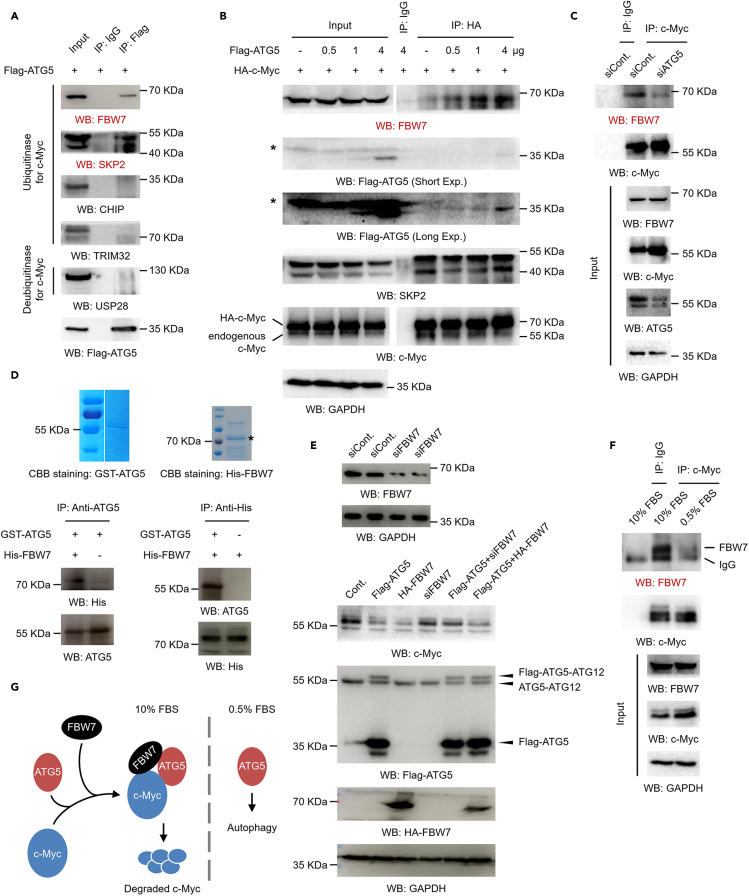
Figure 6.
ATG5 recruits the E3 protein-ubiquitin ligase FBW7 to bind c-Myc
(A) 293T cells were transfected with a Flag-tagged ATG5 for 48 h. Cells were lysed, and co-IP assays were performed with antibodies against Flag. IgG antibodies were used as a negative control.
(B) 293T cells were transfected with an HA-tagged c-Myc construct together with increasing amounts of a Flag-tagged ATG5 construct for 48 h. Cells were lysed, and co-IP assays were performed with antibodies against HA. IgG antibodies were used as a negative control.
(C) 293T cells were transfected with an ATG5-specific siRNA or a control siRNA for 48 h. Cells were lysed, and co-IP assays were performed with antibodies against c-Myc. IgG antibodies were used as a negative control.
(D) GST-tagged ATG5 and His-tagged FBW7 were expressed in E coli and purified (upper). The two proteins were co-incubated, and IP assays with antibodies against ATG5 or His (lower) were performed as indicated to evaluate the direct binding between ATG5 and FBW7 in vitro.
(E) A FBW7-specific siRNA was transfected into 293T cells for 48 h. Cells were then lysed, and the RNAi efficiency was determined by western blot analysis (upper). 293T cells were transfected with a Flag-tagged ATG5 construct, a HA-tagged FBW7 construct, FBW7-specific siRNA, or distinct combinations as indicated for 48 h. Cell extracts were prepared, and western blot analyses were performed (lower).
(F) 293T cells were cultured under normal conditions or serum starvation conditions as indicated. Cells were harvested, and co-IP assays were performed with antibodies against c-Myc. IgG antibodies were used as a negative control.
(G) Molecular model. Under normal culture conditions, ATG5 recruits FBW7 to c-Myc and further promotes c-Myc protein degradation. However, under serum starvation conditions, ATG5 may mainly contribute to the regulation of autophagy.