Abstract
Introduction
Celiac disease (CD) is an autoimmune enteropathy triggered by gluten ingestion in genetically susceptible individuals. In CD, activation of the immune response causes damage of the intestinal mucosa, and a gluten-free diet (GFD) is the only available therapy. Intestinal damage can lead to an increase in the circulation of components of bacteria from the intestinal lumen, such as lipopolysaccharide (LPS). Soluble CD14 (sCD14) and lipopolysaccharide-binding protein (LBP) participate in the recognition of LPS, and their levels are altered in different pathologies. In the present study, the circulating levels of sCD14 and LBP from untreated CD patients were evaluated and compared to CD patients on a GFD and controls.
Material and methods
In total seventy-two adult patients with CD, twenty-three untreated CD patients and forty-nine on a GFD were included. In addition, fifty-five healthy individuals were included as controls. Additionally, the effect of LPS on sCD14 production by both normal and inflamed intestinal tissue culture was explored.
Results
Serum levels of sCD14 were found to be significantly increased in untreated CD patients compared to patients on a GFD and controls. In addition, we found that LPS induced the production of sCD14 by biopsies of intestinal tissue from untreated CD patients.
Conclusions
The data from this study show that circulating levels of sCD14 are increased in the untreated CD patients compared to patients on a GFD. Our data show that LPS induces the production of sCD14 by the intestinal tissue from untreated CD patients.
Keywords: celiac disease, gluten-free diet, lipopolysaccharide-binding protein, soluble CD14
Introduction
Celiac disease (CD) is an autoimmune enteropathy triggered by gluten ingestion in genetically susceptible individuals [1, 2]. Gluten is present in cereals such as wheat, barley, rye, and other related grains, and is composed of prolamins and glutelins. The prevalence of CD worldwide is approximately 1% and the incidence is increasing [3]. CD can affect individuals of any age and the male-to-female ratio is 1 : 3 to 1 : 1.5 [1]. The etiology of CD is complex, involving genetic and environmental factors [4].
In CD, the activation of both the innate and adaptive immune response produces damage of the intestinal mucosa, including infiltration of lymphocytes, villous atrophy, and alteration of intestinal permeability. In a context of increased intestinal mucosal permeability such as that presented in CD, undigested gliadin fragments can reach the lamina propria of the intestine. In the lamina propria, gliadin peptides are deaminated by the transglutaminase 2 (TG2) enzyme, and peptides increase their binding affinity to the HLA class II DQ2 and/or DQ8 molecules present on the surface of antigen presenting cells (APC). Gliadin peptides are presented to CD4+ T lymphocytes, which secrete inflammatory cytokines and promote B lymphocyte activation [4].
A strict lifelong gluten-free diet (GFD) is currently the only available therapy. This treatment allows the resolution of symptoms and the repair of intestinal damage in most cases [5].
Mucosal damage and alteration of intestinal permeability have been associated with microbial translocation, where microorganisms from the intestinal lumen or their products get access to the bloodstream [6]. Thus, the increase in circulating levels of bacterial products such as lipopolysaccharide (LPS) or endotoxin leads to systemic activation of the immune response [6].
Genetic and environmental factors contribute to trigger autoimmunity. There is increasing evidence that intestinal barrier dysfunction and microbial translocation can be found among these factors. Furthermore, microbial translocation and systemic activation of the immune response can not only contribute to trigger autoimmunity but also aggravate autoimmune conditions [7].
In addition, due to the increased levels of circulating LPS in conditions with changed intestinal permeability, the levels of proteins involved in their recognition are altered, including soluble CD14 (sCD14) and lipopolysaccharide-binding protein (LBP) [6, 8]. Because of this, sCD14 and LBP have been proposed as markers of intestinal damage and endotoxemia [6].
sCD14 and LBP are accessory molecules of toll-like receptor (TLR) 4. LBP binds to LPS aggregates and transfers them to the membrane CD14 or sCD14, which then transfers it to the TLR4/myeloid differentiation factor (MD)-2 complex [9].
Altered serum levels of sCD14 and LBP were found in different inflammatory/autoimmune pathologies, such as type 1 diabetes, lupus, multiple sclerosis and Crohn’s disease [10-14]. In CD, studies about sCD14 and LBP proteins are limited. Elevated serum levels of sCD14 were found in untreated CD patients (active CD) compared to CD patients on a GFD and controls [15]. Another study reported no differences between serum levels of sCD14 and LBP in patients with active CD compared to healthy controls [16].
LBP is produced by hepatocytes and epithelial cells [17], while sCD14 is produced by monocytes/macrophages and granulocytes, as well as by keratinocytes, hepatocytes and human intestinal epithelial cells [18-20].
The release of sCD14 by hepatocytes or peripheral mononuclear cells increases in the presence of LPS [21, 22]; however, it is not known whether the production of this molecule in the normal or inflamed intestinal tissue is influenced by exposure to LPS.
In this scenario, more studies are needed to clarify whether these proteins are altered in CD. The aim of this study was to evaluate the serum levels of sCD14 and LBP in untreated CD patients, and to compare them with those of CD patients on a GFD and with healthy controls. In addition, exploring whether LPS, an inflammatory stimulus, induces the production of sCD14 by both normal and inflamed intestinal tissue was proposed.
Material and methods
Patients and controls
Serum samples were collected from 72 adult CD patients (23 untreated and 49 on a GFD) and 55 healthy adults as controls (Table 1). Groups were matched by sex and age. The patients were recruited at the Instituto de Investigaciones en Ciencias de la Salud and Hospital de Clínicas, Universidad Nacional de Asunción, in collaboration with the Fundación Paraguaya de Celíacos (FUPACEL). CD diagnosis was performed by evaluation of clinical presentation, histological examination, and serological evaluation. In summary, all patients included had a diagnosis confirmed by biopsy and histological analysis by a specialist, and they presented total or partial villous atrophy at the time of diagnosis. 90% of celiac patients had intestinal symptoms at the time of diagnosis. The patients included had no other autoimmune disease.
Table 1.
Characteristics of celiac patients and control subjects
| Variable | Controls | CD patients | CD patients treated with GFD | |
|---|---|---|---|---|
| n | 55 | 23 | 49 | |
| Age (years, ±SD) | 34.9 ±12 | 34.7 ±11.9 | 35.2 ±12.5 | |
| Gender, n (%) | ||||
| Male | 14 (25.5) | 7 (30.4) | 10 (20.4) | |
| Female | 41 (74.5) | 16 (69.6) | 39 (79.6) | |
| GFD (%) | N/A | 0 | 100 | |
| Anti-tTG2, n (%) | ||||
| Positive | 0 | 23 (100) | 14 (29) | |
| Indeterminate | 0 | 0 | 4 (8) | |
| Negative | 55 (100) | 0 | 31 (63) | |
CD – celiac disease, GFD – gluten-free diet, tTG2 – tissue transglutaminase, N/A – not applicable
Patients on a GFD had a minimum of 6 months of diet at the time of serum collection (average diet duration of 5.6 years) and they presented negative (63%), undetermined (8%) or positive (29%) immunoglobulin A (IgA) anti-TG2. All patients on a GFD reported that they complied with the diet and received nutritional advice at the beginning of the GFD. Controls were free from any symptoms or signs of disease and were negative for IgA anti-TG2.
This study was approved by the Ethics Committee of the Instituto de Investigaciones en Ciencias de la Salud, Universidad Nacional de Asunción. Written informed consent was obtained from each participant prior to inclusion in the study.
Assessment of serum levels of IgA anti-TG2
Serum levels of IgA anti-TG2 were determined by enzyme-linked immunosorbent assay (ELISA) (AESKULISA,Germany), according to the manufacturer’s protocol.
Determination of sCD14 and LBP in serum
Assessment of sCD14 and LBP serum levels was performed using sandwich ELISA (R&D Systems, USA) following the manufacturer’s instructions. Samples were diluted and incubated in duplicate. The absorbance of the sample was determined by a spectrophotometer and concentrations were calculated from a standard curve.
Production of sCD14 from in vitro cultured small intestinal biopsies
Biopsy specimens were obtained from the second portion of the duodenum during gastroduodenoscopy. Small intestinal biopsy samples from 5 patients with untreated CD (without GFD) and 5 controls (submitted to endoscopy due to suspicion of dyspepsia) were cultured for 24 h in serum-free RPMI in the presence or absence of LPS (5 µg/ml) and the concentration of sCD14 in culture supernatant were measured by ELISA (R&D Systems, USA). Content of sCD14 in the supernatant was adjusted to the total protein content of biopsy determined by Bradford assay (Bio-Rad, USA) and expressed as pg/ml/mg.
Statistical analysis
Normality of distribution was assessed using the Kolmogorov-Smirnov test. Comparison between two groups was performed with the two-sided Mann-Whitney test or t test, as indicated. The correlations between variables were calculated using Spearman’s correlation coefficient. Analyses were conducted using GraphPad Prism 6.01. A p value < 0.05 was considered statistically significant.
Results
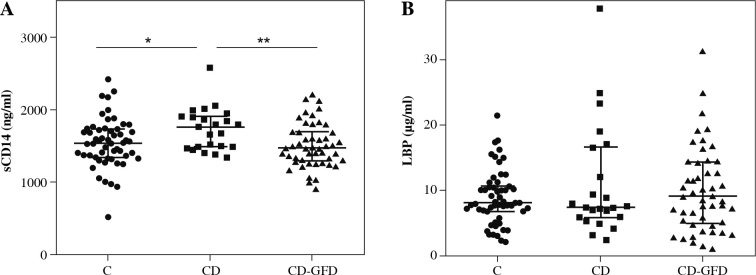
A total of 72 CD patients (23 untreated and 49 on a GFD) were included. Also, 55 healthy adults were included as a control group. Serum sCD14 levels of untreated celiac patients (CD) were found to be increased compared to those in CD patients on a GFD (CD-GFD) (p = 0.0073) and to control subjects (p = 0.0220) (Fig. 1A). The median (IC 25-75%) serum concentration of sCD14 in CD, CD-GFD, and the control group was 1759 (1487-1905) ng/ml, 1472 (1295-1693) ng/ml, and 1543 (1344-1733) ng/ml, respectively. No statistically significant difference was observed between sCD14 levels of CD-GFD and controls. Also, no significant difference was found between the sCD14 levels of females and males in CD patients or the control group (data not shown).
Fig. 1.
Serum sCD14 and LBP levels in CD patients and controls. The sCD14 (A) and LBP (B) concentration in serumsamples from control subjects (C), untreated patients with celiac disease (CD) and patients with celiac disease on a gluten-free diet (CD-GFD). Mann-Whitney test. *p = 0.0220, **p = 0.0073
When analyzing serum concentration of LBP, another soluble protein that participates in the recognition of LPS, no statistically significant difference was observed between levels in untreated CD patients, CD-GFD patients, and the control group (Fig. 1B). The median (IC 25-75%) serum concentration of LBP in CD, CD-GFD and the control group was 7.434 (5.89-16.61) µg/ml, 9.071 (4.93-14.35) µg/ml and 8.129 (6.82-10.64) µg/ml, respectively.
Subsequently, serum sCD14 levels of untreated celiac patients were compared with those of CD patients on a GFD according to the serum value of anti-TG2 IgA antibody (positive or negative). sCD14 levels were found to be significantly increased in untreated CD patients compared to those in CD patients on GFD, regardless of whether the IgA anti-TG2 antibody in the latter group was negative or positive (Fig. 2). Also, no difference in sCD14 levels was observed in the CD-GFD group with a positive or negative value of IgA anti-TG2 antibody.
Fig. 2.
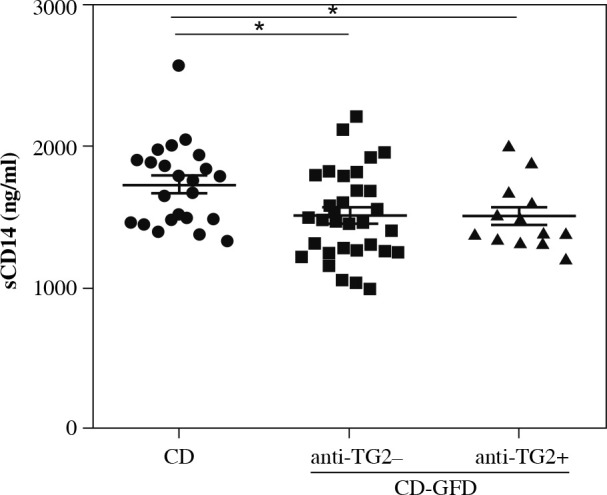
Serum levels of sCD14 in untreated patients with celiac disease (CD) and patients with celiac disease on a gluten-free diet (CD-GFD) according to the value (negative or positive) of IgA anti-transglutaminase (anti-TG2) antibody. Mann-Whitney test. *p < 0.05
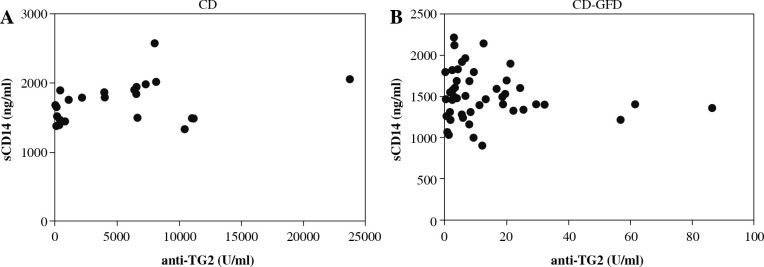
In Figure 3 the results of the correlation between serum levels of sCD14 and IgA anti-TG2 antibody are shown. No significant correlation was observed between sCD14 and the autoantibody from untreated CD patients and CD-GFD patients.
Fig. 3.
Correlation of serum levels of sCD14 and IgA anti-TG2 from untreated celiac disease patients (CD) (A) (Spearman’s r = 0.3478, p = 0.1039) and celiac disease patients on a gluten-free diet (CD-GFD) (B) (Spearman’s r = –0.1307, p = 0.3760)
CD14 is a co-receptor for LPS recognition and is in the plasma membrane. CD14 is shed from the cell surface leading to sCD14 generation. The production of sCD14 from peripheral mononuclear cells increases in the presence of LPS; therefore increased levels of sCD14 could reflect endotoxin exposure. However, it is not known whether LPS influences the production of sCD14 by cells present in the intestine. To elucidate whether LPS affects the production of sCD14 at the level of intestinal tissue, the production of basal and LPS-induced sCD14 was analyzed in supernatants of cultured biopsies from untreated CD patients and control subjects. Intestinal tissue from both controls and CD patients presented a constitutive production of sCD14. The production of sCD14 by the uninflamed intestinal tissue of the control subjects did not increase with the stimulation of LPS. However, when stimulated with LPS, an increase in the production of sCD14 by the intestinal tissue of patients with active CD was observed (Fig. 4).
Fig. 4.

Spontaneous and LPS-stimulated sCD14 production from small intestinal biopsies from control subjects (C) and untreated celiac disease patients (CD), in the presence or not of lipopolysaccharide (LPS). t test, ns – not significant, *p = 0.0353
Discussion
In the present study, elevated serum levels of sCD14 were found in untreated CD patients (active CD) compared to those in CD patients on a GFD and healthy controls. Elevated circulating levels of sCD14 indicate a systemic response to exposure to bacteria or bacterial components [8, 21]. Increased circulating levels of sCD14 in CD patients may be due to increased intestinal permeability and subsequent translocation of bacteria from the intestinal lumen to blood circulation. This microbial translocation may lead to a state of systemic inflammation, which has been reported to promote or affect the development of diverse pathologies [23].
Reports on sCD14 levels in CD are limited. Hoffmanová et al. reported increased levels of sCD14 in CD patients compared to patients on a GFD and a control group [15]. However, Uhde et al. did not find statistically significant differences in sCD14 levels between CD patients and controls; this study did not include patients on a GFD [16]. Our results agree with the first observation, since we detected a statistically significant increase of sCD14 levels in CD patients compared to a control group and with patients on a GFD.
The differences observed between the results from our study and those reported by Udhe et al. could be due to the ethnic differences and/or differences in clinical characteristics of the patients included in both studies. It is noteworthy that none of the patients included in our study manifested any other autoimmune disease aside from CD.
Additionally, no significant difference was observed between sCD14 levels of CD patients on a GFD and controls. This finding could be explained by the fact that once GFD is initiated, inflammation and intestinal damage gradually decrease [3].
Anti-TG2 IgA antibody is an auto-antibody and the preferred serological marker for the diagnosis of CD due to its high sensitivity [1]. As is shown in Figure 3, there was no correlation between anti-TG2 IgA antibody and sCD14 levels in untreated and treated patients, suggesting that these molecules evaluate different components of the inflammatory process.
As was previously reported, we observed that anti-TG2 IgA antibody levels decrease after the beginning of a GFD in most patients. However, seroconversion (negative anti-TG2) in patients on a GFD does not necessarily imply recovery of the damage in the intestinal mucosa [24]. We found that sCD14 was significantly elevated in untreated CD patients compared to those on GFD, independent of the status of anti-TG2 IgA antibody (positive or negative). In this context, our results showed differential production of sCD14 between untreated patients and those on a GFD. This suggests that circulating levels of sCD14 may reflect microbial translocation and stimulation of sCD14 production.
LBP, another protein involved in the recognition of LPS, showed no significant difference in serum concentration between untreated CD patients and the control group; this result is in agreement with a previous report [16]. Likewise, no increased serum levels of LBP were observed in untreated patients compared to those on a GFD. Chronic exposure to LPS has been reported to decrease LBP levels [25]. Since intestinal permeability is altered in CD patients, chronic exposure to LPS may contribute to avoiding the increase of LBP levels in CD patients.
In this study, the production of sCD14 by both untreated CD and normal intestinal tissue was analyzed. We found that LPS induces the production of sCD14 by biopsies from CD patients, in contrast to uninflamed intestinal tissue from controls. These results indicate a role of LPS in the intestinal production of sCD14 in CD patients.
Monocytes/macrophages are an important cellular source of sCD14 [6]. Human intestinal macrophages, in contrast to non-intestinal macrophages, are non-inflammatory and present low expression of receptors of the innate immune system, showing a hyporesponse to LPS [26, 27]. In the presence of an inflammatory environment, as in Crohn’s disease, intestinal macrophages change, with accumulation of CD14hiCD11chi monocytes/immature macrophages. These cells, unlike those that are not found in an inflammatory environment, produce inflammatory cytokines when stimulated with components of bacterial microorganisms, such as LPS [28]. A similar situation could occur in celiac patients, where the sCD14-producing macrophages are found in an inflamed tissue, which may explain the increase in sCD14 production observed in response to LPS stimulus. Further studies are needed to fully elucidate the cause of the increase in sCD14 and the role it plays in the pathogenesis of the disease.
Conclusions
In conclusion, this study shows that circulating levels of sCD14, a co-receptor for LPS and a marker of endotoxemia, are increased in untreated CD patients compared to those in CD patients on a GFD.
Additionally, our data show that LPS induces the production of sCD14 from intestinal tissue of untreated CD patients, indicating a role of LPS in the intestinal production of sCD14 in patients with active CD. This study provides information on the production of proteins related to the exposure of LPS, an important inflammatory stimulus, in CD.
Acknowledgements
The authors thank Fundación Paraguaya de Celíacos (FUPACEL) for their support while conducting the study. This project was co-financed by Consejo Nacional de Ciencia y Tecnología – CONACYT (14-INV-171) with FEEI resources.
Footnotes
The authors declare no conflict of interest.
References
- 1.Al-toma A, Volta U, Auricchio R, et al. (2019): European Society for the Study of Coeliac Disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United Eur Gastroenterol J 7: 583-613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ludvigsson JF, Bai JC, Biagi F, et al. (2014): Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut 63: 1210-1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hujoel IA, Reilly NR, Rubio-Tapia A (2019): Celiac disease: clinical features and diagnosis. Gastroenterol Clin North Am 48: 19-37. [DOI] [PubMed] [Google Scholar]
- 4.Pagliari D, Urgesi R, Frosali S, et al. (2015): The interaction among microbiota, immunity, and genetic and dietary factors is the condicio sine qua non celiac disease can develop. J Immunol Res 2015: 1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kelly CP, Bai JC, Liu E, Leffler DA (2015): Advances in diagnosis and management of celiac disease. Gastroenterology 148: 1175-1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brenchley J, Douek D (2012): Microbial translocation across the GI tract. Annu Rev Immunol 30: 149-173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mu Q, Kirby J, Reilly CM, Luo XM (2017): Leaky gut as a danger signal for autoimmune diseases. Front Immunol 8: 1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kitchens RL, Thompson PA, Viriyakosol S, et al. (2001): Plasma CD14 decreases monocyte responses to LPS by transferring cell-bound LPS to plasma lipoproteins. J Clin Invest 108: 485-493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pugin J, Schürer-Maly CC, Leturcq D, et al. (1993): Lipopolysaccharide activation of human endothelial and epithelial cells is mediated by lipopolysaccharide-binding protein and soluble CD14. Proc Natl Acad Sci U S A 90: 2744-2748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nockher WA, Wigand R, Schoeppe W, Scherberich JE (1994): Elevated levels of soluble CD14 in serum of patients with systemic lupus erythematosus. Clin Exp Immunol 96: 15-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ayyappan P, Harms RZ, Buckner JH, Sarvetnick NE (2019): Coordinated induction of antimicrobial response factors in systemic lupus erythematosus. Front Immunol 10: 658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aravindhan V, Mohan V, Arunkumar N, et al. (2015): Chronic endotoxemia in subjects with type-1 diabetes is seen much before the onset of microvascular complications. PLoS One 10: 1-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brettschneider J, Ecker D, Bitsch A, et al. (2002): The macrophage activity marker sCD14 is increased in patients with multiple sclerosis and upregulated by interferon beta-1b. J Neuroimmunol 133: 193-197. [DOI] [PubMed] [Google Scholar]
- 14.Lakatos PL, Kiss LS, Palatka K, et al. (2011): Serum lipopolysaccharide-binding protein and soluble CD14 are markers of disease activity in patients with Crohn’s disease. Inflamm Bowel Dis 17: 767-777. [DOI] [PubMed] [Google Scholar]
- 15.Hoffmanová I, Sánchez D, Hábová V, et al. (2015): Serological markers of enterocyte damage and apoptosis in patients with celiac disease, autoimmune diabetes mellitus and diabetes mellitus type 2. Physiol Res 64: 537-546. [DOI] [PubMed] [Google Scholar]
- 16.Uhde M, Ajamian M, Caio G, et al. (2016): Intestinal cell damage and systemic immune activation in individuals reporting sensitivity to wheat in the absence of coeliac disease. Gut 65: 1930-1937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ranoa DRE, Kelley SL, Tapping RI (2013): Human lipopolysaccharide-binding protein (LBP) and CD14 independently deliver triacylated lipoproteins to toll-like receptor 1 (TLR1) and TLR2 and enhance formation of the ternary signaling complex. J Biol Chem 288: 9729-9741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zanoni I, Granucci F (2013): Role of CD14 in host protection against infections and in metabolism regulation. Front Cell Infect Microbiol 3: 1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Funda DP, Tucková L, Farré MA, et al. (2001): CD14 is expressed and released as soluble CD14 by human intestinal epithelial cells in vitro: lipopolysaccharide activation of epithelial cells revisited. Infect Immun 69: 3772-3781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Song PI, Park YM, Abraham T, et al. (2002): Human keratinocytes express functional CD14 and toll-like receptor 4. J Invest Dermatol 119: 424-432. [DOI] [PubMed] [Google Scholar]
- 21.Bas S, Gauthier BR, Spenato U, et al. (2004): CD14 is an acute-phase protein. J Immunol 172: 4470-4479. [DOI] [PubMed] [Google Scholar]
- 22.Marcos V, Latzin P, Hector A, et al. (2010): Expression, regulation and clinical significance of soluble and membrane CD14 receptors in pediatric inflammatory lung diseases. Respir Res 11: 1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kelesidis T, Kendall MA, Yang OO, et al. (2012): Biomarkers of microbial translocation and macrophage activation: association with progression of subclinical atherosclerosis in HIV-1 infection. J Infect Dis 206: 1558-1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lanzini A, Lanzarotto F, Villanacci V, et al. (2009): Complete recovery of intestinal mucosa occurs very rarely in adult coeliac patients despite adherence to gluten-free diet. Aliment Pharmacol Ther 29: 1299-1308. [DOI] [PubMed] [Google Scholar]
- 25.Gutsmann T, Müller M, Carroll SF, et al. (2001): Dual role of lipopolysaccharide (LPS)-binding protein in neutralization of LPS and enhancement of LPS-induced activation of mononuclear cells. Infect Immun 69: 6942-6950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang S, Ye Q, Zeng X, Qiao S (2019): Functions of macrophages in the maintenance of intestinal homeostasis. J Immunol Res 2019: 1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ueda Y, Kayama H, Jeon SG, et al. (2010): Commensal microbiota induce LPS hyporesponsiveness in colonic macrophages via the production of IL-10. Int Immunol 22: 953-962. [DOI] [PubMed] [Google Scholar]
- 28.Kamada N, Hisamatsu T, Okamoto S, et al. (2008): Unique CD14 + intestinal macrophages contribute to the pathogenesis of Crohn disease via IL-23/IFN-γ axis. J Clin Invest 118: 2269-2280. [DOI] [PMC free article] [PubMed] [Google Scholar]