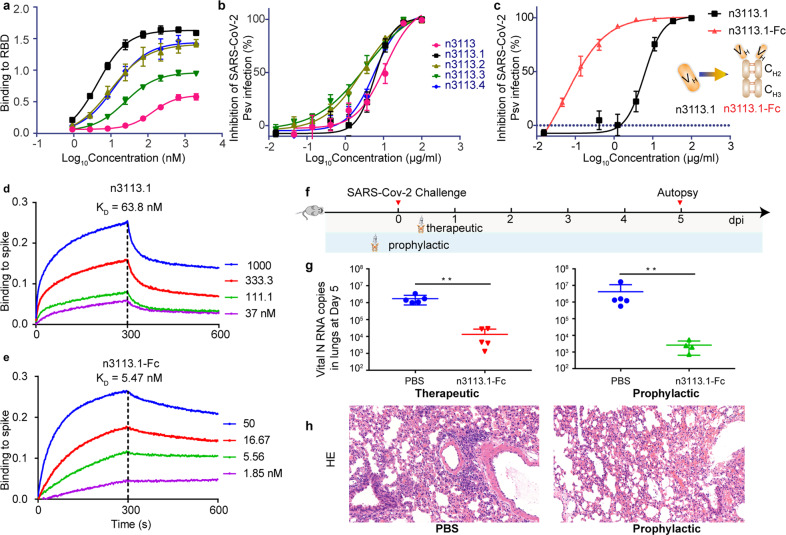
Fig. 1.
Human single-domain antibody shows therapeutic value against SARS-CoV-2. a Binding affinity of n3113 and its variants with SARS-CoV-2 RBD. The mean ± SD from three independent experiments is shown. b Neutralization of SARS-CoV-2 S pseudovirus by n3113 and its variants. The mean ± SD from three independent experiments is shown. c Schematic representation of the generation of n3113.1-Fc and in vitro SARS-CoV-2 S pseudovirus neutralizing activity of n3113.1 and n3113.1-Fc. Neutralizing activities are presented as mean ± SD from three independent experiments. d–e Binding kinetic of n3113.1 (d) and n3113.1-Fc (e) with immobilized prefusion S ectodomain, measured using bio-layer interferometry (BLI). Dissociation constant (KD) values for n3113.1 and n3113.1-Fc were obtained using a 1:1 binding model and 1:2 bivalent model, respectively. The experiments were performed in duplicate with similar results and a representative experiment is shown. f Schematic diagram of n3113.1-Fc treatment in SARS-CoV-2-susceptible mice. g Viral N RNA copies in the therapeutic group and the prophylactic group in the lungs were measured 5 dpi by qRT-PCR. P value was estimated by unpaired t test (**P < 0.01). h Histopathological characterization of the lungs of SARS-CoV-2 infected mice received prophylactic treatment of PBS or n3113.1-Fc