Introduction
Bacteria have long been considered drivers in the development of cutaneous lymphoma, as bacterial antigens may encourage reactive, dysregulated lymphocytic proliferation.1,2 For example, Staphylococcus aureus infection has been implicated in the pathogenesis of mycosis fungoides and Sézary syndrome, whereas Borrelia burgdorferi is clinically associated with primary cutaneous B-cell lymphoma.3, 4, 5, 6, 7 Stenotrophomonas maltophilia is an opportunist gram-negative bacterium, primarily recognized as a cause of nosocomial infection among immunocompromised individuals.8 Ste maltophilia, specifically, has been associated with breast implant–associated anaplastic large-cell lymphoma (BIA-ALCL),9,10 and it has also been reported to be an antigenic driver in cutaneous pseudolymphoma.11 We report a new association of primary cutaneous anaplastic large-cell lymphoma (PC-ALCL) with Ste maltophilia in otherwise immunocompetent men.
Case series
Case 1
An 81-year-old man presented to the Columbia University Comprehensive Skin Cancer Center in August 2018 with an ulcerated tumor on the left side of the upper portion of his back for at least 1 month's duration. The lesion was neither painful nor pruritic. The patient had no history of immunosuppressive conditions or medications. He also reported no history of arthropod bites in the area, skin trauma, unusual environmental exposures, or use of new medications or supplements. Additionally, he denied fever, chills, night sweats, and recent weight loss.
On physical examination, a 5 × 5-cm tumor with central ulceration was noted on the left side of the upper portion of his back (Fig 1). The tumor expressed thick yellow discharge on palpation. No other lesions were observed, and no lymphadenopathy was detected on examination. Wound cultures of the lesion were obtained via a swab of the ulcer base with vigorous probing of its full depth. Cultures grew Ste maltophilia and methicillin-sensitive Sta aureus. The Ste maltophilia isolate was sensitive to trimethoprim/sulfamethoxazole (TMX), minocycline, and levofloxacin.
Fig 1.
Ulcerated tumor on the left side of the upper portion of the back (case 1).
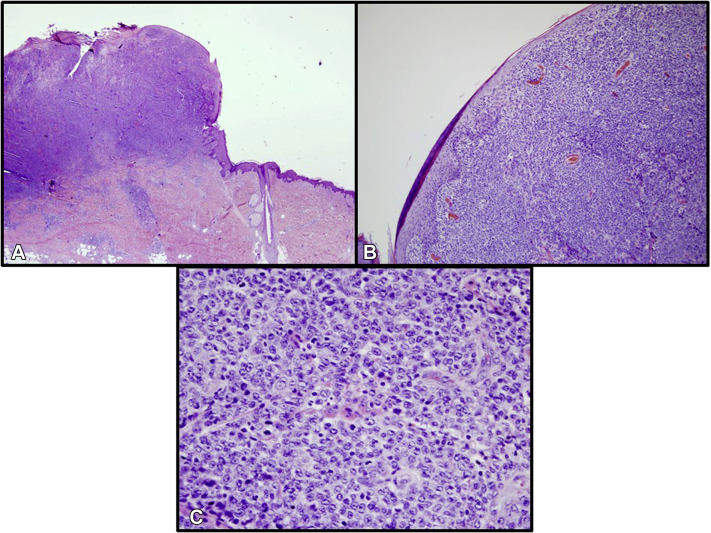
Biopsy of the lesion revealed a dense infiltrate of atypical lymphocytes and scant eosinophils, consistent with a CD30+ lymphoproliferative disorder (Fig 2). CD2, CD3, CD4, CD5, and CD30 staining were strongly positive; 50% of the cells were Ki-67+, and CD7 staining was positive, albeit slightly downregulated. The infiltrate was CD20−, ALK-1−, CD56−, and EBER− (by in situ hybridization). T-cell receptor β gene rearrangements revealed a clonal T cell population. Peripheral blood flow cytometry at this time was negative for clonal involvement, and the positron emission tomography-computed tomography scan showed no evidence of disseminated disease. Staging procedures corroborated by clinical presentation were consistent with a diagnosis of PC-ALCL.
Fig 2.
Histopathology of the tumor on the left side of the upper portion of the back consistent with primary cutaneous anaplastic large-cell lymphoma (case 1). CD30+ immunohistochemistry was observed. (A, CD30 stain; original magnification: B, ×40.)
The patient was treated with TMX for 14 days with interval improvement in tumor size from 5 × 5 cm to 4 × 3.5 cm. Wound cultures after antibiotic therapy were negative. He received electron beam therapy to the residual tumor in October 2018 as standard of care for localized PC-ALCL and was placed on continued TMX therapy until a complete resolution of the tumor was noted in January 2019.
The patient maintained complete remission until July 2019, when the patient presented with an eruption of indurated, erythematous nodules on his left shoulder. Biopsy of these lesions revealed a dense infiltrate of atypical lymphocytes consistent with anaplastic large-cell lymphoma (ALCL). New wound cultures were not obtained at this time. A concurrent positron emission tomography-computed tomography scan revealed multiple involved lymph nodes, new pulmonary nodules, splenic uptake, possible involvement of the right gluteal and right psoas muscles, and osseous involvement of the left scapula. A diagnosis of systemic ALCL was made. The patient was treated with systemic chemotherapy but progressed rapidly and died 1 month later.
Case 2
A 35-year-old man was referred to us in December 2014 for evaluation of suspicious nodules on his body.
Three years prior to evaluation, the patient noted the appearance of firm, painful, and nonpruritic red nodules on his torso and extremities that spontaneously appeared and regressed over time. Outside treatments included intralesional steroid injections to the nodules, resulting in minimal improvement. At the time of presentation to us, he reported no history of eczema, immunosuppression, known trauma to the skin, new exposures, new medications, or supplements. Additionally, he denied fever, chills, night sweats, and recent weight loss, as well as a family history of lymphoma.
On physical examination, he had a 4-cm firm, erythematous, ulcerating tumor of the posterior aspect of the right thigh (Fig 3). There was no palpable lymphadenopathy. A biopsy of the tumor was performed (Fig 4). Immunohistochemical studies revealed positivity for CD2, CD3, and CD30 and negativity for CD4, CD5, CD7, CD8, CD20, and CD79A; a clonal T-cell receptor gene rearrangement was detected. Staging procedures did not reveal disease in lymph nodes or other organs. A diagnosis of primary cutaneous CD30+, ALK−, EMA− large-cell lymphoma was made.
Fig 3.
Ulcerating tumor of the posterior aspect of the right thigh (case 2).
Fig 4.
Histopathology of the tumor on the posterior aspect of the right thigh consistent with primary cutaneous anaplastic large-cell lymphoma (case 2). (Original magnifications: A, ×2; B, ×10; C, ×40.)
Wound culture of the tumor was obtained via a deep swab of its ulcerated component, and at this time, it grew Ste maltophilia and Klebsiella pneumoniae. The Ste maltophilia isolate was sensitive to TMX, minocycline, and levofloxacin. The patient was treated with minocycline for 14 days. His PC-ALCL was subsequently treated with excision with no near-term recurrence.
The patient maintained excellent control of the disease until May 2020. At that time, he presented with a new tumor on the upper portion of the right arm, which did not resolve spontaneously. He underwent wide local excision, and pathology again was consistent with PC-ALCL. Excision was followed by localized electron beam radiation therapy in September and October 2020. He has no evidence of tumor recurrence on follow-up.
Discussion
Sta aureus is the most well-known bacterial pathogen associated with cutaneous T-cell lymphoma. In vitro studies have established that staphylococcal enterotoxins promote maintenance of malignant T-cell clones, while numerous clinical studies have demonstrated that antibiotic therapy significantly improves disease burden in patients with Sta aureus infection and Sézary syndrome or advanced-stage mycosis fungoides.3, 4, 5, 6, 7 To date, a bacterial species has not been identified in association with PC-ALCL. The malignant T cells in ALCL are known to express CD30, a cell membrane protein of the tumor necrosis factor receptor family.12, 13, 14 CD30 expression depends, in part, on costimulation via CD28 or interleukin 4 signaling, suggesting that antigen stimulation or local immune activation may play a role.13 CD30+ T cells are often seen in reactive infiltrates, such as those associated with arthropod bites, eczema, and dermatopathic lymphadenopathy.14 Our cases suggest that the role of chronic antigenic stimulation should be considered.
Kash et al11 previously described a case of Ste maltophilia infection that induced cutaneous γδ T-cell pseudolymphoma, which resolved after a course of TMX. Those findings suggest that Ste maltophilia may serve as an antigenic stimulus for reactive T-cell dyscrasia. Ste maltophilia has also been associated with BIA-ALCL. BIA-ALCL, a CD30+ lymphoma, arises in patients with macrotextured breast implants, which are subject to contamination by gram-negative bacteria.15 Colonies of gram-negative bacteria are thought to contribute to a chronic inflammatory milieu, which promotes the immune dysregulation necessary for the development of ALCL. In 2 studies of the microbiome associated with BIA-ALCL samples, Ste maltophilia comprised a significant portion of the bacterial diversity in tumor samples versus controls, signifying that this species may have a role in the pathogenesis of BIA-ALCL.9,10
Patients with cutaneous T-cell lymphoma often experience a high incidence of bacterial skin and soft tissue infections due to a locally suppressed immune system and compromised skin barriers.2 However, Ste maltophilia is not a commensal organism or a common pathogen in such patients.2,8 Ste maltophilia is typically hospital-acquired. However, neither of the 2 patients in this series had a recent history of hospitalization prior to presentation, leaving in question how cutaneous colonization with Ste maltophilia occurred.
The first patient's primary lesion began to improve after an initial course of TMX, suggesting that bacterial antigen removal may have played a role in resolution. The second patient's lesion was excised at the time of culture, compromising inferences regarding the efficacy of antibiotic therapy. However, that patient did not experience recurrence at the lesion site following antibiotics. There is precedent that removing the pathogen associated with chronic antigenic stimulation may be an important step in treating both B- and T-cell lymphomas. Helicobacter pylori eradication is associated with a remission rate of >75% for gastric mucosa-associated lymphoid tissue lymphomas, while resolution of Borrelia-associated cutaneous B-cell lymphomas and Chlamydophila psittaci–associated ocular adnexal mucosa-associated lymphoid tissue lymphomas has also been reported following antibiotic therapy.5,6 Treatment of Sta aureus infection and colonization has improved cutaneous T-cell lymphoma skin involvement in erythrodermic patients.3,16
Ste maltophilia has a putative role in the development of ALCL. Our report illustrates 2 cases in which this bacterium was clearly associated with biopsy-confirmed PC-ALCL. In addition, Ste maltophilia has formerly been identified as a driver of reactive pseudolymphoma and BIA-ALCL. Further research is necessary to define the antigenic potential of this rare pathogen. However, these cases point to the need for evaluating wound cultures in patients with suspected or newly diagnosed ALCL, as bacterial involvement may serve as a source of chronic antigenic stimulation precipitating dysregulated T-cell proliferation.
Conflicts of interest
None disclosed.
Footnotes
Funding sources: None.
IRB approval status: Not applicable.
References
- 1.Fanok M.H., Sun A., Fogli L.K. Role of dysregulated cytokine signaling and bacterial triggers in the pathogenesis of cutaneous T-cell lymphoma. J Invest Dermatol. 2018;138(5):1116–1125. doi: 10.1016/j.jid.2017.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mirvish E.D., Pomerantz R.G., Geskin L.J. Infectious agents in cutaneous T-cell lymphoma. J Am Acad Dermatol. 2011;64(2):423–431. doi: 10.1016/j.jaad.2009.11.692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jackow C.M., Cather J.C., Hearne V., Asano A.T., Musser J.M., Duvic M. Association of erythrodermic cutaneous T-cell lymphoma, superantigen-positive Staphylococcus aureus, and oligoclonal T-cell receptor V beta gene expansion. Blood. 1997;89(1):32–40. [PubMed] [Google Scholar]
- 4.Woetmann A., Lovato P., Eriksen K.W. Nonmalignant T cells stimulate growth of T-cell lymphoma cells in the presence of bacterial toxins. Blood. 2007;109(8):3325–3332. doi: 10.1182/blood-2006-04-017863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chang A.H., Parsonnet J. Role of bacteria in oncogenesis. Clin Microbiol Rev. 2010;23(4):837–857. doi: 10.1128/CMR.00012-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ferreri A.J., Ernberg I., Copie-Bergman C. Infectious agents and lymphoma development: molecular and clinical aspects. J Intern Med. 2009;265(4):421–438. doi: 10.1111/j.1365-2796.2009.02083.x. [DOI] [PubMed] [Google Scholar]
- 7.Willerslev-Olsen A., Krejsgaard T., Lindahl L.M. Bacterial toxins fuel disease progression in cutaneous T-cell lymphoma. Toxins (Basel) 2013;5(8):1402–1421. doi: 10.3390/toxins5081402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Looney W.J., Narita M., Mühlemann K. Stenotrophomonas maltophilia: an emerging opportunist human pathogen. Lancet Infect Dis. 2009;9(5):312–323. doi: 10.1016/S1473-3099(09)70083-0. [DOI] [PubMed] [Google Scholar]
- 9.Walker J.N., Hanson B.M., Pinkner C.L. Insights into the microbiome of breast implants and periprosthetic tissue in breast implant-associated anaplastic large cell lymphoma. Sci Rep. 2019;9(1):10393. doi: 10.1038/s41598-019-46535-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hu H., Johani K., Almatroudi A. Bacterial biofilm infection detected in breast implant–associated anaplastic large-cell lymphoma. Plast Reconstr Surg. 2016;137(6):1659–1669. doi: 10.1097/PRS.0000000000002010. [DOI] [PubMed] [Google Scholar]
- 11.Kash N., Vin H., Danialan R., Prieto V.G., Duvic M. Stenotrophomonas maltophilia with histopathological features mimicking cutaneous gamma/delta T-cell lymphoma. Int J Infect Dis. 2015;30:7–9. doi: 10.1016/j.ijid.2014.10.026. [DOI] [PubMed] [Google Scholar]
- 12.Pierce J.M., Mehta A. Diagnostic, prognostic and therapeutic role of CD30 in lymphoma. Expert Rev Hematol. 2017;10(1):29–37. doi: 10.1080/17474086.2017.1270202. [DOI] [PubMed] [Google Scholar]
- 13.Horie R., Watanabe T. CD30: expression and function in health and disease. Semin Immunol. 1998;10(6):457–470. doi: 10.1006/smim.1998.0156. [DOI] [PubMed] [Google Scholar]
- 14.Brown R.A., Fernandez-Pol S., Kim J. Primary cutaneous anaplastic large cell lymphoma. J Cutan Pathol. 2017;44(6):570–577. doi: 10.1111/cup.12937. [DOI] [PubMed] [Google Scholar]
- 15.Story S.K., Schowalter M.K., Geskin L.J. Breast implant–associated ALCL: a unique entity in the spectrum of CD30+ lymphoproliferative disorders. Oncologist. 2013;18(3):301–307. doi: 10.1634/theoncologist.2012-0238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Emge D.A., Bassett R.L., Duvic M., Huen A.O. Methicillin-resistant Staphylococcus aureus (MRSA) is an important pathogen in erythrodermic cutaneous T-cell lymphoma (CTCL) patients. Arch Dermatol Res. 2020;312(4):283–288. doi: 10.1007/s00403-019-02015-7. [DOI] [PubMed] [Google Scholar]