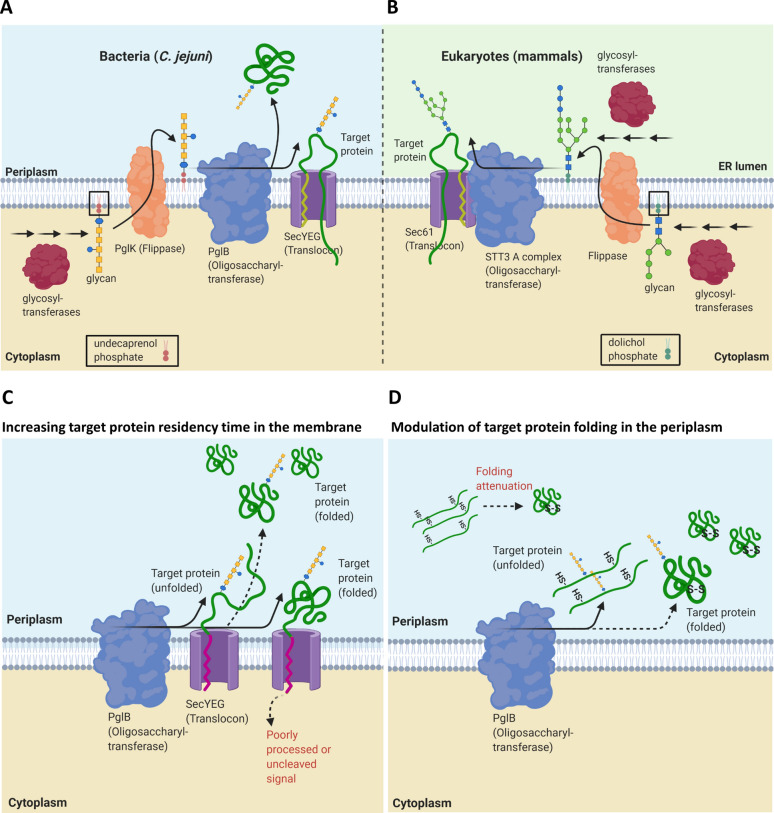
Fig. 1.
Schematic overview of native N-linked protein glycosylation pathway and proposed strategies in this study to improve sequon accessibility of recombinant target protein to PglB during glycosylation in glycoengineered E. coli. N-linked glycosylation in bacteria/C. jejuni (A) compared to early-stage of eukaryotic/mammalian N-linked glycosylation pathway (B). A In C. jejuni, undecaprenol-linked glycan is synthesised by glycosyltransferases in the cytoplasm, flipped to the periplasm by flippase (PglK), and then transferred co or post-translocationally to a target protein by PglB. B In mammalian N-linked glycosylation, dolichol-linked glycan is synthesised both in the cytosol and endoplasmic reticulum (ER) lumen, and glycan is transferred co- or post-translocationally (later not shown) by different STT3 isoforms. C, D Two different strategies are proposed to enhance heterologous protein glycosylation in E. coli. C Approach based on increasing PglB interaction with target protein in the membrane. Increased membrane residence of target protein was achieved by introducing signal peptide mutations with poor processivity or uncleaved signal. D Approach based on increasing sequon accessibility of target proteins to PglB via modulation of target protein folding state during or after translocation. Using a disulphide-bonded model protein, protein folding and maturation in the periplasm are delayed by producing the protein under suboptimal conditions for disulphide formation such as under oxygen-depleted conditions, in the absence of oxidoreductases, or under chemical redox treatment. Solid arrow = increased reaction, dashed arrow = reduced reaction