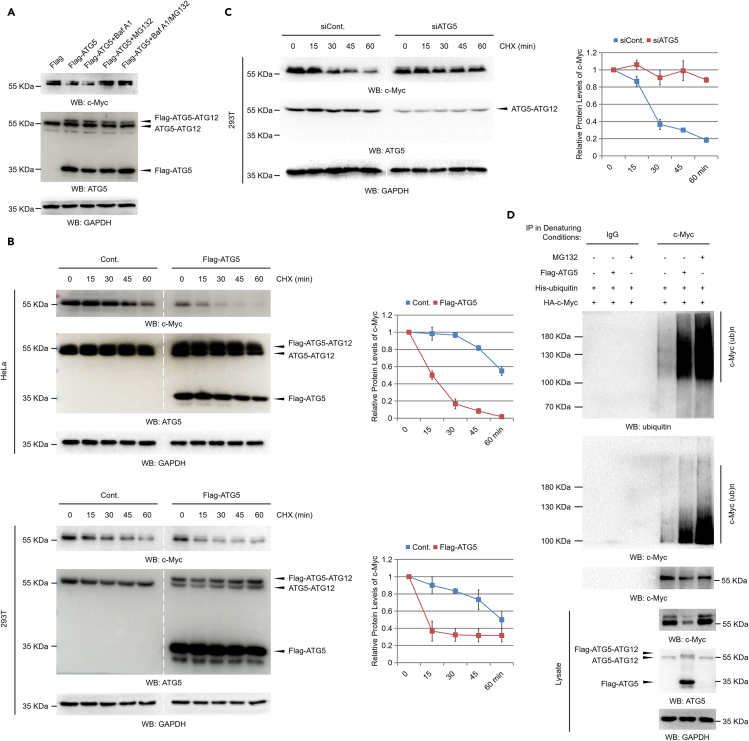
Figure 3.
ATG5 regulates c-Myc protein degradation through the ubiquitination-proteasome pathway
(A) 293T cells were transfected with a Flag control or a Flag-tagged ATG5 construct for 48 h. Cells were then treated with or without 1 μM Baf A1 (bafilomycin A1), 25 μM MG132, or both as indicated for another 10 h. Cell extracts were prepared, and western blot analyses were performed.
(B) HeLa and 293T cells transfected with a Flag control construct or a Flag-tagged ATG5 construct were treated with 50 μg/mL CHX (cycloheximide) for the indicated durations (chase assays). Cells were then lysed and subjected to western blot analyses. Relative protein levels of c-Myc were analyzed with ImageJ software (right).
(C) 293T cells transfected with a control siRNA or an ATG5-specific siRNA were treated with 50 μg/mL CHX for the indicated durations (chase assays). Cells were then lysed and subjected to western blot analyses. Relative protein levels of c-Myc were analyzed with ImageJ software (right).
(D) An HA-tagged c-Myc and an His-tagged ubiquitin construct were transfected with or without a Flag-tagged ATG5 construct into 293T cells as indicated for 48 h. Cells were then treated with or without 25 μM MG132 for another 10 h. Cells were lysed under denaturing conditions, and the c-Myc protein was immunoprecipitated (IP) with c-Myc-specific antibodies. IP with immunoglobulin G (IgG) antibodies was performed as a negative control. The precipitated proteins were subjected to western blot analyses with the indicated antibodies. Statistics: all experiments were repeated more than 3 times. The bars and error bars indicate the mean ± s.d. values; n = 3 independent replicates.