | 13.6–20.4 | 0.5–2 | 1.4–3.0 | 41–52 |
| Flax | 64–71 | 2–5 | 18.6–20.6 | 5 | 0.2–2.0 | 54–57 |
| Ramie | 68.6–76.2 | 0.6–0.7 | 13.1–16.7 | -- | 0.5 | 40–77 |
| Hemp | 57–77 | 9–13 | 14–17 | 0.8 | 2.2–3.0 | 47–60 |
| Kenaf | 44–57 | 15–19 | 22–23 | 2–5 | 1.9–2.2 | 25.4 |
| Sisal | 47–62 | 7–9 | 21–24 | 0.6–1 | 28.6–48.6 | 36–44 |
| Coir | 36–43 | 41–45 | 0.15–0.25 | -- | 50 | 8–18 |
| Sugarcane Bagasse | 32–48 | 19–24 | 27–32 | 1.5–5 | 18.72–54.2 | 7.5–22 |
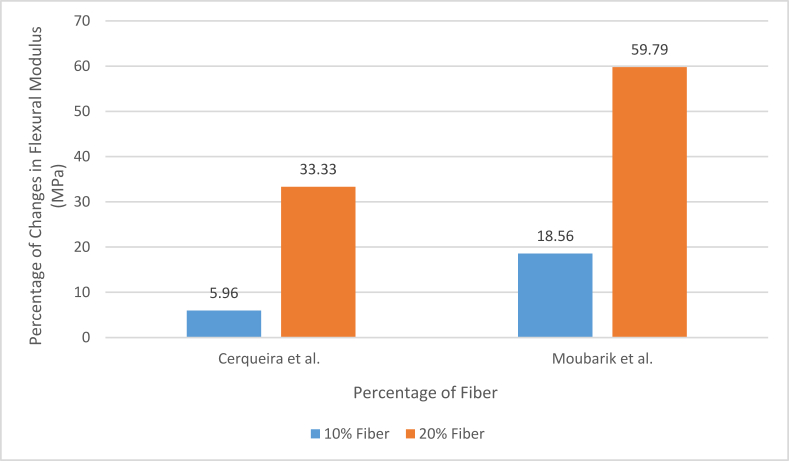
It can be seen from the above table that sugarcane bagasse fiber has meager strength and these fibers are quite coarser in comparison to other fibers such as cotton, flax which are extensively used in textile products. Only coir and sisal fiber has shown some similarities, and these fibers are not used in the textile application that much. But these fibers are not totally unusable. In fact, natural fibers that do not have better properties like cotton or flax are used in composites as reinforcement quite often [30]. Sugarcane bagasse fibers have also been used in composites by several researchers [10, [71], [72], [73], [74], [75], [76], [77]]. Cerqueira et al. prepared bagasse fiber reinforced polypropylene composite [72]. The fibers were pretreated with 10% H2SO4 and 1% NaOH. Addition of 20% fiber increased 15.5% tensile strength, 45.4% impact strength and 32.4% flexural rigidity. Monteiro et al. prepared bagasse fiber reinforced polyester composite and found an increase in flexural rigidity due to reinforcement [75]. Oladele also found an increase in tensile strength with bagasse fiber addition in polyester composite [78]. Moubarik et al. showed that delignified sugarcane bagasse fiber significantly improved the mechanical properties of low-density polyethylene composite [79]. Monterio et al. reported that bagasse fiber-reinforced multilayered epoxy composite could show similar performance to Kevlar multilayered sheet [80].
But polyethylene, polypropylene, epoxy, etc., are synthetic polymers. Composites containing types of the matrix are only partially biodegradable. For producing fully biodegradable material, natural polymers have also been used, among which starch is one of the most common ones. Starch has been used for modifying textile materials, too [81]. There have been some good examples of sugarcane bagasse reinforced starch composites. Gilfillan et al. showed that the addition of sugarcane bagasse fiber increases the crystallinity of the composite [10]. Vallejos et al. demonstrated in their study that the accumulation of sugarcane bagasse fiber increases tensile strength and decreases water absorbency of the starch composite [71]. Jeefferie et al. produced a disposable food container from sugarcane bagasse reinforced starch composite [74]. Guimarães et al. [82] showed that sugarcane bagasse reinforced starch composite has better elongation property than banana fiber reinforced starch composite of the same component ratio. On the other hand, Arrakhiz et al. showed that bagasse fiber (25 and 30% fiber loading) reinforced polypropylene composite has better tensile properties than coir and alfa fiber-reinforced composites even though both coir and alfa fibers are stronger than sugarcane bagasse fiber [73].
Nanocellulose has also been used by several researchers in their studies in recent days to produce eco-friendly composite materials. Gadheri et al. extracted nanocellulose from sugarcane bagasse and used it to reinforce cellulose film for making food packaging material [83]. The nanocellulose was obtained by treating bagasse using sodium hydroxide and anthraquinone at first, then bleaching using sodium chlorite and potassium hydroxide. These processes removed all the contents except alpha-cellulose, and that was then ground to the size of the nanofiber. For making the cellulose matrix, they used DMAc (dimethylacetamide) and LiCl (lithium chloride) solution to dissolve the cellulose contents from bagasse, and then those were used to make the composite material. Considerably good tensile and vapor transmission properties were reported from the test results for those materials. Slavutsky and Bertuzzi prepared nanocellulose reinforced starch composite [11] using sodium chlorite like the previous study. The process was a bit different, but the principle was similar. They reported that the water affinity was significantly decreased with nanocellulose reinforcement. The similarity of the chemical structure of cellulose and starch was also responsible for this improvement.
Achaby et al. produced nanocellulose reinforced polyvinyl alcohol/carboxymethyl cellulose (PVA/CMC) blend composite for food packaging application where the nanocellulose was extracted from sugarcane bagasse [84]. They used the acid hydrolysis process to extract the nanocellulose. The bagasse fibers were treated with alkali and then bleached with chlorite before being hydroseed using sulfuric acid under mechanical stirring to produce nanocellulose. The composites were made using casting techniques. The resulted materials showed that the addition of 5% CNC (weight percentage) within a PVA/CMC improved the tensile strength and modulus by 83% and 141 %, respectively, while the water vapor permeability was reduced significantly by 87%. Gan and Chow also used similar techniques (acid hydrolysis for nanocellulose preparation and casting technique for composite making) to produce nanocellulose reinforced PLA (polylactic acid) composite using sugarcane bagasse [85]. They reported that this combination was helped by the presence of hydrogen bonding among cellulose and PLA in the composite and the end products showed better thermal stability than pure PLA.
Cellulose obtained from sugarcane bagasse can also be of fair use other than in paper industries. Pure cellulose is used to make regenerated cellulosic fibers which can be used as a substitute for cotton fibers in the textile sector [86]. The extracted cellulose from it can be used to make hydroxyethyl cellulose, regenerated fiber like cellulose acetate, etc [9, 44, 46, 87]. Cellulose acetate produced by Shaikh et al. [46] showed a tensile strength of 40–61 Mpa, which matches the tensile strength of commercial cellulose acetate fibers (31–55 Mpa [88]). Other than these, cellulose nanocrystals and nanofibers have been used in composite materials with some good effects [11, 89].
5. Assessment of the properties of some products
Sugarcane bagasse fibers have been found as part of many pieces of research. So, the variation between works is quite common. Various types of works have been mentioned in the previous sections. The variations resulted from various reasons, including the used chemicals, the used procedure, the used condition, etc. If some of the common variations can be compared with similar works to find out some controlling points of the procedures. The comparison starts with the fiber extraction process below.
5.1. Fiber extraction
Collier et al. [12] and Michel et al. [2] both used a similar procedure for fiber extraction but the resulted materials were slightly different in terms of yield percentage, mechanical properties, etc. The properties are shown in the Table 3 below.
Table 3.
Comparative analysis of the fiber extraction processes.
| Pretreatment | Collier et al. [12] |
Michel et al. [2] |
||||||
|---|---|---|---|---|---|---|---|---|
| Alkali Treatment | Prehydrolysis and Alkali Treatment | Alkali Treatment Only | ||||||
| Solution | NaOH | NaOH | Distilled Water | Salty Water | NaOH | |||
| Temperature | 121 °C | 121 °C | 130 °C for Prehydrolysis and 130 °C for Alkali Treatment | 130 °C for Prehydrolysis and 130 °C for Alkali Treatment | 130 °C for Alkali Treatment | |||
| Pressure | 1 atm | 2 atm | 1 atm | 1 atm | 1 atm | |||
| Duration | 1 h | 1 h | 1 h for Prehydrolysis and 1 h for Alkali Treatment | 1 h for Prehydrolysis and 1 h for Alkali Treatment | 1 h for Alkali Treatment | |||
| Alkali Concentration (N) | 0.1 | 1 | 0.1 | 1 | 0.1 | 1 | 0.1 | 1 |
| Properties | ||||||||
| Linear Denisty (Tex) | 54.2 | 24.2 | 49.8 | 25.8 | 39 | 32 | 49 | 35 |
| Tenacity CN/Tex | 28.7 | 21.9 | 23.7 | 15.9 | 14.0 | 7.5 | 22 | 11 |
| Breaking Energy (mJ) | 1.96 | 0.66 | 1.69 | 0.72 | 2.9 | 1.2 | 4.7 | 2.2 |
| Bending Rigidity gf.cm2/fiber bundle | 0.288 | 0.205 | 0.411 | 0.157 | 0.190 | 0.027 | -- | 0.116 |
The results are quite obvious as the indication is towards the use of the pre-hydrolysis process that had some significant impact on the fiber properties. If the outcomes of the two pieces of research are summarised, then a few points can be found.
According to the above-given results, it is clearly found that the pre-hydrolysis process affected the linear density of the fiber bundles. The fibers were extracted in bundle form rather than in single fiber form. The linear density of the bundles was reduced in the case of three cases, when the concentration of the alkali was increased when the atmospheric pressure was increased, and when the duration of treatment was increased. It was down to the degree of the removal of lignin during the alkali treatment of the fibers [12]. Another reason can be added after analyzing the results shown in the table above. It is the pre-hydrolysis that also resulted in finer fiber bundles. The hydrolysis process may have affected the removal of lignin. By removing more lignin, the fiber bundles were made softer, as the reduced bending rigidity of the bundles found in the table above proves that point.
As the linear density, mechanical properties have also been affected due to the removal of lignin. Higher concentration of alkali, longer treatment, higher atmospheric pressure, and pre-hydrolysis, all these processes have resulted in lower tenacity of the fibers. The breaking energy results are proving that theory in the above table.
So the final results from this part of the review are that the higher degree of removal of lignin can be done by increasing atmospheric pressure, temperature, duration of treatment, and introducing a pre-hydrolysis process. The SEM images of these researches also showed that the mentioned points had resulted in a rougher surface of the fibers [2, 12]. And the removal of lignin and other additives from the fibers resulted in a decrement in the linear density of the fibers and deteriorated the mechanical properties of the fibers while the resulted fibers became quite softer with less bending rigidity. But if the results are compared to the mechanical properties of some other common natural fibers. In that case, it is clear that the extracted fibers from sugarcane bagasse are not that good in terms of properties. If the tenacity of the extracted fibers is compared with some other common fibers as shown in Table 4 below, proof of this point can be found.
Table 4.
| Fiber | Tenacity (CN/Tex) |
|---|---|
| Flax | 54–57 |
| Jute | 41–52 |
| Hemp | 47–60 |
| Sisal | 36–44 |
| Coir | 18 |
| Sugarcane Bagasse | 7.5–28.7 |
The tenacity is quite lower than other commonly used fibers like jute, flax, hemp, etc. And sugarcane fiber bundles are quite coarser than those fibers too. It makes the extracted fibers from sugarcane bagasse unsuitable for being used in common applications like spinning to make any kind of fiber or coarser threads. These fibers can only be used along with other fibers or materials; in that case, the composites would be the best option other than extracting cellulose or producing cellulosic products from these bagasse fibers.
5.2. Cellulose extraction
Cellulose extraction involved similar processes like fiber extraction with some addition. The processes being mentioned earlier in this study have had some similarities and some differences that affected the research results. The comparison between the procedures can be started from the very beginning to the end. The following Table 5 will show some comparisons among some researchers.
Table 5.
Comparative analysis of the cellulose extraction processes.
| Name of the Process | Specifications of the Process | Researched by |
||||
|---|---|---|---|---|---|---|
| Sun et al. [17] | Liu et al. [42] | Abdel Halim [44] | ||||
| Choosing the Raw Material | Dried and ground Sugarcane Bagasse | Dried and ground Sugarcane Bagasse | Dried and ground Sugarcane Bagasse | |||
| Dewaxing | Toluene-Ethanol Solution (2:1 v/v) | Chloroform-Ethanol Solution (2:1 v/v) | NaOH | |||
| Duration | 6 h | 6 h | 1 h | |||
| Temperature | Room temperature | Room temperature | 100 °C | |||
| Washing | H2O | H2O | ||||
| Duration | 2 h | 2 h | ||||
| Temperature | 70–80 °C | 55 °C | ||||
| Delignification | NaClO2 | Ultrasonic Irradiation Treatment | NaClO2 | NaClO2 and Hexamethylenetetramine | ||
| Duration | 2 h | 40 min | 2 h | 50–250 min | ||
| Temperature | 75 °C | 55 °C | 75 °C | 50–95 °C | ||
| Bleaching and Cellulose Extraction | KOH and NaOH | NaOH and H2O2 | NaOH and H2O2 | Ultrasonic Irradiation Treatment | ||
| Duration | 10 h | 2 h | 2 h | 30 min | ||
| Temperature | 20 °C | 55 °C | 55 °C | 60 °C | ||
| NaOH or KOH | ||||||
| Duration | 2/12/16 h | |||||
| Temperature | 23 °C | |||||
| Properties | ||||||
| Yield% | 44.2–44.7 | 45.9 | 44.7 | 47.8–55.4 | 45 | |
| Percentage of Weight Loss% | 55.3–55.8 | 54.1 | 55.3 | 44.6–52.2 | 55 | |
| Intrinsic Viscosity(mL/g) | 412.8–415.6 | 400.8 | 356.2 | 534.1–631.6 | ||
| Degree of Polymerisation | 1396–1406.5 | 1351 | 1185.2 | 1913–2238.2 | ||
| FTIR | Absence of lignin band at 1510-1520 cm−1 | Absence of lignin band at 1510-1520 cm−1 | Absence of lignin band at 1510-1520 cm−1 | Absence of lignin band at 1510-1520 cm−1 | Absence of lignin band at 1510-1520 cm−1 | |
From the above comparisons, the possible outcomes to be pointed are,
The ultrasonic irradiation process has some effect on the cellulose extraction process, according to these results. Ultrasonic irradiation is a mechanical process that applies sound as a mechanical force onto the material. It resulted in slightly more purification of cellulose. In fact, in the ultrasonic treatment was used reported more purified but less yield percentage of cellulose [17, 42]. The processes that did not use ultrasonic treatment resulted in a slightly higher yield percentage of cellulose, which means less purified cellulose. The cellulose contents are never affected by the ultrasonic treatment. The mechanical force it created could only attack the outer side of the bagasse made of hemicellulose and lignin. The cellulose is found only on the inner side of the material. The outer part is affected by the external mechanical force created by ultrasonic treatment. That's why the yield percentage was reduced as the lignin, hemicellulose, and other debris is removed from the cellulosic part of the bagasse. When the ultrasonic treatment was not used, the yield percentage increased, which means the presence of some foreign elements in the shape of hemicellulose, lignin, or some other chemicals. And this ultrasonic treatment can be applied before or after the Delignification treatment, as found from the above data.
For dewaxing, two methods have been found; one is to use chloroform-ethanol or toluene-ethanol in soxhlet at room temperature. The other method is using NaOH at boiling temperature. Both processes were found as effective ones. It is difficult to differentiate these processes in terms of duration, temperature; both systems have advantages and disadvantages. But using NaOH at a boil is a bit easier process than using the soxhlet for dewaxing.
For the delignification process, the researchers preferred sodium chlorite, which is quite effective in this action. But to get the best out of it, the introduction of hexamethylenetetramine was referred to by Abdel Halim [44]. In his research, he showed that using hexamethylenetetramine helped decomposing sodium chlorite into sodium hypochlorite. The hexamethylenetetramine decomposes into ammonia and formaldehyde when dissolved in water. Then the liberated formaldehyde accelerates the decomposition of sodium chlorite to produce sodium hypochlorite and formic acid. Sodium hypochlorite is an unstable gas that produces hypochlorous acid. The hypochlorous acid liberates the oxygen, which acts as the bleaching agent. Also, sodium chlorite decomposes easily in an acidic medium and liberates chlorine dioxide gas instead of chlorine gas, and it helps the bleaching process. That's why when the percentage of hexamethylenetetramine was increased; it resulted in more weight loss of bagasse due to better bleaching and cellulose extraction process and increased purity of cellulose. No further alkaline treatment was required when this method was used.
Apart from the above-mentioned points, there were some other findings too. The use of hydrogen peroxide was also found as a quick and effective process of bleaching and cellulose extracting process. It resulted in slightly less intrinsic viscosity, which means more purified cellulose, but the reduced degree of polymerization means the process did harm cellulose to a limited extent. Use of lower temperature but longer treatment resulted in a better degree of polymerization than the shorter treatments at comparatively high temperature, especially when additional alkali treatments, ultrasonic treatments were used.
The sequential treatments with a gradually higher concentration of alkali resulted in comparatively pure cellulose, according to the researchers, while sodium hydroxide was found as slightly more effective than potassium hydroxide due to being slightly stronger alkali than the latter [17, 42]. On the other hand, Sun et al. proposed another process of delignification and removal of noncellulose polysaccharides by a totally chlorine-free system for cellulose isolation and purification using acetic acid and nitric acid mixture. The resulting cellulose was quite pure apart from the formed ester bond with some acid molecules found in the FTIR spectrum (a peak in the region of 1745 cm−1). But the significantly lower degree of polymerization due to breakage of cellulose chain in the acidic medium was found as the main drawback of the system. But still, a chlorine-free method was found nearly as similarly effective as the other methods.
Another point to be mentioned here is the range of the degree of polymerization found in the extracted cellulose of the above-mentioned researches. The range is on the lower side after treatment which means the cleavage of cellulose chains during treatment. But among the mentioned processes, the best results were obtained when no additional bleaching agents were used. It means the bleaching process causes more chain cleavage and reduces the degree of polymerization.
5.3. Nanocellulose extraction
Nanocellulose or cellulose nanocrystals mean cellulosic particles which are sized within the range of nano scale. These types of particles can also be produced from sugarcane bagasse fibers. To do so, an additional process is required, which is the hydrolyzing step of extracted cellulose using acid under vigorous and constant mechanical stirring. In this case, the researchers chose almost a similar system in their researches. It is hydrolyzing the extracted cellulose from sugarcane bagasse using sulphuric acid. Generally, the sulphuric acid solution containing 60–65% sulphuric acid was chosen as the acid medium. The time and temperature of treatment varied a bit along with the cellulose extraction process [11, 50, 51, 54]. Otherwise, the system followed the same steps after hydrolyzing, starting with successive centrifugation at very high-speed rpm for a specific amount of time to remove the acidic solution. Centrifugation was followed by sonication for the removal of acidic solution [11, 51]. Sonication is another mechanical process that applies sound waves to agitate the particles for better separation.
The FTIR of raw sugarcane bagasse extracted cellulose from the bagasse, and the extracted nanocellulose from the bagasse showed similar results for all the researches [50, 51, 54]. The few common points noted from the results were,
-
i)
Raw sugarcane bagasse showed peaks in the spectra at around 1730 cm−1, 1602 cm−1, and 1505 cm−1 which are associated with lignin and hemicellulose. These peaks were not present in neither the spectra of extracted cellulose nor in the spectra of cellulose nano crystals.
-
ii)
Decrement in absorbance within the range of 1030–1160 cm−1 proved breakage of C–O–C glycosidic ether bonds were the results of hydrolysis. It means the cellulose chains were broken at several places, which resulted in reduction in the degree of polymerization of the cellulose polymer in nano form.
The TGA/DTG curves of these studies also showed similar trends. The onset and peak temperatures were found at maximum for extracted pure cellulose, while for raw bagasse and extracted nanocellulose, the temperature went in a downward direction gradually. It happened because of the presence of lignin and hemicellulose in the raw bagasse, which is thermally less stable and degrades at a lower temperature than pure cellulose. The increased crystallinity and the removal of the lignin and hemicellulose from pure cellulose of bagasse showed the best thermal properties as cellulose is generally more thermally strong than lignin and hemicellulose. For nanocellulose, the presence of sulfate (added from sulphuric acid hydrolysis process and this sulfate part is more amorphous) and weakened and reduced sized of the polymer chains of cellulose which created a greater number of free ends of chains showed vulnerability against heat [50, 51, 54]. This was evident in the DTG curves, too, which showed a reduction in the absorbance curve of temperature at the peak of cellulose degradation.
5.4. Properties of the bagasse reinforced composites
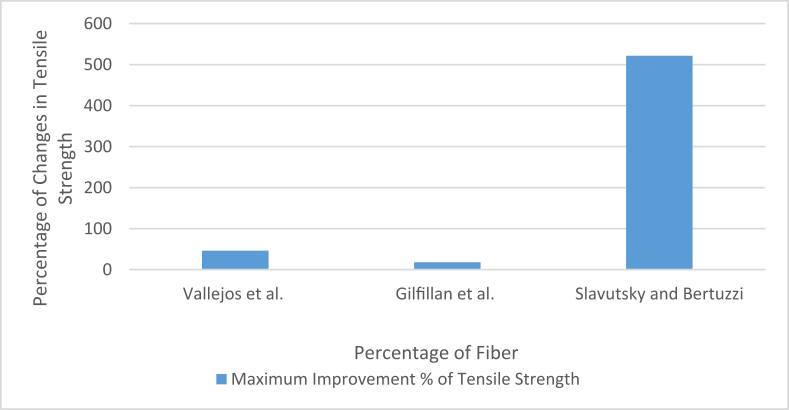
Sugarcane has been used in diverse applications that are discussed briefly in the previous section. It has been found to be useful along with both natural and synthetic matrix materials. Among the biological matrix materials, starch has been quite commonly used with sugarcane bagasse fiber. Vallejos et al. used untreated bagasse fibers (average fiber length was 0.67 mm) with two types of starch (corn and cassava) to produce bagasse fiber-reinforced (depithed fibers) thermoplastic starch composite using the hot pressing method (at 155 °C under 10 Tons of load) [71]. The scanning electron microscope images of their study showed that the fibers were hollow in shape which is responsible for the low density of bagasse fibers [90]. This was one reason for the decrement of tensile strength and increment in the rigidity of the composites with an increasing amount of reinforcing fiber in the material (improvement of mechanical properties took place till 10% fiber addition). Due to having very low density, a lot of fibers could weigh a lot lesser, for which fiber agglomeration occurred when the percentage of fiber was increased beyond 10%. But until that point, the tensile strength increased because of quite uniform dispersion of fibers in the matrix while the fibers were reasonably well coated by matrix materials where minimal voids could be seen. The same also made the material rigid. The moisture absorbency property of the composite went in a downward direction due to the use of untreated fibers (contained lignin).
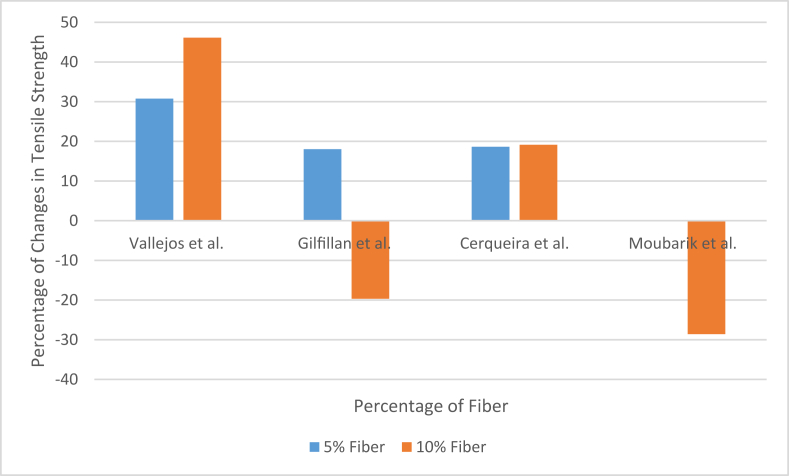
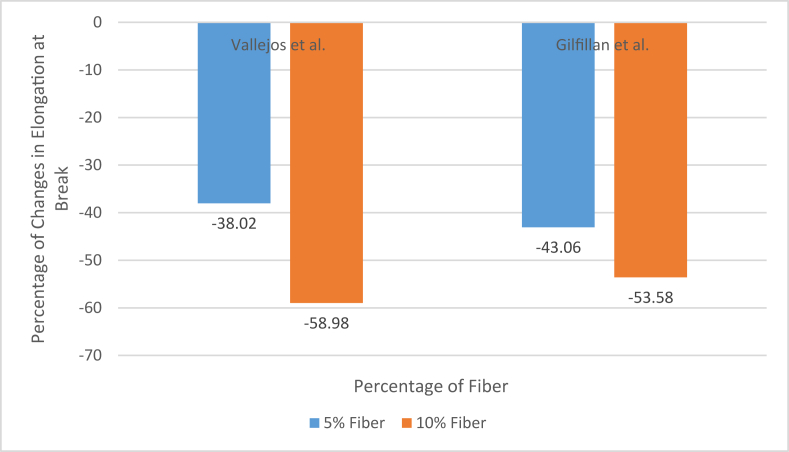
Gilfillan et al. also used depithed bagasse fiber, but the fibers were milled to a lot smaller size for their study [10]. They used a similar hot-pressing technique to produce bagasse fiber-reinforced thermoplastic starch composite. They also found a similar trend as the addition of fiber made the material rigid and decreased tensile strength after 5% fiber addition. But the fiber size can play some role in the mechanical properties of the end product. Cerqueira et al. and Moubarik et al. also reported the effect of fiber loading on the tensile properties of the composites. Cerqueira et al. prepared more like a crude cellulose pulp made from sugarcane bagasse, while Moubarik et al. prepared bleached and purified cellulosic fibers of 1–2 cm lengths to produce the composites. Cerqueira et al. used the hot pressing technique, while Moubarik et al. used the screw extruder to produce the composites. The following Figure 4 and Figure 5 [10, 71, 72, 79] can show the percentage of change in tensile strength and elongation property during fiber loading in the previously mentioned two studies.
Figure 4.
Changes in tensile strength with fiber loading.
Figure 5.
Changes in elongation property with fiber loading.
Even though the materials and procedures were different, but still, the differences were too significant as comparatively longer fibers offered better results according to these results. The reason behind the increment of tensile strength with fiber loading at the initial stage was the formation of only hydrogen bonds between fiber and starch because of similar characteristics of starch and the main component of bagasse, which is cellulose or, more specifically, the presence of –OH group in both cellulose and starch [10, 84]. The FTIR results also showed that no further bonds were created between fiber and resin. It happened for similar molecules of starch and cellulose. And when the resin was changed to synthetic polymer, again, no chemical bonding was recorded in FTIR results which means the properties of the composites made of sugarcane bagasse had to depend entirely on the mechanical bonding between resin and filler [79, 84, 85]. The elongation property also suffered drastically with the addition of fiber because of the very poor elastic properties of bagasse. When long fibers are used, the fibers directly get involved in the tensile properties of the composite as the pull of the tensile test is absorbed by the long fibers within the length of the fibers. On the other hand, when shorter fibers are used, the tensile properties depend more on the cohesive nature of the fibers and the bonding between fiber and resin rather than the tensile properties of the fibers. In fact, in the case of the smaller sized fibers, the resulted composite suffers deterioration of tensile properties at most cases when no chemical bonding is formed between fibers and polymers because the smaller fibers are unable to withstand the pulls or pressure put during the tests, instead the strain is put directly on to the resin, and if the bonding between the resin molecules is affected by the increasing amount of fibers, which is the case most of the times, the tensile properties are found on the wane. Sometimes the mechanical bonding helps to overcome the negative effect created by the fiber loading, but in those cases, extremely high pressure and rough surface of the fibers are essential, which can only be met when suitable techniques of fiber treatment, fiber loading, and pressing are used. The pressing technique is mentioned later in this section.
Meanwhile, the effect on flexural properties of the above-mentioned points can be seen in the research of Cruz-Tirado et al., who showed that the decrement of fiber size from 0.3mm to 0.075mm increased the flexural property of the composite, and it matches the concept explained above [91]. Figure 6 [72, 79] below shows the effect of fiber loading on flexural properties of composites.
Figure 6.
Changes in flexural property with fiber loading.
Here, fiber loading resulted in improved flexural properties in both pieces of research though the resins were different, polypropylene and polyethylene, respectively. So, the flexural properties depend mainly on the flexural property of the resin and the cohesive nature of the fibers in the case of smaller fibers. Fiber loading resulted in better flexural modulus, which means the composites needed more force to bend, but the percentage of deformation was lower. It means fiber loading made the composites stiffer. At the same time, the percentage of increment in the results may mean that polypropylene was improved less than the polyethylene composite. But in reality, the results were not as it is seen here. The polypropylene composite with 20% fiber loading reached about 1200 MPa flexural modulus, while the polyethylene composite reached about 310 MPa. So, as said earlier, the properties depended mainly on the properties of the resin. And the fiber loading improved the results further.
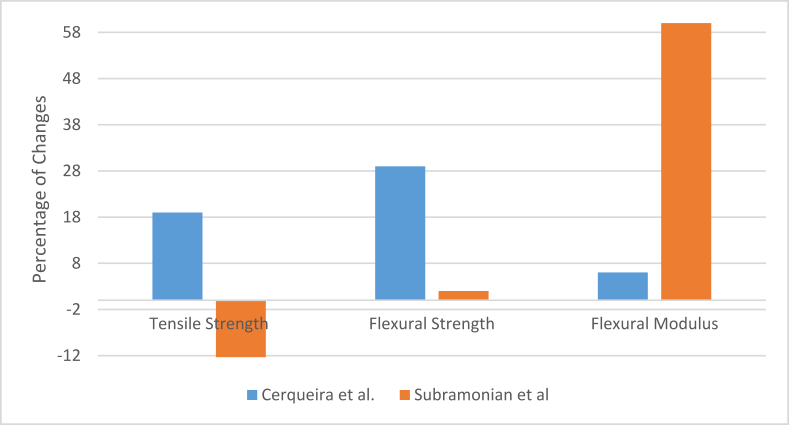
Meanwhile, as mentioned earlier, the composite preparation technique also can have some say in the final properties of the composites. Cerqueira et al. and Subramonian et al. showed that pretreated fiber could improve the tensile properties of polyethylene to a small extent [72, 92]. Scanning electron microscopic images showed pores in the structure and gaps between fiber and matrix, but still, fiber loading increased the tensile strength very slightly. It was due to the composite-making technique and bonding strength of polyethylene. In fact, it applies to all types of synthetic matrix materials. The compression molding process helps to bond between the molecules of these synthetic matrix materials by using a high amount of load. When the fiber percentage remains in small quantities, the matrix molecules can benefit from the added strength of the fiber and its bonding strength. But when the fiber percentage was increased, it becomes difficult for the matrix molecules to form enough bonds where fibers are accumulated in high quantity. It causes a drop in tensile properties. In the above-mentioned researches, the flexural and impact properties were improved by fiber loading because of the load absorption capacity of fibers. It was also reported by Candido et al. in their study where they used bagasse fiber to reinforce thermoset material polyester by casting technique [93]. The absorbency of the material was increased, though, due to having hydrophilic fibers in the structure [94]. Changes in some common mechanical properties in different studies discussed above are shown in Figure 7 below (for 10% fiber loading) [72, 92].
Figure 7.
Changes in mechanical properties with fiber loading.
Gillifilan et al. also reported that the hot-pressing technique was more useful than the casting technique of composite production incase of starch-sugarcane bagasse fiber composite [10]. Since no strong chemical bonds are formed within fiber and resin, the structure depends on the mechanical bonding between the contents. In that case, hot pressing can help to form mechanical bonds by a sufficient amount of pressure, for which the results of pressing techniques have been found in a higher range compared to the other techniques. The same concept was followed by other combinations too. In fact, whenever a natural matrix is used with a natural fiber, there are more chances of some kind of bonding between fiber and matrix than in the cases where a synthetic matrix is reinforced by natural fiber.
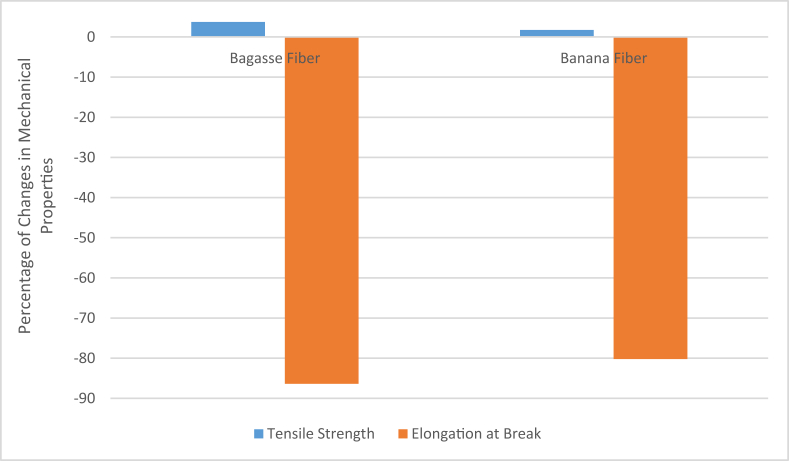
The type of fiber can also affect the properties of the composite due to the structural difference of the fibers, even if only the extracted pure cellulosic parts are used to make the composites. Guimarães et al. reported that sugarcane bagasse fibers could show better performance than banana fiber when used to reinforce the starch composite made in the compression molding method [82]. They used fibers of 3–5 mm in length. The difference was not significantly different, but still, sugarcane bagasse fiber-reinforced composite-edged banana fiber reinforced ones by smaller margins. The comparison of results is shown in Figure 8 [82].
Figure 8.
Changes in mechanical properties with fiber loading.
According to the SEM images and the data found from other researches [95], the banana fiber contains more percentage of cellulose and a lesser amount of lignin, for which it is slightly less rough than the bagasse fibers, which are also hollow fibers. When the pure cellulosic portions of the fibers are extracted, the removal of lignin, hemicellulose, etc., from the surface of the fibers makes the surfaces rougher. When the percentage of removal takes place, the resulted fibers become rougher. And since the mechanical properties of these cellulosic fiber-reinforced composites have been found to form no strong chemical bonds with the resin, the whole structure depends only on the mechanical bonding between fiber and resin. In that case, the rougher surface helps mechanical bonding between the fibers and between fibers and the resin. The resin can cling onto the rougher surfaces better, and the fibers can cling onto each other better when the surfaces are rougher. This reason played a big role in the case of the slightly better mechanical properties of sugarcane bagasse reinforced composite than the banana composite.
Another reason may be the possible intermolecular bonding of the matrix and the cohesive nature of the fiber. Long fibers can withstand more strain than shorter ones in terms of tensile properties. When longer fibers come closer, those can absorb the pulls together so that the material could show comparatively better tensile properties. But shorter fibers also have their advantages too. Shorter fibers can ensure more uniform mixing with the matrix. It helps the matrix material to form bonds between its molecules through accumulated fibers. But it becomes difficult sooner than in the case of longer fibers because shorter fibers can cover an area uniformly without leaving any gaps in between. In contrast, longer fibers can never be arranged in a more uniform nature than shorter fibers, which allows the matrix molecules to penetrate in between fibers and form intermolecular bonds. This amount of gaps also run out with an increasing amount of fiber. And when that happens, the tensile properties start to fall in the downward direction. Longer fibers make the materials comparatively more rigid too. That's why nanocellulose has shown way better tensile properties and flexural properties than fibers of micro or higher length [58]. Nanocellulose has advantages such as low thermal expansion coefficient, high aspect ratio, better tensile strength, and good mechanical properties [49]. In fact, the mechanical properties depend heavily on the size of the fiber. Generally, longer fibers can result in better tensile properties and flexural properties than shorter fibers. But shorter fibers have the advantage of a better degree of orientation in the composite, which makes the composite more uniform in terms of strength and more suitable for practical applications [96]. The pretreated fibers are mainly of high aspect ratio (aspect ratio is the ratio of diameter to length), which can not improve the mechanical properties of the composites generally. Longer fibers can absorb more stress and undergo more strain than the shorter fibers, resulting in better mechanical properties of the composites more often. While Short fibers present more ends where the possibility of cracking increases due to higher distance between fiber to fiber, it reduces cohesion between the fibers, but when the fiber is too short, that is within micro or nano scale, the distance between the ends is reduced, the cohesion between fibers is not affected, and the uniformity of the fibers within the matrix gives better mechanical properties [97]. That's why nanocellulose can improve mechanical properties to a higher degree than other smaller-sized fibers. Figure 9 [10, 11, 71] below can show the comparison between some starch composites reinforced by cellulose of different forms extracted from sugarcane bagasse.
Figure 9.
Changes in tensile properties with different forms of fiber loading.
The above comparison showed the significant improvement of tensile strength of starch composite when nanocellulose was used. At the same time, the tensile modulus also improved while the elongation property went the opposite way when the nanocellulose was used. Another advantage can be achieved by using nanocellulose water barrier properties, which are affected by the orientation and pores in between fibers in the structure. These are significantly affected by nanocellulose as nano particles have a higher degree of orientation and, at the same time it offers dense and least porous structure [11].
So, according to the above-mentioned and explained points, removal of lignin, hemicellulose can help the bonding process between fiber and matrix more chemically and mechanically. It helps chemically when the matrix and fibers are of similar chemical structure (such as starch, PLA, etc., with the cellulose of bagasse fiber) [84]. The mechanical bonds are created due to the resulted rough surface of the fibers after the removal of lignin, hemicellulose, and other contents [98]. This was shown in scanning electron microscopic images by Guedes de Paiva et al. in their study [99]. This helps more in the case of the synthetic matrix. Boontima et al. [100] and Khoo and Chow [101] reported significant improvement of mechanical properties when alkali-treated bagasse fiber was used to reinforce PLA composites.
On the other hand, Subramonian et al. [92] reported that there could never be any chemical bonding between cellulosic bagasse and polymers other than natural polymers that are mentioned above. Alkali treatment removes lignin and hemicellulose, which make the fibers softer and rougher. More fibers can be accumulated when fibers become softer, and it becomes relatively more straightforward for the matrix material to form bonds within itself in between the fibers. But when the quantity of fiber is increased, the gaps in between fibers are decreased. That restricts the bonding between matrix molecules in between the fibers. It caused gradual rigidity and decrement in the tensile properties of the composite. Another reason behind this trend is the different nature of fiber and matrix when the synthetic matrix is used with natural fibers. Natural fibers are hydrophilic in nature, while synthetic matrixes are generally hydrophobic. It is quite impossible to form and the bond between these two polymers of totally opposite character [102, 103]. These concepts were proved by researchers, as mentioned in this section earlier.
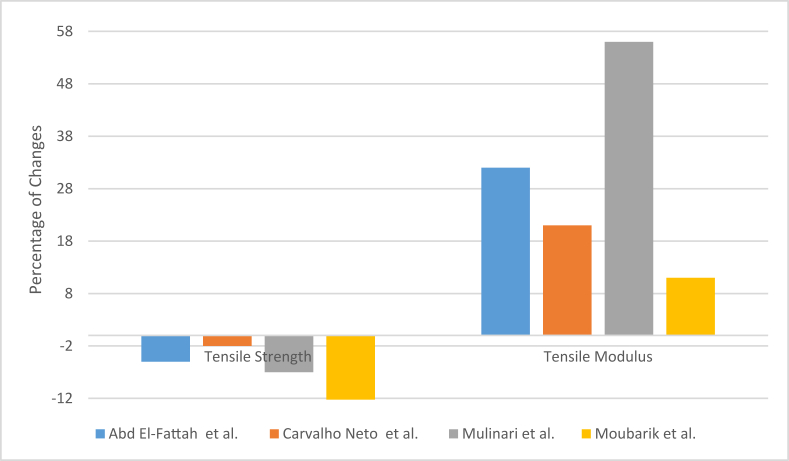
As a solution to the above-mentioned problems, surface modification of fiber using a coupling agent to make it more suited to the hydrophobic matrix can be used. For example, Abd El-Fattah et al. reported that using maleated polyethylene with bagasse fiber to reinforce high-density polyethylene resulted in less tensile strength than composites contain maleated polyethylene [104]. The tensile strength of high-density polyethylene decreased by 27% in the case of 10% fiber loading, but when maleated polyethylene was added, the tensile strength increased by 18% from the reduced value. Carvalho Neto et al. used mercerization (using sodium hydroxide) and acetylation (using acetic anhydride) of bagasse fibers for surface modification [94]. The fibers were used to reinforce the polyethylene sheet by injection molding method. The resulting material showed minimal loss of strength due to fiber loading, but the tensile properties did not improve. Flexural and impact properties were improved while the material became rigid gradually. Mulinari et al. used zirconium oxychloride as the coupling agent with bagasse fiber for high-density polyethylene composite [77]. The addition of fiber reduced the tensile strength of the material by 14%. But the application of zirconium oxide improved the property by 8% from the previous value. But still, the tensile strength was less than the original strength of the matrix. And as in other studies, the composite became stiffer than before. According to the study of Moubarik et al., alkali treatment and bleaching of the bagasse fibers can also follow similar trends, but to a lesser extent, as the decrement in tensile strength was found quite high in comparison to the other studies stated above [79]. But softer and flexible fibers resulted in comparatively lesser stiffness in the composite. Changes in some common mechanical properties in different studies discussed above are shown in Figure 10 below (for 10% fiber loading) [77, 79, 94, 104].
Figure 10.
Changes in mechanical properties with fiber loading.
All these modifications were required because of the hydrophilic nature and the presence of hydroxyl functional group, which act as the main functional group of the cellulose molecule. The hydroxyl groups have a tendency to form hydrogen bonds with adjacent oxygen and hydrogen. Water molecules can get attached to those groups easily by the formation of hydrogen bonds. That's why cellulosic materials have higher moisture regain than any materials. Also, these hydroxyl groups are very often found unable to form new bonds with other functional groups other than the weak hydrogen bonds. The lack of bonding is also a result of the inability of cellulose to withstand too high temperature, which is essential for the formation of most of the bonds. At any temperature, over 150 °C can damage cellulose [81]. For these reasons, the best options for making the cellulose less hydrophilic or engaging those hydroxyl groups to new bond formation or to form any bond with the resin (which is the most difficult process, and in most cases, it is not possible), surface treatment of cellulose or using coupling agents are the easiest and most suitable options most often. The researches mentioned above fall into these categories. Abd El-Fattah et al. used maleated polyethylene as a coupling agent to reduce the fall of tensile strength where the maleted polyethylene acted as the bridge between cellulose and polyethylene by forming bonds with both molecules [104]. Maleted polyethylene contains single-bonded oxygen in the structure, which can participate in the formation of hydrogen bonds with cellulose, while the methyl groups of maleted polyethylene can form covalent bonds with the high-density polyethylene. This system proved quite effective in terms of achieving better tensile properties than before. Mulinari et al. also used a similar system as they applied as a coupling agent in the form of zirconium oxychloride, and they also obtained similar results [77].
Carvalho Neto et al. used mercerization by alkali to remove the lignin and hemicellulose from the bagasse and to produce pure cellulose, while the acetylation by acetic anhydride of the attained cellulose acted as the esterification process of cellulose, which replaced the hydroxyl groups of the cellulose by the acetyl groups [94]. This acetylation made the cellulose hydrophobic in nature as the hydroxyl functional group responsible for the hydrophilic nature of cellulose was replaced in this system. This method was found even more effective when these new materials were used to reinforce polyethylene as both components were of similar hydrophobic characteristics after surface modification.
6. Key findings
Even though all these researches have been done using different materials, different techniques, and various types of machinery. But still, some common trends can be found in these studies. In summary, the results of the assessment done here on several studies are given below.
-
•
Bagasse fibers are not superior in terms of mechanical properties, but their availability and versatile application can make them a handy material, especially in composite industries.
-
•
These fibers can be used in any length, from the nano to the milliliter scale. Different length or size of fiber offers additional features to the final material. But smaller size has been found as a preferable option for the researchers because it is easier to produce a uniform mixture when using shorter fibers, especially fibers within the size range of the nano scale. Better mixing is more important while being employed with hydrophobic matrix material because the hydrophilic nature of the bagasse fiber refrains it from forming bonds with the matrix molecules of the completely opposite character.
-
•
Surface modification of fiber or the use of a coupling agent can help reduce the hydrophilic nature of the fibers and make the fibers compatible to use with any type of polymer. There are many types of surface modification agents available and being used by researchers [30]. But among the studies discussed here, acetylation using acetic anhydride has been found as the most effective one. This modification causes the esterification of cellulose, which is quite a common and useful procedure for reducing the hydrophilic nature of polysaccharides [105]. But still, the addition of fiber may not improve the tensile properties of the material much, but tensile modulus, impact strength, and flexural properties have been improved in most cases.
-
•
Pre-treatment can help to improve mechanical bonding between bagasse fibers and matrix molecules by making the surface of the fibers rougher.
7. Conclusion
Sugarcane bagasse is an excellent source of cellulose as it is a waste material, and any reuse of these materials would be helpful. But its applications are not limited to only paper industries. Bagasse fibers are a source of cellulose that can be used for various applications. The structure of sugarcane bagasse is a bit different than other cellulosic fibers. It has a rind part, which is quite strong, but the inner pith part contains weaker, smaller fibers. That is why it cannot be used easily in its raw state. To extract fibers or pure cellulose or nanocrystals or nanofibers all need different techniques. Some techniques have been summarised in this paper. The applications of fibers, cellulose, etc., have also been discussed in this paper briefly. Some common factors that can affect the properties of bagasse reinforced composite materials have been summarised at the end of the review after assessing the production processes and properties of the end products. The diverse field of application includes both fully and partially biodegradable materials that match the currently ongoing trend of making a substitute for synthetic materials. So, sugarcane bagasse can be an excellent source or raw material for making environmentally friendly products.
Declarations
Author contribution statement
All authors listed have significantly contributed to the development and the writing of this article.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability statement
Data included in article/supplementary material/referenced in article.
Declaration of interests statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
References
- 1.FAOSTAT . 2018. Food and Agriculture Organization of the United Nations.http://www.fao.org/faostat/en/#data/QC [Online]. Available: [Google Scholar]
- 2.Michel D., Bachelier B., Drean J.-Y., Harzallah O. Conference Papers in Materials Science. Guimar˜ aes; 2013. Preparation of cellulosic fibers from sugarcane for textile use. [Google Scholar]
- 3.Chandel A.K., Silva S.S.d., Carvalho W., Singh O.V. Sugarcane bagasse and leaves: foreseeable biomass of biofuel and bio-products. Chem. Technol. Biotechnol. 2012;87:11–20. [Google Scholar]
- 4.Cerqueira D.A., Filho G.R., Meireles C.d.S. Optimization of sugarcane bagasse cellulose acetylation. Carbohydr. Polym. 2007;69:579–582. [Google Scholar]
- 5.Pandey A., Soccol C.R., Nigam P., Soccol V.T. Biotechnological potential of agro-industrial residues. I: sugarcane bagasse. Biores. Technol. 2000;74:69–80. [Google Scholar]
- 6.Hernández-Salas J., Villa-Ramı´rez M.S., Veloz-Rendón J.S., Rivera-Hernández K.N., González-César R.A., Plascencia-Espinosa M.A., Trejo-Estrada S.R. Comparative hydrolysis and fermentation of sugarcane and agave bagasse. Biores. Technol. 2009;100:1238–1245. doi: 10.1016/j.biortech.2006.09.062. [DOI] [PubMed] [Google Scholar]
- 7.Loh Y.R., Sujan D., Rahman M.E., Das C.A. Sugarcane bagasse—the future composite material: a literature review. Res. Cons. Recycl. 2013;75:14–22. [Google Scholar]
- 8.Sn W. Proceedings of the South African Sugar Technologists Association, Durban. 2008. Sugarcane bagasse: how easy is it to measure its constituents? [Google Scholar]
- 9.Costa S.M., Mazzola P.G., Silva J.C.A.R., Pahl R., Pessoa A., Jr., Costa S.A. Use of sugar cane straw as a source of cellulose for textile fiber production. Ind. Crops Prod. 2013;42:189–194. [Google Scholar]
- 10.Gilfillan W.N., Nguyen D.M.T., Sopade P.A., Doherty W.O.S. Preparation and characterisation of composites from starch and sugar cane fibre. Ind. Crops Prod. 2012;40:45–54. [Google Scholar]
- 11.Slavutsky A.M., Bertuzzi M.A. Water barrier properties of starch films reinforced with cellulose nanocrystals obtained from sugarcane bagasse. Carbohydr. Polym. 2014;110:53–61. doi: 10.1016/j.carbpol.2014.03.049. [DOI] [PubMed] [Google Scholar]
- 12.Collier B.J., Collier J.R., Agarwal P., Lo Y.-W. Extraction and evaluation of fibers from sugar cane. Text. Res. J. 1992;62:741–748. [Google Scholar]
- 13.TAPPI . TAPPI; Norcross: 2015. T 10 Wd- 78 (T10 Ts-47), Species Identification of Nonwoody Vegetable Fibers. [Google Scholar]
- 14.van-Dillewijn C. Chronica Botanica Co; Waltham: 1952. Botany of Sugarcane. [Google Scholar]
- 15.Jacobsen S.E., Wyman C.E. Xylose monomer and oligomer yields for uncatalyzed hydrolysis of sugarcane bagasse hemicellulose at varying solids concentration. Ind. Eng. Chem. Res. 2002;41:1454–1461. [Google Scholar]
- 16.Wyman C.E. Biomass ethanol: technical progress, opportunities, and commercial challenges. Ann. Rev. Energy Environ. 1999;24:189–226. [Google Scholar]
- 17.Sun J.X., Zhao X.F.S.H., Sun R.C. Isolation and characterization of cellulose from sugarcane bagasse. Polym. Degrad. Stab. 2004;84:331–339. [Google Scholar]
- 18.Jacobsen S.E., Wyman C.E. Cellulose and hemicellulose hydrolysis models for application to current and novel pretreatment processes. Appl. Biochem. Biotechnol. 2000;81–96:84. doi: 10.1385/abab:84-86:1-9:81. [DOI] [PubMed] [Google Scholar]
- 19.Ding S.Y., Himmel M.E. The maize primary cell wall microfibril: a new model derived from direct visualization. J. Agric. Food Chem. 2006;597–606:54. doi: 10.1021/jf051851z. [DOI] [PubMed] [Google Scholar]
- 20.Kadla J.F., Gilbert R.D. Cellulose structure: a review. Cell. Chem. Technol. 2000;34:197–216. [Google Scholar]
- 21.Amen-Chen C., Pakdel H., Roy C. Production of monomeric phenols by thermochemical conversion of biomass: a review. Biores.Technol. 2001;79:277–299. doi: 10.1016/s0960-8524(00)00180-2. [DOI] [PubMed] [Google Scholar]
- 22.Bon E. FAO Symposium on “The Role of Agricultural Biotechnologies for Production the Role of Agricultural Biotechnologies for Production of Bio-Energy. 2007. Ethanol production via enzymatic hydrolysis of sugarcane bagasse and straw. [Google Scholar]
- 23.Paturau J.M. Elsevier; Amsterdam: 1989. By-products of the Cane Sugar Industry. [Google Scholar]
- 24.Trickett R.C., Neytzell F.G.d.-W. Bagasse heicellulose acid hydrolysis and residue treatment prior to enzymatic hydrolys of cellulose. South Afr. Food Rev. 1982;9:95. [Google Scholar]
- 25.Han Y.W., Catalano E.A., Ciegler A. Chemical and physical properties of sugarcane bagasse irradiated with gamma rays. Journal Agric. Food Chem. 1983;31:34–38. [Google Scholar]
- 26.Institute S.R. Editorial Cient´ıfico-Técnica; Havana: 1980. The Industry of Sugarcane Derivatives. [Google Scholar]
- 27.Saelee K., Yingkamhaeng N., Nimchua T., Sukyai P. The 26 th Annual Meeting of the Thai Society for Biotechnology and International Conference, Chiang Rai. 2014. Extraction and characterization of cellulose from sugarcane bagasse by using environmental friendly method. [Google Scholar]
- 28.Kozłowski R., Władyka-Przybylak M. Flammability and fire resistance of composites reinforced by natural fibers. Polym. Adv. Technol. 2008;19:446–453. [Google Scholar]
- 29.Kozlowski R., Wladyka-Przybylak M. Uses of natural fiber reinforced plastics. In: Wallenberger F.T., Weston N.E., editors. Natural Fibers, Plastics and Composites. Springer; Boston: 2004. pp. 249–274. [Google Scholar]
- 30.Anannya F., Mahmud M.A. Developments in flame-retardant bio-composite material production. Adv. Civil Eng. Mat. 2019;8:9–22. [Google Scholar]
- 31.Bunsell A.R., editor. Handbook of Properties of Textile and Technical Fibres. second ed. Woodhead Publishing; Kidlington: 2018. [Google Scholar]
- 32.Hari P.K. Woven Textiles: Principles, Developments and Applications. Woodhead Publishing Limited; Cambridge: 2012. Types and properties of fibres and yarns used in weaving; pp. 3–34. [Google Scholar]
- 33.Batra S.K. Other long vegetable fibers: abaca, banana, sisal, henequen, flax, hemp, sunn, and coir. In: Lewin M., Pearce E.M., editors. Vol. 4. Marcel Dekker; New York: 1983. pp. 727–807. (Handbook of Fiber Science and Technology). [Google Scholar]
- 34.Lewin M. The middle lamella of the bast fibers. TAPPI. 1958;41:403–415. [Google Scholar]
- 35.Msahli S., Drean J.Y., Sakli F. Evaluating the fineness of agave Americana L. fibers. Text. Res. J. 2005;75:540–543. [Google Scholar]
- 36.Bunsell A.R., editor. Handbook of Tensile Properties of Textile and Technical Fibers. Woodhead Publishing Series in Textiles; Cambridge: 2009. [Google Scholar]
- 37.Harris M. Harris Research Laboratories; Washington DC: 1954. Handbook of Textile Fiber. [Google Scholar]
- 38.Meredit R. 1969. Proceedings of Fifth International Congress on Rheology, Tokyo. [Google Scholar]
- 39.Lewin M. third ed. Taylor & Francis Group; Boca Raton: 2007. Handbook of Fiber Chemistry. [Google Scholar]
- 40.Chiparus O.I., Chen Y. An image method to evaluate bagasse fiber dimensions. Biores. Technol. 2003;90:305–309. doi: 10.1016/s0960-8524(03)00128-7. [DOI] [PubMed] [Google Scholar]
- 41.Cao Y., Shibata S., Fukumoto I. Mechanical properties of biodegradable composites reinforced with bagasse fibre before and after alkali treatments. Comp.: Part A. 2006;37:423–429. [Google Scholar]
- 42.Liu C.F., Ren J.L., Xu F., Liu J.J., Sun J.X., Sun R.C. Isolation and characterization of cellulose obtained from ultrasonic irradiated sugarcane bagasse. J. Agric. Food Chem. 2006;54:5742–5748. doi: 10.1021/jf060929o. [DOI] [PubMed] [Google Scholar]
- 43.Yang Y.Q., Reddy Structure and properties of high quality natural cellulose fibers from cornstalk. Polymer. 2005;46:5494–5500. [Google Scholar]
- 44.Abdel-Halim E. Chemical modification of cellulose extracted from sugarcane bagasse: preparation of hydroxyethyl cellulose. Arab. J. Chem. 2014;7:362–371. [Google Scholar]
- 45.Rocha G.J.M., Gonçalves A.R., Oliveira B.R., Olivares E.G., Rossell C.E.V. Steam explosion pretreatment reproduction and alkaline delignification reactions performed on a pilot scale with sugarcane bagasse for bioethanol production. Ind. Crops Prod. 2012;35:274–279. [Google Scholar]
- 46.Shaikh H.M., Pandare K.V., Nair G., Varma A.J. Utilization of sugarcane bagasse cellulose for producing cellulose acetates:Novel use of residual hemicellulose as plasticizer. Carbohydr. Polym. 2009;75:23–29. [Google Scholar]
- 47.Costa S.M., Gonçalves A.R., Esposito E. Ceriporiopsis subvermispora used indelignification of sugarcane bagasse prior soda/anthraquinone pulping. Appl. Biochem. Biotechnol. 2005;122:695–706. doi: 10.1385/abab:122:1-3:0695. [DOI] [PubMed] [Google Scholar]
- 48.Fink H.P., Weigel P., JPurz H., Ganster J. Structure formation of regenerated cellulose materials from NMMO-solutions. Progr. Polym. Sci. 2001;26:1473–1524. [Google Scholar]
- 49.Sharma A., Thakur M., Bhattacharya M., Mandal T., Goswami S. Commercial application of cellulose nano-composites – a review. Biotechnol. Rep. 2019;21 doi: 10.1016/j.btre.2019.e00316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mandal A., Chakrabarty D. Isolation of nanocellulose from waste sugarcane bagasse (SCB) and its characterization. Carbohydr. Polym. 2011;86:1291–1299. [Google Scholar]
- 51.Kumar A., Negi Y.S., Choudhary V., Bhardwaj N.K. Characterization of cellulose nanocrystals produced by acid-hydrolysis from sugarcane bagasse as agro-waste. J. Mater. Phys. Chem. 2014;2:1–8. [Google Scholar]
- 52.Beck-Candanedo S., Roman M., Gray D.G. Effect of reaction conditions on the properties and behavior of wood cellulose nanocrystal suspensions. Biomacromolecules. 2005;6:1048–1054. doi: 10.1021/bm049300p. [DOI] [PubMed] [Google Scholar]
- 53.Bondeson D., Mathew A., Oksman K. Optimization of the isolation of nanocrystals from microcrystalline cellulose by acid hydrolysis. Cellulose. 2006;13:171–180. [Google Scholar]
- 54.de-Oliveira F.B., Bras J., Pimenta M.T.B., da-Silva Curvelo A.A., Belgacem M.N. Production of cellulose nanocrystals from sugarcane bagasse fibers and pith. Ind. Crops Prod. 2016;93:48–57. [Google Scholar]
- 55.Siqueira G., Abdillahi H., Bras J., Dufresne A. High reinforcing capability cellulose nanocrystals extracted from Syngonanthus nitens (Capim Dourado) Cellulose. 2010;17:289–298. [Google Scholar]
- 56.Teixeira E.M., Bondancia T.J., Teodoro K.B.R., Corrêa A.C., Marconcini J.M., Mattoso L.H.C. Sugarcane bagasse whiskers: extraction and characterizations. Ind. Crops Prod. 2011;33:63–66. [Google Scholar]
- 57.Li J., Wei X., Wang Q., Chen J., Chang G., Kong L., Su J., Liu Y. Homogeneous isolation of nanocellulose from sugarcane bagasse by high pressure homogenization. Carbohydr. Polym. 2012;90:1609–1613. doi: 10.1016/j.carbpol.2012.07.038. [DOI] [PubMed] [Google Scholar]
- 58.Ghaderi M., Mousavi M., Yousefi H., Labbafi M. All-cellulose nanocomposite film made from bagasse cellulose nanofibers for food packaging application. Carbohydr. Polym. 2014;104:59–65. doi: 10.1016/j.carbpol.2014.01.013. [DOI] [PubMed] [Google Scholar]
- 59.Zhang K., Sun P., Liu H., Shang S., Song J., Wang D. Extraction and comparison of carboxylated cellulose nanocrystals from bleached sugarcane bagasse pulp using two different oxidation methods. Carbohydr. Polym. 2016;138:237–243. doi: 10.1016/j.carbpol.2015.11.038. [DOI] [PubMed] [Google Scholar]
- 60.El-Miri N., Abdelouahdi K., Barakat A., Zahouily M., Fihri A., Solhy A., El-Achaby M. Bio-nanocomposite films reinforced with cellulose nanocrystals: rheology of film-forming solutions, transparency, water vapor barrier and tensile properties of films. Carbohydr. Polym. 2015;129:156–167. doi: 10.1016/j.carbpol.2015.04.051. [DOI] [PubMed] [Google Scholar]
- 61.Ferreira F.V., Mariano M., Rabelo S.C., Gouveia R.F., Lona L.M.F. Isolation and surface modification of cellulose nanocrystals fromsugarcanebagasse waste: from a micro- to a nano-scale view. Appl. Surf. Sci. 2018;436:1113–1122. [Google Scholar]
- 62.Rahimi Kord Sofla M., Brown R.J., Tsuzuki T., Rainey T.J. A comparison of cellulose nanocrystals andcellulose nanofibres extracted from bagasseusing acid and ball milling methods. Adv. Nat. Sci. Nanosci. Nanotechnol. 2016;7:1–9. [Google Scholar]
- 63.Feng Y.-H., Cheng T.-Y., Yang W.-G., Ma P.-T., He H.-Z., Yin X.-C., Yu X.-X. Characteristics and environmentally friendly extraction of cellulose nanofibrils from sugarcane bagasse. Ind. Crops Prod. 2018;111:285–291. [Google Scholar]
- 64.Kathiresan M., Sivaraj P. Preparation and characterization of biodegradable sugarcane bagasse nano reinforcement for polymer composites using ball milling operation. Int. J. Polym. Analy. Char. 2016;21:428–435. [Google Scholar]
- 65.Mahmud M.A., Anannya F.R. Jute in geotextiles. Tekstilec. 2018;61:34–40. [Google Scholar]
- 66.Franck R.R., editor. Bast and Other Plant Fibres. Woodhead Publishing Limited; Cambridge: 2000. [Google Scholar]
- 67.Jarman C. ITDG Publishing; 1998. Plant Fibre Processing: A Handbook. [Google Scholar]
- 68.Luniak B. Sir Isaac Pitman & Sons; London: 1953. The Identification of Textile Fibers. [Google Scholar]
- 69.Patni P.C., Shakyawarb D.B., Gupta N.P. Studies on animal fiber blended hand made felts: Part 1-Physical and mechanical properties. Ind. J. Fiber Text. Res. 2006;31:553–558. [Google Scholar]
- 70.Rankilor P.R. Textiles in civil engineering. Part 1 – geotextiles. In: Horrocks A.R., Anand S.C., editors. Handbook of Technical Textiles. Woodhead Publishing Limited; Cambridge: 2000. pp. 358–406. [Google Scholar]
- 71.Vallejos M.E., Curvelo A.A.S., Teixeira E.M., Mendes F.M., Carvalho A.J.F., Felissia F.E., Area M.C. Composite materials of thermoplastic starch and fibers from the ethanol–water fractionation of bagasse. Ind. Crops Prod. 2011;33:739–746. [Google Scholar]
- 72.Cerqueira E.F., Baptista C.A.R.P., Mulinari D.R. Mechanical behaviour of polypropylene reinforced sugarcane bagasse fibers composites. Proc. Eng. 2011;10:2046–2051. [Google Scholar]
- 73.Arrakhiz F.Z., Malha M., Bouhfid R., Benmoussa K., Qaiss A. Tensile, flexural and torsional properties of chemically treated alfa, coir and bagasse reinforced polypropylene. Comp. Part B: Eng. 2013;47:35–41. [Google Scholar]
- 74.Jeefferie A., Fariha O.N., Warikh A.R.M., Yuhazri M.Y., Sihombing H., Ramli J. Preliminary study on the physical and mechanical properties of tapioca starch/sugarcanefiber cellulose composite. ARPN J. Eng. Appl. Sci. 2011;6:7–15. [Google Scholar]
- 75.Monteiro S.N., Rodriquez R.J.S., Souza M.V.D., D’Almedia J.R.M. Sugarcane bagasse waste as reinforcement in low cost composites. Adv. Perf. Mat. 1998;5:183–191. [Google Scholar]
- 76.Stael G.C., Tavares M.I.B., D’almeida J.R.M. Evaluation of sugar cane bagasse waste asreinforcement in EVA matrix composite materials. Polym. Plast. Technol. Eng. 2001;40:217–223. [Google Scholar]
- 77.Mulinari D.R., Voorwald H.J.C., Cioffi M.O.H., da-Silva M.L.C.P., da-Cruz T.G., Saron C. Sugarcane bagasse cellulose/HDPE composites obtained by extrusion. Comp. Sci. Technol. 2009;69:214–219. [Google Scholar]
- 78.Oladele I.O. Effect of bagasse fibre reinforcement on the mechanical properties of polyester composites. J. Assoc. Prof. Eng. Trinidad Tobago. 2014;42:12–15. [Google Scholar]
- 79.Moubarik A., Grimi N., Boussetta N. Structural and thermal characterization of Moroccan sugar cane bagasse cellulose fibers and their applications as a reinforcing agent in low density polyethylene. Comp. Part B: Eng. 2013;52:233–238. [Google Scholar]
- 80.Monteiro S.N., Candido V.S., Braga F.O., Bolzana L.T., Weber R.P., Drelich J.W. Sugarcane bagasse waste in composites for multilayered armor. Eur. Polym. J. 2016;78:173–185. [Google Scholar]
- 81.Mahmud M.A. Development of modified starch coated jute fabric. J. Inst. Eng. (India): Series E. 2018;99:149–156. [Google Scholar]
- 82.Guimarães J.L., Wypych F., Saul C.K., Ramos L.P., Satyanarayana K.G. Studies of the processing and characterization of corn starch and its compositeswith banana and sugarcane fibers from Brazil. Carbohydr. Polym. 2010;80:130–138. [Google Scholar]
- 83.Ghaderi M., Mousavi M., Yousefi H., Labbafi M. All-cellulose nanocomposite film made from bagasse cellulose nanofibers for food packaging application. Carbohydr. Polym. 2014;10:59–65. doi: 10.1016/j.carbpol.2014.01.013. [DOI] [PubMed] [Google Scholar]
- 84.Achaby M.E., Miri N.E., Aboulkas A., Zahouily M., Bilal E., Barakat A., AbderrahimSolhy Processing and properties of eco-friendly bio-nanocomposite films filled with cellulose nanocrystals from sugarcane bagasse. Int. J. Biol. Macromolec. 2017;96:340–352. doi: 10.1016/j.ijbiomac.2016.12.040. [DOI] [PubMed] [Google Scholar]
- 85.Gan I., Chow W.S. Synthesis of phosphoric acid-treated sugarcane bagasse cellulose nanocrystal and its thermal properties enhancement for poly(lactic acid) nanocomposites. J. Thermoplast. Comp. Mat. 2019;32:619–634. [Google Scholar]
- 86.Farha F.I., Iqbal S.M.F., Mahmud M.A. Compositional and structural influence on some weft knitted fabrics comprised of cotton and lyocell yarn. J.Inst. Eng. (India): Series E. 19 March 2019:1–9. [Google Scholar]
- 87.Candido R.G., Godoy G.G., Adilson G.R. Characterization and application of cellulose acetate synthesized from sugarcane bagasse. Carbohydr. Polym. 2017;167:280–289. doi: 10.1016/j.carbpol.2017.03.057. [DOI] [PubMed] [Google Scholar]
- 88.AZoM . 12 June 2002. AZO Materials.www.azom.comhttps://www.azom.com/properties.aspx?ArticleID=1461 [Online]. Available: [Google Scholar]
- 89.Silvério H.A., Neto W.P.F., Dantas N.O., Pasquini D. Extraction and characterization of cellulose nanocrystals from corncob for application as reinforcing agent in nanocomposites. Ind. Crops Prod. 2013;44:427–436. [Google Scholar]
- 90.Amin M.R., Mahmud M.A., Anannya F.R. Natural fiber reinforced starch based biocomposites. Polym. Sci. Series A. 2019;61:533–543. [Google Scholar]
- 91.Cruz-Tirado J.P., Tapia-Blácido D.R., Siche R. Materials Science and Engineering, Telangana. 2017. Influence of proportion and size of sugarcane bagasse fiber on the properties of sweet potato starch foams. [Google Scholar]
- 92.Subramonian S., Ali A., Amran M., Sivakumar L.D., Salleh S., Rajaizam A. Effect of fiber loading on the mechanical properties of bagasse fiber–reinforced polypropylene composites. Adv. Mech. Eng. 2016;8:1–5. [Google Scholar]
- 93.Candido V.S., Silva A.C. R.d., Simonassi N.T., Luz F.S.d., Monteiro S.N. Toughness of polyester matrix composites reinforced with sugarcane bagasse fibers evaluated by Charpy impact tests. J. Mater. Res. Technol. 2017;6:334–338. [Google Scholar]
- 94.Neto A.G. V.d.C., Ganzerli T.A., Cardozo A.L., Fávaro S.L., Pereira A.G.B., Girotto E.M., Radovanovic E. Development of composites based on recycled polyethylene/sugarcane bagasse fibers. Polym. Comp. 2014;35:768–774. [Google Scholar]
- 95.Gupta U.S., Dhamarikar M., Dharkar A., Chaturvedi S., Tiwari S., Namdeo R. Surface modification of banana fiber: a review. Mat. Today: Proc. 2021;43:904–915. [Google Scholar]
- 96.Sathishkumar T.P., Navaneethakrishnan P., Shankar S. Tensile and flexural properties of snake grass natural fiber reinforced. Comp. Sci. Technol. 2012;72:1183–1190. [Google Scholar]
- 97.Al-Maharma A.Y., Sendur P. Review of the main factors controlling the fracture toughness and impact strength properties of natural composites. Mat. Res. Expr. 2018;6:1–33. [Google Scholar]
- 98.Nam T.H., Ogihara S., Tung N.H., Kobayashi S. Effect of alkali treatment on interfacial and mechanical properties of coir fiber reinforced poly(butylene succinate) biodegradable composites. Comp. Part B: Eng. 2011;42:1648–1656. [Google Scholar]
- 99.Paiva F.F. G.d., Maria V.P. K.d., Torres G.B., Dognani G., Santos R.J.d., Cabrera F.C., Job A.E. Sugarcane bagasse fiber as semi-reinforcement filler in natural rubber composite sandals. J. Mater. Cycles Waste Manag. 2019;21:326–335. [Google Scholar]
- 100.Boontima B., Noomhorm A., Puttanlek C., Uttapap D., Rungsardthong V. Mechanical properties of sugarcane bagasse fiber-reinforced soy based biocomposites. J. Polym. Environ. 2015;23:97–106. [Google Scholar]
- 101.Khoo R.Z., Chow W.S. Mechanical and thermal properties of poly(lactic acid)/sugarcane bagasse fiber green composites. J. Thermoplast. Comp. Mat. 2015;30:1091–1102. [Google Scholar]
- 102.Mwasha A. Coir fibre: a sustainable engineering material for the caribbean islands. Int. J. Bahamian Stud. 2009;15:36. [Google Scholar]
- 103.Brahmakumar M., Pavithran C., Pillai R.M. Coconut fibre reinforced polyethylene composites: effect of natural waxy surface layer of the fibre on fibre/matrix interfacial bonding and strength of composites. Comp. Sci. Technol. 2005;65:563–569. [Google Scholar]
- 104.El-Fattah A.A., Demerdash A.G.M.E., Sadik W.A.A., Bedir A. The effect of sugarcane bagasse fiber on the properties of recycled high density polyethylene. J. Comp. Mat. 2015;49:3251–3262. [Google Scholar]
- 105.Amin M.R., Anannya F.R., Mahmud M.A., Raian S. Esterification of starch in search of a biodegradable thermoplastic material. J. Polym. Res. 2020;27:1–12. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data included in article/supplementary material/referenced in article.