Graphical Abstract
Graphical Abstract.
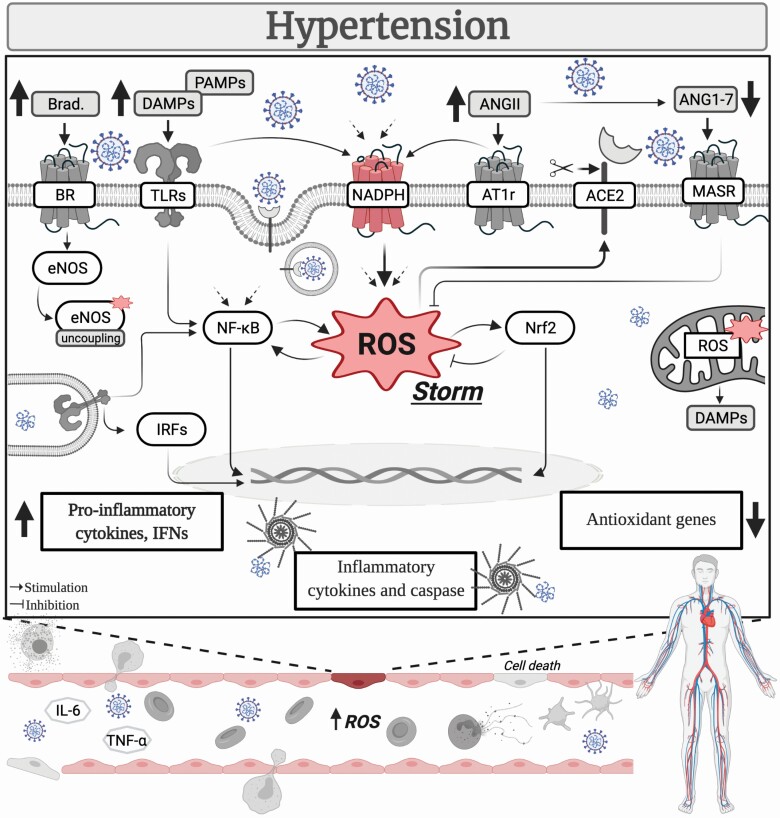
As of 12 May 2021, the Center for Disease Control data tracker reports that more than 32 million Americans were infected with the SARS-CoV-2 virus. The death toll of the ongoing COVID-19 pandemic has surpassed the mark of 580,000, and despite the recent availability of vaccines, it continues to increase on American soil, though at a lower rate. Unfortunately, the full impact of the disease is not yet reflected in many countries, such as Brazil and India, where all victims may not be fully accounted for, and which are battling the virus and witnessing a disproportional toll on their citizens. While our understanding of COVID-19 has drastically improved since its first case in Wuhan (Hubei Province, China), there is still a myriad of open questions regarding the pathophysiology of this disease. Cumulative evidence, however, suggests that patients with cardiometabolic diseases are at a greater risk of developing the severe form of COVID-19, but the specific contribution of each disrupted pathway is unclear. Previous studies highlighted the significance of a cytokine storm to the severity of SARS-CoV-2 infection, and more recently, the possibility of another storm, the bradykinin one, also contributing to the widespread tissue damage observed in some COVID-19 patients, involving those with hypertension comorbidity.1 Under these unprecedented conditions, there is yet, another equally relevant, still overlooked, storm on the forecast for hypertension: the reactive oxygen species (ROS) storm (Figure 1).
Figure 1.
COVID-19 and ROS storm in hypertension. The mechanisms triggering increased oxidative stress are pre-activated in hypertension. Consequently, the release of ROS is most likely intensified in the vasculature of COVID-19 hypertensive patients, giving rise to a dysregulated response, culminating in a ROS storm. This deranged response exacerbates the vascular damage, contributing to disease severity. Abbreviations: ACE2, angiotensin converting enzyme 2; AngII, angiotensin II; Ang(1–7), angiotensin (1–7); AT1r, angiotensin II receptor type 1; Brad., bradykinin; BR, bradykinin receptor; DAMPs, damage-associated molecular patterns; eNOS, endothelial nitric oxide synthase; IFNs, interferons; IL-6, interleukin 6; IRFs, interferon regulatory factor; MASR, MAS receptor; NADPH, nicotinamide adenine dinucleotide phosphate; Nrf2, nuclear factor erythroid 2–related factor 2; NF-κB, nuclear factor-κB; PAMPs, pathogen-associated molecular patterns; ROS, reactive oxygen species; TLRs, toll-like receptors; TNF-α, tumor necrosis factor-α.
ROS are signaling molecules that exert critical biological roles. Conversely, dysregulated ROS production, leading to oxidative stress, contributes to the pathophysiology of several diseases, including hypertension. While different mechanisms could trigger the generation of ROS, it is widely accepted that NADPH oxidase is a main source of these molecules, and not surprisingly, the expression levels of this enzyme are altered during hypertension.2 Additionally, NADPH oxidase could be involved in the host response against the SARS-CoV-2 virus. The circulating levels of soluble NOX2-derived peptide (s-NOX2-dp) are increased in COVID-19 patients, and accordingly, s-NOX2-dp levels are associated with disease severity. In fact, COVID-19 patients receiving intensive care have higher levels of s-NOX2-dp than nonsevere patients,3 but it remains undetermined whether hypertension, a common comorbidity in severe patients, further upregulates ROS levels. Although the relationship between ROS and hypertension per se is very complex, it is accepted that ROS crosstalk with signaling pathways that are known to affect blood pressure in major organs/systems, and high systolic blood pressure is an important predictor of COVID-19 outcome.4 In this sense, the vasculature has become central since it plays an important role in blood pressure regulation and is intrinsically implicated in the mechanisms following SARS-CoV-2 infection. There is growing evidence that COVID-19 is a vascular disease,5 which might shed some light on why hypertension could be associated with poorer outcomes in this previously considered to be only a respiratory disease. Consequently, uncovering hypertension-associated pathways linked to worse outcomes in COVID-19 patients is paramount, mainly because many hypertensive patients do not have the disease under control, leading to end-organ damage, and likely, an adverse prognostic during SARS-CoV-2 infection,6 highlighting the depth of this problem.
The deranged pathways of hypertension partly occur because of excessive ROS production. Vascular NADPH oxidase-derived ROS in response to overstimulation by angiotensin II (AngII), a vasoconstrictor peptide of the renin–angiotensin system, interacts with nitric oxide (NO), stimulates MAPKs (e.g., ERK1/2 and p38), activates the transcriptional factor nuclear factor-κB to induce the release of proinflammatory mediators, modulates proapoptotic pathways, and triggers lipid peroxidation, to name a few.2,7 Unwanted transfer of signals is common between these pathways and is thought to be critical for disease progression. Moreover, disproportionate NADPH oxidase-induced ROS can lead to endothelial nitric oxide synthase uncoupling, xanthine oxidase activation, and mitochondrial dysfunction, further increasing ROS levels.2,7 Mitochondrial damage in response to augmented ROS might also trigger the accumulation of mitochondrial double-stranded RNA and consequently release of self-nucleic material from this organelle. Since mitochondrial double-stranded RNA can act as a damage-associated molecular pattern and activate innate immune receptors, such as toll-like receptors, this process has been implicated in the development of hypertension,8 and subsequently, in a positive feedback loop, it would augment the release of ROS in the vasculature. Noteworthy, dsRNA is produced during the SARS-CoV-2 replication process, which immune receptors might recognize, and in the case of TLR3, it would offer a mechanism to affect blood pressure, including in previously nonhypertensive patients,9 and vascular oxidative status. To aggravate the problem, COVID-19 also impairs the protective arm of the renin–angiotensin system. ACE2, which converts AngII into Ang(1–7), is used by the SARS-CoV-2 virus as its receptor, ultimately leading to a decrease in the activity of the enzyme.10 Upregulation of AngII, in response to reduced ACE2 levels, also could influence the latter’s availability via NADPH oxidase. AngII-derived ROS stimulates MAPK p38 triggering the activation of ADAM17 to actively shed the ACE2 ectodomain.10 Afterward, AngII accumulates, and its detrimental vascular effects prevail, which in the context of COVID-19 hypertensive patients, who already have a dysfunctional vasculature, exacerbates the vascular damage.
ROS production is already exacerbated during hypertension (i.e., the mechanisms triggering increased oxidative stress are pre-activated). In this sense, the release of ROS in COVID-19 hypertensive patients is most likely intensified, giving rise to a dysregulated response, culminating in a ROS storm. This outburst of ROS could be, in turn, a critical modulator of disease severity in hypertensive patients. Furthermore, the ROS storm probably crosstalk with other storms. The mechanisms leading to and/or sustaining the cytokine storm during COVID-19 also induce ROS generation. Immune system dysregulation, evidenced by activation and infiltration of immune cells and overdrive of the inflammasome and toll-like receptors in response to a surge in pathogen-associated molecular patterns and/or damage-associated molecular patterns, AngII overexpression, prothrombotic factor hyperactivation, immune cell infiltration, and ultimately, endothelial cell death, modulate ROS-generating systems. To complete the cycle, excessive oxidative stress reinforces the stimulation of these mediators. Similarly, an indirect interplay can also occur between ROS and the bradykinin storm in the hypertensive vasculature. There is increased release of NO downstream of bradykinin,1 and it is broadly accepted that NO directly interacts with ROS (i.e., superoxide) to form peroxynitrite, a highly toxic molecule to the vasculature. Although it is unknown if the ROS storm precedes or is a consequence of other storms, it is improbable that the mechanisms regulating ROS are functioning normally in the vasculature of COVID-19 hypertensive patients.
The ROS storm might also occur in response to and/or lead to a collapse in the antioxidant system during COVID-19. In support of this argument, a previous study has demonstrated that COVID-19 patients receiving intensive care have a disruption in their oxidative stress status, evidenced by impairment in antioxidant systems and a burst in pro-oxidant pathways. Regarding the antioxidant defenses, levels of vitamin C, thiol proteins, glutathione, γ-tocopherol, and β-carotene were drastically decreased in relation to the reference values. Of note, in this group of patients, hypertension and type 2 diabetes were the most prevalent comorbidities appearing in 66% of the cases.11 Additionally, biopsies of COVID-19 patients displayed reduced expression of genes related to the Nrf2 pathway,12 which could associate with viral oxidative cell damage. Considering the roles of a ROS storm in the hypertensive vasculature during COVID-19, a central question that arises is whether antioxidants could offer a therapeutic strategy to counteract the deleterious effects of increased oxidative stress. According to the ClinicalTrials.gov website, the search for COVID-19 and antioxidants elicit a total of 30 results within the United States. These trials are at different stages and analyze a wide variety of metrics. Up to now, though, only one study (NCT04342728) was completed and reported its results. Unfortunately, in this randomized trial, which was conducted with 214 patients on an outpatient basis, 10 days of zinc gluconate (50 mg), ascorbic acid (8,000 mg), or the combination of these agents did not improve the primary outcome measured (i.e., symptoms duration) compared with the standard of care.13 Nevertheless, it is unclear whether the use of antioxidants could affect other variables and/or offer, in the long run, a benefit for COVID-19 patients. Antioxidants were effective in modulating blood pressure and other parameters in preclinical studies of hypertension. However, the results obtained in clinical trials, similar to what happened in this COVID-19 trial, were discouraging.2,7
In summary, whether the focus is on the ROS storm or other storms in COVID-19, at this point, it seems very unlikely that one happens without the other, and therefore, all of them should be appreciated in the context of the hypertensive vasculature. Moreover, since elevated ROS levels are a hallmark of various pathological conditions, there is a need for additional studies. Finally, while each storm offers valuable mechanistic insights into the pathophysiology of COVID-19, it is acknowledged that much effort is still required to finally obtain a complete picture of the pathophysiology of SARS-CoV-2.
FUNDING
Financial support for this work provided by the National Heart, Lung, and Blood Institute; and NIDDK Diabetic Complications Consortium (RRID: SCR_001415, www.diacomp.org), grants DK076169, DK115255, and NHLBI PO1 HL-134604.
DISCLOSURE
The authors declared no conflict of interest.
REFERENCES
- 1. Wilczynski SA, Wenceslau CF, McCarthy CG, Webb RC. A Cytokine/Bradykinin storm comparison: what is the relationship between hypertension and COVID-19? Am J Hypertens 2021; 34:304–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Paravicini TM, Touyz RM. NADPH oxidases, reactive oxygen species, and hypertension: clinical implications and therapeutic possibilities. Diabetes Care 2008; 31(Suppl 2):S170–S180. [DOI] [PubMed] [Google Scholar]
- 3. Violi F, Oliva A, Cangemi R, Ceccarelli G, Pignatelli P, Carnevale R, Cammisotto V, Lichtner M, Alessandri F, De Angelis M, Miele MC, D’Ettorre G, Ruberto F, Venditti M, Pugliese F, Mastroianni CM. Nox2 activation in Covid-19. Redox Biol 2020; 36:101655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Caillon A, Zhao K, Klein KO, Greenwood CMT, Lu Z, Paradis P, Schiffrin EL. High systolic blood pressure at hospital admission is an important risk factor in models predicting outcome of COVID-19 patients. Am J Hypertens 2021; 34:282–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Siddiqi HK, Libby P, Ridker PM. COVID-19 – a vascular disease. Trends Cardiovasc Med 2021; 31:1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Schiffrin EL. Compendium on hypertension: the microbiome, inflammation and oxidative stress, the sympathetic nervous system and treatment of hypertension. Am J Hypertens 2020; 33:889. [DOI] [PubMed] [Google Scholar]
- 7. Sorriento D, De Luca N, Trimarco B, Iaccarino G. The antioxidant therapy: new insights in the treatment of hypertension. Front Physiol 2018; 9:258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Dela Justina V, Giachini FR, Priviero F, Webb RC. Double-stranded RNA and Toll-like receptor activation: a novel mechanism for blood pressure regulation. Clin Sci (Lond) 2020; 134:303–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Dela Justina V, Giachini FR, Priviero F, Webb RC. COVID-19 and hypertension: Is there a role for dsRNA and activation of Toll-like receptor 3? Vascul Pharmacol 2021; e-pub ahead of print 22 April 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Gheblawi M, Wang K, Viveiros A, Nguyen Q, Zhong JC, Turner AJ, Raizada MK, Grant MB, Oudit GY. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ Res 2020; 126:1456–1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Pincemail J, Cavalier E, Charlier C, Cheramy-Bien J-P, Brevers E, Courtois A, Fadeur M, Meziane S, Goff CL, Misset B, Albert A, Defraigne J-O, Rousseau A-F. Oxidative stress status in COVID-19 patients hospitalized in intensive care unit for severe pneumonia. A pilot study. Antioxidants (Basel). 2021; 10:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Olagnier D, Farahani E, Thyrsted J, Blay-Cadanet J, Herengt A, Idorn M, Hait A, Hernaez B, Knudsen A, Iversen MB, Schilling M, Jørgensen SE, Thomsen M, Reinert LS, Lappe M, Hoang H-D, Gilchrist VH, Hansen AL, Ottosen R, Nielsen CG, Møller C, van der Horst D, Peri S, Balachandran S, Huang J, Jakobsen M, Svenningsen EB, Poulsen TB, Bartsch L, Thielke AL, Luo Y, Alain T, Rehwinkel J, Alcamí A, Hiscott J, Mogensen TH, Paludan SR, Holm CK. SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat Commun. 2020; 11:4938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Thomas S, Patel D, Bittel B, Wolski K, Wang Q, Kumar A, Il’Giovine ZJ, Mehra R, McWilliams C, Nissen SE, Desai MY. Effect of high-dose zinc and ascorbic acid supplementation vs usual care on symptom length and reduction among ambulatory patients with SARS-CoV-2 infection: the COVID A to Z randomized clinical trial. JAMA Netw Open 2021; 4:e210369. [DOI] [PMC free article] [PubMed] [Google Scholar]