Abstract
Objective
The demand for spinal fusion is increasing, with concurrent reports of iatrogenic adult spinal deformity (flatback deformity) possibly due to inappropriate lordosis distribution. This distribution is assessed using the lordosis distribution index (LDI) which describes the upper and lower arc lordosis ratio. Maldistributed LDI has been associated to adjacent segment disease following interbody fusion, although correlation to later-stage deformity is yet to be assessed. We therefore aimed to investigate if hypolordotic lordosis maldistribution was associated to radiographic deformity-surrogates or revision surgery following instrumented lumbar fusion.
Methods
All patients undergoing fusion surgery (≤ 4 vertebra) for degenerative lumbar diseases were retrospectively included at a single center. Patients were categorized according to their postoperative LDI as: “normal” (LDI 50–80), “hypolordotic” (LDI<50), or “hyperlordotic” (LDI>80).
Results
We included 149 patients who were followed for 21±14 months. Most attained a normally distributed lordosis (62%). The hypolordotic group had increased postoperative pelvic tilt (PT) (p<0.001), pelvic incidence minus lumbar lordosis (PI–LL) mismatch (p<0.001) and decreased global lordosis (p=0.007) compared to the normal group. Survival analyses revealed a significant difference in revision surgery (p=0.03), and subsequent multivariable logistic regression showed increased odds of 1-year revision in the hypolordotic group (p=0.04). There was also a negative, linear correlation between preoperative pelvic incidence (PI) and postoperative LDI (p<0.001).
Conclusion
In patients undergoing instrumented lumbar fusion surgery, hypolordotic lordosis maldistribution (LDI<50) was associated to increased risk of revision surgery, increased postoperative PT and PI–LL mismatch. Lordosis distribution should be considered prior to spinal fusion, especially in high PI patients.
Keywords: Lordosis distribution, Spine fusion, Lumbar spine, Ideal lordosis, Adult spinal deformity
INTRODUCTION
Degenerative lumbar spine diseases are common and related to pain and disability. These conditions are among the most important causes of decreased health-related quality of life, and often treated using spinal fusion [1-4]. Meanwhile, iatrogenic causes of adult spinal deformity is a growing concern—possibly due to previous fusion in relative kyphosis (flatback deformity) [5,6]. Adult spinal deformity is a condition related to severe pain, disability, in addition to high procedure-related costs and complications including mechanical complications requiring revision surgery [7-10]. Hypolordosis is established as a risk-factor of iatrogenic deformity while further in-depth assessment, beside the extent, of lordosis is not well understood.
Optimal lordosis is also well established as a cornerstone in treating patients with adult spinal deformity and should reflect pelvic incidence (PI). A PI minus lumbar lordosis (PI–LL) mismatch has been associated with poor postoperative outcome [11,12]. By fusing part of the lumbar segments, the lumbar curvature may be compromised or not adequately restored to suit the ideal sagittal shape [13]. Symptomatic loss of lumbar lordosis can cause sagittal imbalance or deterioration of lordosis and may require major, extensive revision surgery. So-called iatrogenic deformity is considered to be a leading cause of adult spinal deformity besides degenerative ageing processes [5,6]. Lordosis extent and PI–LL interaction are parameters to consider prior to fusion surgery; however, the PI–LL concept may be simplifying a more complex issue. Distribution of lordosis between the upper and lower arc has been proposed as a considerable factor in attaining a well-balanced spine and in reducing postoperative complications [14]. Berthonnaud et al. [15] proposed to separate the lordosis into 2 segments. Roussouly and Pinheiro-Franco [16] further elaborated the concept and demonstrated how the lower arc of lordosis should represent 2/3 of global lordosis, as proposed by Barrey et. al. [17]. Finally, the lordosis distribution index (LDI) was proposed as a ratio of lordosis distribution between the lower arc (L4–S1) and the global lordosis [14]. LDI is calculated as a ratio from 0-100 characterizing the increasing lordosis towards the lower segments (Fig. 1). Normal values span from 50%–80%, LDI<50% suggests hypolordotic maldistribution and LDI> 80% suggests hyperlordotic maldistribution. In a recent study of patients undergoing lumbar interbody fusion, the authors found that maldistributed lordosis was associated to postoperative adjacent segment disease (ASD) [18]. To our knowledge, the LDI has not been assessed in patients undergoing instrumented spine surgery which we sought to assess.
Fig. 1.
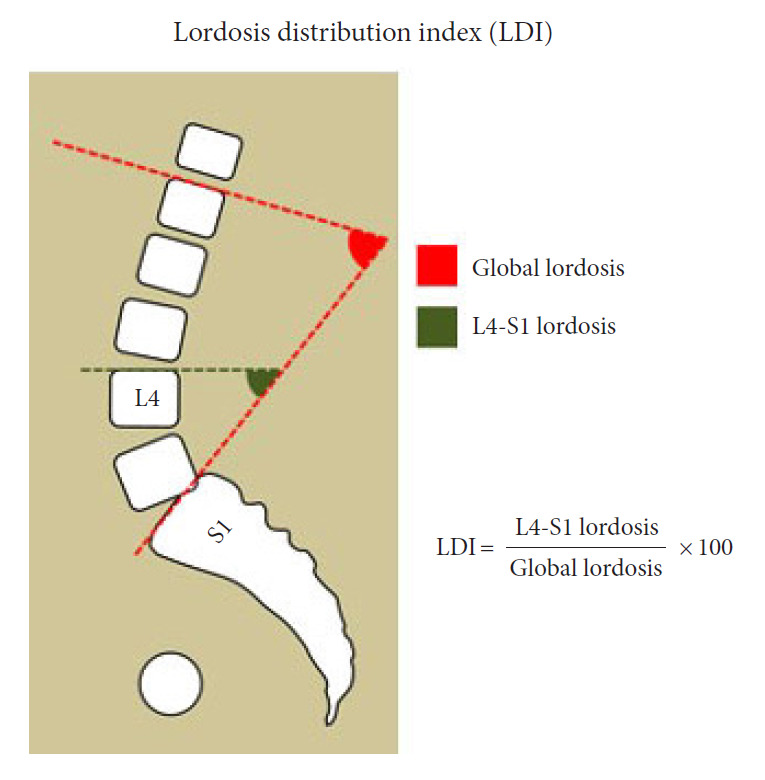
Lordosis distribution index (LDI). The LDI is calculated as a ratio between the lower lordosis (L4–S1) and the global lordosis. The LDI should reflect the increasing lordosis towards the lower spinal segments.
MATERIALS AND METHODS
1. Study Design
We retrospectively screened all patients undergoing short-segment instrumented spine surgery for degenerative lumbar pathologies in a 2-year period from January 1st, 2015 through December 31st, 2016 at a single tertiary institution. Short-segment fusion was defined as instrumented fusion of ≤ 4 vertebrae. Only adult patients (≥ 18 years) were eligible for inclusion. Exclusion criteria were: a history of previous instrumented fusion. Inclusion criteria were sufficient preoperative or postoperative radiographs including both femoral heads, sacral endplate, and all lumbar vertebras up until the inflection point to the thoracic kyphosis. This study was retrospective, noninterventional and LDI was therefore not considered at the time of surgery. This study was approved by the National Health and Medical authority and The National Data Protection Agency.
2. Patient Sample
Patients screened for inclusion underwent surgical treatment for degenerative lumbar spine pathologies including spondylolisthesis (< grade 3), spinal stenosis, disc herniation, degenerative disc disease, or a combination. Surgical treatment included posterior instrumented fusion of 2–4 vertebral levels. Decompression or interbody fusion was performed when deemed necessary. Patient characteristics, medical history, and surgical data were obtained using electronic medical records. Follow-up consisted of clinical and radiographic assessment at 3 months and 1-year following surgery. Radiographic measurements were performed using the online imaging system KEOPS (SMAIO, Lyon, France) [19]. LDI was calculated as the ratio between the lower arc (upper endplate of L4 to S1) and the global lordosis (lordosis/kyphosis inflection point as upper limit). Patients were subcategorized according to their postoperative LDI as: normal (LDI 50–80), hypolordosis (LDI<50), or hyperlordosis (LDI>80). Postoperative complications were registered and subcategorized as minor or major. Major complications were defined as a complication leading to prolonged length of hospital stay (LOS) (> 75th percentile), intensive care, invasive procedures, permanent effect on outcome (e.g., neural injury) or death. Complications were further categorized as “mechanical”: proximal junctional kyphosis or proximal junctional failure; distal junctional failure; rod breakage; or other. The primary outcome was revision surgery excluding revision due to hematoma, wound dehiscence, and infection. Secondary outcomes were other recorded complications and radiographic parameters related to poor outcome in patients with adult spinal deformity. Patient-reported outcome measures were not evaluated.
3. Statistical Analyses
We performed all statistical analyses using the language and environment R (R Core Team 2020, Vienna, Austria) version 4.01 [20-25]. Data distribution was assessed using histograms and reported as means with standard deviations (SDs), medians with interquartile ranges (IQRs), or proportions (%). Student t-test was used to compare approximated Gaussian distributed data and Wilcoxon rank-sum test for non-Gaussian data (paired tests when comparing pre- and postoperative parameters). Categorical variables were analyzed using Pearson chi-square test of independence or Fisher exact test when the expected counts were below 5 for 20% of frequencies. Analysis of variance (ANOVA) was performed when comparing LDI groups, followed by pairwise analyses as described above. Correlation between preoperative PI and postoperative LDI was assessed using linear regression. Uni- and multivariable logistic regression was used to assess postoperative LDI on 1-year revision, adjusted for number of instrumented vertebra and preoperative PI. Overall revision was estimated using Kaplan-Meier (KM) survival analyses [26,27], plotted as cumulative incidences (1-KM), stratified by LDI subgroups and compared using log-rank tests. Estimated parametric accelerated failure was further assessed for all 3 groups using a Weibull regression model (proportional and accelerated) for relative event rates and relative extension in survival time [28,29]. Results were presented as odds ratios (ORs) with 95% confidence intervals (CIs). All p-values are 2-sided, unadjusted for multiple comparisons and were considered significant if < 0.05.
RESULTS
We identified 249 adult patients undergoing instrumented fusion of which 75 (30%) were excluded due to history of previous fusion. Out of 174 eligible patients, 23 (13%) were excluded due to insufficient preoperative radiographs and 2 (1%) due to insufficient postoperative radiographs leaving 149 for final analyses (eligibility ratio: 86%). Table 1 details patient characteristics. Mean age±SD at time of surgery was 59±13 and a majority of patients were female (66%, n=98). The most common etiology was a combination of multiple pathologies (34%), followed by spondylolisthesis (22%), disc herniation (14%), and spinal stenosis (12%). Previous noninstrumented spinal surgery was common (43%, n=64). Surgery was performed for a mean of 177±51 minutes, most cases involved interbody cages (79%, n=118) and the median (IQR) number of instrumented vertebrae was 3 (3–4). A majority of procedures included the S1 (n=108, 72%). Median (IQR) LOS was 4 days (4–6 days) and patients were followed for a mean±SD of 21±14 months.
Table 1.
Patient characteristics and surgical data (n=149)
| Characteristic | Value |
|---|---|
| Age (yr) | 59 ± 13 |
| Female sex | 98 (66) |
| Etiology | |
| Multiple | 51 (34) |
| Listhesis | 32 (22) |
| Disc herniation | 21 (14) |
| Degenerative disc disease | 18 (12) |
| Stenosis | 16 (11) |
| Other | 11 (7) |
| Carlson Comorbidity Index | 2± 2 |
| ASA PS classification | 2± 1 |
| Surgery time (min) | 177 ± 51 |
| Interbody cage | 118 (79) |
| Minimal invasive | 4 (3) |
| Instrumented vertebra | 3 (3–4) |
| Length of stay | 4 (4–6) |
| Follow-up (mo) | 21 ± 14 |
| Previous spine surgery | 64 (43) |
Values are presented as mean±standard deviation, number (%), or median (interquartile range).
ASA PS, American Society of Anesthesiologists physical status.
1. Sagittal Radiographic Parameters
Differences between preoperative and postoperative radiographic measurements were assessed and found with significant increase in pelvic tilt (PT) (18°±9° vs. 20°±9°, p<0.001) and PI–LL (4°±14° vs. 7°±14°, p<0.001). These remained significant at 1-year follow-up (p<0.001). A modest, although significant, decrease was seen in sacral slope (37°±11° vs. 35°±11°, p<0.001) and without significant difference at 1-year follow-up (p=0.072). Global lordosis decreased from 53°±14° preoperatively to 49°±13° postoperatively (p<0.001) and remained decreased at 1-year follow-up (50°±15°, p<0.001). Long-standing radiographs were only available in select patients, as deemed by the surgeon, although analyses were performed when available and without significant differences between groups.
2. LDI and Sagittal Parameters
Mean±SD postoperative LDI was 59%±22% and without significant difference when compared to preoperative (62%±23%) and 1-year measurements (59%±22%). Patients were further subcategorized according to their postoperative LDI: 93 (62%) were classified as normal (LDI 50–80), 36 (24%) as hypolordotic (LDI<50) and 20 (13%) as hyperlordotic (LDI>80). Table 2 details radiographic parameters according LDI groups and ANOVA suggested differences between groups in both preoperative and postoperative PI, PT, sacral slope, global lordosis, and PI–LL. Patients with normal postoperative LDI had predominantly normal preoperative LDI (74%). Similarly, patients with hypolordotic postoperative LDI were mainly hypolordotic preoperatively (71%) and patients with postoperative hyperlordosis were also hyperlordotic preoperatively (75%). Subsequent pairwise analyses were performed comparing the postoperative normal and hypolordotic groups. Results showed that the hypolordotic LDI group, compared to the normal LDI group, had increased preoperative PT (22°±10° vs. 17°±8°, p=0.03) and 50% had a preoperative PT > 20° compared to 29% in the normal group (p=0.039). This increase remained significant postoperatively (25°±11° vs. 18°±8°, p<0.001). PI–LL was also increased in the hypolordotic group, both preoperatively (12°±17° vs. 0°±12°, p<0.001) and postoperatively (16°±17° vs. 3°±11°, p<0.001). Finally, postoperative global lordosis was smaller in the hypolordotic group compared to the normal (45°±13° vs. 52°±12°, p=0.007) whilst remaining parameters were without significant differences.
Table 2.
Radiographic parameters and complications according to lordosis distribution index (LDI) groups
| Variable | Normal (LDI: 50–80) (n = 93) | Hypolordosis (LDI < 50) (n = 36) | Hyperlordosis (LDI > 80) (n = 20) | Total (n = 149) | p-value† | p-value‡ | |
|---|---|---|---|---|---|---|---|
| Preoperative radiographic measurements | |||||||
| Pelvic incidence (°) | 54.5 ± 13.7 | 58.8 ± 13.5 | 47.1 ± 11.2 | 54.6 ± 13.7 | 0.007* | 0.112 | |
| Pelvic tilt (°) | 16.5 ± 8.1 | 22.3 ± 10.1 | 18 ± 6 | 18.1 ± 8.7 | 0.002* | 0.003* | |
| Pelvic tilt > 20° | 27 (29.0) | 18 (50.0) | 7 (35.0) | 52 (34.9) | 0.081 | 0.039* | |
| Sacral slope (°) | 38 (11.4) | 36.6 (8.2) | 29.1 (10.9) | 36.5 (11) | 0.003* | 0.426 | |
| Global lordosis (°) | 55.7 ± 14.7 | 50.8 ± 13.2 | 43.9 ± 10.9 | 53 ± 14.4 | 0.001* | 0.069 | |
| SVA (mm)§ | 34.5 ± 49 | 61.1 ± 51.5 | 47.3 ± 32.6 | 43.3 ± 47.8 | 0.111 | 0.064 | |
| SVA > 40 mm§ | 15 (33.3) | 11 (57.9) | 7 (46.7) | 33 (41.8) | 0.174 | 0.096 | |
| PI–LL (°) | 0.1 ± 11.5 | 11.7 ± 17.2 | 8 ± 11.7 | 4 ± 14 | < 0.001* | < 0.001* | |
| PI–LL ≥ 10° | 12 (12.9) | 16 (44.4) | 7 (35.0) | 35 (23.5) | < 0.001* | < 0.001* | |
| LDI | 65.2 ± 14.4 | 38.5 ± 24.8 | 85.8 ± 15.7 | 61.5 ± 22.9 | < 0.001* | < 0.001* | |
| LDI groups | < 0.001* | < 0.001* | |||||
| 50–80 | 69 (74.2) | 9 (25.7) | 5 (25.0) | 83 (56.1) | |||
| < 50 | 12 (12.9) | 25 (71.4) | 0 (0.0) | 37 (25.0) | |||
| > 80 | 12 (12.9) | 1 (2.9) | 15 (75.0) | 28 (18.9) | |||
| Postoperative radiographic measurements | |||||||
| Pelvic incidence (°) | 54.6 ± 14.1 | 58.5 ± 13.5 | 47 ± 10.8 | 54.5 ± 13.9 | 0.009* | 0.145 | |
| Pelvic tilt (°) | 17.8 ± 7.9 | 25.1 ± 11.2 | 20.8 ± 5.5 | 20 ± 9 | < 0.001* | < 0.001* | |
| Pelvic tilt > 20° | 38 (40.9) | 22 (61.1) | 12 (60.0) | 72 (48.3) | 0.063 | 0.049* | |
| Sacra slope (°) | 36.9 ± 10.1 | 33.5 ± 8.9 | 26.2 ± 9.7 | 34.6 ± 10.4 | < 0.001* | 0.065 | |
| Global lordosis (°) | 52.2 ± 12 | 45.3 ± 12.6 | 40.6 ± 11.3 | 48.9 ± 12.8 | < 0.001* | 0.007* | |
| SVA (mm)§ | 68.8 ± 54.4 | 127.5 ± 41.9 | 88.7 ± 51.8 | 91.4 ± 53.1 | 0.195 | 0.092 | |
| SVA > 40 mm§ | 4 (66.7) | 4 (100.0) | 3 (100.0) | 11 (84.6) | 0.252 | 0.467 | |
| PI–LL (°) | 3.2 ± 11.1 | 16.1 ± 16.9 | 11 ± 11.4 | 7.4 ± 13.9 | < 0.001* | < 0.001* | |
| PI–LL ≥ 10° | 21 (22.6) | 19 (52.8) | 11 (55.0) | 51 (34.2) | < 0.001* | 0.001* | |
| LDI | 63 ± 8.6 | 32.8 ± 22.3 | 90.6 ± 8.5 | 59.4 ± 22 | < 0.001* | < 0.001* | |
Values are presented as mean±standard deviation or number (%).
SVA, sagittal vertical axis; PI–LL, pelvic incidence minus lumbar lordosis.
A p-value was derived using †analysis of variance comparing all 3 LDI groups, followed by pairwise comparison of the ‡“normal” and “low (LDI<50)” LDI groups using either Student t-test or Fisher exact test.
p<0.05.
SVA was available in n=79 preoperatively and n=13 postoperatively.
Linear regression analysis (Fig. 2) was found with a linear, negative correlation between preoperative PI and postoperative LDI (OR, -0.51; 95% CI, -0.76 to -0.27; p<0.001).
Fig. 2.
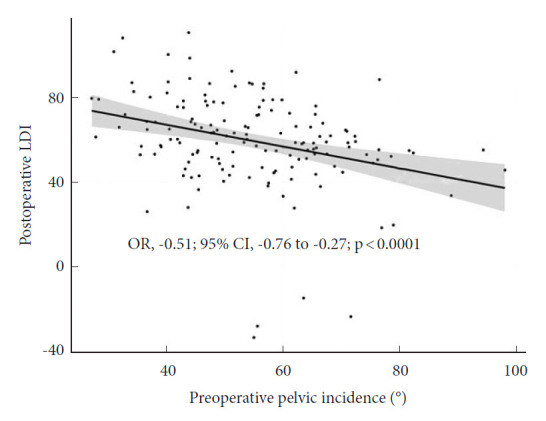
Linear regression model of postoperative lordosis distribution index (LDI) and preoperative pelvic incidence (PI). We found a negative linear correlation between PI and postoperative LDI illustrating the complexity of achieving adequate lower arc lordosis in high PI patients. OR, odds ratio; CI, confidence interval.
3. Complications and All-Cause Revision
We found no differences in complications across LDI groups (Table 3). Regarding all-cause revision (including infection, hematoma, and wound dehiscence), we found an overall 1-year all-cause revision rate of 11%; 22% at 2 years and 32% at any time point in follow-up (mean, 21±14 months). Comparing LDI groups, results showed a significantly increased 1-year allcause revision rate in the hypolordotic group compared to the normal LDI group (19.4% vs. 6.5%, p=0.046). Similarly, the 2-year rate was lowest in the normal LDI group, although the difference was not significant (Table 3).
Table 3.
Complications and revision surgery according to lordosis distribution index (LDI) groups
| Variable | Normal (LDI: 50–80) (n = 93) | Hypolordosis (LDI < 50) (n = 36) | Hyperlordosis (LDI > 80) (n = 20) | Total (n = 149) | p-value† | p-value‡ | |
|---|---|---|---|---|---|---|---|
| Complications | |||||||
| Early (< 3 mo) | 33 (35.5) | 16 (44.4) | 7 (35.0) | 56 (37.6) | 0.621 | 0.419 | |
| Late (> 3 mo) | 43 (46.2) | 13 (36.1) | 8 (40.0) | 64 (43.0) | 0.558 | 0.328 | |
| Minor | 45 (48.4) | 16 (44.4) | 7 (35.0) | 68 (45.6) | 0.544 | 0.700 | |
| Major | 36 (38.7) | 16 (44.4) | 10 (50.0) | 62 (41.6) | 0.600 | 0.556 | |
| Mechanical complications | |||||||
| Major | 5 (5.4) | 5 (13.9) | 3 (15.0) | 13 (8.7) | 0.126 | 0.140 | |
| Minor | 2 (2.2) | 1 (2.8) | 0 (0) | 3 (2.0) | 1.00 | 1.00 | |
| Any | 7 (7.5) | 6 (16.7) | 3 (15) | 16 (10.7) | 0.229 | 0.188 | |
| Adjacent segment disease | |||||||
| Major | 8 (8.6) | 3 (8.3) | 0 (0) | 11 (7.4) | 0.587 | 1.00 | |
| Minor | 1 (1.1) | 0 (0.0) | 0 (0) | 1 (0.7) | 1.00 | 1.00 | |
| Any | 9 (9.7) | 3 (8.3) | 0 (0) | 12 (8.1) | 0.550 | 1.00 | |
| Any complication | 57 (61.3) | 25 (69.4) | 12 (60.0) | 94 (63.1) | 0.659 | 0.422 | |
| Revision§ | |||||||
| Revision 1 yr | 6 (6.5) | 7 (19.4) | 3 (15.0) | 16 (10.7) | 0.082 | 0.046* | |
| Revision 2 yr | 15 (16.1) | 10 (27.8) | 7 (35.0) | 32 (21.5) | 0.100 | 0.143 | |
| Revision any time | 27 (29) | 12 (33.3) | 9 (45.0) | 48 (32.0) | 0.378 | 0.672 | |
Values are presented as number (%).
A p-value was derived using †analysis of variance comparing all 3 LDI groups, followed by pairwise comparison of the ‡“normal” and “low (LDI<50)” LDI groups using either Student t-test or Fisher exact test.
p<0.05.
Revision surgery due to infection, hematoma, or wound complications was not included.
4. Revision Excluding Infection, Hematoma, and Wound Dehiscence
The main outcome of this study was revision due to other causes than infection, hematoma or wound dehiscence and rates were assessed across LDI groups (Fig. 3). One- and 2-year revision rates were highest in the hyperlordotic group and lowest in the normal LDI group. KM survival models plotted as 1-KM (Fig. 4) was with similar results, suggesting lowest revision incidence in the normal LDI group (p=0.030, Fleming-Harington weighted p=0.036). Incidence of revision remained highest in the hyperlordotic group, although an apparent increase was seen at the 2-year mark (Fig. 4). For clinically relevant extrapolation, an accelerated time and event model was performed using Weibull survival regression and plotted over a 4-year period following surgery (Fig. 5). Results suggested lower risk of revision in patients with normal postoperative LDI and highest in the hypolordotic group. Logistic regression analyses were used to assess the degree of association between predefined parameters and revision surgery (Table 4). Univariate analyses showed increased odds of revision with increased LOS; increased preoperative SVA and PI–LL; and increased postoperative SVA, PI–LL, and PT. The parameters with greatest increased odds were postoperative PT>20° (OR, 3.65; 95% CI, 1.20–13.59; p=0.032), and postoperative LDI<50 (OR, 3.50; 95% CI, 1.08–11.70; p=0.036). Postoperative LDI was further assessed for correlation with revision using a multivariable model adjusted for number of instrumented vertebrae and preoperative PI and remained significantly associated (OR, 3.37; 95% CI, 1.03–11.39; p=0.043).
Fig. 3.
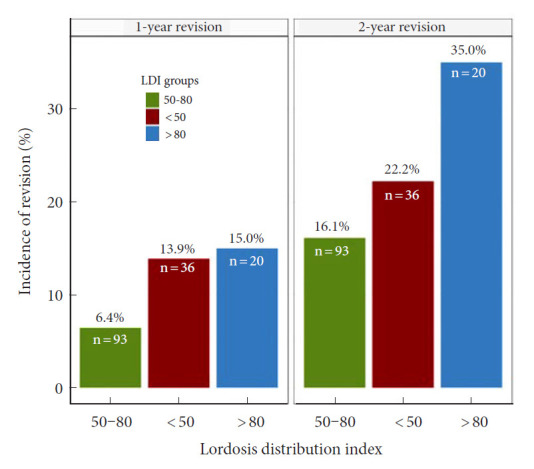
Revision surgery according to postoperative lordosis distribution index (LDI). Revision surgery excluding hematoma, infection, and wound dehiscence according to postoperative LDI. Revision rates were lowest in patients with “normal” postoperative LDI (LDI 50–80). LDI ratios refer to the following LDI groups: 50–80, normal distribution; < 50, hypolordotic maldistribution; > 80, hyperlordotic maldistribution.
Fig. 4.
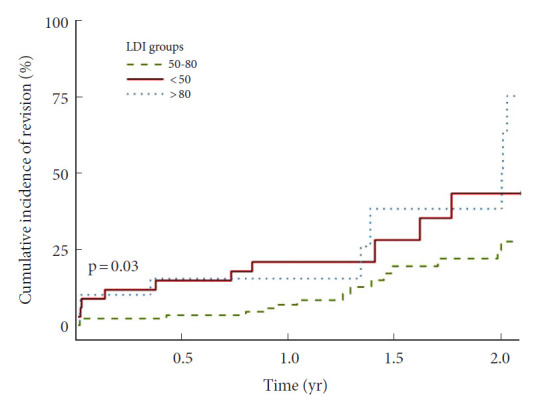
Incidence of revision surgery according to postoperative lordosis distribution index (LDI). Cumulative incidence of revision surgery, using 1-Kaplan Meier survival analyses, according to postoperative LDI groups showed a significant difference in revision between groups. The lowest incidence of revision was found in patients with “normal” postoperative LDI (LDI 50–80). Revision surgery was defined as revision for any reason except hematoma, infection, or wound dehiscence. The provided p-value is derived from an unweighted log-rank test (p=0.030). Fleming-Harington weighted log-rank test: p=0.036. LDI ratios refer to the following LDI groups: 50–80, normal distribution; < 50, hypolordotic maldistribution; > 80, hyperlordotic maldistribution.
Fig. 5.
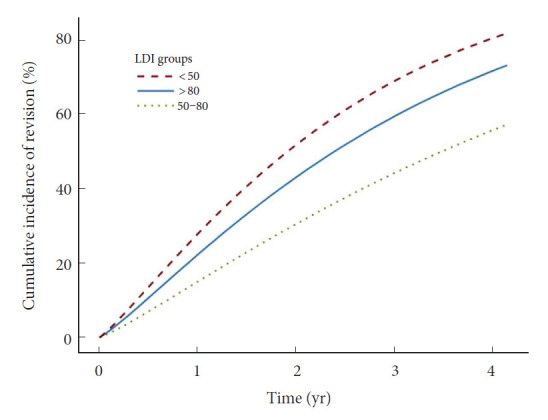
Weibull regression model for revision surgery according to LDI groups. The Weibull regression model for accelerated, and proportional, relative event rates and relative extension survival time showed lowest incidence of revision surgery in patients with “normal” postoperative lordosis distribution index (LDI) defined as LDI of 50–80. Revision surgery was defined as revision for any reason except hematoma, infection, or wound dehiscence. LDI ratios refer to the following LDI groups: 50–80, normal distribution; < 50, hypolordotic maldistribution; > 80, hyperlordotic maldistribution.
Table 4.
Logistic regression analyses of 1-year revision†
| Variable | Univariable |
Multivariable |
|||
|---|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| Age (yr) | 1.04 (0.99–1.09) | 0.140 | - | - | |
| Male sex | 1.57 (0.53–4.50) | 0.398 | - | - | |
| CCI | 1.06 (0.78–1.38) | 0.688 | - | - | |
| ASA PS classification | 1.27 (0.52–3.08) | 0.590 | - | - | |
| Surgery time (min) | 1.00 (0.99–1.01) | 0.784 | - | - | |
| Interbody cage | 0.53 (0.18–1.82) | 0.282 | - | - | |
| Instrumented vertebra (per 1 increase) | 0.71 (0.28–1.26) | 0.374 | 0.71 (0.27–1.33) | 0.398 | |
| Sacral fusion | 0.75 (0.07–8.55) | 0.820 | |||
| Previous spine surgery | 1.37 (0.48–3.95) | 0.548 | - | - | |
| Length of hospital stay | 1.10 (1.02–1.18) | 0.010* | - | - | |
| Preoperative radiographic measurements | |||||
| Pelvic incidence | 1.01 (0.97–1.04) | 0.752 | 1.00 (0.96–1.04) | 0.906 | |
| Pelvic tilt | 1.05 (0.99–1.11) | 0.108 | - | - | |
| Pelvic tilt ≥ 20 | 2.69 (0.94–7.99) | 0.065 | - | - | |
| Sacral Slope | 0.97 (0.92–1.02) | 0.263 | - | - | |
| Global lordosis | 0.97 (0.93–1.00) | 0.065 | - | - | |
| SVA (mm) | 1.01 (1.00–1.03) | 0.024* | - | - | |
| SVA ≥ 40 | 3.94 (1.15–15.82) | 0.036* | - | - | |
| PI–LL | 1.04 (1.00–1.07) | 0.029* | - | - | |
| PI–LL ≥ 10 | 1.56 (0.46–4.66) | 0.441 | - | - | |
| LDI | 0.98 (0.96–1.00) | 0.104 | - | - | |
| LDI groups (reference: 50-80) | - | - | - | - | |
| LDI < 50 | 2.10 (0.63–6.82) | 0.213 | - | - | |
| LDI > 80 | 0.84 (0.12–3.72) | 0.829 | - | - | |
| Postoperative radiographic measurements | |||||
| Pelvic incidence | 1.00 (0.97–1.04) | 0.853 | - | - | |
| Pelvic tilt | 1.05 (1.00–1.11) | 0.058 | - | - | |
| Pelvic tilt ≥ 20 | 3.65 (1.20–13.59) | 0.032* | - | - | |
| Sacral Slope | 0.96 (0.91–1.01) | 0.144 | - | - | |
| Global lordosis | 0.95 (0.91–0.99) | 0.024* | - | - | |
| SVA (mm) | 1.01 (0.98–1.03) | 0.574 | - | - | |
| SVA ≥ 40 | NA | 0.997 | - | - | |
| PI–LL | 1.03 (1.00–1.07) | 0.076 | - | - | |
| PI–LL ≥ 10 | 3.74 (1.30–11.64) | 0.016* | - | - | |
| LDI | 0.99 (0.97–1.02) | 0.596 | - | - | |
| LDI groups (reference: 50–80) | - | - | - | - | |
| LDI < 50 | 3.50 (1.08–11.70) | 0.036* | 3.37 (1.03–11.39) | 0.043* | |
| LDI > 80 | 2.56 (0.50–10.75) | 0.213 | 2.72 (0.52–11.98) | 0.198 | |
OR, odds ratio; CI, confidence interval; CCI, Carlson Comorbidity Index; ASA PS, American Society of Anesthesiologists physical status; SVA, sagittal vertical axis; PI–LL, pelvic incidence minus lumbar lordosis; LDI, lordosis distribution index. NA, upper or lower CI=infinity.
p<0.05.
Revision surgery due to infection, hematoma, or wound complications were not included.
DISCUSSION
The demand for spinal fusion is increasing, with concurrent reports of iatrogenic causes of adult spinal deformity. The role of lordosis maldistribution following short-segment fusion is not fully understood. Therefore, we aimed to assess postoperative LDI after short-segment fusion for degenerative lumbar disease resulting in 3 main findings. Firstly, most patients had adequate postoperative distribution of their lordosis. Secondly, radiographic parameters related to poor outcome in adult spinal deformity were more frequent in patients with maldistributed postoperative LDI. Thirdly, LDI maldistribution was associated to increased risks of revision surgery. In addition, postoperative LDI was inversely, and significantly, correlated to PI.
1. Radiographic Parameters
Overall, surgical alteration in radiographic parameters related to poor outcome was found in several analyzed measurements, including increased PT suggesting compensated sagittal imbalance and inadequate attention to lordosis [30-32]. Similarly, global lordosis decreased during surgery and PI–LL mismatch increased [12,33,34]. Failure to restore adequate lordosis has previously been proposed as a leading cause of iatrogenic deformity [5]. Joelson et al. [35] found that 10% of patients treated for high-grade spondylolisthesis developed sagittal imbalance and compensated deformity was even more common.
The ideal lordosis can be estimated as roughly equal to the PI (PI–LL mismatch) [13,36,37]. In adult spinal deformity, recent studies suggests that sagittal spinal curvatures are more complex [13,38,39]. It is now well established that the lordosis is unequally distributed with an increasing lordosis towards the lower segments [14-16,40,41]. The LDI was; therefore, proposed as part of the Global Alignment and Proportion score for predicting postoperative mechanical complications [14,39].
2. Lordosis Distribution Index
Recently, Zheng et al. [18] assessed the LDI in 215 consecutive patients undergoing posterior lumbar interbody fusion. They found that LDI maldistribution was associated to postoperative ASD. Similarly, we found that postoperative LDI<50% was associated to poor radiographic outcome in several measured parameters, including high PT, increased PI–LL mismatch and decreased global lordosis. These parameters have previously been related to poor outcome in patients with adult spinal deformity and are possible precursors to later-stage sagittal imbalance. The main outcome of the current study was revision surgery (for other causes than hematoma, infection, or wound dehiscence) which was considerably more common in patients with maldistributed postoperative LDI. The 1-year revision incidence was similar in the LDI<50 and LDI>80 groups, and considerably higher compared to the normal group (Fig. 3). At 2-year following surgery, the incidence for the LDI>80 groups increased considerably while remaining lowest in the normal group. The 1-KM plot (Fig. 4) suggested that this sudden deviation was due to a possibly random peek in the LDI>80 group, close to the 2-year mark. Therefore, extrapolation using Weibull accelerated event and time regression was performed in efforts to smoothen the effect of the low sample size (Fig. 5). Results suggested that risk of revision surgery was highest in the LDI< 50 (hypolordotic), similar to the findings of Zheng et al. [18].
3. The High PI Controversy
Sagittal spine shape is not only complex but also varying between individuals. Using asymptomatic individuals, Roussouly et al. [13] assessed this variation and proposed 4 spine shapes correlated to PI. The system has since been revised, and accompanied by studies of surgical algorithms and assessments of postoperative outcome in adult spinal deformity [38,42-44]. Individual PI is often considered important, as high PI requires larger lordosis in efforts to avoid mismatch. Achieving such adequate lordosis may prove difficult in complex cases. In the current study, we found an inverse linear correlation between PI and postoperative LDI suggesting that achieving adequate lordosis distribution may be even more complicated, and important, in patients with high PI.
4. Limitations
The results of the present study should be evaluated in light of several limitations. The current cohort consisted of patients with various spinal pathologies, complicating interpretation. However, the aim was to assess LDI as a viable concept and we acknowledge that further detailed estimates are required in different etiologies as effects may vary. The retrospective nature both reduces external validity and increases risks of selection and sampling bias. Although, the fully disclosed inclusion process and the short enrollment period add to reducing these risks. Further, the short minimum follow-up is unsatisfactory as previous studies have suggested up to 5-year follow-up in efforts to appropriately detect postoperative complications [45]. Results of our Weibull analyses suggested declining incidence just prior to 2 years after surgery which may be an appropriate follow-up in future studies. Also, patient-reported outcome measures were not assessed in the current study and assessing the effect of lordosis maldistribution on measures of health-related quality of life would be of most interest in future studies. Finally, data on preoperative disc or facet degeneration was not obtained in the current study which could influence the effect of postoperative ASD.
CONCLUSION
Most patients undergoing short-segment fusion for lumbar degenerative spine disease had adequate postoperative lordosis distribution. Patients with postoperative maldistributed LDI had increased PT, increased PI–LL mismatch and an overall reduced global lordosis following surgery. Revision surgery was most frequent in patients with postoperative hypolordotic maldistribution (LDI<50). Our results suggest that inadequate lordosis distribution may predispose iatrogenic deformity and should be considered in short-segment spinal fusion. Special care should be allocated to high PI patients as LDI maldistribution and PI were linearly correlated.
Footnotes
BD (consulting fees from Stryker outside of the submitted work), MG (institutional grants from K2M and Medtronic outside of the submitted work), the remaining authors report no conflicts of interest.
REFERENCES
- 1.Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2163–96. doi: 10.1016/S0140-6736(12)61729-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fingar KR, Stocks C, Weiss AJ, et al. Healthcare Cost and Utilization Project (HCUP) statistical briefs [Internet] Rockville (MD): Agency for Healthcare Research and Quality (US); Most frequent operating room procedures performed in U.S. hospitals, 2003–2012: statistical brief #186. 2014 Dec. 2006 Feb–. Available from: https://www.ncbi.nlm.nih.gov/books/NBK274246/ [PubMed] [Google Scholar]
- 3.Deyo RA, Mirza SK. Trends and variations in the use of spine surgery. Clin Orthop Relat Res. 2006;443:139–46. doi: 10.1097/01.blo.0000198726.62514.75. [DOI] [PubMed] [Google Scholar]
- 4.Weinstein JN, Lurie JD, Olson PR, et al. United States’ trends and regional variations in lumbar spine surgery: 1992–2003. Spine (Phila Pa 1976) 2006;31:2707–14. doi: 10.1097/01.brs.0000248132.15231.fe. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Potter BK, Lenke LG, Kuklo TR. Prevention and management of iatrogenic flatback deformity. J Bone Joint Surg Am. 2004;86:1793–808. doi: 10.2106/00004623-200408000-00027. [DOI] [PubMed] [Google Scholar]
- 6.Diebo BG, Shah NV, Boachie-Adjei O, et al. Adult spinal deformity. Lancet. 2019;394:160–72. doi: 10.1016/S0140-6736(19)31125-0. [DOI] [PubMed] [Google Scholar]
- 7.Pellisé F, Vila-Casademunt A, Ferrer M, et al. Impact on health related quality of life of adult spinal deformity (ASD) compared with other chronic conditions. Eur Spine J. 2014;24:3–11. doi: 10.1007/s00586-014-3542-1. [DOI] [PubMed] [Google Scholar]
- 8.Good CR, Auerbach JD, O’Leary PT, et al. Adult spine deformity. Curr Rev Musculoskelet Med. 2011;4:159–67. doi: 10.1007/s12178-011-9101-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ames CP, Scheer JK, Lafage V, et al. Adult spinal deformity: epidemiology, health impact, evaluation, and management. Spine Deform. 2016;4:310–22. doi: 10.1016/j.jspd.2015.12.009. [DOI] [PubMed] [Google Scholar]
- 10.Hallager DW, Karstensen S, Bukhari N, et al. Radiographic predictors for mechanical failure after adult spinal deformity surgery. Spine (Phila Pa 1976) 2017;42:E855–63. doi: 10.1097/BRS.0000000000001996. [DOI] [PubMed] [Google Scholar]
- 11.Schwab F, Ungar B, Blondel B, et al. Scoliosis Research Society—Schwab Adult Spinal Deformity Classification. Spine (Phila Pa 1976) 2012;37:1077–82. doi: 10.1097/BRS.0b013e31823e15e2. [DOI] [PubMed] [Google Scholar]
- 12.Rothenfluh DA, Mueller DA, Rothenfluh E, et al. Pelvic incidence-lumbar lordosis mismatch predisposes to adjacent segment disease after lumbar spinal fusion. Eur Spine J. 2015;24:1251–8. doi: 10.1007/s00586-014-3454-0. [DOI] [PubMed] [Google Scholar]
- 13.Roussouly P, Gollogly S, Berthonnaud E, et al. Classification of the normal variation in the sagittal alignment of the human lumbar spine and pelvis in the standing position. Spine (Phila Pa 1976) 2005;30:346–53. doi: 10.1097/01.brs.0000152379.54463.65. [DOI] [PubMed] [Google Scholar]
- 14.Yilgor C, Sogunmez N, Yavuz Y, et al. Relative lumbar lordosis and lordosis distribution index: individualized pelvic incidence-based proportional parameters that quantify lumbar lordosis more precisely than the concept of pelvic incidence minus lumbar lordosis. Neurosurg Focus. 2017;43:E5. doi: 10.3171/2017.8.FOCUS17498. [DOI] [PubMed] [Google Scholar]
- 15.Berthonnaud E, Dimnet J, Roussouly P, et al. Analysis of the sagittal balance of the spine and pelvis using shape and orientation parameters. J Spinal Disord Tech. 2005;18:40–7. doi: 10.1097/01.bsd.0000117542.88865.77. [DOI] [PubMed] [Google Scholar]
- 16.Roussouly P, Pinheiro-Franco JL. Sagittal parameters of the spine: biomechanical approach. Eur Spine J. 2011;20:578–85. doi: 10.1007/s00586-011-1924-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Barrey C. Equilibre sagittal pelvi-rachidien et pathologies lombaires dégénératives. Etude comparative à propos de 100 cas. Lyon (France): TUniversité Claude-Bernard; 2004. [Google Scholar]
- 18.Zheng G, Wang C, Wang T, et al. Relationship between postoperative lordosis distribution index and adjacent segment disease following L4-S1 posterior lumbar interbody fusion. J Orthop Surg Res. 2020;15:129. doi: 10.1186/s13018-020-01630-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Maillot C, Ferrero E, Fort D, et al. Reproducibility and repeatability of a new computerized software for sagittal spinopelvic and scoliosis curvature radiologic measurements: Keops®. Eur Spine J. 2015;24:1574–81. doi: 10.1007/s00586-015-3817-1. [DOI] [PubMed] [Google Scholar]
- 20.R Core Team . R: A Language and Environment for Statistical Computing. [Vienna]: R Foundation for Statistical Computing; 2020. [Google Scholar]
- 21.Wickham H. ggplot2: elegant graphics for data analysis. Cham: Springer International Publishing; 2016. [Google Scholar]
- 22.Kassambara A, Kosinski M, Biecek P. survminer: drawing survival curves using “ggplot2” [Internet] Montpellier (France): DataNovia; 2020. Available from: https://rpkgs.datanovia.com/survminer/ [Google Scholar]
- 23.Gerds TA. prodlim: product-limit estimation for censored event history analysis [Internet] Vienna (Austria): R Foundation for Statistical Computing; 2019. Available from: https://cran.r-project.org/web/packages/prodlim/index.html. [Google Scholar]
- 24.Therneau TM. A Package for Survival Analysis in R [Internet] Vienna (Austria): R Foundation for Statistical Computing; 2020. Available from: https://cran.r-project.org/web/packages/survival/vignettes/survival.pdf. [Google Scholar]
- 25.Jackson C. {flexsurv}: a platform for parametric survival modeling in {R} J Stat Softw. 2016;70:1–33. doi: 10.18637/jss.v070.i08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81. [Google Scholar]
- 27.Southern DA, Faris PD, Brant R, et al. Kaplan-Meier methods yielded misleading results in competing risk scenarios. J Clin Epidemiol. 2006;59:1110–4. doi: 10.1016/j.jclinepi.2006.07.002. [DOI] [PubMed] [Google Scholar]
- 28.Carroll KJ. On the use and utility of the Weibull model in the analysis of survival data. Control Clin Trials. 2003;24:682–701. doi: 10.1016/s0197-2456(03)00072-2. [DOI] [PubMed] [Google Scholar]
- 29.Wei LJ. The accelerated failure time model: a useful alternative to the cox regression model in survival analysis. Stat Med. 1992;11:1871–9. doi: 10.1002/sim.4780111409. [DOI] [PubMed] [Google Scholar]
- 30.Lafage V, Schwab F, Patel A, et al. Pelvic tilt and truncal inclination. Spine (Phila Pa 1976) 2009;34:E599–606. doi: 10.1097/BRS.0b013e3181aad219. [DOI] [PubMed] [Google Scholar]
- 31.Lazennec JY, Brusson A, Rousseau MA. Hip–spine relations and sagittal balance clinical consequences. Eur Spine J. 2011;20:686–98. doi: 10.1007/s00586-011-1937-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lazennec JY, Ramaré S, Arafati N, et al. Sagittal alignment in lumbosacral fusion: relations between radiological parameters and pain. Eur Spine J. 2000;9:47–55. doi: 10.1007/s005860050008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Aoki Y, Nakajima A, Takahashi H, et al. Influence of pelvic incidence-lumbar lordosis mismatch on surgical outcomes of short-segment transforaminal lumbar interbody fusion. BMC Musculoskelet Disord. 2015;16:213. doi: 10.1186/s12891-015-0676-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zachary J, Tempel M, Gurpreet S, et al. The influence of pelvic incidence and lumbar lordosis mismatch on development of symptomatic adjacent level disease following single-level transforaminal lumbar interbody fusion. Neurosurgery. 2017;80:14–6. doi: 10.1093/neuros/nyw073. [DOI] [PubMed] [Google Scholar]
- 35.Joelson A, Danielson BI, Hedlund R, et al. Sagittal balance and health-related quality of life three decades after in situ arthrodesis for high-grade isthmic spondylolisthesis. J Bone Joint Surg Am. 2018;100:1357–65. doi: 10.2106/JBJS.17.01415. [DOI] [PubMed] [Google Scholar]
- 36.Duval-Beaupere G, Schmidt C, Cosson PH. A Barycentremetric study of the sagittal shape of spine and pelvis: the conditions required for an economic standing position. Ann Biomed Eng. 1992;20:451–62. doi: 10.1007/BF02368136. [DOI] [PubMed] [Google Scholar]
- 37.Boulay C, Tardieu C, Hecquet J, et al. Sagittal alignment of spine and pelvis regulated by pelvic incidence: standard values and prediction of lordosis. Eur Spine J. 2006;15:415–22. doi: 10.1007/s00586-005-0984-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Laouissat F, Sebaaly A, Gehrchen M, et al. Classification of normal sagittal spine alignment: refounding the Roussouly classification. Eur Spine J. 2018;27:2002–11. doi: 10.1007/s00586-017-5111-x. [DOI] [PubMed] [Google Scholar]
- 39.Yilgor C, Sogunmez N, Boissiere L, et al. Global Alignment and Proportion (GAP) Score: development and validation of a new method of analyzing spinopelvic alignment to predict mechanical complications after adult spinal deformity surgery. J Bone Joint Surg Am. 2017;99:1661–72. doi: 10.2106/JBJS.16.01594. [DOI] [PubMed] [Google Scholar]
- 40.Roussouly P, Nnadi C. Sagittal plane deformity: an overview of interpretation and management. Eur Spine J. 2010;19:1824–36. doi: 10.1007/s00586-010-1476-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pizones J, Perez-Grueso FJS, Moreno-Manzanaro L, et al. Ideal sagittal profile restoration and ideal lumbar apex positioning play an important role in postoperative mechanical complications after a lumbar PSO. Spine Deform. 2020;8:491–8. doi: 10.1007/s43390-019-00005-3. [DOI] [PubMed] [Google Scholar]
- 42.Pizones J, Moreno-Manzanaro L, Sánchez Pérez-Grueso FJ, et al. Restoring the ideal Roussouly sagittal profile in adult scoliosis surgery decreases the risk of mechanical complications. Eur Spine J. 2020;29:54–62. doi: 10.1007/s00586-019-06176-x. [DOI] [PubMed] [Google Scholar]
- 43.Sebaaly A, Gehrchen M, Silvestre C, et al. Mechanical complications in adult spinal deformity and the effect of restoring the spinal shapes according to the Roussouly classification: a multicentric study. Eur Spine J. 2020;29:904–13. doi: 10.1007/s00586-019-06253-1. [DOI] [PubMed] [Google Scholar]
- 44.Bari TJ, Hansen LV, Gehrchen M. Surgical correction of adult spinal deformity in accordance to the Roussouly classification: effect on postoperative mechanical complications. Spine Deform. 2020;8:1027–37. doi: 10.1007/s43390-020-00112-6. [DOI] [PubMed] [Google Scholar]
- 45.Kumar M, Baklanov A, Chopin D. Correlation between sagittal plane changes and adjacent segment degeneration following lumbar spine fusion. Eur Spine J. 2001;10:314–9. doi: 10.1007/s005860000239. [DOI] [PMC free article] [PubMed] [Google Scholar]


