Abstract
Background
Immune checkpoint inhibitors (ICIs) as a cancer immunotherapy have emerged as a treatment for multiple advanced cancer types. Because of enhanced immune responses, immune-related adverse events (irAEs), including endocrinopathies such as hypophysitis, have been associated with the use of ICIs. Most underlying mechanisms of ICI-related hypophysitis remain unclear, especially for programmed cell death-1 (PD-1)/PD-1 ligand 1 (PD-L1) inhibitors. We hypothesized that ICI-related hypophysitis is associated with paraneoplastic syndrome caused by ectopic expression of pituitary-specific antigens.
Methods
Twenty consecutive patients with ICI-related hypophysitis between 2017 and 2019 at Kobe University Hospital were retrospectively analyzed. Circulating anti-pituitary antibodies were detected using immunofluorescence staining and immunoblotting. Ectopic expression of pituitary autoantigens in tumor specimens was also examined.
Results
Eighteen patients were treated with PD-1/PD-L1 inhibitors, and two were treated with a combination of cytotoxic T-lymphocyte antigen-4 (CTLA-4) and PD-1 inhibitors. All patients showed adrenocorticotropic hormone (ACTH) deficiency and additionally, three showed thyroid-stimulating hormone (TSH) deficiency, and one showed gonadotropin-releasing hormone (GnRH) deficiency. Among these patients, three exhibited anti-pituitary antibodies, two with anti-corticotroph antibody and one with anti-somatotroph antibody. Interestingly, the anti-corticotroph antibody recognized proopiomelanocortin (POMC) and those two patients exhibited ectopic ACTH expression in the tumor, while the patients without anti-corticotroph antibody did not.
Conclusions
We demonstrated 10% of PD-1/PD-L1 inhibitors-related hypophysitis were associated with the autoimmunity against corticotrophs and maybe caused as a form of paraneoplastic syndrome, in which ectopic expression of ACTH in the tumor was observed. It is also suggested that the pathophysiology is heterogenous in ICI-related hypophysitis.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00262-021-02955-y.
Keywords: Autoimmunity, Immune checkpoint inhibitor, Hypopituitarism, Hypophysitis, Paraneoplastic syndrome
Introduction
The discovery of immune checkpoint inhibitors (ICIs) has revolutionized cancer treatment and has been shown to be effective for several types of advanced cancer [1]. However, these agents are associated with significant potential toxicities termed immune-related adverse events (irAEs). Particularly, several endocrinopathies, including hypophysitis, are often observed with the use of these agents [2]. However, the underlying mechanisms of these irAEs remain largely unknown.
Cytotoxic T-lymphocyte antigen-4 (CTLA-4) expressed on T cells suppresses T-cell activation, and the inhibition of CTLA-4 leads to T-cell activation and the inhibition of regulatory T cells [3, 4]. Interestingly, CTLA-4 is also expressed in the pituitary gland and may be directly involved in the development of hypophysitis [5]. On the other hands, programmed cell death-1 (PD-1) is mainly expressed on effector T cells [6] and binds to programmed cell death-1 ligand 1 (PD-L1) expressed by tumor cells.
Secretion of thyroid-stimulating hormone (TSH) and luteinizing hormone (LH)/follicle-stimulating hormone (FSH) is frequently impaired in the hypophysitis associated with CTLA-4 inhibitor therapy, along with impairment in adrenocorticotropic hormone (ACTH) secretion [7]. In contrast, PD-1 inhibitor also induces hypophysitis, but less frequently [8], with most of these patients developing isolated ACTH deficiency (IAD) [9]. Patients with PD-L1 inhibitor-related hypophysitis also develop IAD [10]. In addition, at the diagnosis of sellar masses in patients treated with ICIs, it should be taken into account that not PD-1/PD-L1-related hypophysitis but CTLA-4 inhibitor-related hypophysitis often reveals pituitary enlargement with headache; however, it is important to exclude a metastasis of the malignancy [11]. These data strongly suggest that the underlying mechanisms of PD-1 or PD-L1 inhibitor-related hypophysitis are different from those in CTLA-4 inhibitor therapy.
The importance of anti-pituitary antibodies (APAs) in pituitary autoimmunity associated with hypophysitis has been widely recognized [12]. In fact, several kinds of pituitary autoantibodies against thyrotrophs, corticotrophs, and gonadotrophs have been reported in patients with ICI-related hypophysitis [5]; however, it is currently unknown whether these autoantibodies play a causal role. Anti-corticotroph antibody has also been detected in patients with IAD, in which autoimmunity has been considered to be involved [13, 14]. One patient with IAD exhibited circulating anti-corticotroph antibody, as well as cytotoxic T cells that specifically recognize proopiomelanocortin (POMC) [15]. Interestingly, this patient’s complicated with a tumor that ectopically expressed POMC, suggesting that the IAD was caused by a form of paraneoplastic syndrome in the case.
In the current study, we hypothesized that ICI-related hypophysitis was caused as a paraneoplastic syndrome and aimed to clarify the significance of APAs.
Materials and methods
Patients
This study was approved by the ethics committee of Kobe University Graduate School of Medicine (#29–62). All methods were performed in accordance with the guidelines of the approved protocol. Patients provided written informed consent. Most patients were treated in Hyogo Cancer Center, and the diagnosis of ICI-related hypophysitis was performed in Kobe University Hospital. Twenty consecutive patients who diagnosed with ICI-related hypophysitis were enrolled. Most patients were treated with PD-1/PD-L1 inhibitors rather than CTLA-4 inhibitors because of the historical background in Japan.
Diagnosis of ICI-related hypophysitis and hormone assays
For the screening of hypopituitarism, basal levels of pituitary and peripheral hormones were measured [16]. In patients with a suspicion of hypopituitarism, provocation tests for anterior pituitary hormones and pituitary MRI were performed [17]. Provocative test was performed as previously described [17, 18] using insulin (0.05 unit/kg) or corticotropin-releasing hormone (CRH) (100 µg), thyroid-releasing hormone (TRH) (200 µg), luteinizing hormone-releasing hormone (LHRH) (100 µg), and growth hormone-releasing peptide-2 (GHRP-2) (100 µg). ICI-related hypopituitarism was defined and diagnosed according to the guidelines for endocrine-irAEs [16, 19, 20]. If basal level of serum cortisol was less than 4 µg/dL and there was no increase in basal ACTH level, we diagnosed ACTH deficiency. Growth hormone (GH), prolactin (PRL), TSH, LH, and FSH deficiencies were diagnosed by baseline hormone levels of the pituitary and its peripheral hormones. We diagnosed ACTH deficiency by decreased peak serum cortisol value (< 18 µg/dL) and impaired responses of ACTH (< twofold of baseline) in CRH test and TSH deficiency by impaired responses of TSH (< 10 µIU/mL) in TRH test. No patients had a history of exogenous steroid administration. Patients who underwent radiotherapy to the hypothalamus and/or pituitary area and have a history of pituitary surgery were excluded. Case 2 received Cyberknife radiosurgery for metastatic lesions in the brain; however, the hypothalamus and pituitary area were not exposed to radiation. The plasma ACTH, cortisol, and GH levels were measured by a chemiluminescent enzyme immunoassay (CLEIA; TOSOH, Tokyo, Japan), and PRL, TSH, free thyroxine 4 (T4), LH, FSH, estradiol (E2), and, testosterone levels were measured by a chemiluminescent immunoassay (CLIA; Abbott, Tokyo, Japan), and IGF-I level was assayed by an electrochemiluminescence immunoassay (ECLIA; Roche, Tokyo, Japan), respectively.
Animals
Experiments with animal tissues were performed according to the guidelines of the Animal Ethics Committee of Kobe University Graduate School of Medicine. The experimental protocols were approved by the Institutional Animal Care and Use Committee. Mouse pituitary tissues were used for immunohistochemical analyses.
Immunofluorescence staining and immunohistochemistry
For immunofluorescence staining of mouse pituitary specimens, tissues were fixed by perfusion with 4% paraformaldehyde, underwent heat-induced antigen retrieval with tris-ethylenediaminetetraacetic acid (EDTA) buffer (10 mM Tris base, 1 mM EDTA solution, 0.1% Tween 20, pH 9.0), and were permeabilized for 15 min at room temperature using phosphate-buffered saline (PBS) supplemented with 0.3% Triton X-100. The specimens were blocked using Blocking One buffer (Cat# 05,999–84, Nacalai Tesque, Kyoto, Japan), Fc Receptor Blocker (Cat# NB335, Innovex Biosciences, Richmond, CA, the USA), and True-Black Quencher (Cat# 23,007, Biotium, Hayward, CA, the USA). Subsequently, specimens were incubated with patient sera (1:20) for 24 h at 4 °C, washed three times with PBS supplemented with 0.05% Tween-20 (PBS-T), and then incubated with goat anti-human-IgG-Alexa Flour 488 antibody (Cat# ab70328, Abcam) for 2 h at room temperature (RT). This was followed by 24 h incubation with pituitary hormone antibodies, including anti-ACTH (Cat# Ab74976, Abcam, Cambridge, MA, the USA), anti-GH (Cat# sc-166696, Santa Cruz), anti-PRL (Cat# A0569, Dako, Carpinteria, CA), anti-TSH (Cat# M3503, Dako), anti-LH (Cat# M350201-2, Dako), anti-FSH (Cat# M3504, Dako) and anti-S100β (Cat# Ab52642, Abcam) antibodies. The secondary antibodies were donkey anti-mouse Alexa Flour 546 (Cat# ab10036, Abcam) and donkey anti-rabbit Alexa Flour 546 (Cat# ab10040, Abcam) as appropriate and were used. The nuclei were counterstained with Hoechst 33,342.
For the POMC absorption test, each serum sample (1:20) was incubated 24 h at 4 °C with or without 150 μg/mL of recombinant human POMC (rhPOMC) protein (Cat# GWB-P0950A, GenWay Biotech) prior to use for immunofluorescence staining.
In the immunohistochemistry for the detection of ectopic hormone expression, at least three slices that covered most of tissues were analyzed because the ectopic expression was sometimes confined in a part of the tumor. The fixed tissues underwent antigen retrieval, were permeabilized, and blocked above-mentioned. Subsequently, specimens were incubated with primary antibodies anti-ACTH (Cat# M3501, Dako) and anti-GH (Cat# A0570, Dako). Color development was performed using 3,3'-diaminobenzidine (DAB) as a chromogen. We obtained all images using a BZ-X710 fluorescence microscope (Keyence, Osaka, Japan) and then reconstructed the images using BZ-H3A software (Keyence). The representative results from a part of patients without anti-POMC antibody are shown.
For autoantibody detection, 0.1 µg of rhPOMC (GenWay Biotech) was applied for SDS-PAGE and immunoblotted with the serum from each patient or with six healthy control sera as primary antibody (1:50). Anti-POMC antibody (Cat# SAB1410992; SIGMA) was used as a control. The representative results are shown.
Results
Clinical characteristics of patients with ICI-related hypophysitis
Twenty consecutive patients who diagnosed with ICI-related hypophysitis [20] were enrolled in the study. Sixteen patients were treated with PD-1 inhibitor, two were treated with PD-L1 inhibitor, and two were treated with a combination of CTLA-4 and PD-1 inhibitors (Table 1). The average duration of treatment before the onset of hypophysitis was 9.0 wk (range, 8.5–9.5 wk) for the combination therapy, 20.5 wk (range, 12.3–36.3 wk) for the PD-1 inhibitor therapy, and 54.0 wk (range, 53.0–55.0 wk) for the PD-L1 inhibitor therapy. With respect to anterior pituitary hormone impairment at the time of the diagnosis of the hypophysitis, all patients exhibited ACTH deficiency, and three additionally exhibited TSH deficiency and one LH/FSH deficiency, in which the ACTH deficiency in all patients was comparable with the previous report [8] (Table 1). In the six patients being treated with levothyroxine, the treatment had been started before the diagnosis of hypophysitis (Table 1). Pituitary MRI was performed in 17 patients. A slight enlargement was observed in three patients, slight atrophy in three patients, and no obvious abnormalities in 11 patients.
Table 1.
Clinical characteristics of the patients
| Age | Cease No | Sex | ICI | Primary disease | Duration of ICI administration before onset (weeks) | Pituitary Hormone deficiency | Anti-pituitary antibody | Previous endocrine irAE | ACTH (pg/mL) |
|---|---|---|---|---|---|---|---|---|---|
| 76 | 1 | M | PD-1 + CTLA4 | Non-small cell lung cancer | 8 | ACTH | − | 2.3 | |
| 76 | 2 | M | PD-1 | Malignant melanoma | 20 | ACTH + TSH | Corticotroph | 3.6 | |
| 65 | 3 | M | PD-L1 | Non-small cell lung cancer | 56 | ACTH | − | 4.1 | |
| 71 | 4 | M | PD-L1 | Non-small cell lung cancer | 52 | ACTH | − | 4.8 | |
| 87 | 5 | M | PD-1 | Renal cell carcinoma | 7 | ACTH | − | Primary hypothyroidism | 4.3 |
| 68 | 6 | M | PD-1 | Renal cell carcinoma | 12 | ACTH | − | Primary hypothyroidism | 2.7 |
| 64 | 7 | F | PD-1 | Non-small cell lung cancer | 23 | ACTH | − | 12.2 | |
| 73 | 8 | M | PD-1 | Malignant melanoma | 12 | ACTH | − | 7.5 | |
| 65 | 9 | F | PD-1 | Stomach cancer | 21 | ACTH | − | Thyrotoxicosis | < 0.2 |
| 70 | 10 | F | PD-1 | Renal cell carcinoma | 25 | ACTH | − | 4.5 | |
| 53 | 11 | M | PD-1 | Stomach cancer | 17 | ACTH + TSH | Somatotroph | 10.7 | |
| 60 | 12 | M | PD-1 | Stomach cancer | 48 | ACTH + TSH | − | < 0.2 | |
| 71 | 13 | M | PD-1 | Renal cell carcinoma | 4 | ACTH | − | Primary hypothyroidism | 7.3 |
| 64 | 14 | M | PD-1 | Renal cell carcinoma | 28 | ACTH | Corticotroph | 9.3 | |
| 70 | 15 | M | PD-1 | Urinary tract cancer | 12 | ACTH | − | 3.8 | |
| 75 | 16 | M | PD-1 + CTLA4 | Esophageal cancer | 10 | ACTH | − | 4.9 | |
| 68 | 17 | M | PD-1 | Submandibular gland cancer | 47 | ACTH | − | 10.6 | |
| 35 | 18 | F | PD-1 | Large cell neuroendocrine carcinoma | 29 | ACTH + LH + FSH | − | Primary hypothyroidism | < 2 |
| 55 | 19 | M | PD-1 | Renal cell carcinoma | 9 | ACTH | − | Primary hypothyroidism | 3.2 |
| 72 | 20 | M | PD-1 | Non-small cell lung cancer | 28 | ACTH | − | Primary hypothyroidism | 3.6 |
| Cortisol (µg/dL) | GH (ng/mL) | IGF-I (ng/mL) | TSH (µIU/mL) | FT4 (ng/dL) | PRL (ng/mL) | LH (µIU/mL) | FSH (µIU/mL) | T (pg/mL) | E2 (pg/mL) |
|---|---|---|---|---|---|---|---|---|---|
| 0.3 | 0.4 | 95 | 2.2 | 1.2 | 16.1 | 6.5 | 12.8 | N/A | N/A |
| 0.8 | 0.2 | 78 | 4.1 | 0.7 | 13.6 | 4.7 | 6.9 | 2.7 | N/A |
| < 0.2 | 0.4 | 47 | 1.5 | 0.8 | 17.8 | 4.4 | 9.4 | 6.4 | N/A |
| 3.1 | 0.1 | 61 | 1.3 | 1.1 | 11.1 | 24.3 | 50.4 | 5 | N/A |
| 1.9 | 4.1 | 79 | 2.0 | 1.08* | 12.2 | 22.3 | 35.3 | 6.6 | N/A |
| 0.7 | N/A | 157 | 1.3 | 1.0* | 25.6 | 25.6 | 49.6 | 3.4 | N/A |
| 0.8 | 0.2 | 40 | 4.3 | 0.9 | 17.1 | 10.6 | 24.1 | N/A | < 10 |
| 0.7 | 0.5 | 91 | 4.9 | 1.0 | 13.2 | 2.8 | 7 | 6.7 | N/A |
| 2.4 | 2.4 | 49 | 0.05 | 2.5 | 21.1 | 36.6 | 95.7 | N/A | < 10 |
| 7.2 | 0.4 | 69 | 4.5 | 1.0 | 132 | 34.9 | 49.5 | N/A | < 10 |
| 2.5 | 1.3 | 92 | 0.4 | 0.8* | 4.1 | 2.5 | 23.3 | 2.8 | N/A |
| 0.9 | N/A | 56 | 2.3 | 1.1 | 21.1 | 6.6 | 11.8 | 5.2 | N/A |
| 7.8 | 0.1 | 110 | 1.8 | 1.0* | 10.1 | 7.9 | 20 | 7.8 | N/A |
| 2.9 | 0.6 | 46 | 3.2 | 1.1 | 14.5 | 9 | 21.2 | 7.7 | N/A |
| 0.9 | 0.1 | 164 | 2.4 | 0.8 | 17.5 | 8.3 | 16.1 | 7.4 | N/A |
| 0.6 | 0.3 | 104 | 9.9 | 0.7 | 14.4 | 10.5 | 22.2 | 5.5 | N/A |
| 1.8 | 0.2 | 109 | 1.4 | 1.1 | 10.4 | 2.6 | 3.9 | 8.2 | N/A |
| 3.1 | 0.6 | 113 | 1.9 | 1.1* | 13.4 | 0.1 | 1.4 | N/A | < 10 |
| 1.8 | N/A | 100 | 3.9 | 1.0* | 11.1 | 4.6 | 15.5 | 5.6 | N/A |
| 7.7 | N/A | 63 | 18.8 | 1.0 | 13.2 | 6 | 12.9 | 0.1 | N/A |
Anti-pituitary antibodies in patients with ICI-related hypophysitis
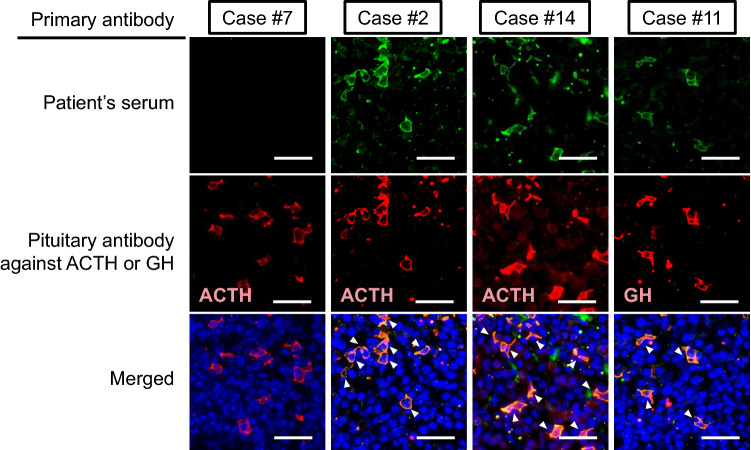
We evaluated the presence of circulating anti-pituitary antibodies using mouse pituitary tissue by immunofluorescent staining. Among the 20 patients, we detected anti-pituitary antibodies in three patients (15%) (Fig. 1 upper panel, Table 1). Next, we performed double staining analysis for each pituitary hormone (ACTH, TSH, GH, PRL, LH, and FSH) and S100β, demonstrating that the sera of two patients recognized corticotrophs and one recognized somatotrophs, while no merged signals were detected in the double staining with other pituitary hormone antibodies (Fig. 1 middle and lower panel, Supplementary File 1).
Fig. 1.
Immunofluorescence staining using patients’ serum and antibodies against pituitary hormones. Mouse pituitary tissue was stained with patients’ serum and anti-pituitary hormone antibodies. Serum of cases #2 and #14 recognized corticotrophs and that of case #11 recognized somatotrophs. Case #7 showed negative for circulating anti-pituitary antibody. Autoantibodies against other pituitary hormones were not detected (Supplementary Fig. 1). The representative results are shown. Scale bars; 100 µm
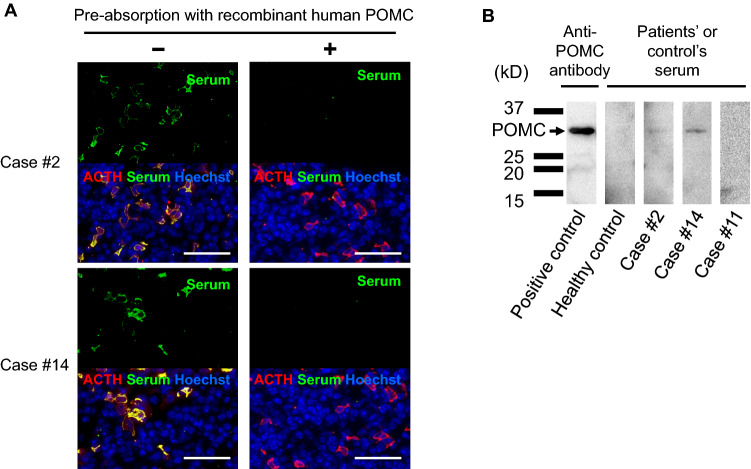
Autoimmunity against POMC in the patients with anti-corticotroph antibody
To clarify the corticotroph autoantigen in these patients, we performed a pre-absorption test using recombinant human POMC protein. Interestingly, pre-absorption of the sera with POMC protein diminished the reactivity against corticotrophs (Fig. 2a). In addition, immunoblotting using the patient sera (case #2 and #14) clearly demonstrated recognition of the POMC protein (Fig. 2b), indicating that the autoantibody was specific for POMC. Sera from the other patients and from healthy subjects did not show any signals (Fig. 2b, data not shown), indicating the specificity of this autoantibody.
Fig. 2.
Antigen-absorption experiments using rhPOMC protein in immunofluorescence and immunoblotting analysis using patients’ serum. a Pre-absorption of the serum with rhPOMC protein diminished the signal in cases #2 and #14, indicating that the autoantibody specifically recognized POMC protein. Scale bar: 50 µm. b The sera in the cases #2 and #14 specifically recognized a 29-kDa protein that corresponded to POMC protein (arrow). The representative results are shown. No sera from other cases nor healthy subjects exhibited anti-POMC antibody
ACTH was ectopically expressed in tumor tissues of the patients with anti-POMC antibody
Because it has been reported that ectopic ACTH expression is not rare in various cancers [14, 15], we hypothesized that the tumor ectopically expressed ACTH, resulting in the evocation of autoimmunity against ACTH. Interestingly, the neoplastic cells of malignant melanoma in patient #2 and renal cell carcinoma in patient #14 specifically exhibited ectopic ACTH expression, while the other tumors of patients without anti-corticotroph antibody did not (Fig. 3). In contrast, ectopic GH expression was not detected in the patient with anti-GH antibody (data not shown).
Fig. 3.
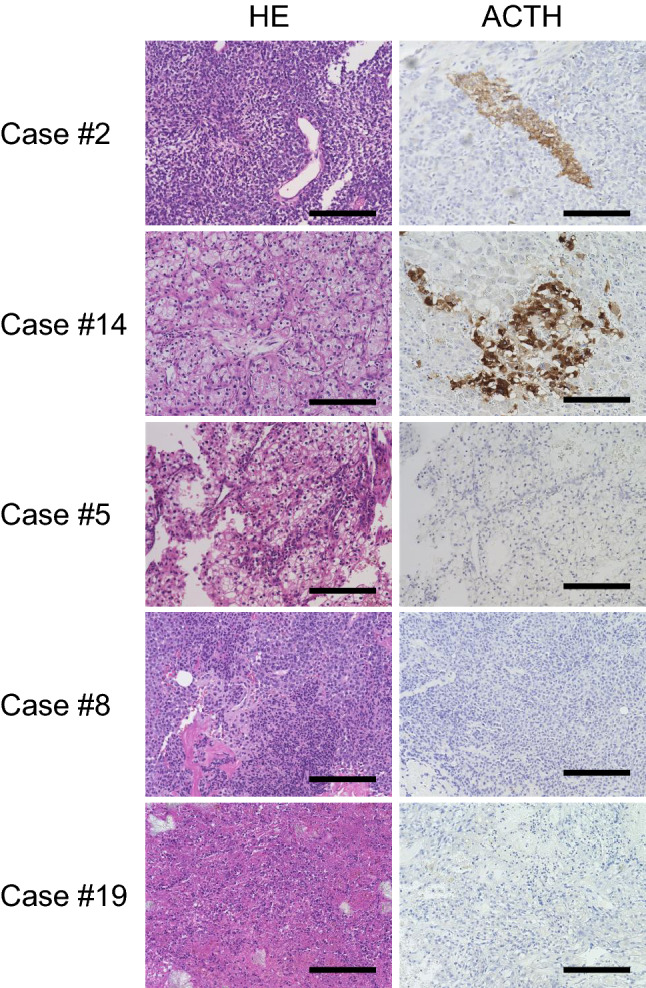
Specific ectopic ACTH expression was detected in the tumor specimens, in which case revealed anti-POMC antibody. Staining and immunohistochemistry for ACTH in the tumor tissues. Tumor specimen in the cases #2 and #14 specifically demonstrated ectopic ACTH expression. In contrast, tumor specimen in other cases was negative for ACTH. The representative results from a part of patients without anti-POMC antibody are shown. Scale bars: 200 µm
Discussion
Currently, the precise mechanisms of irAEs remain unclear; however, several biomarkers associated with the mechanisms have been suggested, including immune cell phenotypes [20], genetic variability of immune systems [21], and autoantibodies [5, 21]. Interestingly, in patients with metastatic melanoma treated with ipilimumab who developed irAEs, the repertoire of autoantibodies against self and cancer antigens preceded the development of irAEs [22]. In addition, increased T-cell activity against antigens present in tumors and normal tissues has been proposed as one of the underlying mechanisms [23]. These reports strengthened the importance of immune cross-reactivity between the tumor and involved organs as the mechanisms for irAE.
In the current study, we screened for circulating anti-pituitary antibodies as well as the ectopic antigen expressions in consecutive 20 patients with ICI-related hypophysitis who showed ACTH deficiency and demonstrated that two patient sera exhibited anti-corticotroph antibodies. Interestingly, these autoantibodies recognized POMC protein, and ectopic ACTH expression was specifically detected in the tumors of these patients. These data are in line with the hypothesis that ectopic ACTH expression in tumors can evoke autoreactive T-cell activation and ICI administration can enhance the autoimmunity, ultimately resulting in the specific injury of corticotrophs and ACTH deficiency.
In CTLA-4 inhibitor-related hypophysitis, it has been reported that the CTLA-4 expression in the anterior pituitary cells evoked a direct interaction of anti-CTLA-4 antibody with these cells and induced a complement-dependent cell injury in the pituitary [5]. In contrast, the underlying mechanisms in PD-1/PD-L1 inhibitor-related hypophysitis in the present study seem to be different because PD-L1 or PD-1 is not expressed in the pituitary [24, 25]. Also, the result that only a part of patients exhibited anti-corticotroph autoantibody suggested that there are other mechanisms.
It has been reported that in patients with IAD, circulating anti-corticotroph antibody has been detected in a portion of patients [26]. In addition, an epitope of endogenous proteins has been presented by MHC class I molecules in the anterior pituitary cells [27], which enables the recognition of specific T-cell receptors on cytotoxic T cells. Interestingly, a case of acquired IAD as a form of paraneoplastic syndrome was caused by autoimmunity against corticotrophs with the ectopic ACTH expression in the complicated tumor [15], and several cases of IAD were complicated with malignant tumors have been reported [28, 29]. Interestingly, ectopic ACTH, expression is not rare in various cancers, regardless of the type and ectopic expression of the other pituitary hormones is extremely rare [15].
These data may explain, at least in part, the reason for the preference of ACTH deficiency in ICI-related hypophysitis. We also detected anti-somatotroph antibody in one patient. ACTH, TSH, and LH/FSH deficiency, but not GH deficiency, were observed in this patient, and the ectopic expression of GH was not detected in the tumor. Although further investigation is necessary to clarify the significance of this anti-GH antibody, it is speculated that it did not play a pivotal role in the development of hypophysitis, rather, it may be results of destruction of the pituitary.
Generally, pharmacological dose of glucocorticoids is used as first-line therapy for treatment of irAEs. However, in terms of ICI-related hypophysitis, it is recommended to use physiological doses of glucocorticoids as a replacement therapy unless the presence of severe visual disturbance or intolerable headache because pharmacological dose of glucocorticoids does not restore pituitary function and recent study suggested high-dose glucocorticoids in patients with ICI-related hypophysitis might impair the effect of ICIs and survival [30, 31].
One limitation of the study is the sensitivity of autoantibody. The sensitivity for detecting the anti-pituitary antibody may not be enough because of the use of mouse pituitary tissue. The other limitation is the number of patients. However, considering the prevalence of hypophysitis with a strict diagnosis, it may be difficult to recruit more number of patients.
Conclusion
We demonstrated that 10% of PD-1/PD-L1 inhibitors-related hypophysitis were associated with the autoimmunity against corticotrophs and maybe caused as a form of paraneoplastic syndrome, in which ectopic expression of ACTH in the tumor was observed. It is also suggested that the pathophysiology is heterogenous in ICI-related hypophysitis. Although further investigation is necessary, these data provide insight into a novel mechanism for the etiology of irAE.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We would like to thank C. Ogita and K. Imura for their technical assistance.
Abbreviations
- ACTH
Adrenocorticotropic hormone
- APA
Anti-pituitary antibody
- CLEIA
Chemiluminescent enzyme immunoassay
- CLIA
Chemiluminescent immunoassay
- CRH
Corticotropin-releasing hormone
- CTLA-4
Cytotoxic T-lymphocyte antigen-4
- ECLIA
Electrochemiluminescence immunoassay
- EDTA,
Ethylenediaminetetraacetic acid
- E2
Estradiol
- Free T4
Free thyroxine 4
- FSH
Follicle-stimulating hormone
- GnRH
Gonadotropin-releasing hormone
- GHRP-2
Growth hormone-releasing peptide-2
- irAE
Immune-related adverse event
- IAD
Isolated ACTH deficiency
- ICI
Immune checkpoint inhibitor
- LH
Luteinizing hormone
- LHRH
Luteinizing hormone-releasing hormone
- PD-1
Programmed cell death-1
- PD-L1
Programmed cell death 1-ligand 1
- POMC
Proopiomelanocortin
- PRL
Prolactin
- TSH
Thyroid-stimulating hormone
- TRH
Thyroid-releasing hormone
Author Contributions
KK, GI, and YT designed the studies. KK performed the experiments. KK, GI, HB, SU, HS, YF, RM, KS, MY, HF, WO, and YT interpreted the data. KK and YT drafted the manuscript.
Funding
This work was supported by the Japan Society for the Promotion of Science (KAKENHI, Grant Numbers 23659477 and 26670459 [YT], 15K09431, 18K08514 [G.I.], and 17K16165 [H.B.]), Foundation for Growth Science (GI), Japan Agency for Medical Research and Development (17bm0804012h0001 [YT]), Uehara Memorial Foundation (YT), and Naito Foundation (YT).
Data availability
All data associated with this paper can be found in the main text.
Declarations
Conflict of interests
The authors declare that the study was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval and consent to participate
The Ethics Committee of the Kobe University Graduate School of Medicine reviewed and approved all research procedures involving human or animal participants. All patients expressed informed consent.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018;359(6382):1350–1355. doi: 10.1126/science.aar4060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy - immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13(4):195–207. doi: 10.1038/nrendo.2016.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Collins AV, Brodie DW, Gilbert RJC, Iaboni A, Manso-Sancho R, Walse B, Stuart DI, van der Merwe PA, Davis SJ. The interaction properties of costimulatory molecules revisited. Immunity. 2002;17(2):201–210. doi: 10.1016/S1074-7613(02)00362-X. [DOI] [PubMed] [Google Scholar]
- 5.Iwama S, De Remigis A, Callahan MK, Slovin SF, Wolchok JD, Caturegli P. Pituitary expression of CTLA-4 mediates hypophysitis secondary to administration of CTLA-4 blocking antibody. Sci Transl Med. 2014;6(230):230ra245. doi: 10.1126/scitranslmed.3008002. [DOI] [PubMed] [Google Scholar]
- 6.Fife BT, Bluestone JA. Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunol Rev. 2008;224:166–182. doi: 10.1111/j.1600-065X.2008.00662.x. [DOI] [PubMed] [Google Scholar]
- 7.Caturegli P, Di Dalmazi G, Lombardi M, Grosso F, Larman HB, Larman T, Taverna G, Cosottini M, Lupi I. Hypophysitis secondary to cytotoxic T-lymphocyte-associated protein 4 blockade: insights into pathogenesis from an autopsy series. Am J Pathol. 2016;186(12):3225–3235. doi: 10.1016/j.ajpath.2016.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kobayashi T, Iwama S, Yasuda Y, Okada N, Okuji T, Ito M, Onoue T, Goto M, Sugiyama M, Tsunekawa T, Takagi H, Hagiwara D, Ito Y, Suga H, Banno R, Yokota K, Hase T, Morise M, Hashimoto N, Ando M, Fujimoto Y, Hibi H, Sone M, Ando Y, Akiyama M, Hasegawa Y, Arima H. Pituitary dysfunction induced by immune checkpoint inhibitors is associated with better overall survival in both malignant melanoma and non-small cell lung carcinoma: a prospective study. J Immunother Cancer. 2020;8(2):e000779. doi: 10.1136/jitc-2020-000779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ohara N, Ohashi K, Fujisaki T, Oda C, Ikeda Y, Yoneoka Y, Hashimoto T, Hasegawa G, Suzuki K, Takada T. Isolated Adrenocorticotropin deficiency due to nivolumab-induced hypophysitis in a patient with advanced lung adenocarcinoma: a case report and literature review. Intern Med. 2018;57(4):527–535. doi: 10.2169/internalmedicine.9074-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kanie K, Iguchi G, Bando H, Fujita Y, Odake Y, Yoshida K, Matsumoto R, Fukuoka H, Ogawa W, Takahashi Y. Two cases of atezolizumab-induced hypophysitis. J Endocr Soc. 2018;2(1):91–95. doi: 10.1210/js.2017-00414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Prodam F, Caputo M, Mele C, Marzullo P, Aimaretti G. Insights into non-classic and emerging causes of hypopituitarism. Nat Rev Endocrinol. 2021;17(2):114–129. doi: 10.1038/s41574-020-00437-2. [DOI] [PubMed] [Google Scholar]
- 12.Ricciuti A, De Remigis A, Landek-Salgado MA, De Vincentiis L, Guaraldi F, Lupi I, Iwama S, Wand GS, Salvatori R, Caturegli P. Detection of pituitary antibodies by immunofluorescence: approach and results in patients with pituitary diseases. J Clin Endocrinol Metab. 2014;99(5):1758–1766. doi: 10.1210/jc.2014-1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sauter NP, Toni R, McLaughlin CD, Dyess EM, Kritzman J, Lechan RM. Isolated adrenocorticotropin deficiency associated with an autoantibody to a corticotroph antigen that is not adrenocorticotropin or other proopiomelanocortin-derived peptides. J Clin Endocrinol Metab. 1990;70(5):1391–1397. doi: 10.1210/jcem-70-5-1391. [DOI] [PubMed] [Google Scholar]
- 14.Sugiura M, Hashimoto A, Shizawa M, Tsukada M, Maruyama S, Ishido T, Kasahara T, Hirata Y. Heterogeneity of anterior pituitary cell antibodies detected in insulin-dependent diabetes mellitus and adrenocorticotropic hormone deficiency. Diabetes Res. 1986;3(3):111–114. [PubMed] [Google Scholar]
- 15.Bando H, Iguchi G, Kanie K, Nishizawa H, Matsumoto R, Fujita Y, Odake Y, Yoshida K, Suda K, Fukuoka H, Tanaka K, Ogawa W, Takahashi Y. Isolated adrenocorticotropic hormone deficiency as a form of paraneoplastic syndrome. Pituitary. 2018;21(5):480–489. doi: 10.1007/s11102-018-0901-7. [DOI] [PubMed] [Google Scholar]
- 16.Schneider HJ, Aimaretti G, Kreitschmann-Andermahr I, Stalla GK, Ghigo E. Hypopituitarism. Lancet. 2007;369(9571):1461–1470. doi: 10.1016/S0140-6736(07)60673-4. [DOI] [PubMed] [Google Scholar]
- 17.Fleseriu M, Hashim IA, Karavitaki N, Melmed S, Murad MH, Salvatori R, Samuels MH. Hormonal replacement in hypopituitarism in adults: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2016;101(11):3888–3921. doi: 10.1210/jc.2016-2118. [DOI] [PubMed] [Google Scholar]
- 18.Chihara K, Shimatsu A, Hizuka N, Tanaka T, Seino Y, Katofor Y. A simple diagnostic test using GH-releasing peptide-2 in adult GH deficiency. Eur J Endocrinol. 2007;157(1):19–27. doi: 10.1530/EJE-07-0066. [DOI] [PubMed] [Google Scholar]
- 19.Arima H, Iwama S, Inaba H, Ariyasu H, Makita N, Otsuki M, Kageyama K, Imagawa A, Akamizu T. Management of immune-related adverse events in endocrine organs induced by immune checkpoint inhibitors: clinical guidelines of the Japan Endocrine Society. Endocr J. 2019;66(7):581–586. doi: 10.1507/endocrj.EJ19-0163. [DOI] [PubMed] [Google Scholar]
- 20.Albarel F, Castinetti F, Brue T. Management of endocrine disease: immune check point inhibitors-induced hypophysitis. Eur J Endocrinol. 2019;181(3):R107–r118. doi: 10.1530/EJE-19-0169. [DOI] [PubMed] [Google Scholar]
- 21.Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS, Miller ML, Rekhtman N, Moreira AL, Ibrahim F, Bruggeman C, Gasmi B, Zappasodi R, Maeda Y, Sander C, Garon EB, Merghoub T, Wolchok JD, Schumacher TN, Chan TA. Cancer immunology mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Sci (New York, NY) 2015;348(6230):124–128. doi: 10.1126/science.aaa1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Da Gama DJ, Parakh S, Andrews MC, Woods K, Pasam A, Tutuka C, Ostrouska S, Blackburn JM, Behren A, Cebon J. Autoantibodies may predict immune-related toxicity: results from a phase I study of intralesional bacillus calmette-guérin followed by ipilimumab in patients with advanced metastatic melanoma. Front Immunol. 2018;9:411–411. doi: 10.3389/fimmu.2018.00411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378(2):158–168. doi: 10.1056/NEJMra1703481. [DOI] [PubMed] [Google Scholar]
- 24.Zhao J, Roberts A, Wang Z, Savage J, Ji RR. Emerging role of PD-1 in the central nervous system and brain diseases. Neurosci Bull. 2021 doi: 10.1007/s12264-021-00683-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pollack R, Kagan M, Dresner-Pollak R, Neuman T. PD-L1 expression in normal endocrine tissues is not increased despite high incidence of PD-1 inhibitor-associated endocrinopathies. Endocr Pract. 2021;27(1):34–37. doi: 10.1016/j.eprac.2020.11.004. [DOI] [PubMed] [Google Scholar]
- 26.Hannon MJ, O'Halloran DJ. Isolated acquired ACTH deficiency and primary hypothyroidism: a short series and review. Pituitary. 2011;14(4):358–361. doi: 10.1007/s11102-008-0164-9. [DOI] [PubMed] [Google Scholar]
- 27.Kanie K, Bando H, Iguchi G, Muguruma K, Matsumoto R, Hidaka-Takeno R, Okimura Y, Yamamoto M, Fujita Y, Fukuoka H, Yoshida K, Suda K, Nishizawa H, Ogawa W, Takahashi Y. Pathogenesis of Anti-PIT-1 antibody syndrome: PIT-1 presentation by HLA class I on anterior pituitary cells. J Endocr Soc. 2019;3(11):1969–1978. doi: 10.1210/js.2019-00243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kamiya Y, Murakami M. Type 2 diabetes mellitus accompanied by isolated adrenocorticotropic hormone deficiency and gastric cancer. Intern Med. 2009;48(12):1031–1035. doi: 10.2169/internalmedicine.48.1972. [DOI] [PubMed] [Google Scholar]
- 29.Yamaguchi H, Nakamura H, Mamiya Y, Yamamoto Y, Tajika K, Sugihara H, Gomi S, Inokuchi K, Hasegawa S, Shibazaki T, Dan K, Wakabayashi I. Acute lymphoblastic leukemia with isolated adrenocorticotropic hormone deficiency. Intern Med. 1997;36(11):819–821. doi: 10.2169/internalmedicine.36.819. [DOI] [PubMed] [Google Scholar]
- 30.Cooksley T, Girotra M, Ginex P, Gordon RA, Anderson R, Blidner A, Choi J, Dougan M, Glezerman I, Gupta D, Johnson D, Shannon VR, Suarez-Almazor M, Rapoport BL. Multinational association of supportive care in cancer (MASCC) 2020 clinical practice recommendations for the management of immune checkpoint inhibitor endocrinopathies and the role of advanced practice providers in the management of immune-mediated toxicities. Support Care Cancer. 2020;28(12):6175–6181. doi: 10.1007/s00520-020-05709-1. [DOI] [PubMed] [Google Scholar]
- 31.Faje AT, Lawrence D, Flaherty K, Freedman C, Fadden R, Rubin K, Cohen J, Sullivan RJ. High-dose glucocorticoids for the treatment of ipilimumab-induced hypophysitis is associated with reduced survival in patients with melanoma. Cancer. 2018;124(18):3706–3714. doi: 10.1002/cncr.31629. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data associated with this paper can be found in the main text.