Abstract
Background
Performing a cerebrospinal fluid (CSF) drainage challenge can be used to measure the pressure equalization (PE) ratio, which describes the extent to which CSF drainage can equalize pressure to the height of the external ventricular drain and may serve as a correlate of cerebral edema. We sought to assess whether treatment with mannitol improves PE ratio in patients with severe traumatic brain injury (TBI) with elevated intracranial pressure (ICP).
Methods
We studied consecutive patients with TBI and brain edema on computed tomography scan and an external ventricular drain (EVD), admitted to the neurointensive care unit. PE ratio, defined as ICP prior to CSF drainage minus ICP after CSF drainage divided by ICP prior to CSF drainage minus EVD height, was measured as previously described. Patients were treated with mannitol for raised ICP based on clinical indication and PE ratio measured before and after mannitol administration.
Results
We studied 20 patients with severe TBI with raised ICP. Mean ICP prior to mannitol treatment was 29 ± 7 mm Hg. PE ratio rose substantially after mannitol treatment (0.62 ± 0.24 vs. 0.29 ± 0.20, p < 0.0001), indicating an improved ability to drain CSF and equalize ICP with the preset height of the EVD. The combination of mannitol and CSF drainage led to an improved reduction in ICP compared with that seen before mannitol therapy (11 ± 2 mm Hg vs. 6 ± 2 mm Hg, p < 0.01), and led to a decrease in ICP below the 20 mm Hg threshold in 77% of cases.
Conclusions
Treatment with mannitol leads to a substantial improvement in PE ratio that reflects the ability to achieve a greater decrease in ICP when CSF drainage is performed after mannitol administration. This preliminary study raises the possibility that PE ratio may be useful to follow response to therapy in patients with cerebral edema and raised ICP. Further studies to determine whether PE ratio may serve as an easily obtained and clinically useful surrogate marker for the extent of brain edema are warranted.
Keywords: Pressure equalization ratio, Traumatic brain injury, Intracranial pressure, Mannitol, Response to therapy, External ventricular drain
Introduction
The external ventricular drain (EVD) first described by Guillaime and Janny [1, 2] remains the gold standard to measure intracranial pressure (ICP) and offers the ability to therapeutically drain cerebrospinal fluid (CSF). Although CSF drainage patterns may differ markedly between patients, few methods have characterized those differences in a quantitative fashion. In a recent work, we described the use of a CSF drainage challenge to measure the pressure equalization (PE) ratio, a parameter that can characterize the response to CSF drainage in a quantitative manner in patients with an EVD [3]. The pressure equalization ratio, by its definition, reflects the ability of CSF drainage to achieve a pressure equal to the preset height of the EVD. We demonstrated that patients with nontraumatic brain injury, in whom the underlying pathology is often one of CSF outflow obstruction, had substantially higher PE ratio than that in patients with traumatic brain injury (TBI), in whom cerebral swelling predominates. The higher PE ratio in patients with non-TBI indicates a better ability to drain CSF and to equalize ICP with the height of the EVD. We hypothesized that the degree of cerebral edema may account for the substantial differences in PE ratio observed between patients with TBI and those in whom the primary cause of elevated ICP is related to CSF outflow obstruction.
To further explore the hypothesis that PE ratio lends insight into the degree of brain swelling, we sought to assess whether treatments that aim to decrease cerebral edema, such as administration of hypertonic fluids, may influence the PE ratio. Patients with severe TBI, in whom the underlying pathology is often cerebral edema that leads to raised ICP, present a clinical opportunity to assess this hypothesis. A wealth of data have documented the substantial reduction in ICP achieved by hypertonic fluid administration in patients with severe TBI [4–15]. Our clinical experience suggests that treatment with hypertonic agents leads to concomitant improvement in drainage of CSF from the EVD that, in turn, has a synergistic effect on ICP reduction. We hypothesized that in patients with severe TBI, treatment with an osmotic agent given as part of standard care may lead to an improved ability to drain CSF and reduce ICP that can be quantified by measuring the PE ratio. In this study, we sought to determine whether PE ratio improves following mannitol administration in patients with severe TBI with cerebral edema treated for raised ICP.
Methods
We prospectively studied consecutive patients with severe TBI admitted to the neurosurgical intensive unit at our institution that had an EVD placed per clinical indication. The recruitment period for this study began after the initial study of PE ratio was completed, with no overlap of participants between studies. EVD was placed on the side determined by the treating neurosurgeon. EVD height and all treatment interventions were determined by institutional protocols. Our treatment protocols for patients with TBI prescribe an EVD height set to 10 cm and kept closed with opening of the EVD to drainage when ICP rises above 22 mm Hg. This study was approved by the Hadassah-Hebrew University Medical Center Institutional Review Board (approval number 19384). Waiver of informed consent was approved for this study, as no deviations from standard of care were involved.
In all patients, a CSF drainage challenge was performed to assess the ability of CSF drainage to equalize pressure with the height of the EVD. As described in our previous report, CSF drainage challenge was performed after the EVD had been closed for 30 min prior to the challenge. Before a CSF drainage challenge was performed, all patients were evaluated to assure adequate sedation. If any resistance to the ventilator, spontaneous motor movements, or other signs of wakefulness were present, sedation was increased until these ceased. Once adequate sedation was achieved, no changes in the degree of sedation were made and no additional doses of sedative medications were administered during the period of PE ratio measurement. In addition, no changes in bed position were allowed before or during measurement of the PE ratio because changes in the relationship between the position of the patient’s head and the EVD may lead to changes in venous outflow from the cranium. Such changes in venous outflow may, in turn, result in alterations in the relative volumes of brain, blood, and CSF within the cranium that might potentially affect the PE ratio. Lastly, no changes in the ventilator settings were made immediately prior to or during the measurement period of the PE ratio. Specifically, neither the fraction of inspired oxygen, positive end-expiratory pressure (PEEP), ventilatory rate, degree of pressure support, or tidal volume were altered. The CSF drainage challenge and assessment of PE were performed as previously described.
The ICP immediately prior to opening of the EVD was recorded (ICPinitial). The drain height zeroed at the level of the external auditory meatus was also recorded (EVDheight). The drain was opened to allow drainage of CSF until CSF stopped draining. Immediately on cessation of CSF drainage the EVD was closed and the postdrainage ICP (ICPpost) was recorded. The difference between initial ICP and the postdrainage ICP measured immediately after EVD closure was calculated as follows:
In cases of pure obstruction to the outflow of CSF end ICP after drainage (ICPpost) would be expected to equalize with the preset height of the EVD (EVDheight). Therefore, when elevated ICP results from pure outflow obstruction, the maximal expected decrease in ICP is defined as follows:
PE ratio is defined as the ratio between the actual and expected decrease in ICP as follows:
As such, PE ratio generally varies between 0 and 1, reflecting the degree to which equalization is achieved between postdrainage ICP and the height of the EVD. When no CSF is drained, the PE ratio is 0, and when CSF is drained until ICP (post) is equal to EVD height, PE ratio is 1.
Treatment with 100 gm of mannitol was initiated per clinical indication as determined by the treating neurocritical care team. In general, our institutional protocols call for mannitol administration to treat elevated ICP above a threshold of 22 mm Hg in patients with cerebral swelling when elevated ICP does not respond to adequate sedation and other first-tier therapies and when serum osmolality is below 320 milliosmoles per liter. The threshold above 22 mm Hg is used per the recommendations in the 4th Edition of the Brain Trauma Foundation Guidelines [16]. At our institution, we generally prefer mannitol to hypertonic saline as first-line treatment, due to concerns regarding the development of hyperchloremic metabolic acidosis with repeated dosing of hypertonic saline. Prior to mannitol administration, we measured the PE ratio as described above.
At our institution, we attempt to follow the Seattle International Brain Injury Consensus Conference algorithm for treatment of elevated ICP which classifies mannitol administration and CSF drainage as tier 1 therapeutic options. The Seattle International Brain Injury Consensus Conference algorithm specifically does not rank the order of therapies within tiers. When ICP rises to higher thresholds in patients with severe TBI, it has been our institutional approach to administer mannitol first, followed by CSF drainage from the EVD. The rationale for using this approach is that severe cerebral edema may cause displacement of CSF out of the cranium and decrease the intracranial CSF volume reserve. By administering mannitol first, our intention is to attain some decrease in the degree of cerebral edema, which may in turn allow for an increase in the intracranial CSF volume reserve and enhance the ability to drain CSF from the EVD to achieve a synergistic effect in lowering ICP. After mannitol administration, the EVD was kept closed and ICP recorded. The EVD was opened again when ICP again rose above threshold values. At that point, PE ratio was again measured as described above. In patients who underwent primary surgical intervention (craniotomy or primary decompressive craniectomy), measurements of the PE ratio were made after surgery, whereas in patients who underwent secondary decompressive craniectomy measurements of the PE ratio were made prior to surgical intervention.
We evaluated the admission computed tomography (CT) scan for all patients, calculating the Rotterdam score [17]. In all patients, we assessed the presence and degree of midline shift and the status of the basal cisterns as either: open, compressed, or completely effaced.
Statistics
All continuous parameters are reported as mean and standard deviation. Noncontinuous parameters are reported as median and interquartile range. A paired Student’s t-test was used to compare data premannitol and postmannitol administration. A p value of less than 0.05 was considered significant.
Results
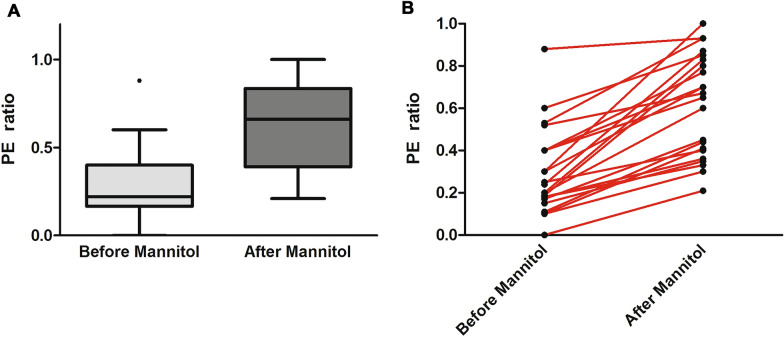
We studied 20 patients with severe TBI with cerebral swelling and raised ICP who were treated with mannitol. Patient characteristics are detailed in Table 1. As seen in Table 1, all patients presented with compressed or effaced cisterns on initial CT scan and 16 of 20 presented with a Rotterdam CT score of 4 or greater, indicating evidence of severe injury and cerebral swelling. No patient had a substantial amount of intraventricular blood that might impede the free flow of CSF within the ventricular system or cause obstruction of CSF flow. We performed 22 paired CSF drainage challenges with measurements of CSF drainage parameters and PE ratio at a mean of 2 ± 1 days post injury. Systemic and cerebral physiological parameters and CSF drainage parameters from the EVD prior to and following mannitol administration are detailed in Table 2. With the EVD kept closed after mannitol administration, ICP decreased transiently but rose above threshold values within 45 ± 11 min, at which point the EVD was opened to drain CSF and PE ratio was again measured. As expected, most patients drained CSF more briskly and in larger volume after mannitol administration when compared with drainage immediately prior to mannitol treatment. Mean ICP reduction following CSF drainage improved substantially after mannitol administration (11 ± 2 mm Hg) compared with the reduction seen before mannitol (6 ± 2 mm Hg, p < 0.01). Across all patients, PE ratio increased substantially after mannitol administration, indicating that mannitol therapy and CSF drainage led to an improved ability to reduce ICP compared with CSF drainage alone (Fig. 1a). In nearly all patients, mannitol improved PE ratio (Fig. 1b). Combining mannitol treatment with CSF drainage led to a decrease in ICP below the 20 mm Hg threshold in 17 of 22 cases (77%). Figure 2 demonstrates the improved CSF drainage attained after mannitol treatment in an individual patient. Within individual patients, PE ratio improved by a mean of 0.33 ± 0.18 following mannitol administration, indicating that PE ratio may serve as a marker of treatment efficacy in patients with TBI with cerebral swelling and raised ICP. We did not find an association between improvement of PE ratio following mannitol and the degree of midline shift or an association with surgical intervention.
Table 1.
Patient characteristics
| Characteristic | Patients |
|---|---|
| Age, (Mean ± SD (yr)) | 43 ± 23 |
| Sex, nGender | |
| Male | 14 |
| Female | 6 |
| Mechanism of Injury, n | |
| MVA | 11 |
| FALL | 9 |
| Admission GCS score, (median (, IQR) | 7 (4–—8) |
| Surgical intervention, n | |
| Craniotomy | 2 |
| Primary decompressive craniectomy | 4 |
| Secondary decompressive craniectomy | 2 |
| EVD, days (mean ± sd (d)) | 10 ± 5 |
| ICU, days (mean ± sd (d)) | 17 ± 7 |
| Midline Shift, n | |
| No Midline shift | 2 |
| Midline shift > 1 MM AND < 5 MM | 13 |
| Midline shift > 5 MM | 5 |
| Basal cisterns, n | |
| Open | 0 |
| Compressed | 13 |
| Effaced | 7 |
| Rotterdam score, n | |
| 1 | 0 |
| 2 | 0 |
| 3 | 4 |
| 4 | 9 |
| 5 | 6 |
| 6 | 1 |
SD: standard deviation; IQR: interquartile range; EVD: external ventricular drain; GCS: Glasgow Coma Score; ICU: intensive care unit, IQR: interquartile range, MVA: motor vehicle accident; SD: standard deviation
Table 2.
Physiological and EVD drainage parameters during CSF drainage challenge
| Pre-CSF drainage (mm Hg) | Post-CSF drainage (mm Hg) | |
|---|---|---|
| MAP | ||
| BEFORE mannitol | 77 ± 10 | 78 ± 6 |
| AFTER mannitol | 76 ± 8 | 75 ± 11 |
| ICP | ||
| BEFORE mannitol | 29 ± 7 | 23 ± 6* |
| AFTER mannitol | 27 ± 6 | 16 ± 4* |
| CPP | ||
| BEFORE mannitol | 46 ± 2 | 54 ± 9 |
| AFTER mannitol | 47 ± 9 | 60 ± 10 |
| Volume OF CSF drained (ML) | ||
| BEFORE mannitol | 4 ± 1 | |
| AFTER mannitol | 6 ± 3* | |
CPP: cerebral perfusion pressure; CSF: cerebrospinal fluid; EVD: external ventricular drain; ICP: intracranial pressure; MAP: mean arterial pressure
*p < 0.05
Fig. 1.
a PE ratio in patients with severe TBI with elevated ICP before and after mannitol administration. PE ratio rises substantially after mannitol treatment (0.62 ± 0.24 vs. 0.29 ± 0.20), indicating an improved ability to drain CSF and equalize ICP with the preset height of the external ventricular drain. The improved PE ratio following mannitol suggests that PE ratio may serve as a correlate of the degree of cerebral edema and may potentially be useful as a marker of the response to treatment interventions to reduce cerebral edema. *p < 0.0001. b PE ratio before and after mannitol administration in individual patients. In most patients, an improved PE ratio is observed following mannitol regardless of the pretreatment PE ratio value. CSF, cerebrospinal fluid, ICP, intracranial pressure, PE, pressure equalization, TBI, traumatic brain injury
Fig. 2.
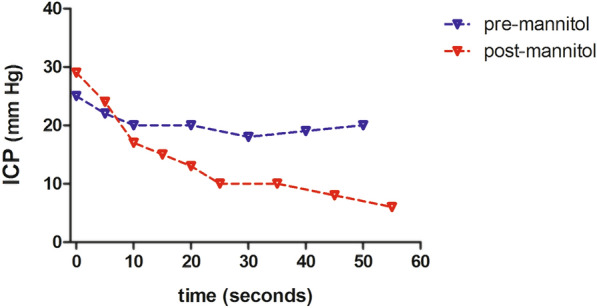
Example of the response to a CSF drainage challenge in a patient before and after treatment with mannitol. Following mannitol administration, CSF drained more briskly and a greater decrease in ICP is observed, leading to an improved PE ratio. CSF, cerebrospinal fluid, ICP, intracranial pressure, PE, pressure equalization
Discussion
Our previous study demonstrated that performing a CSF drainage challenge to measure PE ratio can quantify CSF drainage characteristics in patients who are neurocritically ill with an EVD and can distinguish between patients with TBI and those with different pathologies in which raised ICP results primarily from CSF outflow obstruction. However, whether treatment interventions to reduce brain swelling can affect PE ratio remained unclear. Our current findings clearly indicate that patients with severe TBI with elevated ICP treated with mannitol demonstrate a marked improvement in PE ratio. Because treatment with mannitol is a well-accepted method to mitigate brain swelling, the observed improvement in PE ratio following mannitol suggests that it has the potential to serve as an easily obtained surrogate marker for the degree of brain swelling. Estimating the intracranial volume-reserve capacity is an important goal in neurocritical care that can help advise decision making in the intensive care unit. Because CSF within the cranium provides the primary buffering capacity for any increase in intracranial volume, measuring the PE ratio (by estimating the degree to which CSF drainage can reduce ICP against a set EVD height) may provide a standardized method to assess this buffering capacity and how close to it is to being exhausted at a given time point. Although this observational study was limited to observing the effect of mannitol administration on the PE ratio, future investigations should aim to determine whether this index may help guide treatment interventions by serving as a surrogate marker of the degree of brain edema and the intracranial volume-reserve capacity.
There is a real clinical need for improved markers for the degree of brain swelling and for response to treatment. In current clinical practice, ICP alone is often used to guide many important decisions regarding medical and surgical interventions. Several authors and Consensus Conference statements have pointed out the limitations of using a single univariate parameter to guide clinical care and have advised incorporating other measures to help inform decision making in the neurointensive care unit [18–20]. PE ratio, which is derived from the relationship between ICP, the response to a CSF drainage challenge, and EVD height, can be measured easily at bedside. As such, it may hold promise in providing a readily obtained measure that may inform both the need for intervention to treat cerebral swelling and to follow the response to treatment. Interestingly, the improved PE ratio we observed with mannitol treatment while substantial, rising from a pretreatment level of 0.29 to a posttreatment level of 0.62, was still lower than the mean PE ratio of 0.86 measured in patients with aneuyrsmal subarachnoid hemorrhage (SAH) in our previous study, indicating that PE ratio maintains an ability to distinguish between patients in whom cerebral edema has been ameliorated with treatment and those in whom the primary pathology is CSF outflow obstruction. The range of values for PE ratio that indicate different degrees of cerebral edema and may support specific medical or surgical interventions will need to be defined in future studies.
Although it is not surprising that mannitol treatment led to an improved ability to drain CSF and reduced ICP, our results indicate that it may be possible to use the PE ratio to better define the response to treatment in patients with severe TBI. Measuring PE ratio in patients treated with a hyperosmolar agent may help to characterize how effectively cerebral swelling and raised ICP can be treated with a combination of hyperosmolar therapy and CSF drainage. The recently published algorithm for treatment of elevated ICP in patients with severe TBI includes both CSF drainage and treatment with hyperosmolar agents as tier 1 therapy for elevated ICP [21]. Our findings lend support to the efficacy of these interventions in reducing ICP, especially when used in combination. Measuring PE ratio before and after treatment at each stage of a tiered therapy protocol may provide a quantitative method to evaluate the benefit of interventions to treat cerebral edema in patients with severe TBI. The effect of higher-tier interventions, especially decompressive craniectomy may have on PE ratio is yet unclear. Although decompressive surgery in and of itself does not affect the degree of cerebral edema, it improves intracranial compliance and intracranial volume-reserve capacity. How these effects interact to influence PE ratio will require further investigations with measurements of PE ratio before and after decompressive surgery.
The importance of cerebral interstitial fluid flow in maintaining normal cerebral hemostasis has received great attention in recent years [22–26]. Traumatic mass lesions with surrounding edema may lead to an increased resistance to flow of interstitial fluid within the edematous brain parenchyma that further promotes compression of the ventricles in the already swollen brain. Theoretically, mannitol administration which promotes cellular shrinkage through its osmotic effect, may help to alleviate diminution of the extracellular space that occurs with severe brain edema [27–29], which may in turn facilitate the movement of cerebral interstitial fluid. The combination of these effects may reduce ventricular compression and promote improved drainage of CSF. Although the current study cannot shed light on the pathophysiological mechanisms that account for our findings, it supports the need for further investigations of the interaction between cerebral edema, cerebral interstitial fluid flow, and CSF drainage since an improved understanding of these interrelationships may help to refine treatment strategies in patients with severe cerebral swelling. Although we hypothesize that the primary effect of mannitol administration is to reduce cerebral swelling via its accepted mechanism of action of drawing fluid from the brain parenchyma into the cerebral vasculature, it is important to point out that we did not perform direct measurements of the degree of brain edema before and after this intervention. Such measurements are difficult to obtain in clinical practice and often require imaging studies, such as MRI, that are difficult to obtain at frequent intervals in patients who are neurocritically ill. Precisely because it is difficult to obtain measures of the degree of cerebral swelling over time in these patients, the volume-reserve capacity in individual patients is often difficult for the clinician to estimate.
Our data provide insight into the ICP reduction that is achieved when mannitol is used in conjunction with CSF drainage to treat elevated ICP. Dose–response data on mannitol is surprisingly rare. In a recent study, Poole et al. [7], performed a meta-analysis using individual patient data from multiple studies to assess the magnitude of ICP reduction following mannitol treatment. They found that ICP decreased from a mean of 22.1 mm Hg to 16.8 mm Hg at 60 min post mannitol administration. Their data indicated that a higher initial ICP was associated with a greater ICP reduction, however, the dose of mannitol was not related to the magnitude of decrease in ICP. In contrast, Sorani and colleagues demonstrated a clear dose–response relationship between mannitol and ICP, with a longer lasting therapeutic effect of 100-gm dose of mannitol in comparison with a 50-gm dose [30]. With both doses, they reported a decrease in ICP occurring over approximately 30 min and rising slowly but steadily after that. ICP returned to near pretreatment values after a mean of 100 min in those receiving the 50-gm dose and to 75% of pretreatment levels in those receiving the 100-gm dose within 100 min. In our study, mean pretreatment ICP was higher (29 mm Hg) than in the study by Sorani (22 mm Hg) and returned to near its pretreatment value at a mean of 45 min following mannitol administration. Sorani and Manley also performed a meta-analysis of the published reports of the dose–response relationship between mannitol and ICP [31]. They found that a pretreatment ICP more than 30 mm Hg was associated with a greater reduction in ICP when mannitol was given. Revealingly, they report a lack of uniformity in the manner which the response to mannitol is reported between studies and a paucity of detailed data to allow assessment of both the magnitude and time course of the dose–response relationship between mannitol and ICP. In many of the works little or no data regarding concomitant treatments interventions, including CSF drainage, are reported. In the current study, we studied the specific combination of mannitol therapy followed by CSF drainage to reduce ICP. This combination is an inherent part of measuring PE ratio since the ratio cannot be calculated without draining CSF. Our results indicate a substantial reduction in ICP following mannitol administration and CSF drainage, suggesting that the combination of hyperosmolar therapy and CSF drainage may be a particularly effective therapeutic strategy in TBI patients with cerebral edema and raised ICP. Further investigations to assess whether protocols that combine hyperosmolar therapy with CSF drainage performed in succession offer added benefit in controlling elevated ICP may help to assess the efficacy of in-tandem treatment strategies.
Limitations
Our study has several limitations. We studied a small number of patients with severe TBI with high ICP at an early stage after injury. Our findings will need to be confirmed in a larger group of patients with TBI over a wide range of ICP and throughout the ICU course. Future studies are required to ascertain how PE ratio changes from day to day along the hospital course as cerebral edema waxes and wanes. It will be important to determine whether measuring the PE ratio is an effective means to follow the response to treatment over time and to assess the severity of cerebral swelling over time. It will also be important to determine whether the observed effect of mannitol administration on PE ratio is dependent on the dose administered per patient weight (gm mannitol per kg patient weight). This small exploratory study was also not designed to determine clinically relevant thresholds for PE ratio that are associated with untoward clinical events or those requiring intervention. Further studies will be required to ascertain whether a persistently low PE ratio that does not respond positively to intervention may be indicative of a low intracranial volume reserve that may lead to impending herniation or necessitate surgical intervention. In this study, mannitol was the sole osmotic agent given to treat elevated ICP. Further investigations will be needed to see whether the same degree of improvement in PE ratio is seen when hypertonic saline is used as the osmotic agent rather than mannitol. The purpose of this study was to study the effect of mannitol treatment in patients with severe TBI. Other studies will be required to assess how the PE ratio responds to interventions in other pathological conditions in which cerebral edema predominates. A further consideration is that changes in the volume of mass lesions may impact the PE ratio. Another potential limitation in this work is that we did not measure cerebral blood flow or cerebral blood volume. Because mannitol may affect flow in the cerebral microcirculation it may influence the cerebral vasodilatory response and cerebral blood volume. Future studies should seek to measure the possible interactions of mannitol’s effect on the cerebral microcirculation with its effect on brain edema to determine how these interactions may influence the PE ratio. Lastly, the current study aimed to describe the effect of mannitol administration on a physiological parameter, the PE ratio. It was not designed to assess the association between PE ratio and clinical or radiological outcome parameters. Exploring a potential link between PE ratio and outcome parameters requires future large studies.
Conclusions
Pressure equalization ratio improves substantially following mannitol administration in patients with severe TBI with raised ICP. These preliminary findings raise the possibility that PE ratio may potentially be useful as a measure to follow response to treatment in patients with cerebral edema treated with hyperosmolar agents and CSF drainage. Further studies are needed to assess whether the PE ratio may serve as a readily obtained surrogate marker for the degree of brain edema.
Author contributions
Conception and design: Doron, Manley, Rosenthal. Acquisition of data: Doron. Analysis and interpretation of data: Doron, Hemphill, Rosenthal. Drafting the article: Doron, Rosenthal. Critically revising the article: Doron, Rosenthal, Hemphill, Manley. Statistical analysis: Doron. Study supervision: Rosenthal. All authors approve the final manuscript.
Source of support
None.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval/informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Guillaume J, Janny P. Continuous intracranial manometry; physiopathologic and clinical significance of the method. Presse Med. 1951;59(45):953–955. [PubMed] [Google Scholar]
- 2.Guillaume J, Janny P. Continuous intracranial manometry; importance of the method and first results. Rev Neurol (Paris) 1951;84(2):131–142. [PubMed] [Google Scholar]
- 3.Candanedo C, Doron O, Hemphill JC, 3rd, et al. Characterizing the response to cerebrospinal fluid drainage in patients with an external ventricular drain: the pressure equalization ratio. Neurocrit Care. 2019;30(2):340–347. doi: 10.1007/s12028-018-0612-y. [DOI] [PubMed] [Google Scholar]
- 4.Schwimmbeck F, Voellger B, Chappell D, Eberhart L. Hypertonic saline versus mannitol for traumatic brain injury: a systematic review and meta-analysis with trial sequential analysis. J Neurosurg Anesthesiol. 2021;33(1):10–20. doi: 10.1097/ANA.0000000000000644. [DOI] [PubMed] [Google Scholar]
- 5.Chen H, Song Z, Dennis JA. Hypertonic saline versus other intracranial pressure-lowering agents for people with acute traumatic brain injury. Cochrane Database Syst Rev. 2019;12: CD010904. [DOI] [PMC free article] [PubMed]
- 6.Kamel H, Navi BB, Nakagawa K, Hemphill JC, 3rd, Ko NU. Hypertonic saline versus mannitol for the treatment of elevated intracranial pressure: a meta-analysis of randomized clinical trials. Crit Care Med. 2011;39(3):554–559. doi: 10.1097/CCM.0b013e318206b9be. [DOI] [PubMed] [Google Scholar]
- 7.Poole D, Citerio G, Helbok R, et al. Evidence for mannitol as an effective agent against intracranial hypertension: an individual patient data meta-analysis. Neurocrit Care. 2020;32(1):252–261. doi: 10.1007/s12028-019-00771-y. [DOI] [PubMed] [Google Scholar]
- 8.Marshall LF, Smith RW, Rauscher LA, Shapiro HM. Mannitol dose requirements in brain-injured patients. J Neurosurg. 1978;48(2):169–172. doi: 10.3171/jns.1978.48.2.0169. [DOI] [PubMed] [Google Scholar]
- 9.Kirkpatrick PJ, Smielewski P, Piechnik S, Pickard JD, Czosnyka M. Early effects of mannitol in patients with head injuries assessed using bedside multimodality monitoring. Neurosurgery. 1996;39(4):714–20; discussion 20–1. [DOI] [PubMed]
- 10.McGraw CP, Alexander E, Jr, Howard G. Effect of dose and dose schedule on the response of intracranial pressure to mannitol. Surg Neurol. 1978;10(2):127–130. [PubMed] [Google Scholar]
- 11.Smith HP, Kelly DL, Jr, McWhorter JM, et al. Comparison of mannitol regimens in patients with severe head injury undergoing intracranial monitoring. J Neurosurg. 1986;65(6):820–824. doi: 10.3171/jns.1986.65.6.0820. [DOI] [PubMed] [Google Scholar]
- 12.Soustiel JF, Mahamid E, Chistyakov A, et al. Comparison of moderate hyperventilation and mannitol for control of intracranial pressure control in patients with severe traumatic brain injury--a study of cerebral blood flow and metabolism. Acta Neurochir (Wien). 2006;148(8):845–51; discussion 51. [DOI] [PubMed]
- 13.Vialet R, Albanese J, Thomachot L, et al. Isovolume hypertonic solutes (sodium chloride or mannitol) in the treatment of refractory posttraumatic intracranial hypertension: 2 mL/kg 7.5% saline is more effective than 2 mL/kg 20% mannitol. Crit Care Med. 2003;31(6):1683–7. [DOI] [PubMed]
- 14.Ware ML, Nemani VM, Meeker M, et al. Effects of 23.4% sodium chloride solution in reducing intracranial pressure in patients with traumatic brain injury: a preliminary study. Neurosurgery. 2005;57(4):727–36;discussion 36. [PubMed]
- 15.James HE, Langfitt TW, Kumar VS, Ghostine SY. Treatment of intracranial hypertension. Analysis of 105 consecutive, continuous recordings of intracranial pressure. Acta Neurochir (Wien). 1977;36(3–4):189–200. [DOI] [PubMed]
- 16.Carney N, Totten AM, O'Reilly C, et al. Guidelines for the management of severe traumatic brain injury, fourth edition. Neurosurgery. 2017;80(1):6–15. [DOI] [PubMed]
- 17.Maas AI, Hukkelhoven CW, Marshall LF, Steyerberg EW. Prediction of outcome in traumatic brain injury with computed tomographic characteristics: a comparison between the computed tomographic classification and combinations of computed tomographic predictors. Neurosurgery. 2005;57(6):1173–82;discussion 82. [DOI] [PubMed]
- 18.Chesnut RM. A conceptual approach to managing severe traumatic brain injury in a time of uncertainty. Ann N Y Acad Sci. 2014;1345:99–107. doi: 10.1111/nyas.12483. [DOI] [PubMed] [Google Scholar]
- 19.Guiza F, Depreitere B, Piper I, et al. Visualizing the pressure and time burden of intracranial hypertension in adult and paediatric traumatic brain injury. Intensive Care Med. 2015;41(6):1067–1076. doi: 10.1007/s00134-015-3806-1. [DOI] [PubMed] [Google Scholar]
- 20.Helbok R, Meyfroidt G, Beer R. Intracranial pressure thresholds in severe traumatic brain injury: Con: the injured brain is not aware of ICP thresholds! Intensive Care Med. 2018;44(8):1318–1320. doi: 10.1007/s00134-018-5249-y. [DOI] [PubMed] [Google Scholar]
- 21.Hawryluk GWJ, Aguilera S, Buki A, et al. A management algorithm for patients with intracranial pressure monitoring: the Seattle International Severe Traumatic Brain Injury Consensus Conference (SIBICC) Intensive Care Med. 2019;45(12):1783–1794. doi: 10.1007/s00134-019-05805-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nakada T, Kwee IL. Fluid dynamics inside the brain barrier: current concept of interstitial flow, glymphatic flow, and cerebrospinal fluid circulation in the brain. Neuroscientist. 2019;25(2):155–166. doi: 10.1177/1073858418775027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abbott NJ. Evidence for bulk flow of brain interstitial fluid: significance for physiology and pathology. Neurochem Int. 2004;45(4):545–552. doi: 10.1016/j.neuint.2003.11.006. [DOI] [PubMed] [Google Scholar]
- 24.Brinker T, Stopa E, Morrison J, Klinge P. A new look at cerebrospinal fluid circulation. Fluids Barriers CNS. 2014;11:10. doi: 10.1186/2045-8118-11-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Iliff JJ, Chen MJ, Plog BA, et al. Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury. J Neurosci. 2014;34(49):16180–16193. doi: 10.1523/JNEUROSCI.3020-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thrane AS, Rangroo Thrane V, Nedergaard M. Drowning stars: reassessing the role of astrocytes in brain edema. Trends Neurosci. 2014;37(11):620–628. doi: 10.1016/j.tins.2014.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Matsuoka Y, Hossmann KA. Brain tissue osmolality after middle cerebral artery occlusion in cats. Exp Neurol. 1982;77(3):599–611. doi: 10.1016/0014-4886(82)90231-X. [DOI] [PubMed] [Google Scholar]
- 28.Hossmann KA. Cortical steady potential, impedance and excitability changes during and after total ischemia of cat brain. Exp Neurol. 1971;32(2):163–175. doi: 10.1016/0014-4886(71)90060-4. [DOI] [PubMed] [Google Scholar]
- 29.Hrabetova S, Nicholson C. Contribution of dead-space microdomains to tortuosity of brain extracellular space. Neurochem Int. 2004;45(4):467–477. doi: 10.1016/j.neuint.2003.11.011. [DOI] [PubMed] [Google Scholar]
- 30.Sorani MD, Morabito D, Rosenthal G, Giacomini KM, Manley GT. Characterizing the dose-response relationship between mannitol and intracranial pressure in traumatic brain injury patients using a high-frequency physiological data collection system. J Neurotrauma. 2008;25(4):291–298. doi: 10.1089/neu.2007.0411. [DOI] [PubMed] [Google Scholar]
- 31.Sorani MD, Manley GT. Dose-response relationship of mannitol and intracranial pressure: a metaanalysis. J Neurosurg. 2008;108(1):80–87. doi: 10.3171/JNS/2008/108/01/0080. [DOI] [PubMed] [Google Scholar]