Summary
Background
Concerns about long-lasting symptoms attributed to Lyme borreliosis (LB) are widespread in the Western world, while such symptoms are highly prevalent in the general population.
Methods
In the largest prospective study to date, adults with physician-confirmed LB were included at the start of antibiotic treatment. Primary outcomes, prevalence of persistent symptoms and symptom severity, were assessed using three-monthly standardised questionnaires during one year. Persistent symptoms were defined as impaired scores for fatigue (CIS, subscale fatigue), cognitive impairment (CFQ) or pain (SF-36, subscale bodily pain) ≥6 months, with onset <6 months. Outcomes were compared with a longitudinal general population and a tick-bite cohort without LB as a reference.
Findings
Of 1135 LB patients (94•8% erythema migrans, 5•2% disseminated LB), 1084 fulfilled primary analysis criteria, as well as 1942 population and 1887 tick-bite controls. Overall prevalence of persistent symptoms in LB patients was 27•2% (95%CI, 24•7%-29•7%); 6•0% and 3•9% higher than in population (21•2%, 95%CI, 19•3%-23•1%; p < 0•0001) and tick-bite (23•3%, 95%CI 21•3%-25•3%; p = 0•016) cohorts, respectively. At 12 months, fatigue, cognitive impairment, and pain were significantly more severe in erythema migrans patients than in reference cohorts, while in disseminated LB patients, only pain was more severe.
Interpretation
In treated LB patients, persistent symptoms were significantly more prevalent and symptoms were more severe than in individuals without LB, although the background prevalence was substantial. This suggests an association, either direct or indirect, between persistent symptoms and LB in a relatively small subset of patients.
Funding
ZonMw; Dutch Ministry of Health, Welfare and Sport.
Abbreviations: ACA, Acrodermatitis chronica atrophicans; CFQ, Cognitive Failure Questionnaire; CIS, Checklist Individual Strength; EM, Erythema migrans; LB, Lyme borreliosis; PCR, Polymerase Chain Reaction; PHQ-15, Patient Health Questionnaire; PTLDS, Post-treatment Lyme Disease Syndrome; SF-36, SF-36 item Health Survey; s.l., sensu lato; TiC-P, Treatment Inventory of Costs in Patients with psychiatric disorders
Research in context.
Evidence before this study
In 2014, at the development of the LymeProspect study proposal, and at the start of the analyses in January 2019, PubMed was consulted using the following search terms: “Borrelia Infections", "Borrelia", “borrelia”, “borreliosis”, “Erythema Chronicum Migrans”, “Erythema Migrans”, “lyme”, “Lymes”, “Lyme's”, “Neuroborreliosis”. In between, and during the writing phase, new literature was searched periodically. In order to obtain a broad overview of existing literature, all studies on prevalence of persistent symptoms related to Lyme borreliosis (LB) were reviewed, without restrictions on design or date. Concerns about the perceived sequelae of LB are widespread in the Western world, but at the same time the attributed symptoms are highly prevalent in the general population. Reported prevalences in LB patients varied widely between 0 and 48%. This wide range may have been due to the variety in study characteristics such as a prospective or retrospective design, LB manifestations included, and divergent case definitions or follow-up durations. Furthermore, of many studies the power was limited, or the results were not controlled for background prevalence of symptoms and symptom severity. We designed the LymeProspect study based on the published data and on the strengths and lessons learned from previous studies.
Added value of this study
Prevalence of persistent symptoms and symptom severity were assessed during one year of follow-up in the largest prospective cohort of LB patients so far, and compared with two reference cohorts who had undergone the same longitudinal assessments. The prevalence of persistent symptoms was approximately 5% higher than the background prevalence of more than 20% in these two reference cohorts of individuals without LB. Moreover, symptoms were also significantly more severe in LB patients. Several sensitivity analyses confirmed the robustness of the primary study outcomes.
Implications of all the available evidence
These findings suggest an association between LB and persistent symptoms, albeit in a relatively small subset of patients reporting these symptoms, and underscore the need for identification of determinants for these symptoms, whether or not directly related to the disease.
Alt-text: Unlabelled box
1. Introduction
Lyme borreliosis (LB), or Lyme disease, is an emerging tick-borne infectious disease both in Europe and Northern America and is caused by Borrelia burgdorferi sensu lato (s.l.). Despite appropriate antimicrobial therapy for LB, there are patients reporting persistent symptoms for months to years. These include fatigue, pain, cognitive impairment, and other constitutional symptoms, which may greatly impact quality of life.1 When initial signs and symptoms of a confirmed LB manifestation have resolved or stabilized, and the aforementioned symptoms have developed within six months after treatment and persist for at least six months, this complex of symptoms is often referred to as Post-Treatment Lyme Disease Syndrome (PTLDS).2
Reported prevalence of persistent symptoms after treatment for LB varies between 0 and 48%.[3], [4], [5], [6], [7], [8], [9], [10], [11], [12] This wide range may be influenced by study characteristics, such as duration of follow-up, and definition or assessment of persistent symptoms, as well as inclusion of patients with localised (e.g., erythema migrans) versus disseminated (e.g., Lyme neuroborreliosis) manifestations. Furthermore, small prospective studies have described incidences of persistent symptoms after LB that do not exceed those reported in healthy matched controls.4,9
The variety in incidence rates and definition of PTLDS, potential causative factors and optimal treatment strategies, are topics of ongoing debate and research.[13], [14], [15] The LymeProspect study was initiated upon a citizens' initiative, reflecting public concerns about persistent symptoms attributed to LB.3,16 Prevalence and severity of persistent symptoms were assessed in the largest prospective cohort of strictly defined LB patients so far, with a follow-up of one year, using a clear definition of persistent symptoms based on standardised questionnaires. To control for background prevalence, results were compared with two cohorts of individuals without LB.
2. Methods
2.1. Study design and patients
LymeProspect is a nationwide prospective cohort study on post-treatment persistence of symptoms in LB patients in the Netherlands. The study was approved by the medical ethics review committee Noord-Holland (NL50227.094.14) and conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent. Between April 2015 and October 2018, adult patients with a new LB diagnosis, either physician-confirmed erythema migrans (EM) or disseminated disease, were eligible for inclusion (Table S1).16 Patients were included after online self-registration (www.tekenradar.nl) or through participating clinical LB centers (Supplementary Methods). For patients with EM included after online self-registration, their general practitioner was asked to confirm the diagnosis. Patients were requested to upload a photograph of their EM, which was classified by three independent experts (one dermatologist and two infectious diseases specialists). All patients were included before, or within a maximum of seven days after the start of antibiotic treatment. Inclusion did not interfere with standard diagnostic and therapeutic procedures.
2.2. Procedures
All LB patients were assessed with a comprehensive set of online questionnaires at baseline, 3, 6, 9 and 12 months after inclusion, primarily to determine the presence of persistent symptoms, i.e. fatigue, self-reported cognitive impairment or pain. Using the Checklist Individual Strength (CIS), fatigue was assessed by the subscale fatigue.[17], [18], [19] Cognitive impairment was assessed by the Cognitive Failures Questionnaire (CFQ),20 and pain by the bodily pain subscale of the RAND SF-36 Health Status Inventory (SF-36).[21], [22], [23] The SF-36 physical and social functioning subscales were used at all time points to assess the impact of symptoms on functioning. In addition, questionnaires included assessment of somatic symptoms (PHQ-15), health care use, absenteeism from paid work and comorbidity (adapted from TiC-P). Blood samples for B. burgdorferi s.l. serology were collected at baseline and after six weeks. Skin biopsies were obtained for culture and PCR upon additional consent in patients with skin manifestations who visited the clinical LB centers.
To control for background prevalence of persistent symptoms in individuals without evidence of LB, data on fatigue, cognitive impairment and pain, as measured with the same set of questionnaires, from two reference cohorts were gathered. One cohort consisted of adults randomly selected from the general population database, based on frequency matching to the LB cohort by age, sex, geographical region, and month of enrollment. The other cohort comprised individuals who had reported a tick bite online.24 Eligibility for inclusion in the primary analysis was assessed according to specific in- and exclusion criteria (Table S2). In addition, a cohort of patients with chronic symptoms of unknown etiology attributed to LB, but without a confirmed LB diagnosis, was included through the clinical LB centers to compare the course and severity of their chronic symptoms with that of persistent symptoms in the LB cohort. For inclusion criteria, see Table S3.
2.3. Outcome measures
Primary outcomes were prevalence of persistent symptoms and symptom severity following treatment in the prospective cohort of LB patients. Symptoms were defined as persistent if at least one symptom score was impaired (based on previously published norm scores) for fatigue, cognitive impairment or pain (CIS, subscale fatigue, score ≥35, CFQ, self-reported cognitive impairment, score ≥44, and SF-36, subscale bodily pain, score ≤55)18,20,22 during at least six subsequent months and with onset within six months after initial antibiotic treatment (Panel 1). The definition of persistent symptoms in the population and tick bite cohorts was based on the same questionnaire scores and duration of symptoms. Additional predefined outcomes included the prevalence of impaired questionnaire scores at individual time points at baseline or during follow-up, and the prevalence of symptoms lasting ≥3 but <6 months.
Panel 1.
Definition of persistent symptoms.
| Patient-reported fatigue, cognitive impairment or pain within six months after antibiotic treatment for a physician-confirmed diagnosis of Lyme borreliosis and consistently persisting for a minimum of six months. The presence and severity of symptoms was assessed at baseline and after 3, 6, 9, and 12 months, using the following questionnaires: CIS, subscale fatigue (fatigue if score ≥35), CFQ (cognitive impairment if score ≥44), and SF-36, subscale bodily pain (pain if score ≤55). |
Definition based on the case definition for Post-Treatment Lyme Disease Syndrome (PTLDS) as proposed by the Infectious Diseases Society of America (IDSA)2.
2.4. Statistical analysis
Statistical procedures were predefined in a statistical analysis plan. All participants who had completed the CIS (subscale fatigue), CFQ, and SF-36 (subscale bodily pain) at least at one time point were included in the analyses. For some analyses, the cohort of LB patients was divided into patients with EM or disseminated LB. The prevalence of persistent symptoms in all cohorts was assessed with a primary analysis scenario for substitution of missing data, if primary outcome questionnaires were completed at ≥2 time points. Missing scores were substituted by linear interpolation of available preceding and following continuous scores of the particular questionnaire. Missing first or final observations were substituted by carrying backward the first or forward the last available questionnaire score. Sensitivity analyses included three alternative substitution techniques for missing primary outcome questionnaires and three alternative analysis populations (Table S4). Primary outcomes were standardised to the distribution of the pre-defined confounders sex, age, educational level and self-reported comorbidity in the LB cohort (Supplementary Methods). Because of low numbers of patients with disseminated LB, only sex and comorbidity were used as confounders for analysis in that group.
Severity of symptoms was assessed by evaluating the mean questionnaire score for each symptom at baseline and during follow-up. Symptom severity over time of EM and disseminated LB patients was compared with the severity in the population and tick bite cohorts using linear mixed effects models, where the score was considered a function dependent on cohort, time and confounders (Supplementary Methods). In addition, differences in mean severity scores at the end of follow-up were assessed between cohorts. As a secondary outcome, severity of symptoms in the cohort of patients with chronic symptoms attributed to unconfirmed LB was compared with that of LB patients with persistent symptoms.
For the analyses of severity and prevalence of impaired questionnaire scores at individual time points, missing questionnaire scores were not substituted and standardization was performed for each specific time point. Differences in the prevalence of persistent symptoms and in severity scores at the 12 months’ time point between cohorts were assessed using permutation tests. Two-sided 5% significance levels were used to identify statistical significance. Analyses were performed with R version 3.6.0.
2.5. Role of the funding source
The funding sources (ZonMw and the Dutch Ministry of Health, Welfare and Sport) had no role in the collection, analysis and interpretation of data; the writing of the report; and the decision to submit for publication. ZonMw requested inclusion of the additional cohort of patients with chronic symptoms attributed to unconfirmed LB. All authors have full access to all study data and take responsibility for the decision to submit for publication.
3. Results
In the LB cohort, 1135 patients were included, 1076 (94•8%) with EM and 59 (5•2%) with disseminated manifestations. As a reference for background symptom prevalence, data were available from 4000 individuals from the general population, and 2405 individuals with a tick bite without clinical LB (Fig. 1). Of these participants, 1084 LB patients, 1942 participants from the population cohort, and 1887 participants from the tick bite cohort fulfilled the criteria for the primary analysis scenario. Baseline characteristics of the LB and reference cohorts are shown in Table 1. For LB patients, the median duration of symptoms at inclusion was six days (EM five days; disseminated LB 208 days). The availability of primary outcome questionnaire data per cohort is shown in Table S5. The prospectively assessed prevalence of persistent symptoms (Panel 1) in patients with physician-confirmed LB was 27•2% (95%CI, 24•7%−29•7%) (27•2% (95%CI, 24•6%−29•8%) in EM and 34•3% (95%CI, 21•7%−46•9%) in disseminated LB patients). This was significantly higher than the prevalence in the population (21•2%, 95%CI, 19•3%−23•1%, p < 0•0001) and tick bite cohorts (23•3%, 95%CI, 21•3%−25•3%, p = 0•016) (Fig. 2A). No significant difference in the prevalence of persistent symptoms was observed between the population and tick bite cohorts (p = 0•10). Sensitivity analyses with alternative substitution techniques for missing data and alternative analysis populations showed outcomes similar to those in the primary analysis (Table S4).
Fig. 1.
Enrollment and follow-up *Of which 557 individuals were screened online (www.tekenradar.nl). ‘Tekenradar’ (www.tekenradar.nl) is an online platform where individuals can report tick bites and LB diagnoses. This website was used for both inclusion and follow-up (questionnaires). **Some participants were excluded for more than one reason. ¶The sum of manifestations exceeds the number of included patients with Lyme borreliosis, as one patient was diagnosed with two concurrent manifestations. ‡1 patient had two concurrent skin lesions, denoted as EM as inclusion criteria for multiple EM were not met. #According to the inclusion criteria as defined in Table S1. Abbreviations: CFQ = Cognitive Failure Questionnaire, CIS = Checklist Individual Strength, EM = erythema migrans, LB = Lyme borreliosis, SF-36 = SF-36 item Health Survey.
Table 1.
Baseline characteristics.
| Reference cohorts |
|||||
|---|---|---|---|---|---|
| Characteristic | Population cohort (n = 4000) |
Tick bite cohort (n = 2405) |
All LB patients (n = 1135) |
EM (n = 1076) |
Disseminated LB (n = 59) |
| Demographic | |||||
| Male sex - no. (%) | 1892 (47.3) | 1064 (44.2) | 465 (41.0) | 432 (40.1) | 33 (55.9) |
| Age (years) | 57 (50–63) | 50 (39–60) | 55 (45–63) | 55 (45–63) | 53 (47–63) |
| Educational level - no. (%) Low Medium High |
505 (12.6) 1970 (49.2) 1525 (38.1) |
54 (2.2) 777 (32.3) 1574 (65.4) |
61 (5.4) 455 (40.1) 619 (54.5) |
57 (5.3) 428 (39.8) 591 (54.9) |
4 (6.8) 27 (45.8) 28 (47.5) |
| Status during past 3 months | |||||
| Paid work - no. (%) Absenteeism1 - no. (%) |
2515 (62.9) 135 (3.4) |
NA NA |
719 (63.4) 36 (3.2) |
682 (63.4) 25 (2.3) |
37 (62.7) 11 (18.6) |
| Healthcare consumption (number of visits) | 6 (0–54) | 1 (0–3) | 2 (0–4) | 2 (0–4) | 7 (4–12) |
| General health status | |||||
| Self-reported severe symptoms during >3 months in the past 12 months2 Fatigue - no. (%) Pain - no. (%) Concentration disorder - no. (%) |
192 (4.8) 116 (2.9) 78 (1.9) |
86 (3.6) 46 (1.9) 47 (2.0) |
45 (5.5) 36 (4.4) 21 (2.6) |
41 (5.2) 33 (4.2) 18 (2.3) |
4 (16.0) 3 (12.0) 3 (12.0) |
| Concomitant diagnoses (past 12 months)3 Yes - no. (%) Number of diagnoses |
2078 (51.9) 1 (0–1) |
1082 (45.0) 0 (0–1) |
613 (54.0) 1 (0–1) |
577 (53.6) 1 (0–1) |
36 (61.0) 1 (0–2) |
| Lyme borreliosis in history - no. (% of total cohort) | 253 (6.3) | 315 (13.1) | 101 (8.9) | 94 (8.7) | 7 (11.9) |
| Current Lyme borreliosis episode | |||||
| Noticed tick bite - no. (%) | NA | NA | 663 (58.8) | 647 (60.6) | 16 (27.1) |
| Duration of LB manifestation (days) | NA | NA | 6 (3–18) | 5 (3–14) | 208 (50–665) |
| Antibiotic treatment - no. (%) Doxycycline Amoxicillin Azithromycin Ceftriaxone (i.v.) Other - no. (%)4 |
NA |
NA |
1134 (99.9) 1042 (91.9) 48 (4.2) 22 (2.0) 14 (1.2) 8 (0.8) |
1078 (100) 1000 (92.9) 47 (4.4) 21 (2.0) 0 8 (0.7) |
58 (98.3) 42 (72.4) 1 (1.7) 1 (1.7) 14 (24.1) 0 |
| Current symptoms - no. (%)5 Pain Dizziness Collapse Palpitations Dyspnea Gastro-intestinal symptoms Sleeping disturbances Fatigue Cognitive symptoms |
2745 (68.6) 562 (14.1) 37 (0.9) 774 (19.4) 603 (15.1) 1169 (29.2) 1517 (37.9) 1636 (40.9) 988 (24.7) |
1620 (67.4) 353 (14.7) 19 (0.8) 403 (16.8) 273 (11.4) 643 (26.7) 742 (30.9) 902 (37.5) 579 (24.1) |
805 (70.9) 292 (25.7) 15 (1.3) 257 (22.6) 205 (18.1) 450 (39.6) 529 (46.6) 646 (56.9) 446 (39.3) |
755 (70.2) 273 (25.3) 12 (1.1) 236 (21.9) 188 (17.5) 424 (39.4) 489 (45.4) 600 (55.8) 410 (38.1) |
50 (84.7) 19 (32.2) 3 (5.1) 21 (35.6) 17 (28.8) 26 (44.1) 40 (67.8) 46 (78.0) 36 (61.0) |
Data are medians with interquartile ranges (IQR) for continuous values. For calculating percentages, participants with missing data were excluded, unless mentioned otherwise.
1Absenteeism ≥3 weeks from paid work in the past 3 months due to illness.
2Based on three questions on severe fatigue, pain or concentration disorder as indicated by the participant. This question was introduced during the course of study. Therefore, data are available from 811 LB patients (786 EM and 25 disseminated LB), and for the total population and tick bite cohorts. Data represent proportion of participants that answered this question.
3Reported comorbidity as listed in the Tic-P.
4Category 'other': amoxicillin/clavulanic acid, clarithromycin, clindamycin, phenoxymethylpenicillin, tetracycline. One patient with ACA refused antibiotic treatment.
5Based on the PHQ-15, assessing common (transient) symptoms in the past four weeks, to which a question on cognitive symptoms was added. The ordinal questionnaire scale (0–1–2), was converted to binary (0–1). The PHQ-15 symptoms related to pain (abdominal pain, back pain, arthralgia, dysmenorrhea, dyspareunia, head ache, and chest pain) were categorized as 'pain' in this table; nausea and diarrhea were grouped as 'gastro-intestinal symptoms'.
Abbreviations: ACA = acrodermatitis chronic atrophicans, EM = erythema migrans, i.v. = intravenously, LB = Lyme borreliosis, no. = number, PHQ-15 = Patient Health Questionnaire, TiC-P = Treatment Inventory of Costs in Patients with psychiatric disorders.
Fig. 2.
Prevalence of persistent symptoms Standardised prevalence (with 95% confidence intervals) of participants with persistent symptoms per cohort, comparing all LB patients, as well as the subset of patients with EM or disseminated LB, with the population (1) and tick bite (2) cohorts, using the primary scenario for substitution of missing data. (A) Percentage of participants meeting the definition of persistent symptoms (i.e., having at least one persistent symptom). (B-D) Percentage of participants reporting each individual persistent symptom. Participants may have reported two or more symptoms, therefore the sum of the percentages does not equal the overall prevalence of persistent symptoms. * p ≤ 0•05; ** p ≤ 0•01; *** p ≤ 0•001; ns = not significant. P values are provided in Table S6. Abbreviations: EM = erythema migrans, LB = Lyme borreliosis.
Fatigue was the most frequently reported persistent symptom in the LB, population and tick bite cohorts, followed by cognitive impairment and pain, which were often accompanied by fatigue. In disseminated LB patients, persistent pain was more frequently reported than cognitive impairment (Fig. 2B, Fig.S1A-C). In all cohorts, the majority of participants with persistent symptoms reported these symptoms from baseline through the final time point (Fig.S1D). While persistent symptoms were defined as lasting ≥6 months, prevalence of symptoms lasting ≥3 but <6 months was almost twice as high in LB patients (6•4%, 95%CI, 4•9%−7•8%) as in the population (3•3%, 95%CI, 2•4%−4•2%, p = 0•00,037) and tick bite cohorts (3•5%, 95%CI, 2•6%−4•3%, p = 0•00,026) (Table S7).
The mean severity scores for fatigue, cognitive impairment and pain during follow-up per cohort are shown in Fig. 3. In EM patients, baseline fatigue, cognitive impairment and pain were more severe compared with the population (p < 0•005) and tick bite (p<0•0001) cohorts, and still significantly more severe at the 12 months’ time point (Fig.S2, Table S8). In disseminated LB patients, baseline fatigue was more severe compared with both reference cohorts (p < 0•0001), but not at 12 months. Cognitive impairment severity was not significantly different compared with the reference cohorts. Pain was more severe at baseline (p < 0•0001), and although mean pain scores decreased over time, it was still significantly different from the reference cohorts at the end of follow-up. SF-36 physical and social functioning scores, reflecting the burden of symptoms, were particularly lower in disseminated LB patients during the first months of follow-up compared with the reference cohorts (Fig.S3A). LB patients who fulfilled the definition of persistent symptoms consistently reported higher symptom severity at all time points for all three symptoms than those without persistent symptoms (Fig. 3D-F), as well as lower SF-36 physical and social functioning scores (Fig.S3B).
Fig. 3.
Symptom severity Standardised mean severity scores for the CIS (subscale fatigue), CFQ, and SF-36 (subscale bodily pain) at five time points, with 95% confidence intervals. CIS subscale fatigue scores range between 8 and 56, a higher score indicating more fatigue, and a norm score of <35. CFQ scores range from 0 to 100, a higher score indicating more impaired cognitive functioning, and a norm score of <44. Scores on the SF-36, subscale bodily pain, range between 0 and 100, a higher score indicating less pain, and a norm score of >55. Dashed lines indicate norm scores thresholds. (A-C) Mean scores for LB patients with EM or disseminated LB, and the population and tick bite cohorts. (D-F) Mean scores for all LB patients who meet the definition of persistent symptoms and those who did not meet the definition of persistent symptoms. Abbreviations: CFQ = Cognitive Failure Questionnaire, CIS = Checklist Individual Strength, EM = erythema migrans, LB = Lyme borreliosis, SF-36 = SF-36 item Health Survey.
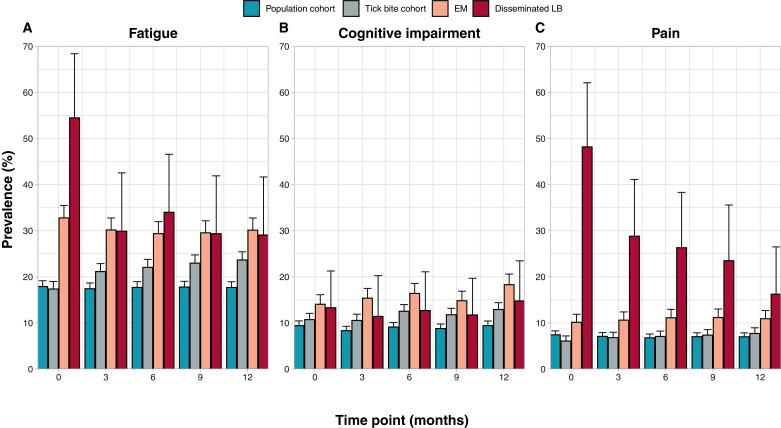
The mean prevalence of each symptom at individual time points was assessed as well (Fig. 4). Fatigue was the most prevalent symptom in all cohorts. In patients with EM, the prevalence of each symptom was stable over time, 30•1% (n = 263/873, 95%CI, 27•5%−32•8%) for fatigue, 18•3% (n = 160/873, 95%CI, 16•0%−20•6%) for cognitive impairment, and 10•9% (n = 95/873, 95%CI, 9•1%−12•7%) for pain at 12 months. About half of the disseminated LB patients reported fatigue or pain at baseline. From the 3 months’ time point onward, the prevalence of fatigue was similar to that of EM patients, while pain gradually declined. The prevalence of fatigue in the tick bite cohort increased from 17•4% (n = 418/2405, 95%CI, 15•8%−19•0%) at baseline to 23•7% (n = 263/1111, 95%CI, 21•9%−25•4%) at 12 months, while cognitive impairment and pain in this cohort were similar between time points.
Fig. 4.
Prevalence of impaired questionnaire scores at each time point Standardised prevalence of reported symptoms (with 95% confidence intervals), based on available impaired questionnaire scores, on each time point per cohort, for fatigue (A), cognitive impairment (B), and pain (C). Abbreviations: EM = erythema migrans, LB = Lyme borreliosis.
Finally, 65 patients with chronic symptoms attributed to unconfirmed LB were included. These patients were significantly younger than the 295 physician-confirmed LB patients who reported persistent symptoms (median age, 39y versus 54y), had less frequently paid work (39•1% versus 60•0%), and absenteeism in the subgroup of patients with paid work was significantly higher (36•0% versus 9•6%). The median duration of their symptoms was over four years. At baseline, fatigue was more severe than in LB patients with persistent symptoms, whereas cognitive impairment was less severe, and pain scores were similar. During follow-up, fatigue and pain severity slightly decreased, but remained impaired with respect to the norm scores (Fig.S4, Table S8). Physical and social functioning were more impaired in the patients with chronic symptoms attributed to unconfirmed LB throughout the entire follow-up (Fig.S5).
4. Discussion
In this large prospective observational cohort of patients treated for physician-confirmed LB, prevalence of persistent fatigue, cognitive impairment or pain during one year of follow-up was 3•9% and 6•0% higher than the background prevalence in two reference cohorts. The mean symptom severity in LB patients was significantly higher as well, compared with the reference cohorts. Furthermore, persistent symptoms were associated with impaired physical and social functioning, reflecting a substantial burden of symptoms.
The reference cohorts, a population and a tick bite cohort undergoing the same prospective questionnaire surveys, enabled correction for the substantial background prevalence of persistent symptoms (21•2% and 23•3%, respectively), regardless of their numerous possible causes. The prevalence of severe fatigue (CIS score ≥35) at baseline in both reference cohorts was consistent with a recent study in which 18% of 78,363 individuals from the general population reported severe fatigue on a single measurement.25 The higher prevalence of persistent symptoms in LB patients compared with the reference cohorts points at an association with LB, albeit in a subset of patients. The observation that around 70% of participants with persistent symptoms reported persistent symptoms from baseline onward suggests that those symptoms may be related to pre-existent conditions. However, the prevalence of persistent symptoms in subgroups of individuals without self-reported severe fatigue, pain or concentration disorders prior to baseline remained significantly higher in LB patients, further supporting an association of the LB episode with increased prevalence of persistent symptoms. Whether these symptoms are specific for LB or may be due to a general post infectious syndrome or other mechanism requires further research. Other infectious diseases, such as Q-fever or Epstein-Barr virus infection, and more recently COVID-19, as well as various non-infectious diseases, have also been associated with long-term persistent symptoms.[26], [27], [28], [29]
The tick bite cohort was included in the study because it was comparable to the LB cohort in terms of the online methods for inclusion and follow-up. Individuals with a subsequent diagnosis of LB were excluded from this cohort, and potential effect of unnoticed or non-reported LB manifestations was limited at most, since the primary outcomes did not differ significantly between tick bite and population cohorts.
The primary outcome was persistent symptoms lasting for at least six months, but approximately 6% of LB patients reported shorter-term symptoms (≥3 and <6 months). This prevalence was twice as high as in the reference cohorts, suggesting that a more substantial part of these shorter-lasting symptoms might be specifically related to the LB episode.
Of the three symptoms assessed, pain was the least frequently reported symptom by EM patients and both reference cohorts. However, almost 50% of disseminated LB patients reported severe pain at baseline, gradually decreasing during follow-up. The majority of these patients was diagnosed with acrodermatitis chronica atrophicans (ACA), followed by Lyme neuroborreliosis and Lyme arthritis, which are commonly accompanied by pain.2
In the cohort of patients with chronic symptoms self-attributed to unconfirmed LB, who were included if they had reported symptoms for six or more months prior to baseline, the mean severity of symptoms was substantial throughout the one year follow-up. In these relatively young individuals, SF-36 physical and social functioning scores were even lower than in the group of LB patients meeting the definition of persistent symptoms. This underscores that symptoms in these patients, regardless of the etiology, are severe, and greatly impact daily life.
Severe long-lasting symptoms in patients treated for LB have been described before, although in widely varying frequencies. Differences in study design, geographical location, LB case definition, outcome measures, follow-up duration and inclusion of control groups have contributed to differences between study outcomes. Prospective studies in patients with early localised LB have identified persistent symptoms in 5•7% to 11•0% of patients.5,7,30 Studies in patients specifically diagnosed with Lyme neuroborreliosis have generally demonstrated higher frequencies of residual symptoms averaging 28% in 34 studies with up to 20 years of follow-up, although findings are inconsistent with regard to quality of life and fatigue measures.8,31 Prospective studies in Europe and the USA comparing the prevalence of persistent symptoms after EM with symptom prevalence in control groups have found no increased frequency of symptoms.4,9,32 However, most of these studies were not powered to detect significant differences against a high background symptom prevalence, as the present study was, enrolling a total of 7540 participants.
In this unique and largest prospective LB study to date, specific efforts were made to select patients meeting strict, pre-defined definitions for LB and persistent symptoms, and to select appropriate longitudinal reference cohorts. The substantial background prevalence observed in this study underscores the need for such reference cohorts in future studies addressing persistent symptoms after treatment for LB. This accounts for other (infectious) diseases as well, which has become even more relevant in light of the COVID-19 pandemic.29 A strict laboratory confirmation was required for disseminated LB manifestations. For EM, clinical diagnoses were physician-confirmed following international clinical case definitions33, and EM photographs were reviewed by an expert panel. To assess the potential impact of clinical misdiagnoses, a sensitivity analysis on patients with highly probable or proven EM yielded a similar prevalence of persistent symptoms as the primary analysis. As the definition of persistent symptoms may greatly impact study outcomes, the primary outcome in this study was based on the PTLDS criteria, that have been used in multiple other studies.2 Typical PTLDS symptoms (fatigue, pain and cognitive impairment) were systematically assessed by standardised questionnaires, with pre-defined cut-off scores for clinical relevance. Outcomes of these questionnaires reflect the patients’ perception of symptoms, and thus the impact on their daily life, rather than objectify reported symptoms. In line with previous studies, this study indicates that self-reported symptoms are clinically relevant with regard to daily functioning.34 We recommend that this definition of persistent symptoms is used in future studies into this topic, allowing for comparison of findings between studies. Lastly, the longitudinal assessment of symptoms enabled describing the course and severity of symptoms over time.
This study has several limitations. First, missing observations occurred during follow-up, that were substituted according to pre-defined scenarios. Robustness of the prevalence of persistent symptoms was confirmed by similar findings in several sensitivity analyses, including one without substitution of missing observations. Second, follow-up ended 12 months after treatment, whereas the majority of patients with persistent symptoms reported ongoing symptoms at the last time point. Although sequelae have been reported many years post treatment,1 and may disappear over time, insights into the course and impact of persistent symptoms more than one year after LB remain limited. Third, it could be hypothesized that ongoing infection despite treatment may underlie persistent symptoms. Therefore, at all follow-up time points, LB patients were asked to report new LB diagnoses or manifestations. Nine patients reported LB manifestations during follow-up, none of which was suspicious for relapse of the prior infection. Thus, it seems unlikely that treatment failure has a large share in the total number of patients with persistent symptoms. Fourth, because of the relatively small number of disseminated LB patients in this study, the results contribute particularly to the insights into the prognosis after EM, the most frequent LB manifestation.3 Fifth, fatigue, cognitive impairment and pain are widespread in the general population, as reflected by the reference cohorts. Elaborating the origin and explainability of symptoms in individual patients was therefore not possible. The significant difference in prevalence of persistent symptoms between LB patients and reference cohorts points at an association of symptoms with LB in only a subset of the LB cohort. Finally, due to widespread attention for chronic symptoms after LB, patients diagnosed with LB may have had more focus on perceiving symptoms than individuals in the reference cohorts. However, rather than a response bias, this may reflect a potential cognitive-behavioral mechanism for persistence of symptoms in LB patients in general. As part of ongoing analyses that will be reported separately, potential cognitive-behavioral determinants for persistence of symptoms in this cohort will be investigated, besides numerous other predefined factors.
In conclusion, this study shows that the prevalence of persistent symptoms in patients treated for Lyme borreliosis was 3•9% and 6•0% higher than the substantial background prevalence in two reference cohorts. Furthermore, symptoms were more severe in LB patients than in the reference cohorts. These findings suggest that Lyme borreliosis may induce persistent symptoms, albeit in a small proportion of patients. Since many patients seek medical help for their often disabling long-lasting symptoms attributed to LB, these findings are relevant for clinicians confronted with patients with persistent symptoms after treatment for Lyme borreliosis. In addition, these observations call for further research into the underlying mechanisms of persistent symptoms after treatment for LB – whether or not directly related to an infection with B. burgdorferi s.l. – which may lead to the development of more specific preventive and treatment strategies.
Data sharing
De-identified data that underlie the results reported in this article, as well as the statistical analysis plan, are available upon request for a period of 36 months after publication of intended manuscripts. Requests from qualified researchers with appropriate ethics board approvals and relevant data use agreements will be processed after contact by email with the corresponding author.
Declaration of Competing Interest
All authors have completed the ICMJE uniform disclosure form and declare: JWH, LABJ, and CCvdW report grants from the Netherlands Organization for Health Research and Development (ZonMw), and the Dutch Ministry of Health, Welfare and Sport (VWS); LABJ has a patent on Lyme diagnosis issued; JWH was supported by the European Union's regional development fund (INTERREG) as part of the NorthTick project; all other authors have no conflicts to declare.
Acknowledgements
This study was funded by the Netherlands Organization for Health Research and Development, (ZonMw) (project number 522001003), and the Dutch Ministry of Health, Welfare and Sport (VWS). We gratefully acknowledge the following persons for their contributions to this study: M.E. Baarsma, K. van Beurden, R. van Boxtel, H. Dijkstra, J. Ersöz, E. Franz, H. ter Hofstede, M. Kerstholt, B. van Kooten, K. Kremer, H. Lemmers, G.A. Oei, F. van de Schoor, M. Starink, A. van Vliet, H. van Weert, A. Wijmenga, M. Zwerink; and all the study participants for their involvement and use of their data in these analyses.
Footnotes
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.lanepe.2021.100142.
Appendix. Supplementary materials
References
- 1.Mac S., Bahia S., Simbulan F. Long-term sequelae and health-related quality-of-life associated with lyme disease: a systematic review. Clin Infect Dis: An Off Pub Of The Infect Dis Soc of Am. 2019 doi: 10.1093/cid/ciz1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wormser G., Dattwyler R., Shapiro E. The clinical assessment, treatment, and prevention of lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the infectious diseases society of america. Clin Infect Dis. 2006;43(9):1089–1134. doi: 10.1086/508667. [DOI] [PubMed] [Google Scholar]
- 3.Hofhuis A., Harms M., Bennema S., van den Wijngaard C.C., van Pelt W. Physician reported incidence of early and late lyme borreliosis. Paras Vect. 2015;8:161. doi: 10.1186/s13071-015-0777-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cerar D., Cerar T., Ruzic-Sabljic E., Wormser G.P., Strle F. Subjective symptoms after treatment of early lyme disease. Am J Med. 2010;123(1):79–86. doi: 10.1016/j.amjmed.2009.05.011. [DOI] [PubMed] [Google Scholar]
- 5.Weitzner E., McKenna D., Nowakowski J. Long-term assessment of post-treatment symptoms in patients with culture-confirmed early lyme disease. Clin Infects Dis: An Off Pub Of The Infect Dis Soc of Am. 2015;61(12):1800–1806. doi: 10.1093/cid/civ735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eikeland R., Mygland A., Herlofson K., Ljostad U. European neuroborreliosis: quality of life 30 months after treatment. Acta Neurol Scand. 2011;124(5):349–354. doi: 10.1111/j.1600-0404.2010.01482.x. [DOI] [PubMed] [Google Scholar]
- 7.Aucott J.N., Crowder L.A., Kortte K.B. Development of a foundation for a case definition of post-treatment lyme disease syndrome. Int J Of Infect Dis: IJID: Off Pub Of The Int Soc For Infect Dis. 2013;17(6):e443–e449. doi: 10.1016/j.ijid.2013.01.008. [DOI] [PubMed] [Google Scholar]
- 8.Dersch R., Sommer H., Rauer S., Meerpohl J.J. Prevalence and spectrum of residual symptoms in lyme neuroborreliosis after pharmacological treatment: a systematic review. J Neurol. 2016;263(1):17–24. doi: 10.1007/s00415-015-7923-0. [DOI] [PubMed] [Google Scholar]
- 9.Wormser G.P., McKenna D., Karmen C.L. Prospective evaluation of the frequency and severity of symptoms in lyme disease patients with erythema migrans compared with matched controls at baseline, 6 months, and 12 months. Clin Infect Dis: An Off Pub Of The Inf Dis Soc of Am. 2020 doi: 10.1093/cid/ciz1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ljostad U., Mygland A. Remaining complaints 1 year after treatment for acute lyme neuroborreliosis; frequency, pattern and risk factors. Eur J Neurol. 2010;17(1):118–123. doi: 10.1111/j.1468-1331.2009.02756.x. [DOI] [PubMed] [Google Scholar]
- 11.Wormser G.P., Weitzner E., McKenna D., Nadelman R.B., Scavarda C., Nowakowski J. Long-term assessment of fatigue in patients with culture-confirmed lyme disease. Am J Med. 2015;128(2):181–184. doi: 10.1016/j.amjmed.2014.09.022. [DOI] [PubMed] [Google Scholar]
- 12.Kalish R.A., Kaplan R.F., Taylor E., Jones-Woodward L., Workman K., Steere A.C. Evaluation of study patients with lyme disease, 10-20-year follow-up. J Infect Dis. 2001;183(3):453–460. doi: 10.1086/318082. [DOI] [PubMed] [Google Scholar]
- 13.Baker P.J. Straight talk about chronic lyme disease. Am J Med. 2018;131(6):592–594. doi: 10.1016/j.amjmed.2017.11.050. [DOI] [PubMed] [Google Scholar]
- 14.Berende A., ter Hofstede H.J., Vos F.J. Randomized trial of longer-term therapy for symptoms attributed to lyme disease. The New Eng J Of Medi. 2016;374(13):1209–1220. doi: 10.1056/NEJMoa1505425. [DOI] [PubMed] [Google Scholar]
- 15.Kullberg B.J., Vrijmoeth H.D., van de Schoor F., Hovius J.W. Lyme borreliosis: diagnosis and management. BMJ. 2020;369:m1041. doi: 10.1136/bmj.m1041. [DOI] [PubMed] [Google Scholar]
- 16.Vrijmoeth H.D., Ursinus J., Harms M.G. Prevalence and determinants of persistent symptoms after treatment for lyme borreliosis: study protocol for an observational, prospective cohort study (lymeprospect) BMC Infect Dis. 2019;19(1):324. doi: 10.1186/s12879-019-3949-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vercoulen J., Swanink C., Fennis J., Galama J., van der Meer J., Bleijenberg G. Dimensional assessment of chronic fatigue syndrome. J Psychosom Res. 1994;38(5):383–392. doi: 10.1016/0022-3999(94)90099-x. [DOI] [PubMed] [Google Scholar]
- 18.Worm-Smeitink M., Gielissen M., Bloot L. The assessment of fatigue: psychometric qualities and norms for the Checklist individual strength. J Psychosom Res. 2017;98:40–46. doi: 10.1016/j.jpsychores.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 19.Vercoulen J., Alberts M., Bleijenberg G. De checklist individual strength (CIS) Gedragstherapie. 1999;32:131–136. [Google Scholar]
- 20.Ponds R., Van Boxtel M., Jolles J. De cognitive failure questionnaire als maat voor subjectief cognitief functioneren. Tijdschrift voor Neuropsychologie. 2006;2:37–45. [Google Scholar]
- 21.VanderZee K.I., Sanderman R., Heyink J.W., de Haes H. Psychometric qualities of the RAND 36-Item Health Survey 1.0: a multidimensional measure of general health status. Int J Behav Med. 1996;3(2):104–122. doi: 10.1207/s15327558ijbm0302_2. [DOI] [PubMed] [Google Scholar]
- 22.Aaronson N., Muller M., Cohen P. Translation, validation, and norming of the Dutch language version of the SF-36 Health Survey in community and chronic disease populations. J Clin Epidemiol. 1998;51(11):1055–1068. doi: 10.1016/s0895-4356(98)00097-3. [DOI] [PubMed] [Google Scholar]
- 23.van der Zee K.I., Sanderman R. Research Institute SHARE; 2012. Het meten van de algemene gezondheidstoestand met de RAND-36, een handleiding: umcg /Rijksuniversiteit groningen. [Google Scholar]
- 24.Hofhuis A., van de Kassteele J., Sprong H. Predicting the risk of Lyme borreliosis after a tick bite, using a structural equation model. PLoS ONE. 2017;12(7) doi: 10.1371/journal.pone.0181807. e0181807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Goërtz Y.M.J., Braamse A.M.J., Spruit M.A. 2021. Fatigue in patients with chronic disease: towards a trans-diagnostic risk prediction model. in preparation. [Google Scholar]
- 26.Morroy G., Keijmel S.P., Delsing C.E. Fatigue following Acute Q-Fever: a Systematic Literature Review. PLoS ONE. 2016;11(5) doi: 10.1371/journal.pone.0155884. e0155884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hickie I., Davenport T., Wakefield D. Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study. BMJ. 2006;333(7568):575. doi: 10.1136/bmj.38933.585764.AE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Menting J., Tack C.J., Bleijenberg G. Is fatigue a disease-specific or generic symptom in chronic medical conditions? Health Psychol. 2018;37(6):530–543. doi: 10.1037/hea0000598. [DOI] [PubMed] [Google Scholar]
- 29.Huang C., Huang L., Wang Y. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397(10270):220–232. doi: 10.1016/S0140-6736(20)32656-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sjowall J., Fryland L., Nordberg M. Decreased Th1-type inflammatory cytokine expression in the skin is associated with persisting symptoms after treatment of erythema migrans. PLoS ONE. 2011;6(3):e18220. doi: 10.1371/journal.pone.0018220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dersch R., Sarnes A.A., Maul M. Quality of life, fatigue, depression and cognitive impairment in Lyme neuroborreliosis. J Neurol. 2015;262(11):2572–2577. doi: 10.1007/s00415-015-7891-4. [DOI] [PubMed] [Google Scholar]
- 32.Stupica D., Lusa L., Ruzic-Sabljic E., Cerar T., Strle F. Treatment of erythema migrans with doxycycline for 10 days versus 15 days. Clin Infect Diss: An Off Pub Of The Infect Dis Soc of Am. 2012;55(3):343–350. doi: 10.1093/cid/cis402. [DOI] [PubMed] [Google Scholar]
- 33.Stanek G., Fingerle V., Hunfeld K.P. Lyme borreliosis: clinical case definitions for diagnosis and management in europe. Clin Microbiol And Infect: The Off Pub Of The Euro Soc Of Clin Microbiol And Infect Dis. 2011;17(1):69–79. doi: 10.1111/j.1469-0691.2010.03175.x. [DOI] [PubMed] [Google Scholar]
- 34.Berende A., Agelink van Rentergem J., Evers A.W.M. Cognitive impairments in patients with persistent symptoms attributed to Lyme disease. BMC Infect Dis. 2019;19(1):833. doi: 10.1186/s12879-019-4452-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.