Abstract
Exosomes play a role as mediators of cell-to-cell communication, thus exhibiting pleiotropic activities to homeostasis regulation. Exosomal non-coding RNAs (ncRNAs), mainly microRNAs (miRNAs), long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs), are closely related to a variety of biological and functional aspects of human health. When the exosomal ncRNAs undergo tissue-specific changes due to diverse internal or external disorders, they can cause tissue dysfunction, aging, and diseases. In this review, we comprehensively discuss the underlying regulatory mechanisms of exosomes in human diseases. In addition, we explore the current knowledge on the roles of exosomal miRNAs, lncRNAs, and circRNAs in human health and diseases, including cancers, metabolic diseases, neurodegenerative diseases, cardiovascular diseases, autoimmune diseases, and infectious diseases, to determine their potential implication in biomarker identification and therapeutic exploration.
Subject terms: Predictive markers, Cancer, Endocrine system and metabolic diseases, Cardiovascular diseases, Neurodevelopmental disorders
Introduction
Exosomes are a class of extracellular vesicles (EVs) ~30–150 nm in diameter.1,2 Exosomes were first reported in sheep reticulocytes in 1983. Further studies reported that exosomes are derived from most cell types and are present in the cell-conditioned medium and distinct biological fluids such as serum, plasma, urine, saliva, ascites, cerebrospinal fluid, and amniotic fluid.3 Exosomes were initially regarded as means of cellular waste disposing until further studies reported their role in mediating cell-to-cell communication, thus attracting significant attention of scholars worldwide.4,5 Several functions of exosomes have been characterized including cellular proliferation, differentiation, apoptosis, angiogenesis, and immune regulation.6,7 Exosomes exhibit these functions by interacting with surface receptors of recipient cells thus transmitting biomolecules such as lipids, proteins, messenger RNAs (mRNA), and non-coding RNAs (ncRNAs) to recipient cells.8 Notably, ncRNAs are the components of exosomes that have attracted particular attention.9
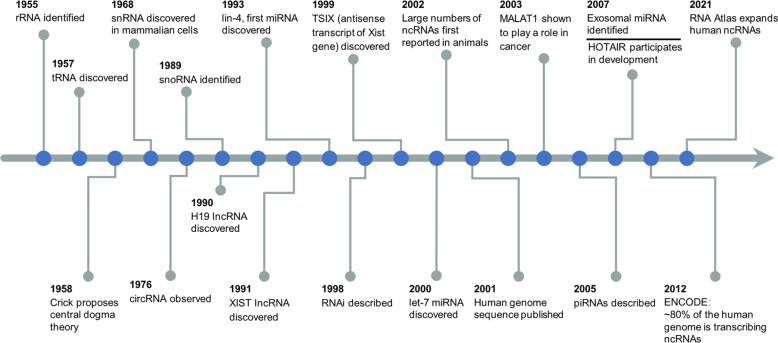
NcRNAs refer to molecules that lack protein-coding regions, which have become a hot topic of increasing concern. The correlation of ncRNAs with human diseases has primarily been identified in the function and expression of miRNA found in cancers. However, the extent of ncRNAs involvement in diseases is only just being explored. To better understand the discovery and research history of ncRNAs in human health and diseases, it is helpful to review the timeline of ncRNAs (Fig. 1). Discovery of housekeeping ncRNAs, such as ribosomal RNA (rRNA)10 and transfer RNA (tRNA)11 in the 1950s, supported Crick’s “central dogma” theory, which stated that genetic information can proceed from DNA to RNA to protein.12 Further studies reported new ncRNAs, such as small nuclear RNAs (snRNA),13,14 small nucleolar RNAs (snoRNA)15,16 and circular RNAs (circRNAs).17 In the late 1980s, studies first reported long non-coding RNAs (lncRNAs), such as H1918 and Xist.19,20 The human genome sequence was published in 2001,21 and the findings showed that genes that encoded proteins only accounted for 1.2% of the genome, whereas the rest were considered as “non-coding”.22 The first small temporal RNAs, lin‑4,23 and let‑7,24 were discovered in Caenorhabditis elegans in 1993 and 2000, respectively. Analysis showed that ncRNA can act as conserved functional molecules needed for development. Further studies showed that large numbers of gene do not encode proteins but encode various unique transcripts.25–28 NcRNAs can regulate gene expression at transcriptional, post-transcriptional, and translational levels, thereby modulating associated signaling networks.29 In addition, different kinds of ncRNAs can interact with each other to regulate their stability or abundance.30 In 2007, it was reported that exosomes contain microRNAs (miRNAs) and mRNA and can transfer them to other cells.31 Since then, accumulating evidence demonstrates that a variety of ncRNAs can be encapsulated and transported by exosomes, the most attractive of which are miRNAs, lncRNAs, and circRNAs, explaining their roles in intercellular communication.32,33 Notably, exosomal ncRNAs exhibit diverse expression patterns in different cells or various physiological and pathological conditions, indicating the potential role of these exosomal biomolecules in occurrence and development of different diseases.34,35 These differences in expression levels in pathological states indicate that exosomal ncRNAs are promising diagnostic and therapeutic tools for various human diseases.9,36–38 A large number of studies dealing with circulating exosomes and their cargoes prove that exosomal miRNAs, lncRNAs and circRNAs are closely involved in human health and the initiation and development of various diseases.39 Therefore, we specifically focus on effects of exosomal ncRNAs (miRNAs, lncRNAs, and circRNAs) in physiopathology, clinical diagnosis, and therapy of human diseases, such as metabolic diseases, cancers, neurodegenerative diseases, cardiovascular diseases, autoimmune diseases, and infectious diseases.
Fig. 1.
Timeline of the discovery and research history of ncRNAs in human health and diseases. Key discoveries are highlighted. XIST X (inactive)-specific transcript, RNAi RNA interference, piRNA PIWI-interacting RNA, HOTAIR HOX transcript antisense RNA, ENCODE Encyclopedia of DNA Elements
Potential regulatory mechanisms of exosomes in human diseases
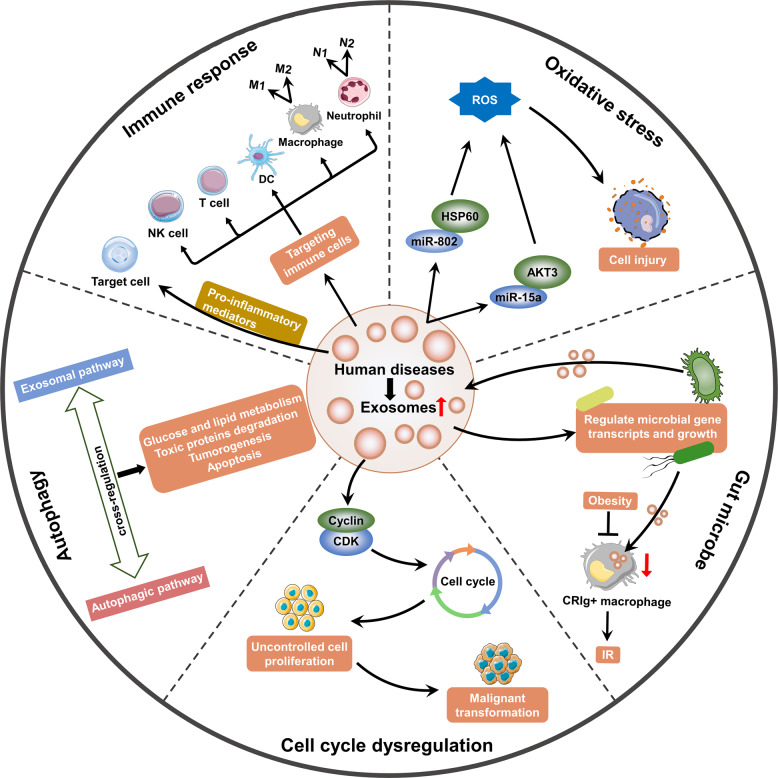
Several studies reported the importance of exosomes in human health and diseases.40,41 In addition, recent studies demonstrated that exosomes could exert their roles by modulating immune response, oxidative stress, autophagy, gut microbe, and cell cycle.42–45 This section provides a comprehensive overview of the current understanding of exosomes research and discusses potential mechanisms of exosomes in human diseases (Fig. 2).
Fig. 2.
The potential regulatory mechanisms of exosomes in human diseases. Several mechanisms of the occurrence of human diseases are modulated by exosomes, including immune response, oxidative stress, autophagy, gut microbe, and cell cycle. This figure was created with the aid of Servier Medical Art (https://smart.servier.com/). ROS reactive oxygen species, HSP heat shock protein, AKT3 AKT serine/threonine kinase 3, IR insulin resistance, CDK cyclin-dependent kinase, NK natural killer, DC dendritic cell
Immune response
Exosomes released from immune and non-immune cells exert a pivotal effect in immune regulation.46–48 Recent studies reported the functions of exosomes in triggering or inhibiting immune response, indicating their potential roles in the development and progression of autoimmune and inflammatory diseases.49–54
Exosomes can regulate immune response via the transfer and presentation of antigenic peptides. Exosomes derived from antigen-presenting cells (APCs) activate T cells by carrying major histocompatibility complex class II (MHC-II) that binds to antigenic peptides.48 Notably, APC-released exosomes with MHC-II bearing tumor peptides, significantly inhibit tumor growth in mice in a T cell-dependent manner.55 In addition, exosomes from APCs carrying bacterial antigens promote activation of anti-bacterial immunity. For instance, macrophage-derived exosomes carrying mycobacterial antigens protect mice against mycobacterium tuberculosis infection by inducing CD4+ and CD8+ T cells to produce IFN-γ and IL-2.56 However, excessive immune response mediated by exosomes can cause normal tissue damage thus promoting onset and development of diseases. For example, circulating exosomes from patients with Hashimoto thyroiditis can present antigens to dendritic cells (DCs), thus inducing DC activation through the NF-κB signaling pathway, contributing to imbalanced differentiation in CD4+ T cells, and potentially leading to Hashimoto thyroiditis onset.54
Exosomes can mediate tumor or pathogen immune escape by affecting gene expression in immune cells, mainly by delivering miRNAs, thus promoting progression of cancers and infection. For instance, tumor-derived exosomal miR-212-3p downregulates expression of regulatory factor X-associated protein (RFXAP), which inhibits MHC-II and promotes immune tolerance of dendritic cells.57 Moreover, virus-infected cell can transmit viral miRNAs to uninfected host immune cells through exosomes, thus downregulating immunomodulatory genes.58 Therefore, exosomes present a novel intersection between immune response and disease.
Moreover, exosomes seem to be involved in the regulation of macrophage and neutrophil polarization, which ultimately can induce the pathophysiologic process of several diseases. Macrophages comprise a population of heterogeneous cells which are classified into two classes including pro-inflammatory M1 or anti-inflammatory M2 macrophages based on their activation status.59 Exosomes can induce macrophage differentiation into M1 or M2 phenotypes, a critical regulatory mechanism of inflammation, which has essential effects on homeostasis.60–63 For example, adipose-derived exosomes exacerbate insulin resistance (IR) and atherosclerosis by inducing macrophage M1 polarization.62,64 Moreover, exosomes derived from cancer cells can also promote M1 macrophage polarization. Xiao and coworkers reported that exosomal thrombospondin-1 (THBS1) secreted by oral squamous cell carcinoma cells can activate M1 macrophages polarization to promote malignant migration.65 Notably, exosomes can induce activation of M2 macrophages thus inhibiting inflammatory response, leading to abrogation of many diseases.66,67 Studies reported that exosomes from mesenchymal stem cells (MSCs) ameliorate cardiac damage in myocardial infarction rats and ischemia/reperfusion mice by activating macrophage M2 polarization.68,69 However, M2 macrophages polarization can be detrimental. For example, M2 macrophages support tumor growth and survival.70 Studies report that tumor-derived exosomes promote occurrence and development of tumor by activating M2 phenotypes. Exosomal DLX6-AS1 from HCC cells triggers M2 macrophage polarization to provoke tumor invasion and migration through the miR-15a-5p/CXCL17 axis.71 Besides, exosomes can play a crucial role in disease progression by inducing neutrophil polarization.72,73 Laboratory tests have confirmed the existence of N1 (antitumoral) and N2 (protumoral) tumor-related neutrophils, parallel to M1 and M2 macrophage polarization.74 Moreover, tumor-derived exosomes can induce N2 polarization of neutrophils to promote cancer progression.72 Conversely, exosomes that inhibit neutrophil inflammatory response can alleviate the tissue injury and have the therapeutic potential.75
In addition, exosomes can modulate immune response by transporting cytokines or other pro-inflammatory mediators thus directly acting on target organs.76–78 Macrophage-derived exosomes containing miR-21-5p promote inflammatory activation and regulate podocyte injury in diabetic nephropathy mice.79 Fabbri et al. found that tumor-secreted exosomal miRNAs induce inflammatory response that may contribute to tumor growth and metastasis.80 Conversely, some exosomes exert anti-inflammatory effects that can be targeted for development of therapies.81 For instance, exosomal miR-192 significantly attenuates tumor metastasis by suppressing secretion of proangiogenic factors, such as interleukin (IL)-8, intercellular cell adhesion molecule (ICAM), and C-X-C motif chemokine ligand 1(CXCL1).82
Collectively, exosomes have been identified to regulate immune responses by carrying biomolecules to targeted cells, thereby affecting the phenotype and immunomodulatory functions of immune cells, or directly acting on target organs. Particularly, exosomes derived from immune cells or non-immune cells exert pivotal roles in immunotherapy. In this section, we discuss the roles of exosomes as carriers for regulating immune responses and as predictive biomarkers for immune activation.
Oxidative stress
Oxidative stress refers to an imbalance in oxidative-antioxidative systems and leads to excessive accumulation of reactive oxygen species (ROS), which contributes to various disorders by inducing cell and tissue dysfunction.
Nutrition stress promotes oxidative stress.83 Advanced glycation end-products (AGEs) play an important role in ROS production and promote oxidative stress in diabetes individuals.84 Exosomes exhibit an essential role in oxidative stress. Exosomal miR-802-5p derived from hypertrophic adipocyte causes cardiac IR by suppressing expression of heat shock protein 60 (HSP60), which is implicated in promoting oxidative stress.85 Kamalden and coworkers reported that pancreatic β-cells-derived exosomal miR-15a can travel through the circulation and induce oxidative stress by targeting AKT serine/threonine kinase 3 (AKT3), leading to retinal injury in T2DM subjects.43 Furthermore, exosomal-miR-21-5p from macrophages induces podocyte injury in diabetic nephropathy mice partially by promoting ROS production.79 In addition, oxidative stress is associated with age-related bone loss that increases the risk of osteoporosis.86 Exosomes from serum of aged normal individuals can exert a protective effect on bone health by inhibiting aging-related oxidative stress.87 Recent studies have documented that exosomal circHIPK3 produced by hypoxia-pretreated cardiomyocytes decreases oxidative stress-induced cardiac microvascular endothelial cells dysfunction.88
To sum up, these findings indicate the relationship between exosomes and oxidative stress in human disease. Exosomes have been shown to play a vital part in modulation of oxidative stress during the occurrence and development of various diseases.
Autophagy
Under normal physiological conditions, autophagy serves as a protective mechanism to remove protein aggregates, impaired organelles, and invading pathogens, and is implicated in recycling amino acids, lipids, and sugars to maintain cellular renovation and homeostasis.89,90 However, autophagy dysfunction is associated with several diseases, such as cancers, neurodegenerative diseases, and metabolic diseases. The crosstalk between autophagy and exosome biogenesis varies with the type of disease.91 Autophagy can reduce release of exosomes through multivesicular bodies (MVB) degradation.92 In addition, exosome release and autophagy can act synergistically against cell stress.93
Reduced autophagy level is reported in multiple diseases, such as obesity, hyperglycemia, and osteoporosis.90,94–96 Li and colleagues revealed that defective autophagy induced by adipose sirtuin1 (SIRT1) deficiency increases exosome release from adipose tissue, thus promoting decreased glucose tolerance, diminished insulin sensitivity, and impaired lipid metabolism.97 Mammalian sterile 20-like kinase 1 (Mst1)-enclosed exosomes from cardiac microvascular endothelial cells (ECs) are implicated in inhibition of autophagy, promotion of apoptosis and suppression of glucose uptake in diabetic cardiomyopathy.98 Notably, exosomes play a cell protective role by activating intracellular autophagy.91 MSC transplantation after myocardial infarction improves cardiac function and infarct size partially through release of exosomes that improve autophagic flux. Accumulating evidence indicates that MSC-derived exosomes alleviate T2DM complications including diabetic ulcers and nephropathy by inducing autophagy.44,99–101 A similar effect is reported in exosomes released from M2 macrophage.102
Neuronal cells can eliminate protein aggregates to ameliorate proteotoxicity through autophagic degradation and exosome release. Conversely, abnormal accumulation and aggregation of proteins are manifestations of various neurodegenerative diseases. Exosome secretion can be elevated to attenuate the toxic proteins during autophagic or lysosomal dysfunction. For instance, Yang and coworkers identified secretory carrier membrane protein 5 (SCAMP5) as an autophagy inhibitor that promote exosomal secretion of alpha-synuclein (α-SYN).103
Studies report that cancer cell-derived exosomes affect autophagy in recipient cells.104 In addition, exosome regulate drug resistance and tumor microenvironment in an autophagy-dependent manner.105 For instance, exosomal circ-PVT1 promotes cisplatin resistance in gastric cancer cells by inducing cell autophagy and invasion and inhibiting apoptosis.106 Furthermore, gastric cancer cell-derived exosomes trigger autophagy and promote activation of neutrophils, ultimately promoting gastric cancer cell migration.72
Altogether, these findings indicate that exosomes play important roles in multiple physiological and pathological processes by regulating autophagy. Besides, the biogenesis and release of exosomes are closely associated with autophagy in diseases. Autophagy dysfunction is one of the important potential mechanisms of exosomes in many diseases, such as metabolic diseases, neurodegenerative diseases, and cancers.
Gut microbe
Gut microbes exert important roles in physiological processes, such as providing essential nutrients, assisting in cellulose digestion, regulating integrity of gut barriers and immune response.107 However, dysbiosis, the imbalance in microbiota composition and diversity, in response to internal changes or external stimuli is associated with several chronic diseases, such as autoimmune, metabolic, cardiovascular diseases, and cancer.108–110 Intestinal homeostasis relies on dynamic and coordinated interactions between microbes, epithelium, and host immune system. Accumulating evidence has supported that exosomes provide a link between the host and gut microbial community. Liu and coworkers uncovered a new effect of fecal exosomal miRNAs on shaping gut microbiota. Intestinal ECs-derived miRNAs-containing exosomes can enter bacteria and regulate gene transcripts and growth, and their loss leads to dysbiosis and aggravation of colitis.111 These findings indicate the key roles of fecal exosomal miRNAs on maintaining normal gut microbiota.
Exosomes from beneficial microbes can improve metabolic functions through various mechanisms. Recent studies report that exosomes can restore intestinal and metabolic homeostasis in HFD-induced obesity mice by reversing the adverse effects of obesity including adipose and gut inflammation, intestinal mucosal barrier permeability, and fat weight gain.45,112 Conversely, maleficent bacteria is implicated in impairing of metabolic homeostasis. Stool exosomes from Pseudomonas panacis in HFD mice promote glucose intolerance and IR in healthy mice.113 Liver CRIg+ (complement receptor of the immunoglobulin superfamily) macrophage can clear bacteria and their products from the bloodstream. A recent study reported a decrease in CRIg+ macrophage population in obese subjects. As a result, less circulating gut microbial DNA-containing exosomes were eliminated, and more exosomes diffused to distant metabolic tissues, thus aggravating tissue inflammation and IR.114 In addition, gut microbiota are involves in modulation of bone metabolism. A recent study reported that gut microbiota in children and Akkermansia muciniphila release exosomes to bone tissues to ameliorate osteoporosis via promoting osteogenic activity and decreasing osteoclast formation.115
With the relevant cumulative findings, we summarize and indicate the role of exosomes as a link between gut microbiota and diseases. Moreover, these findings suggest that the effects of exosomes on the microbiome may be utilized to target specific host processes to ameliorate diseases.
Cell cycle dysregulation
Cell proliferation and division are basic cell physiological activities. Growth factors, hormones, and oncogene products can induce or inhibit cell proliferation, thereby influencing the cell cycle. Dysregulation of the cell cycle is associated with multiple diseases. Accelerated cell cycle can lead to carcinogenesis.116 Recent studies have revealed the essential role of exosomes in regulating cell cycle. Exosomal circRNA_100284 accelerates cell cycle and promotes proliferation by targeting miR-217, which induces malignant transformation of human hepatic cells.117 In addition, exosomal lncRNA ZFAS1 promotes gastric cancer progression by shortening cell cycle and epithelial-mesenchymal transition (EMT).118 Conversely, exosomes that block cell cycle in cancer can serve as therapeutic targets. ADSC-released exosomal-miRNAs present inhibitory effects on ovarian cancer cells by blocking cell cycle and inducing apoptosis signaling.119 These findings show the dual effects of exosomes on cell cycle regulation in the initiation and progression of cancers.
Differences of exosomal and non-exosomal ncRNAs in health and diseases
It has been confirmed that ncRNAs exist not only in cells, but also in different body fluids such as serum, plasma, urine, saliva, and so on. The ncRNAs in biofluids are often referred to as extracellular ncRNAs or circulating ncRNAs. Notably, the RNase activity is high in the extracellular environment, but extracellular ncRNAs remain relatively stable in plasma, suggesting that circulating ncRNAs may be protected and circumvented from harsh conditions. One intriguing mode of transport of circulating ncRNAs is related to exosomes. In this section, we try our best to investigate the differences between ncRNAs in exosomes and non-exosomes in regulating physiological homeostasis and pathological processes in health and diseases (Fig. 3).
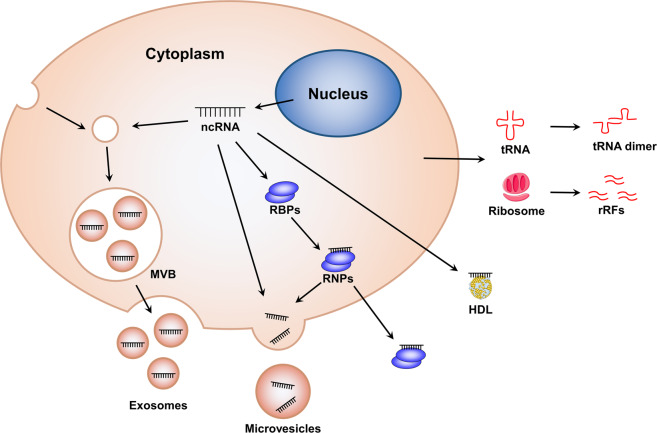
Fig. 3.
Different mechanisms underlying the stability of extracellular ncRNAs. NcRNAs can be protected from harsh extracellular environment through extracellular vesicles encapsulation (such as exosomes and microvesicles), ribonucleoprotein (RNP) complex formation, and high-density lipoprotein (HDL) transportation. Moreover, some extracellular RNA fragments that generate from non-vesicular ncRNAs in extracellular space can form self-protecting dimers. The source of these non-vesicular RNAs remains uncertain. This figure was created with the aid of Servier Medical Art (https://smart.servier.com/). MVB multivesicular bodies, RBPs RNA-binding protein, rRFs rRNA-derived fragments
NcRNAs can be encapsulated by EVs (including exosomes, microvesicles, and apoptotic bodies) and secreted out of cells to act as mediators for intercellular communication, thereby regulating different diseases according to the target cells. In addition to the vesicle-dependent pattern, a considerable number of extracellular ncRNAs are present in the form of ribonucleoprotein (RNP) complexes with RNA-binding proteins (RBPs) such as argonaute-2 (AGO2) that modulate mRNA inhibition in cells.120,121 These RBPs can affect RNA sorting into EVs in an indirect manner.122 Moreover, high-density lipoprotein (HDL) can also transport endogenous ncRNAs to recipient cells with functional targeting capabilities, the effect of which may vary depending on the disease state.123 Further, recent studies have revealed a post-release processing of ncRNAs. Some extracellular RNA fragments that generate from non-vesicular ncRNAs, such as ribosomes and full-length tRNAs, in extracellular space can form self-protecting dimers to resist RNases.124,125
The roles of exosomal ncRNAs in human diseases
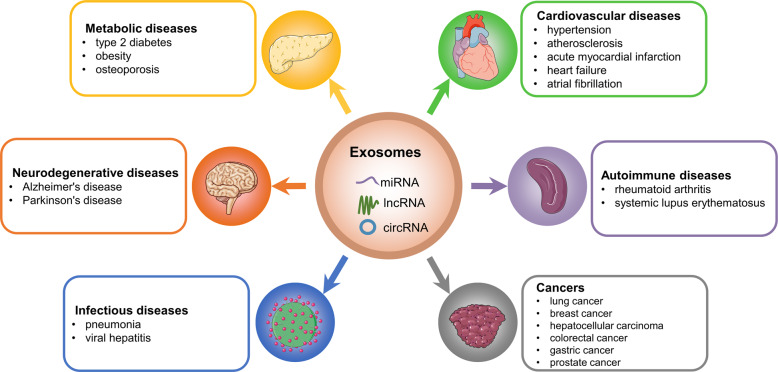
Accumulating data suggest that exosomal ncRNAs exert pleiotropic effects on human diseases.126,127 Among these ncRNAs cargoes, the most intriguing ones are miRNAs, lncRNAs, and circRNAs. MiRNAs are small, highly conserved ncRNAs.128 LncRNAs are poorly conserved ncRNAs with a length of more than 200 nucleotides.30 CircRNAs are a subset of ncRNAs with covalently closed structures which are implicated in the regulation of gene expression.129 In this section, the roles of exosomal miRNAs, lncRNAs, and circRNAs in different human diseases (including cancers, metabolic diseases, cardiovascular diseases, neurodegenerative diseases, autoimmune diseases, and infectious diseases) are explored (Fig. 4).
Fig. 4.
The roles of exosomal ncRNAs in human diseases. The figure showed examples of human diseases where exosomal ncRNAs exert pivotal function. This figure was created with the aid of Servier Medical Art (https://smart.servier.com/)
The roles of exosomal ncRNAs in cancers
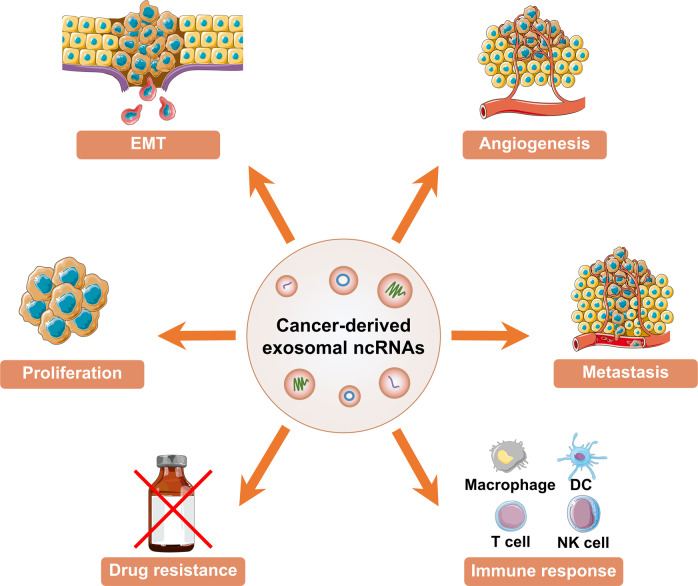
GLOBOCAN statistics report that approximately 14.1 million new cancer cases were diagnosed and 8.2 million deaths occurred in 2012. Prevalence of cancer is raising owing to the increase in population growth and aging population, creating a huge health burden for both patients and society.130 Recent studies have explored tumor-associated exosomal ncRNAs.131 Emerging studies revealed that exosomal ncRNAs are implicated in progression of human cancers, such as lung cancer,132 breast cancer (BC),133 and hepatocellular carcinoma (HCC).134 Exosomal ncRNAs play a role in cancers, including EMT, proliferation, angiogenesis, metastasis, drug resistance, and immune-inflammation (Fig. 5). In this section, the roles of exosomal miRNAs, lncRNAs, and circRNAs in various human cancers, including lung cancer, BC, HCC, colorectal cancer (CRC), gastric cancer (GC), and prostate cancer (PCa) are summarized.
Fig. 5.
The roles of exosomal ncRNAs in cancer. Exosomal ncRNAs play a role in cancers, including EMT, proliferation, angiogenesis, metastasis, drug resistance, and immune response. This figure was created with the aid of Servier Medical Art (https://smart.servier.com/). EMT epithelial-mesenchymal transition, DC dendritic cell, NK natural killer
Exosomal ncRNAs in lung cancer
Lung cancer is the leading cause of cancer-related mortality globally. Approximately 70% of lung cancer patients present with complex complications at the time of diagnosis and surgical resection is the primary treatment option for lung cancer.135 Association of exosomal ncRNAs and lung cancer can be explored to identify novel biomarkers for tumor targeted diagnosis and therapy.132 Numerous lines of publications have indicated that exosomes and exosomal ncRNAs exert important roles in multiple cellular and molecular processes linked to lung cancer, thus providing new diagnostic biomarkers and therapeutic targets in lung cancer.
EMT is tightly associated with tumor invasion and metastasis by promoting lung cancer cells infiltration and migration.136 Kim et al. reported that miR-23a is significantly enriched in TGF-β1-treated human lung adenocarcinoma (LUAD) cells and is involved in EMT.137 Notably, bone marrow-derived mesenchymal stem cells (BMSCs) are essential components of cancer microenvironment and are involved in development of lung cancer. Zhang et al. demonstrated that BMSCs-derived exosomal miR-193a-3p, miR-210-3p, and miR-5100 promote lung cancer cells invasion by activating signal transducer and activator of transcription 3 (STAT3) signaling pathway and triggering EMT.138 Furthermore, miR-499a-5p is upregulated in highly metastatic cells-derived exosomes and enhances cell proliferation, migration, and EMT by targeting the rapamycin (mTOR) pathway.139
Cellular proliferation and migration are implicated in cancer onset and development.140 It has been reported that exosomal miR-96 plays a role in lung cancer cells proliferation and migration by targeting LIM-domain only protein 7 (LMO7).141 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases.142 Exosomes released from gemcitabine-resistant A549 cells can transfer miR-222-3P to target cells and promote cell proliferation, migration, and invasion by targeting suppressor of cytokine signaling 3 (SOCS3).143 On the contrary, EC-derived exosomes are characterized by high level of miR-126 and play a role in inhibiting cell proliferation and reducing loss of malignancy of NSCLC cells.144 Mechanistically, exosomal miR-126 suppress NSCLC development by targeting insulin receptor substrate 1 (IRS1) and vascular endothelial growth factor (VEGF). A549 NSCLC cells-derived exosomal miR-208a inhibits NSCLC cell proliferation by targeting p21 thus activating the AKT/mTOR pathway.145 In addition, exosomal miR-512 suppresses cell proliferation by targeting TEA domain family member 4 (TEAD4).146
Oxygen and nutrients are necessary for survival of cancer cells, thus angiogenesis is essential for tumor growth and metastasis.147 Zhuang et al. reported that exosomal miR-9 enhances angiogenesis by activating JAK/STAT pathway.148 Moreover, human bronchial epithelial (HBE) cells-derived exosomal miR-21 can promote angiogenesis by activating STAT3 and increasing expression of VEGF.149 Furthermore, circulating exosomal miR-23a levels are positively correlated with lung cancer proangiogenic activities. Hypoxic lung cancer cells promote angiogenesis by repressing the tight junction protein ZO-1 through exosomal miR-23a.150 Exosomal miR-126 secreted by NSCLC cells can trigger angiogenesis and accelerate lung cancer progression.144 In addition, miR-210 packaged in exosomes from tumor cells stimulates angiogenesis.151
Metastasis is a primary feature of cancer. Bone metastasis is common in patients with lung cancer.131 MiR-21 in A549 cells-derived exosomes promotes tumorigenesis and osteoclastogenesis by targeting the PDCD4 pathway.152 In addition, immune responses are involved in cancer progression. Fabbri et al. demonstrated that NSCLC cells-derived exosomes are characterized by high expression levels of miR-21, miR-27b, and miR-29a. Notably, exosomal miR-21 and miR-29a can promote tumor growth and metastasis by targeting Toll-like receptor (TLR) and inducing pro-metastatic inflammatory responses.80 Moreover, overexpression of exosomal miR-192 significantly appeased osseous metastasis by suppressing secretion of proangiogenic factors.82
Exosomal lncRNAs have been reported to be highly correlated with lung cancer. Exosomal lncRNAs such as H19, MALAT1, HOTAIR, UCA1, lnc-MMP2-2, GAPLINC, TBILA, AGAP2-AS1, and SOX2-OT play several roles in pathological processes including cell proliferation, migration, invasion, and EMT linked to lung cancer.153 Li et al. reported that GAS5 in exosomes is not only important for cancer development, but a promising biomarker for diagnosis patients with early NSCLC.154 Teng et al. found that exosomal SOX2-OT can be used as an effective noninvasive plasma-based tumor marker for lung squamous cell carcinoma (LSCC).155
Exosomal circRNAs are involved in the development of lung cancer. CircSATB2 are enriched in NSCLC cells and can be transported to other cells by exosomes to facilitate cell proliferation, migration, and invasion of NSCLC cells, and trigger abnormal proliferation of bronchial epithelial cells.156 Exosomal has-circ-0014235 promotes NSCLC progression by targeting miR-520a-5p/cyclin-dependent kinase 4 (CDK4) axis.157 Besides, circRNA-002178 shuttled by exosomes can be transferred to CD8+ T cells thus promoting generation of programmed death-ligand 1 (PDL1)/programmed cell death protein 1 (PD1) in LUAD.158 In addition, the expression levels of exosomal circ-0007761, circ-0047921, circ-0056285, circ-0008928, circRNA-102481, circ-MEMO1, circ-ARHGAP10, circ-PIP5K1A, and circ-FARSA are changed in NSCLC,159–165 while that of exosomal circ-0000690, circ-0001346, circ-0001439, and circ-0001492 are significantly increased in LUAD, which have potential as promising diagnostic biomarkers.166
Exosomal ncRNAs in BC
BC is the most frequent female malignant tumor globally and 70–80% patients present with early, non-metastatic BC which is considered curable.167 Approximately 2.1 million cases of BC were diagnosed in women and 626,679 breast cancer-related deaths were reported in 2018.168 Over the past decades, increasing evidence has shown that exosomal ncRNAs are closely associated with BC development.169
Exosomal miRNAs play roles in cellular proliferation, migration, and invasion of BC. A previous study reported that exosomal miR-10b is highly expressed in BC cells and promotes invasion by inhibiting expression of HOXD10 and KLF4.170 Exosomal miR-1246 is highly expressed in breast cancer cells and enhances cell proliferation and invasion by targeting CCNG2.171 On the contrary, exosomal miR-134 downregulates and inhibits cell proliferation, migration and invasion by targeting STAT5B in BC.172 Overexpression of miR-130a-3p in BC stem cells inhibits cellular proliferation, migration, and invasion by targeting RAB5B.173 In addition, miR-127, miR-197, miR-222, and miR-223 shuttled by exosomes inhibits cell proliferation by suppressing CXCL12.
Exosomal miRNAs are also involved in metastasis of BC. Cancer-associated fibroblasts (CAFs) are essential components of tumor microenvironment and play important roles in tumor development and metastasis. Exosomal miR-9 is highly expressed in breast CAFs and promotes switch of fibroblasts to CAF phenotype.174 Wu et al. revealed that exosomal miR-16 and miR-148a from focal adhesion kinase knockout CAFs are upregulated and ameliorate tumor cell metastasis.175 CAFs-derived exosomal miR-21, miR-143, and miR-378e promote stemness and EMT phenotype of BC cells.176 Moreover, miR-1910-3p shuttled by exosomes promotes cell proliferation and metastasis by targeting MTMR3 and NF-κB signaling pathway.104 In addition, exosomal miR-503-3p, miR-4269, miR-30e-3p, miR-105, miR-122, miR-200, miR-939, and miR-940 play crucial roles in promoting BC progression and metastasis.177–182
Apart from these, exosomal miRNAs participate in other cellular process of BC. For instance, BC cells-derived exosomal miR-20a-5p enhances differentiation of osteoclasts by targeting SRCIN1.183 MiR-23b is upregulated in BC cells-derived exosomes and reduces expression of MARCKS, a key regulator of cell cycling and motility.184 Notably, miR-210 from BC cells-derived exosomes can promote angiogenesis.185 On the contrary, miR-16 and miR-100 in exosomes from mesenchymal stem cells (MSCs) reduce secretion of VEGF in tumor cells and inhibit angiogenesis.186,187
Exosomal lncRNAs play key roles in BC progression and are potential diagnostic biomarkers for BC. LncRNA MALAT1 shuttled by BC exosomes can promote cell proliferation.188 Exosomal lncRNA GS1-600G8.5 are highly expressed in brain metastatic breast cancer cells and are implicated in destroying the BBB system and promoting transfer of cancer cells across the BBB.189 Moreover, expression of exosomal lncRNAs, including H19, SUMO1P3, XIST, and HOTAIR is upregulated in patients with BC indicating that they can serve as promising diagnostic biomarkers for BC.190–194
Exosomal circRNAs exhibit various roles in BC. Yang et al. found that serum exosomal circPSMA1 from BC is highly upregulated and promotes BC tumorigenesis, migration, and migration through miR-637/Akt1/β-catenin (cyclin D1) axis.195 Besides, circHIF1A (circ-0032138) is highly expressed in hypoxic CAFs-derived exosomes and modulates stem cell properties of BC through miR-580-5p/CD44 axis.196 Besides, exosomal circHIF1A significantly promotes BC growth and metastasis by activating AKT/STAT3 signaling pathway and suppressing expression of P21.197 On the contrary, Wang et al. reported that expression of several exosomal circRNAs was downregulated in BC cells.198
Exosomal ncRNAs in HCC
HCC is a major cause of cancer-related deaths worldwide.199 Notably, HCC is commonly diagnosed in cirrhosis patients.200 Recent studies have explored the biological functions of exosomal ncRNAs in initiation and development of HCC and report that exosomal ncRNAs can be used as non-invasive biomarkers for HCC.201
Exosomal miRNAs are closely associated with the pathology of HCC. Cui et al. reported that exosomal miR-224 promotes cell proliferation by targeting glycine N-methyltransferase.202 In addition, exosomal miR-93 can stimulate proliferation and invasion of HCC by suppressing TIMP2/TP53INP1/CDKN1A.203 Moreover, HCC cell-derived exosomal miR-665 promotes tumor cell proliferation by targeting MAPK/ERK signal pathway.204 Studies report that serum exosomal miR-1247-3p is implicated in lung metastasis in HCC patients. Exosomal miR-1247-3p released from high-metastatic HCC cells promotes tumor development by activating the β1-integrin-NF-κB signaling pathway and releasing pro-inflammatory cytokines, including IL-6 and IL-8.205 HCC cell-derived exosomal miR-210 can be transferred into ECs and enhances angiogenesis by targeting SMAD4 and STAT6.206 Similarly, miR-155 shuttled by exosomes from hypoxia-treated HCC cells is involved in tube formation of ECs and tumor angiogenesis.207 In addition, overexpression of exosomal miRNAs, including miR-224, miR-21, miR-92b, miR-93, miR-10b-5p, hsa-miRNA-1298, and miR-215-5p can be used as diagnostic markers for patients with HCC.202,203,208–212 On the contrary, levels of some exosomal miRNAs, including miR-9-3p, miR-125b, miR-638, miR-718, miR-101, miR-106b, miR-122, miR-195, and miR-744 are downregulated in HCC patients.213–218
LncRNAs shuttled by exosomes play key roles in regulating tumor cell proliferation, angiogenesis, invasion, and metastasis.219 Li et al. revealed that lncRNA FAL1 is highly expressed in serum exosomes from HCC patients and promotes HCC cell proliferation and metastasis through competitively binding to miR-1236.220 H19 is upregulated in exosomes from propofol-treated HCC cells and stimulates tumor cell proliferation, migration, and invasion through the miR-520a-3p/LIMK1 axis.221 Moreover, exosomal lncRNA H19 is significantly upregulated in CD90+ liver cancer cells and is implicated in promoting angiogenesis and regulating tumor microenvironment.222 In addition, lncRNA TUC339 is highly expressed in exosomes from HCC cells and it stimulates M2 macrophage polarization, thus enhancing tumor cell migration, invasion, and EMT.223 Similarly, HCC-derived exosomal DLX6-AS1 triggers M2 macrophage polarization by targeting the miR-15a-5p/CXCL17 axis.71 LincRNA VLDLR encapsulated in extracellular vesicles was highly expressed in HCC cells and promotes cellular stress responses.224 Sorafenib-treated HCC cell-derived exosomal lincRNA ROR is upregulated and suppresses death of recipient HCC cells by targeting the p53 signaling pathway.225 Ma et al. reported that expression of exosomal ASMTL-AS1 was highly correlated with the stage, metastasis, and prognosis in HCC.226 High expression level of exosomal lncRNA-ATB is significantly correlated with lower overall survival in HCC patients. Therefore, exosomal lncRNA-ATB is a promising prognostic biomarker for HCC.208 In addition, expression levels of LINC00161, LINC00635, lncRNA-RP11-583F2.2, lnc-FAM72D-3, lnc-EPC1-4, and lncRNA-HEIH in exosomes are high in HCC patients.212,227–230 On the contrary, SENP3-EIF4A1 and linc-FAM138B are downregulated in plasma exosomes in HCC patients. Exosomal SENP3-EIF4A1 can be transferred into HCC cells thus inhibiting tumor cell growth, and attenuate invasion and migration of HCC cells,231 while exosomal linc-FAM138B plays a role in repressing HCC growth by targeting miR-765.232
Exosomal circRNAs play a role in cellular processes of HCC, such as cell proliferation, angiogenesis, metastasis, and EMT. Huang et al. claimed that exosomal circRNA-100338 secreted by highly metastatic HCC cells is significantly upregulated and can be transferred to human umbilical vein endothelial cells (HUVECs). Exosomal circRNA-100338 can stimulate cell proliferation, angiogenesis, and vasculogenic mimicry formation of HUVECs and promote tumor metastasis.233 In addition, exosomal circRNA Cdr1as is highly expressed in HCC cells and can be transferred to surrounding cells. Overexpression of Cdr1as stimulates cell proliferation and migration by targeting the miR-1270/AFP axis.234 Circ-ZNF652 is highly expressed in exosomes from HCC cells and in serum of HCC patients. Exosomal circ-ZNF652 is implicated in stimulating cell proliferation, migration, invasion, and glycolysis by targeting miR-29a-3p/GUCD1 axis.235 Moreover, expression of circFBLIM1 is upregulated in HCC serum exosomes and promotes tumorigenesis and glycolysis by targeting miR-338.236 Exosomal circ-0004277 plays a role in inducing malignant phenotype of HCC by suppressing expression of ZO-1 and increasing tumor cell progression, migration, and EMT.237 In addition, the level of serum exosomal CircPTGR1 is upregulated in HCC patients and plays a role in facilitating tumor metastasis by targeting miR449a/MET pathway.238 Overexpression of exosomal has-circ-0039411 increases secretion of matrix metallopeptidase 2 (MMP2) by sponging miR-136-5p. High expression levels of exosomal has-circ-0039411 and MMP2 are correlated with tumor metastasis and low overall survival of HCC patients.239 HCC cell-derived exosomes exhibit a high expression level of circUHRF1. Exosomal circUHRF1 can trigger dysfunction of natural killer (NK) cells thus inducing immunosuppression in HCC patients.240 Expressions of exosomal circRNAs, such as circAKT3, has-circ-0004001, has-circ-0004123, has-circ-0075792, circ-0061395, and circTMEM45A are also highly upregulated in HCC patients.241–243 However, exosomal circ-0051443 is downregulated in plasma exosomes from HCC patients, which exerts a role in inducing cell apoptosis and inhibiting malignant behaviors.244 Wang et al. reported that level of exosomal has-circ-0074854 is also downregulated in HCC patients and plays a role in inhibiting tumor cell migration and invasion by repressing M2 macrophage polarization.245
Exosomal ncRNAs in CRC
CRC is a common cause of cancer-associated deaths worldwide. Approximately 1.2 million CRC cases are diagnosed and 600,000 people die with CRC every year.246 Previous studies report that exosomal ncRNAs play important roles in CRC.
Exosomal miRNAs exert vital effects on different progression of CRC.247 First, exosomal miR-183-5p promotes tumor development and induces cell proliferation, invasion, and tube formation of ECs.248 Second, exosomal miR-25-3p and miRNA-146a-5 are corelated with angiogenesis and tumorigenesis of CRC, respectively.249,250 Third, high expressed miR-17-5p in exosomes promotes CRC metastasis.251 Fourth, exosomal miR-210 plays a role in promoting EMT and anoikis resistance.252
Besides, several exosomal miRNAs modulate the metastasis of CRC. CRC-derived exosomal miR-934 is involved in inducing liver metastasis of CRC by mediating cellular communication between tumor-associated macrophages and CRC cells.253 In addition, miR-1255b-5p is highly expressed in hypoxic-treated exosomes from mouse CRC. Exosomal miR-1255b-5p inhibits EMT, CRC development, and liver metastasis by regulating expression of human telomerase reverse transcriptase (hTERT).254 Furthermore, high-metastatic CRC-derived exosomal miR-106b-3p induces tumor cell migration, invasion, EMT, and lung metastasis by targeting deleted in liver cancer-1 (DLC-1).255
Numerous studies have found that expression levels of serum exosomal miRNAs, including let-7a, miR-1229, miR-1246, miR-150, miR-21, miR-223, miR-23a, miR-301a, miR-17-5p, miR-92a-3p, miR-6803-5p, and miR-320d are significantly upregulated in primary CRC patients.256–260 In addition, some plasma exosomal miRNAs, such as miR-27a and miR-130a are upregulated in CRC and can serve as noninvasive biomarkers for CRC.261 On the contrary, low expression levels of exosomal miRNAs, including miR-874, miR-30a-5p, and miR-128-3p are highly correlated with tumor metastasis, differentiation, and advanced TNM stage.262–264
Moreover, exosomal lncRNAs play important roles in CRC. Exosomal RPPH1, MALAT1, NNT-AS1 promotes tumor progression, including cell proliferation, invasion, migration, and metastasis,265,266 while exosomal LINC02418 and H19 induces tumorigenesis and development.267,268 Exosomal HOTTIP is highly expressed in mitomycin-resistant CRC cells and promotes mitomycin resistance.269 Notably, low levels of exosomal HOTTIP are highly correlated with poor overall survival of CRC patients.270 Besides, studies report that exosomal lncRNAs, including GAS5, CRNDE-h, CRNDE-p, LINC02418, CCAT2, LNCV6-116109, LNCV6-98390, LNCV6-38772, LNCV-108266, LNCV6-84003, LNCV6-98602, FOXD2-AS1, NRIR, and XLOC-009459 are high expressed in CRC patients.267,271–276 In addition, serum exosomal lncRNA UCA1 is downregulated in CRC patients.277
Recent studies reported that exosomal circRNAs are involved in pathophysiology of CRC. Shang et al. determined that exosome-encapsulated circPACRGL from CRC patients stimulates tumor cell proliferation, migration, invasion, and metastasis, as well as neutrophil differentiation.278 Moreover, exosomal circFMN2 mediates cell proliferation and migration,279 while exosomal circIFT80 is implicated in promoting CRC development.280 Exosomal has-circ-0005963 and circ-133 regulate the process of CRC by targeting miR-122/M2 isoform of pyruvate kinase (PKM2) axis281 and miR-133a/GEF-H1/RhoA axis,282 respectively. Circulating exosomal hsa-circ-0004771 is significantly upregulated in CRC patients and is a novel potential diagnostic biomarker of CRC.283
Exosomal ncRNAs in GC
GC is a common cause of cancer-related death worldwide. In 2018, approximately 784,000 GC-related deaths were reported.284 Emerging evidence suggested that exosomal ncRNAs are involved in development of GC.
Cellular proliferation, migration, and invasion are closely associated with GC. Exosomal miR-1290 is upregulated in GC patients and stimulates proliferation, migration, and invasion of GC cells by downregulating expression of naked cuticle homolog 1 (NKD1).285 GC tissue-derived mesenchymal stem cells (GC-MSCs)-derived exosomal miR-221 play a role in tumor cell growth and migration.286 Exosomal miR-301a-3p promotes progression and metastasis of GC cells by regulating PHD3/HIF-1α.287 Exosomal miR-15b-3p promotes tumor cell proliferation, migration, and invasion by targeting DYNLT1/Caspase-3/Caspase-9 pathway.288 In addition, miR-34 packaged in exosomes from gastric CAFs shows low expression levels. Notably, overexpression of exosomal miRNA-34 can inhibit tumor cell proliferation, invasion, and motility.289
Besides, exosomal miRNAs take part in the progression of metastasis and angiogenesis of GC. MiR-let-7 is highly expressed in metastatic GC cell-derived exosomes. Exosomal miR-let-7 promotes tumorigenesis and metastasis by targeting RAS and HMGA2.290 Exosomal miR-21-5p promotes GC peritoneal metastasis by stimulating mesothelial-to-mesenchymal transition of peritoneal mesothelial cells (PMCs). Mechanistically, exosomal miR-21-5p induces metastasis by targeting SMAD7 and activating TGF-β/Smad signaling pathway.291 Wang et al. reported that exosomes released from GC cells exhibit a high level of miR-27a. Exosomal miR-27a induces CAFs and promotes tumor cell motility and metastasis by targeting CSRP2.292 Gastric CAFs-derived exosomal miR-139 suppresses gastric cancer metastasis and development by reducing matrix metalloproteinase 11 (MMP11).293 GC cell-derived exosomal miR-130a can be delivered into ECs to induce angiogenesis and tumor growth by regulating c-MYB.294 Studies report that exosomal miRNAs, such as miR-19b-3p, miR-106a-5p, miR-1246, miR-107, miR-196a-1, miR-106a, and miR-155-5p are all upregulated in GC patients.295–300
Exosomal lncRNAs regulate the progression of GC through diverse mechanisms. Overexpression of exosomal HOTAIR induces proliferation, migration, and invasion of GC cells by increasing KRAS.301 Exosomal LINC01559 promotes tumor progression by regulating miR-1343-3p/ phosphoglycerate kinase 1 (PGK1) and activating PI3K/AKT pathway.302 GC cells-derived exosomal lncRNA HEIH can be delivered into normal gastric cells and promote malignant transformation of GC by promoting expression of EZH2.303 Wang et al. reported that exosomal HOTTIP is implicated in cisplatin resistance in GC patients by targeting miR-218/HMGA1 axis.304 Numerous lines of evidence suggested that exosomal lncRNAs, such as UEGC1, HOTTIP, GC1, MIAT, H19, lnc-SLC2A12-10:1, CEBPA-AS1, ZFAS1 are highly expressed.118,305–311 On the contrary, exosomal lncRNAs, such as GNAQ-6:1 and PCSK2-2:1 show low expression levels in GC patients.312,313
Exosome-mediated circRNAs play important roles in GC. Xie et al. demonstrated that circSHKBP1 is highly expressed in exosomes from GC patients and the level of exosomal circSHKBP1 was reduced after gastrectomy. Exosomal circSHKBP1 promotes proliferation, migration, invasion, and angiogenesis of GC cells by targeting miR-582-3p/HUR/VEGF axis and through inhibition of HSP90 degradation.314 CiRS-133 encapsulated in exosomes secreted by GC cells can be transferred to preadipocytes. Exosomal ciRS-133 stimulates preadipocytes differentiation into brown-like cells by regulating miR-133/ PRDM16 pathway.315 Exosomal circNEK9 promotes proliferation, migration, invasion, and motility of recipient GC cells by modulating miR-409-3p/MAP7 axis.316 Moreover, circ-PVT1 is highly expressed in cisplatin-resistant GC cells-derived exosomes. Exosomal circ-PVT1 promotes cisplatin resistance in GC cells by inducing cell autophagy and invasion and by inhibiting apoptosis. Overexpression of exosomal circ-PVT1 induces low expression of miR-30a-5p and high expression of YAP1 in GC cells.106 Circ29 packaged in exosomes can be transferred from GC cells to ECs and promote proliferation, migration, and tube formation of ECs by targeting the miR-29a/VEGF pathway.317 In addition, circNHSL1 is highly expressed in exosomes released from GC cells. Exosomal circNHSL1 promotes migration, invasion, and glutaminolysis of GC cells by targeting miR-149-5p/YWHAZ axis.318 Overexpression of exosomal circ-0032821from oxaliplatin-resistant GC cells promotes proliferation, migration, and invasion of GC cells by modulating miR-515-5p/SOX9 pathway.319 Notably, exosomal has-circ-0065149 is downregulated in GC patients.320
Exosomal miRNAs in PCa
PCa is a heterogeneous disease.321 Approximately 160,000 PCa cases are diagnosed each year in the United States.322 Accumulating studies indicate that exosomes are implicated in PCa tumor development. Dysregulated exosomal ncRNAs are involved in tumor initiation and progression of PCa.323
Exosomal miRNAs are implicated in PCa. MiR-217 encapsulated in exosomes exerts roles in promoting tumor cell proliferation, invasion, and EMT.324 MiR-1246, a tumor inhibitor, was downregulated in PCa cell-derived exosomes. Overexpression of exosomal miR-1246 inhibits tumor cell proliferation, migration, and invasion and promoted cell apoptosis by suppressing EMT. Exosomal miR-205 released from human bone marrow mesenchymal stem cells (hBMSCs) promotes tumor cell apoptosis and inhibits proliferation, migration, and invasion of PCa cells by targeting RHPN2.325 Exosomal miR-26a exerts a vital role in mediating tumor growth and tumor cell metastasis.326 Tumor-associated macrophages enhances PCa progression through exosomal miR-95.327 MiR-183 encapsulated in exosomes is upregulated in PCa patients and promotes proliferation, migration, and invasion of tumor cell by downregulating expression of TPM1.328 Ye et al. reported that exosomal miR-141-3p promotes osteoblastic metastasis of PCa by modulating activity of osteoblasts.329 Huang et al. suggested that overexpression of plasma exosomal miR-1290 and -375 was significantly correlated with poor overall survival of PCa.330 Plasma exosomal miRNAs such as miR-1285, miR-622, miR-221, miR-145, and serum exosomal miRNAs such as miR-1274a, miR-1207-5p, miR-885-5p, miR-874, miR-766, miR-640, miR-636, miR-486-5p, miR-375, miR-346, and miR-141 are significantly upregulated in PCa patients and are promising biomarkers for diagnosis of PCa.331,332
Exosomal lncRNAs are implicated in diverse molecular processes involved in PCa progression. LncRNA MYU shuttled by exosomes is highly expressed in PCa patients and can be delivered into adjacent cells. Notably, exosomal lncRNA MYU induces cell proliferation and migration by competitively binding miR-184 and promoting expression of c-Myc.333 PCa-derived exosomal SChLAP1 is highly expressed and significantly correlated with the level of prostate specific antigen (PSA) and tumor cell invasion.334 Ozgur et al. demonstrated that exosomal H19 is involved in PCa by regulating androgen receptor pathway.335
CircRNAs highly expressed in exosomes can be transferred from tissues into various body fluids and are potential diagnostic biomarkers for PCa.336 Has-circ-0044516 shuttled by exosomes induces tumor cell metastasis and inhibits tumor cell apoptosis by sponging miR-29a-3p.337 Exosomal circ-XIAP is upregulated in docetaxel-resistance PCa cells and promotes docetaxel resistance by targeting miR-1182/TPD52 axis.338
In general, exosomal ncRNAs are involved in pathological cellular processes including cell proliferation, migration, invasion, metastasis, angiogenesis, and EMT associated with diverse cancers. Accumulating evidence over the last decade has further revealed that exosomal ncRNAs can participate in multiple processes contributing to cancer development, diagnosis biomarkers, and therapeutic effects, showing the dual characteristics of promoting and suppressing cancer. In this part, we mainly discuss the roles of exosomal miRNAs, lncRNAs, and circRNAs in lung cancer, BC, HCC, CRC, GC, and PCa. Notably, studies have explored roles of exosomal ncRNAs in other cancers, including esophageal cancer, pancreatic cancer, ovarian cancer, and leukemia.339
The roles of exosomal ncRNAs in metabolic diseases
Previous studies report that exosomal ncRNAs play important roles in remote tissues. NcRNAs-encapsulated exosomes are implicated in various processes that involved in development of metabolic diseases, such as T2DM, obesity, and osteoporosis (Fig. 6).
Fig. 6.
The role of exosomal ncRNA in the pathological process of metabolic diseases. Exosomes secreted by different tissues can be released into the circulation and transported to other organs, where they are internalized by recipient cells, mediating metabolic regulation. This figure was created with the aid of Servier Medical Art (https://smart.servier.com/)
Exosomal ncRNAs in T2DM
T2DM is a prevalent chronic disease that causes cardiovascular, renal, retinal, and neurological complications, and is a major cause of death and disability worldwide.340,341 T2DM is the most common clinical type of diabetes accounting for over 90% of diabetes cases, which is characterized by relative insulin deficiency as a result of progressive inadequate insulin secretion and varying degrees of IR in peripheral tissues, such as adipose tissues, skeletal muscle, and liver tissues.9,342 Aging, obesity, and other cardiovascular risk factors promote the development of T2DM.340 However, the roles of these factors in promoting the pathological process in T2DM has not been fully elucidated. Recent studies on exosomes and their ncRNA cargoes have been conducted to explore the underlying mechanisms.
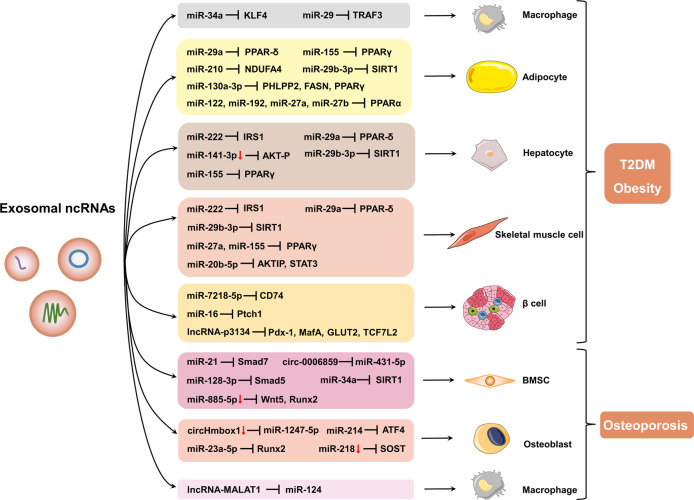
MiRNAs are the most explored ncRNAs, and several exosomal miRNAs are closely linked to T2DM. Adipose tissue is an endocrine organ that functionally regulates systematic energy homeostasis by releasing various endocrine factors which modulate glucose and lipid metabolism.9,343 In addition, adipose tissue is a major source of circulating exosomal miRNAs, which modulate glucose tolerance and the expression of fibroblast growth factor 21 (FGF21) in the liver.344 MiRNAs profiles in adipocyte-derived exosomes vary under different physiopathological conditions of adipose tissue.345 Adipocyte-derived exosomal miRNAs, such as miR-34a,346 miR-222,347 miR-27a,348 and miR-802-5p85 can promote IR via regulating Krüppel-like factor 4 (KLF4), insulin receptor substrate 1 (IRS1), peroxisome proliferator-activated receptor γ (PPARγ), and HSP60. Hepatocyte uptake of exosomes from adipocytes in obese mice containing less miR-141-3p than that in healthy mice, and the reduced absorption of miR-141-3p resulted in reduced glucose uptake in hepatocytes.349 A recent study reported that exosomal miR-130b-3p from adipocytes isolated from epididymal fat of HFD mice or incubated with high glucose/high lipid level aggravates myocardial ischemia/reperfusion injury in nondiabetic mice, by inhibiting various antiapoptotic and cardioprotective signaling, mainly AMP-activated protein kinase (AMPK).350 In addition to adipocytes, macrophages can release exosomes containing miRNAs that regulate glucose metabolism. Adipose tissue macrophage (ATM)-derived exosomal miR-155 in obese mice promotes glucose intolerance and IR by targeting PPARγ.351 Similarly, ATM-derived exosomal miR-29a can be transported into adipocytes, myocytes, and hepatocytes, thus inducing IR in vitro and in vivo through PPAR-δ.352 MiR-690 in exosomes derived from M2-polarized macrophages can alleviate glucose tolerance and IR in obese mice by targeting Nadk, a gene encoding NAD+ kinase.353 Exosomal miR-210 released from high glucose (HG)-induced ATM promotes the development of diabetic obesity in mice by inhibiting NADH dehydrogenase ubiquinone 1 alpha subcomplex 4 (NDUFA4) in adipocytes, leading to reduced glucose uptake and mitochondrial complex IV (CIV) activity.354 Exosomal miR-21-5p derived from HG-stimulated macrophages induces inflammation, ROS production, and podocyte injury in diabetic nephropathy (DN) mice by modulating A20.79
Liver-derived exosomal miR-130a-3p ameliorates glucose intolerance and IR by repressing pleckstrin homology domain leucine-rich repeat protein phosphatases 2 (PHLPP2) thus triggering AKT/Akt substrate of the 160 kDa (AS160)/glucose transporter 4 (GLUT4) axis in adipocytes.355 Expression level of hepatocyte-released exosomal miR-7218-5p in HFD mice is lower compared with that in normal diet mice. MiR-7218-5p mimic transfection decreases islet β cells proliferation by regulating CD74.356
Furthermore, skeletal muscle-derived exosomal miRNAs play important roles in T2DM, although only a few studies have explored these roles. Jalabert and colleagues found that exosomes extracted from quadriceps muscle in mice fed with high palmitate diet promoted proliferation of β cells and isolated islets. MiR-16 is upregulated in these exosomes and can modulate Ptch1 associated with pancreas development.357
It has been reported that islet β cells can secretes different miRNAs. When encapsulated by exosomes, the miRNAs can be transported to functionally act on different recipient cells.358 Specifically, it has been shown that β cell-derived exosomal miR-29s attenuate hepatic insulin sensitivity and regulate glucose homeostasis as a result of high levels of free fatty acids.359 Another study shows that exosomal miR-29 induces recruitment and activation of monocytes and macrophages as well as consequent inflammation in HFD mice. This promotes progression of diabetes by decreasing tumor receptor-associated factor 3 (TRAF3).42 The abundance of miR-26a is decreased in serum exosomes of overweight human and obese mice and is negatively correlated with clinical characteristics of T2DM. Several experiments have proven that exosomal miR-26a from β cells can ameliorate peripheral IR induced by obesity. Moreover, upregulation of miR-26a in β cells reduces glucose-stimulated insulin secretion (GSIS) by inhibiting actin cytoskeleton remodeling and thus prevents obesity-induced islet hyperplasia.360 Exosomal miR-15a released from pancreatic β-cells can travel through the circulation and be absorbed by Müller cells. This triggers oxidative stress and causes retinal injury and apoptotic cell death under T2DM conditions.361 Additionally, β cell-derived exosomal miR-127 enhances the migration and ability of islet ECs to form tubes.362
Bone marrow-derived MSC (BMSC)-derived exosomal miR-29b-3p in aged mice can be absorbed by adipocytes, myocytes, and hepatocytes, which subsequently decreases insulin sensitivity both in vivo and in vitro via targeting SIRT1. Intriguingly, downregulation of nanocomplex-mediated miR-29b-3p in exosomes from BMSCs attenuates IR in aged mice.40
Circulating exosomal miR-20b-5p attenuates insulin-stimulated glycogen accumulation through regulating AKT-interacting protein (AKTIP) and STAT3.363 MiRNA profiles in plasma exosomes were altered in obese mice, including increased miR-122, miR-192, miR-27a-3p, and miR-27b-3p. By targeting PPARα, the treatment of lean mice with exosomes containing obesity-related miRNAs induces glucose intolerance and IR.78 Additionally, altered miRNAs encapsulated in circulating exosomes correlates with the adiponectin pathway in T2DM patients.364
In addition to miRNAs, exosomal lncRNAs are also emerging as significant factors in T2DM. Expression of serum exosomal lncRNA-p3134 was elevated in T2DM patients.365 This lncRNA has been revealed to be interrelated with levels of fasting blood glucose and homeostasis model assessment β-cell function (HOMA-β). Overexpression of lncRNA-p3134 can promote GSIS and reduce apoptosis in β cells, which provides a novel mechanism of glucose homeostasis regulation by lncRNAs.365 The circulating level of exosomal lncRNA-MALAT1 significantly decreases in patients with T2DM.366
Exosomal lncRNAs also exert protective effect. Li and coworkers have shown that exosomal lncRNA H19 from MSCs can induce fibroblast proliferation and migration, as well as inhibit apoptosis and inflammation via abrogating miR-152-3p-mediated PTEN repression and hence facilitates diabetic wound healing.367 Exosomes-mimetic nanovesicles carrying a high level of H19 can serve as a nano-drug delivery system to neutralize the inhibitory effect of hyperglycemia on regeneration and speed up chronic wound healing.368 MSC-released exosomal lncRNA SNHG7 inhibits HG-stimulated endothelial-mesenchymal transition and tube formation of human retinal microvascular ECs by targeting miR-34a-5p/XBP1 axis. This provides a promising therapeutic method for diabetic retinopathy.369
Emerging evidence has unveiled the important role of exosomal circRNAs in T2DM complications. Our recent study has revealed that HG-stimulated ECs release exosomes containing circRNA-0077930 to vascular smooth muscle cells (VSMCs) causing VSMC senescence,370 which may lead to diabetic vascular complications. Further, circRNAs in serum exosomes were different among patients with diabetic foot ulcer (DFU), non-DFU diabetes, and healthy cases.371 Among these modified circRNAs, the abundance of exosomal has-circ-0000907 and has-circ-0057362 were significantly increased in early DFU. Further experiments showed their role as promising biomarkers in the early diagnosis of DFU.371 It has been reported that circRNA cPWWP2A can be transported from retinal pericytes to ECs by exosomes in a paracrine manner. Further, high expression of cPWWP2A attenuates diabetes-induced retinal vascular dysfunction in vivo.372 Besides, a recent study has verified that serum exosomal circRNA DLGAP4 was elevated in patients with DN and rat models as compared with T2DM individuals without DN and normal rats. Furthermore, exosomal circRNA DLGAP4 have been shown to induce mesangial cell proliferation and fibrosis, as well as exacerbating DN in vivo by sponging miR-143 to motivate Erb-b2 receptor tyrosine kinase 3 (ERBB3)/NF-κB/matrix metalloproteinase-2 (MMP-2) axis.373
Collectively, exosomal miRNAs from a variety of sources, including adipose tissue, liver, skeletal muscle, islet β cells, BMSC, are all involved in the pathological process of T2DM. Exosomal lncRNAs not only act as modulators (lncRNA-p3134 and MALAT1), but also have therapeutic effects on T2DM and its complications (H19 and SNHG7). Exosomal circRNAs mainly play vital roles in T2DM complications, such as DFU, DN, and retinal vascular disorders.
Exosomal ncRNAs in obesity
Obesity is defined as abnormal or excessive accumulation of body fat that presents a health risk to an individual. According to World Health Organization, obesity is diagnosed with a body mass index (BMI) greater than 30 kg/m.2,374 Obesity has grown into an epidemic with approximately 604 million adults affected worldwide in 2015.375 This disorder has caused enormous social burden due to multiple comorbidities such as diabetes, hyperlipidemia, cardiovascular diseases, and diverse cancers.376 Understanding the pathogenesis of obesity contributes to effective management of this harmful disorder.
Emerging evidence reveals that exosomes, as a mediator that intercellularly transports ncRNAs, play a pivotal role in the development of obesity and associated metabolic disorders. Santamaria-Martos and coworkers detected several plasma exosomal miRNAs candidates associated with BMI (e.g., let-7b, miR-146a), dyslipidemia (miR-29c) and fasting insulin (e.g., miR-222/223, miR-26b) in obese and non-obese women.377 Moreover, it has been reported that exosomal lncRNA-H19 expression is correlated with waist circumference.366 Elsewhere, exosomal miR181b‑5p and miR219‑5p of immune cell origin that are induced by elafin (an anti-inflammatory protein) promote leptin expression in adipocytes to reduce food consumption, obesity, and hyperglycemia in HFD male mice.378 It has been reported that aerobic exercise regulates serum exosomal miRNAs in obese mice. It decreases the levels of miR-122, miR-192, and miR-22, which are associated with improved adipogenesis, insulin sensitivity, and hepatic steatosis.379 Similarly, exosomal miRNAs profiles are modified in obese patients after bariatric surgery.380,381
Obesity is recognized as a state with chronic low-grade inflammation.59 Adipose tissue is an active endocrine organ that releases a variety of cytokines, such as adiponectin, interleukins, and TNF-α. It also secretes other pro-inflammatory biomolecules including miRNAs, which leads to metabolic dysfunction, particularly IR.126 MiRNAs contained in exosomes have been identified as important mediators of the inflammation caused by obesity. Some exosomal miRNAs are identified to be differentially expressed in visceral adipose of obese subjects compared to lean subjects. Besides, these miRNAs might target genes associated with obesity-induced inflammation, such as transforming growth factor-β (TGF-β) and Wingless and int-1 (Wnt)/β-catenin signaling.382,383 Moreover, circulating exosomal miRNAs from obese mice have also been found to trigger adipose tissue inflammation in lean mice.78 Elsewhere, exosomal miR-34a from obese mice have been found to promote inflammation by regulating the M1-to-M2 macrophage ratio.346 Additionally, exosomal miR-690 from M2-polarized macrophages regulates inflammation in obese mice.353
There is direct or indirect association of obesity with IR disorder.384 It has been reported that exosomal miRNAs profiles are modified by dysregulation of glucose metabolism in obese patients.385 On the other hand, obesity can contribute to IR through transporting exosomal miRNAs to target cells and tissues, which participates in a variety of pathophysiological processes including inflammation response and insulin signaling pathway.76,384 As previously stated, exosomal miRNAs from adipose tissue, particularly miR-155, miR-34a, miR-222, miR-27a, miR-29a, miR-210, and miR-141-3p, are crucial regulators in the physiopathology of obesity-induced IR.346–349,351,352,354 In addition, miR-192, miR-122, miR-27a-3p, and miR-27b-3p are upregulated in plasma exosomes in obese mice. Injection of exosomes transfected with these increased miRNAs contributes to central obesity in lean mice by targeting PPARα.78 On the contrary, miR-26a from β cells and miR-690 from M2-polarized macrophages which are contained in exosomes can ameliorate IR induced by obesity.353,360
In summary, exosomal ncRNAs participate in regulation of obesity and are related to BMI, dyslipidemia, and waist circumference. In addition, they are considered to be crucial mediators of inflammation caused by obesity and IR disorder.
Exosomal ncRNAs in osteoporosis
Osteoporosis is characterized by loss of bone mass, degradation of bone microstructure. The disease contributes to increased bone fragility and risk of fracture.386,387 As the global population ages, the incidence of osteoporotic fractures is increasing with consequent enormous economic costs, reduced quality of life and lifespan.388,389 The maintenance of bone mass relies on the strictly coordinated balance between bone formation and bone resorption, and the main cells involved are osteoblasts and osteoclasts. Osteoporosis is accompanied by an increase in bone resorption and a decrease in bone formation. Recent studies have demonstrated that some exosomal ncRNAs play a pivotal part in the modulation of osteogenesis and bone resorption.390,391
Exosomal miRNAs derived from MSCs exert a potential regulatory effect on osteogenesis.392 During osteogenic differentiation in human BMSCs, the miRNAs profile contained in exosomes is changed during osteogenic differentiation in human BMSCs. Among the effected changes, miR-885-5p is expression of miR-885-5p is lowered and proven to serve as a negative regulator of BMSCs osteogenic differentiation by suppressing Wnt5 and runt-related transcription factor 2 (Runx2).393 According to Jiang et al., miR-21-bearing exosomes in osteoporosis patients repress osteogenesis by targeting small mothers against decapentaplegic homolog 7 (Smad7).394 Further, a study by Xu et al. reported that exosomal miR-128-3p derived from MSCs in aged rats inhibits osteogenesis and fracture healing through dampening Smad5.390
Emerging evidence indicates that osteoclast-derived exosomes can transfer miRNAs to osteoblast that regulate bone formation. Elsewhere, Sun et al. revealed that osteoclast-derived exosomes shuttling miR-214 represses the osteoblasts activity.395 Further, Li et al. demonstrated that exosomal miR-214-3p could reduce osteoblast activity in vitro and impair bone formation in vivo. On the contrary, inhibition of miR-214-3p in osteoclast was reported to facilitate bone formation in aging ovariectomized mice.396 Moreover, Yang and colleagues showed that osteoclast-released exosomes enriched with miR-23a-5p inhibits osteogenic differentiation via repressing Runx2.397
Exosomes can mediate muscle-bone crosstalk through transporting their miRNAs cargoes. Fulzele et al. proved that circulating muscle-derived exosomal miR-34a increases with age in mice. High expression of miR-34a in the exosomes from myoblasts impaired BMSC viability and promoted cellular senescence, as well as decreased SIRT1 expression in BMSCs.398 Other studies have also reported that Sirt1 promotes differentiation of osteoblast.399,400 These findings suggests that there is a potential pattern of inter-organ crosstalk which causes physiopathology of bone with age. Myostatin, a muscular-secreted myokine, has been revealed to exert modulating effects on bone mass. Notably, it was found that downregulation of exosomal miR-218 derived from osteocytes could mediate inhibition of osteoblastic differentiation that is induced by myostatin through repression of sclerostin (SOST).401
Exosomal lncRNAs also exert important roles in osteoporosis. Teng et al. detected the levels of circulating exosomal lncRNAs in osteoporotic patients compared with normal subjects and identified 393 differentially expressed lncRNAs.402 Further bioinformatics analysis suggested that these lncRNAs may be associated with several osteoporosis pathways.402 Elsewhere, Cui et al. reported that lncRNA MALAT1 encapsulated by exosomes from endothelial progenitor cells (EPCs) could promote osteoclastogenesis of bone marrow-derived macrophages.403 This effect is achieved by MALAT1 serving as a miR-124 sponge to upregulate integrin subunit β 1 (ITGB1), which presents a pivotal role in osteoclastogenesis.403
Recently, exosomal circRNAs are recognized as a novel player in the pathophysiology of osteoporosis. CircRNAs in exosomes are differentially expressed in patients with osteoporosis. Among these circRNAs, exosomal hsa-circ-0006859 were upregulated and could differentiate individuals with osteopenia or osteoporosis from healthy individuals with high sensitivity and specificity. Mechanistically, hsa-circ-0006859 represses osteoblastic differentiation and induced adipogenic differentiation of human BMSCs through sponging miR-431-5p to promote ROCK1 expression.404 Exosomes containing a low concentration of circHmbox1 derived from TNF-α-treated osteoclasts decrease osteoblasts differentiation mainly by targeting miR-1247-5p. Overexpression of circHmbox1 remarkably mitigates the osteoporotic phenotypes in ovariectomized mice, which might function as a promising treatment strategy for postmenopausal osteoporosis.405
In a brief summary, exosomal miRNAs exert regulatory effects on osteogenesis, bone formation, and muscle-bone crosstalk. Exosomal lncRNAs are associated with a variety of osteoporosis pathways, such as MALAT1, which promotes osteoclastogenesis. Exosomal circRNAs are emerging as new regulators in the pathophysiology of osteoporosis. For instance, exosomal hsa-circ-0006859 is a highly sensitive and specific marker for patients with osteoporosis, while exosomal circHmbox1 exhibits a potential therapeutic effect for postmenopausal osteoporosis.
The roles of exosomal ncRNAs in cardiovascular diseases
Despite the continuous progress in the treatment of cardiological ailments, cardiovascular diseases, such as hypertension, atherosclerosis (AS), acute myocardial infarction (AMI), heart failure (HF), and atrial fibrillation (AF), are still the leading cause of the morbidity and mortality worldwide.406,407 Recently, accumulating exosomal ncRNAs has been identified to be involved in the pathogenesis of cardiovascular diseases, which provides a new insight into the mechanisms and therapeutic targets for the diagnosis and treatment of these diseases.408–413 Moreover, exosomal ncRNAs serve as emerging regulators in dyslipidemia, thereby leading to an increase in risk of atherosclerotic cardiovascular diseases.414 For example, miR-26a is downregulated in circulating exosomes of overweight humans and obese mice, whereas, upregulation of miR-26a in mice reduces the abundance of plasma cholesterol, low-density lipoprotein (LDL) and HDL, hepatic triglyceride, as well as lipid droplets in adipose tissue.360 This study summarizes the current evidence about the roles of exosomal ncRNAs in cardiovascular diseases.
Exosomal ncRNAs in hypertension
Hypertension is an important risk factor for total mortality and cardiovascular disease, such as stroke, myocardial infarction, coronary heart disease, and HF.415–417 Emerging evidence indicates that diverse exosomal ncRNAs were implicated in the development of hypertension through modulating multiple cellular and molecular events, including renin-angiotensin-aldosterone system (RAAS), endothelial dysfunction, angiogenesis, VSMCs proliferation, vascular remodeling, inflammation, and oxidative stress.418–420
Current evidence reveals that exosomal miRNAs from different sources can modulate the initiation and progression of hypertension. Next-generation sequencing was used to detect the exosomal miRNA expression profile in spontaneously hypertensive rats (SHRs) and normotensive Wistar-Kyoto rats (WKYs). Liu et al. found that 23 exosomal miRNAs were significantly upregulated and 4 exosomal miRNAs were downregulated in SHRs compared to WKYs. The levels of exosomal miR-17-5p and miR-425-5p were markedly elevated in SHRs plasma and tightly associated with inflammation and blood pressure homeostasis, respectively.421 Besides, exosomal miR-155-5p derived from aortic adventitial fibroblast was downregulated in SHRs compared to WKYs. Low expression of exosomal miR-155-5p enhanced the expression of vascular angiotensin-converting enzyme and angiotensin II (Ang II) and promoted VSMCs proliferation, vascular remodeling, and hypertension.422
Macrophages-derived exosomal miRNAs play crucial roles in hypertension. It has been reported that THP-1 cells-derived exosomal miR-27a impaired vasodilation and increased rat blood pressure through inhibiting the expression of Mas receptor in ECs and endothelial nitric oxide synthase (eNOS) phosphorylation in mesenteric arteries.423 Additionally, miR-17 was decreased in Ang II-treated THP-1-derived exosomes and promoted ECs inflammation through increasing the expression of intercellular adhesion molecule-1 (ICAM-1) and plasminogen activator inhibitor-1 (PAI-1).424 The transfer of exosomal miR-106b-5p from macrophages to renal juxtaglomerular cell stimulated inflammation-induced hypertension by inhibiting transcription factors E2f1 and Pde3b.425
Exosomal ncRNAs in AS
AS is a chronic immune-inflammatory and age-related disorder which is characterized by lipid-rich plaques accumulated in the arterial wall.426 It is one of the main causes of cardiovascular diseases that leads to severe clinical outcomes like myocardial infarction and stroke.427–429 Accumulating evidence shows that various exosomal ncRNAs play an important regulatory role in the pathophysiological process of atherosclerosis. They were involved in the occurrence and development of atherosclerosis through regulating vascular inflammation, lipid metabolism, and cell survival.430–432
Several studies have demonstrated that exosomal miRNAs released by various types of cells are detected in circulation and are involved in the regulation of pathogenic AS.410,433 It has been reported that the levels of exosomal miR-223, miR-339 and miR-21 derived from thrombin-activated platelet were significantly upregulated.434,435 They could be transferred into VSMCs and inhibited the VSMCs proliferation stimulated by platelet derived growth factor.434 Besides, Li et al. also demonstrated that miR-223 inhibited TNF-α-stimulated endothelial cells (ECs) inflammation by decreasing the expression of ICAM-1.435 These findings indicate that exosomal miR-223 may play a protective role in AS through inhibiting the vascular inflammatory response.
The communication between exosomal miRNAs and ECs plays a vital role in the pathogenesis of AS.436 Exosomal miR-92a, upregulated by the combination of low shear stress and oxidized LDL in atherosclerotic mice model, promoted endothelial inflammation and atherosclerotic plaque formation.437 Besides, Xing et al. reported that exosomal miR-342-5p released by adipose-derived MSCs exerted an anti-atherosclerotic effect by promoting H2O2-induced ECs apoptosis and protecting against ECs injury.438 On the contrary, miR-155 derived from VSMCs destroyed the tight junction and integrity of ECs, which led to the ECs injury and might promote AS progress.430
The dysfunction of VSMCs mediated by macrophages-derived exosomal miRNAs also plays an important role in AS.432,439 A study by Zhu et al. reported that the expression of exosomal miR-21-3p was increased in nicotine-stimulated macrophages and could be assimilated by neighboring VSMCs.432 This effect resulted in an increase in capacities of VSMCs proliferation and migration and thus accelerated the development of AS. Further, it was found that exosomal miR-106a-3p derived from oxidized LDL-incubated macrophages can promote VSMCs proliferation and inhibit their apoptosis, which may protect individuals against AS.439
Macrophages are the main types of cells responsible for the inflammation reaction in AS. The role of macrophages is regulated by exosomal miRNAs which has attracted a lot of attention. It has been found that miR-let7 family is highly enriched with exosomes released by MSCs and inhibited the macrophages infiltration via IGF2BP1 pathway.67 Besides, it also promotes the polarization of M2 macrophages via HMGA2/NF-κB pathway, which ameliorates the progression of AS.67
In addition to miRNAs, it has been demonstrated that the expression of lncRNA GAS5 significantly increases in exosomes collected from patients or animals with AS.440 Further in vitro study revealed that lncRNA GAS5 promoted the apoptosis of macrophages and ECs induced by oxidized LDL, which may lead to the aggravation of AS.440 Besides, the expression of MALAT1, a lncRNA involved in tumorigenesis, was downregulated in oxidized LDL-induced ECs. Overexpression of exosomal MALAT1 derived from ECs increases the expression of MALAT1 in dendritic cells and inhibits the production of ROS and maturation of dendritic cells through activating NRF2 signaling pathway.441 This indicates that downregulation of exosomal MALAT1 derived from oxidized LDL-induced ECs may accelerate the development of AS through inducing dendritic cells maturation. Intriguingly, another study found an increase in the expression of ECs-derived exosomal MALAT1, which promotes the M2 macrophage polarization and may protect individuals against AS.442 However, another study found that the increase in expression of exosomal MALAT1 derived from ECs promotes the formation of neutrophil extracellular traps and accelerates the progression of AS.443
A few studies have found an association between exosomal circRNAs and the pathogenesis of AS.444,445 Wen et al. suggested that the expression of exosomal circRNA‑0006896 in circulation is positively associated with the levels of TG, LDL, and C-reactive protein (CRP) in patients with unstable plaque atherosclerosis.445 The study also reported that the elevated circulating exosomal circRNA‑0006896 leads to high expression of circRNA‑0006896 in ECs, which results in an increases in capacities of proliferation and migration in ECs. This effect is accompanied with the upregulation of DNMT1 and phosphorylation of STAT3, which may provide protection against the vulnerable plaque formation.445
Exosomal ncRNAs in AMI
AMI is characterized by the loss of cardiomyocyte and the necrosis of myocardial. It results from acute obstruction of the coronary artery and can leads to severe clinical consequences.446 Recent studies have revealed that exosomal ncRNAs play a vital role in AMI and may become effective biomarkers for diagnosis and treatment of AMI.447–449
During the pathogenesis of AMI, a wide range of exosomal miRNAs such as miR-125b, miR-499, miR-133, miR-22, miR-21, and miR-301 are found to be upregulated.450–454 Recent evidence has indicated that exosomal miRNAs are involved in the regulation of occurrence and development of AMI. This could be through regulation of pathophysiology pathways such as apoptosis, autophagy, inflammation, and angiogenesis.447,455–457
It has been demonstrated that miR-125b, miR-25-3p, miR-144, miR-126, and miR-146a encapsulated in exosomes from myocardial cells are enriched in expression and exert an anti-atherosclerotic role partly by inhibiting myocardial apoptosis and facilitating ischemic cardiac repair.450,458–461 Besides, exosomal miR-25-3p and miR-146a derived from myocardial cells were also found to restrain the inflammation response, whereas exosomal miR-301 inhibits myocardial autophagy.454,458,461 On the contrary, it was reported that circulating levels of exosomal miR-499 and miR-133a derived from cardiomyocytes are elevated in patients with acute coronary syndromes than those with stable coronary artery disease or without coronary artery disease.451 This implies that exosomal miR-499 and miR-133a may promote the progression of AMI.
In addition, exosomal miRNAs released by other types of cells other than cardiomyocytes also plays an important role in the pathogenesis of AMI. According to Feng et al. the expression of miR-22 in exosomes from MSCs is upregulated under ischemic condition.452 It is also revealed that exosomal miR-22 could be internalized by cardiomyocytes and promote the protection of cardiomyocytes against apoptosis via targeting methyl-CpG-binding protein 2 in vitro. Further in vivo experiments shows that delivery of exosomal miR-22 remarkably reduces cardiac fibrosis thus indicating their significant benefit in treating myocardial fibrosis after AMI.452 Besides, Wang et al. reported that enhanced expression of exosomal miR-21 derived from MSCs have a cardiac protective role through inhibiting apoptosis and promoting angiogenesis of ECs.453
Emerging evidence demonstrates an involvement of exosomal lncRNAs in AMI.448,449,462,463 According to Shyu et al. the expression of exosomal MALAT1 derived from hyperbaric oxygen-induced cardiomyocytes is significantly increased after AMI.448 The increase in MALAT1 then suppresses the expression of miR-92a and enhances the neovascularization. Huang et al. found an elevation in exosomal lncRNA H19 expression released by MSCs after treatment with atorvastatin. This led to an improvement of cardiac function recovery, a reduction of infarct size and cardiomyocyte apoptosis.449 Further, Wang et al. reported that the expression of exosomal lncRNA AK139128 from cardiomyocytes under hypoxic condition increases and this promotes the apoptosis but inhibits the proliferation, migration and invasion of cardiac fibroblasts.463
Exosomal ncRNAs in HF
HF results from dysfunction of myocardial systole or diastole. It is referred to as the decrease in cardiac blood output and the insufficient blood flow in the pulmonary or systemic circulation.464 The main mechanisms involved in HF include myocardial inflammation, autophagy, apoptosis, and remodeling. Recent studies have revealed that exosomal ncRNAs are involved in the regulation of pathogenesis of HF.462,463,465
During the pathogenesis of HF, a number of exosomal miRNAs are reported to be altered in the serum levels. It has been reported that exosomal miR-425 and miR-744 are lowered in the circulation of patients with HF.466 The downregulation of exosomal miR-425 and miR-744 are also reported in angiotensin II-treated cardiac fibroblasts and is positively related to the expression of collagen 1 and α-SMA, which leads to cardiac fibroblast fibrosis. Further in vitro experiments have revealed that exosomal miR-425 and miR-744 protect against myocardial remodeling by inhibiting angiotensin-II-induced collagen formation and fibrogenesis by targeting TGFβ1.466
Exosomal ncRNAs in AF
AF is a type of cardiac arrhythmia and is correlated with the structural and electrical remodeling.467,468 Recently, some studies have revealed that ncRNAs, especially miRNAs play an important role in pathological process of AF.469–471 According to Liu et al. there is low expression of miR-320d in AF cardiomyocytes, accompanied with an increase in apoptosis and impaired cell viability in cardiomyocytes.469 However, transfecting with exosomal miR-320d derived from adipose tissue-derived mesenchymal stem cells significantly reverses the effects of AF on cardiomyocytes, which is dependent on inhibition of STAT3. Besides, Wang et al. reported that the expression of exosomal miR-107 derived from patients with AF was significantly higher than the expression from patients without AF.470 Incubation of ECs with AF-derived exosomes or miR-107 mimics significantly inhibits cell viability and migration, while enhancing cell apoptosis by regulating miR-107/USP14 pathway. Furthermore, Li et al. demonstrated that miR-21-3p loaded in exosomes derived from myofibroblasts contributes to an increase in vulnerability of AF through upregulation of the expression of L-type calcium channel Cav1.2.471
In summary, exosomal ncRNAs are identified as key regulators in diverse cardiovascular diseases such as hypertension, AS, AMI, HF, and AF. Exosomal miRNAs are relatively well studied in cardiovascular diseases, which exert functions by inhibiting vascular inflammatory response, modulating cellular pathophysiology pathways, and so on. Exosomal lncRNAs (GAS5, MALAT1, H19, AK139128) are associated with AS and AMI. However, only a small number of exosomal circRNAs have been determined to be involved in the pathogenesis of AS.
The roles of exosomal ncRNA in neurodegenerative diseases
The incidence of neurodegenerative diseases, including Alzheimer’s disease (AD) and Parkinson’s disease (PD) increases with age. Deviation of ncRNA levels in neurodegenerative diseases has been widely reported.472–478 Exosomes are demonstrated to carry ncRNAs and establish cell-to-cell communication in neurons. They are involved in the pathogenesis and progression of neurodegenerative diseases as mediators.
Exosomal ncRNA in AD
AD is the most common cause of dementia in elderly people. The pathological features of AD are extracellular Aβ deposition, intracellular neurofibrillary tangles, and neuronal loss.479 Exosomal ncRNAs have been shown to regulate the expression and function of amyloid precursor proteins (APP) and tau proteins.480
Growing evidence shows that exosomal miRNAs play an important role in AD.481,482 MiRNA expression profile alters in the brains of patients with AD.483 APP, β-site amyloid precursor protein-cleaving enzyme 1 (BACE1) and microtubule-associated protein tau (MAPT) are pathologically related proteins of AD.484,485 Neuropathological changes in AD are the consequence of diverse cellular processes, such as alterations of AD relevant proteins and oxidative stress.
The interaction between exosomal miRNAs and APP plays an important role in the pathogenesis of AD. Mir-34a, a miRNA strongly related to cognitive dysfunction, is highly expressed in postmortem brain tissue of AD patients. It has been found that miR-34a promotes amyloid processing of APP, whereas knockdown of miR-34a reduces APP accumulation in brain tissues.486,487 Exosomes secreted by miR-34a-overexpressing neurons can be absorbed by adjacent neurons, resulting in the inhibition of target genes in neural network.488 Overexpression of miR-34a in AD tissues is correlated with simultaneous inhibition of target genes of synaptic plasticity, oxidative phosphorylation, and glycolysis. Moreover, miR-193b can also target the 3′ untranslated regions of mRNA (UTR) of APP to exert a regulatory role and further influence the progression of AD. The peripheral blood-derived exosomal miR-193b was measured in the normal population, patients with mild cognitive impairment (MCI) and patients with AD, and it was found that miR-193b is significantly lower in patients with MCI and AD than in the control group and its expression level was negatively correlated with Aβ42.489
Several research reports have clarified the roles of oxidative stress in the pathogenesis of neurodegenerative diseases,490–492 because oxidative stress has been shown to play an important role in enhancing beta-amyloid and tau hyperphosphorylation.493 Low concentrations of miR-141-3p have been observed in plasma exosomes from patients with AD.494 A large amount of miR-141-3p was also found in the exosomes of astrocytes stimulated by inflammation.495 Further studies have confirmed that miR-141-3p can damage the antioxidant defense system and upregulate oxidative stress by inhibiting PTEN.496 In addition, exosomal miR-125b-5p is also involved in the process of AD.497 Transfection of miR-125b significantly promoted neuronal apoptosis and Tau phosphorylation by activating cyclin-dependent kinase 5 (CDK5) and p35/25.498 Besides, inhibition of miR-125b-5p reduces ROS levels, showing a neuroprotective effect against oxidative stress.499
Exosomal lncRNAs also take part in the pathogenesis of AD. LncRNA BACE1-AS is upregulated in the brain of patients with AD. The levels of lncRNA BACE1-AS in plasma derived exosomes of AD and healthy individuals have been determined. No significant difference was observed between the two groups. However, in the whole plasma sample, there was significant difference between AD and the control groups.500
Exosomal ncRNA in PD
PD is characterized by bradykinesia, resting tremor and postural and gait disorders. Typical pathological features of PD are loss of dopaminergic (DAergic) neurons in the dense part of substantia nigra and aggregation of α-SYN protein in Lewy bodies and neurites.501,502 Exosomes-derived ncRNAs are involved in the pathophysiology of PD.503–506
Multiple miRNAs are reported to be upregulated in exosomes of PD cell model, including miR-210-5p, miR-128-1-5p, miR-505-5p, miR-325-5p, miR-16-5p, miR-1306-5p, miR-669b-5p, miR-125b5p, miR-450b-3p, miR-24-2-5p, and miR-2 -6516-3p and miR-1291. These exosomal miRNAs regulate important pathways in the pathogenesis of PD, such as autophagy, inflammation, and protein aggregation.507
Similarly, changes in exosomal miRNAs are found in the cerebrospinal fluid (CSF) of PD patients. Among them, exosomal miR-153, miR-409-3p, miR-10a-5p, and let-7g-3p were significantly increased in CSF of PD patients, while miR-1 and mir19b-3p were significantly decreased.508 Let-7 miRNA family is highly conserved in animal species. It has been reported that let-7 was highly expressed in the PD model.509 The expression of exosomal let-7 in CSF of PD patients was upregulated.510 When exosomal let-7 is absorbed by neurons, it leads to neurodegenerative changes by activating TLR7.511 In addition, in C. elegans PD model, silence of let-7 decreases the accumulation of α-SYN protein, thereby alleviating the progression of PD.512
In addition, the levels of exosomal miRNAs altered in the circulating plasma of PD patients. MiR-195, miR-24, and miR-331-5p were upregulated whereas miR-19b and miR-505 were downregulated in circulating exosomes of patients with PD.513,514 Moreover, the increase of miR-137 was observed in the plasma of PD patients.515,516 Downregulation of exosomal miR-137 can upregulate oxidative resistance 1 (OXR1) in PD mice model, thereby generating a neuroprotective effect.517
When the level of lncRNA extracted from exosomes in plasma samples from PD patients and control group was measured, it was found that 15 and 24 exosomal lncRNAs were upregulated and downregulated, respectively. Among them, lnc-MKRN2-42:1 was positively correlated with MDS-UPDRS III score, which is used to evaluate the severity of dyskinesia in PD patients.518 Compared with the control group, the concentrations of lnc-POU3F3 and α-SYN increased in neuro-derived L1CAM exosomes in PD patients. There was a significant correlation between L1CAM exosomal lnc-POU3F3 levels and PD severity, including motor/cognitive impairment.519 Four lncRNAs (SNCA-AS1, MAPT AS1, AK127687, and AX747125) were detected in exosomes from human cerebrospinal fluid, providing preliminary evidence that these lncRNAs may be of potential use as a diagnostic tool for PD.520 However, further research is needed to elucidate their possible role in PD.
In this chapter, we review recent research on the roles of ncRNAs, especially miRNAs and lncRNAs, in neurodegenerative diseases with an emphasis on AD and PD, whereas exosomal circRNAs have been poorly studied. This fascinating area needs to be explored further.
The roles of exosomal ncRNA in autoimmune diseases
Autoimmune diseases refer to the diseases caused by the immune response of human body to its own antigens hence causing damage to its own tissues. Rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) are the most common systemic autoimmune diseases. The pathogenesis of autoimmune diseases is complex and a lot about it is still unknown. The current research studies shows that ncRNAs encapsulated in exosomes play a critical role in autoimmune diseases.
Exosomal ncRNA in RA
RA is a chronic autoimmune disease characterized by infiltration of leukocyte into joints, causing production of inflammatory mediators and destruction of bone and cartilage tissue.521 Synovitis mediated articular cartilage destruction is associated with upregulation of matrix metalloproteinases (MMPs). This cartilage injury is irreversible and forms a key step in RA joint injury.522 Angiogenesis mediates the delivery of nutrients and inflammatory factors. Continuous angiogenesis leads to chronic changes of synovium in RA.523 Part of the role of MMPs in regulating vascular remodeling is that they activate the secretion of VGF-β in the stromal matrix by activating VEGF.524
MiR-150-5p is associated with T cell maturation and is therefore involved in autoimmune diseases.525 It was also confirmed that miR-150-5p could regulate angiogenesis.526 Compared with patients with osteoarthritis, there is a decrease in expression of miR-150-5p and an increase in expression of VEGF and MMP-14 as well as angiogenesis in RA patients.527 The previous study showed that exosomal micRNA-150 alleviates RA symptoms by downregulating MMP14 and VEGF as well as inhibiting angiogenesis.527 In addition, the miRNA microarray analysis showed that miR-548a-3p was significantly reduced in serum exosomes of RA patients. Serum exosomal miR-548a-3p was negatively correlated with serum levels of CRP, rheumatoid factor (RF), and erythrocyte sedimentation rate (ESR) in RA patients. Further research has proven that exosomal miR-548a-3p is involved in the regulation of macrophage mediated inflammation through the TLR4/NF-κB signaling pathway in RA. Therefore, exosomes may be an important factor in predicting RA disease activity.528
LncRNAs profiles in RA serum exosomes were analyzed, and it was found that Hotair, LUST, anti-NOS2a, MEG9, SNHG4, TUG1, and NET1 were upregulated in RA serum exosomes and Hotair expression level was increased by an average of about 4 times in RA exosomes. Further studies confirmed that the overexpression of Hotair in RA exosomes may be involved in the pathogenesis of RA in two ways. First, it attracts activated macrophages and induces an immune response. Secondly, Hotair may be involved in the production of MMP in osteoclasts and RA synovial cells.529
Exosomal ncRNA in SLE
SLE is an autoimmune disease involving multiple systems and organs, which is characterized by persistent inflammation and autoantibody production. MiRNAs carried by exosomes are involved in the pathogenesis of SLE, especially in regulating inflammation and immune imbalance.
Exosomal miRNAs play an important role in the immune-pathophysiology of SLE.530,531 Dysregulation of miRNA has been found in SLE, and exosomal miRNA from patients with SLE can regulate inflammation and adaptive immune response.532 Previous studies have found that miR-155 promotes autoimmune process by inhibiting suppressor of cytokine signaling-1 (SOCS-1),533 or by repressing transcription of PU.1 and TNF-α.534 Exosomal miR-155 expression was significantly upregulated in patients with SLE, which was higher in patients with lupus nephritis (LN).535 Defects in miR-155 ameliorate autoimmune inflammation in SLE.536 Moreover, plasma miR-21 levels in patients with SLE were higher than with healthy controls.537 Further studies have confirmed that miR-21 modulates abnormal T-cell responses in SLE patients.538 MiR-21 was significantly upregulated in exosomes of SLE patients.535 Moreover, exosomal miR-21 from SLE patients can induce the production of type I interferon by dendritic cells.532
The expression of serum exosomal miR-451a is correlated with SLE disease activity and renal injury. The level of serum exosome miR-451a in patients with active SLE was significantly lower than that in patients with inactive SLE and control group, especially in those with renal damage. Glucocorticoid or hydroxychloroquine increases the expression of exosomal miR-451a in CD4+ T cells.539 The aging of MSCs plays an important role in the occurrence and progression of SLE. The expression of miR-146a in serum exosomes of patients with SLE decreases significantly compared with healthy controls. MiR-146a is internalized into MSCs through exosomes and is involved in MSCs senescence by targeting TRAF6/NF-κB signaling pathway.540 In addition, it also found that miR-146a was associated with SLE activity and proteinuria.541
LN occurs in 40 to 75% of patients with SLE, which is one of the leading causes of death in these patients.542 Compared with other biological samples, urine samples are easy to obtain hence less costly. Urinary exosomal miRNAs can accurately reflect renal dysfunction and structural damage.543,544 Multiple urinary exosomal miRNAs have been found to be associated with renal fibrosis,545,546 and three of these were associated with renal fibrosis in LN, including miR-410, miR-29c, and miR-150.547–549 Among them, miR-410 directly interacts with IL-6 3’- UTR to reduce its expression and inhibits renal fibrosis548 whereas miR-150 promotes renal fibrosis by downregulating SOCS1.547 MiR-29c levels were inversely correlated with histological chronicity index and glomerulosclerosis, but not with renal function.549 In addition, let-7 and miR-21 were downregulated in patients with active LN compared with patients with inactive LN. It was reported that let-7 and miR-21 are downregulated during disease flare and upregulated after treatment. It is suggested that urinary exosomal miRNAs (let-7a and miR-21) can be used to guide the clinical staging of patients with LN.550
In this section, we discuss the roles of exosomal ncRNAs in autoimmune diseases including RA and SLE. Exosomal miR-150-5p alleviates RA symptoms by regulating VEGF, MMP-14, and angiogenesis, while exosomal miR-548a-3p regulates macrophage-mediated inflammation in RA. Exosomal lncRNAs Hotair, LUST, anti-NOS2a, MEG9, SNHG4, TUG1, and NET1 may be involved in the pathogenesis of RA. In addition, exosomal miRNAs have been implicated in the pathogenesis of SLE, especially in the regulation of inflammation and immune imbalance, activity, and complication, mainly associated with LN.
The roles of exosomal ncRNAs in infectious diseases
Infectious diseases refer to a group of diseases that are caused by viruses, bacteria, fungi, or other pathogens. Among many infectious diseases, pneumonia and viral hepatitis are very common in the human population. Recently, accumulating evidence revealed that exosomal ncRNAs participated in the pathogenesis of infectious diseases.551–553 In this study, we summarize the current evidence about the roles of exosomal ncRNAs in pneumonia and viral hepatitis.
Exosomal ncRNAs in pneumonia
Pneumonia is a very common disease worldwide. Severe community-acquired pneumonia remains a life-threatening disease especially in children and elderly.554 It has been demonstrated that exosomal ncRNAs levels are markedly different in patients with pneumonia.553,555
Exosomal miRNAs are the most studied ncRNAs involved in the development of pneumonia. Recently, it was reported that abundant exosomes selectively loaded with miR-155 are presented in circulation from sepsis-related acute lung injury (ALI) mice.555 Injection of exosomes harvested from ALI mice significantly increased the number of M1 macrophages in the lung and led to the inflammation in healthy mice through activating the NF-κB signaling pathway and the downstream upregulation of TNF-α and IL-6. Besides, circulating exosomal miR-155 was also found to promote macrophage proliferation and inflammation by regulating SHIP1 and SOCS1.555 According to Quan et al. the expression of exosomal miR-371b-5p derived from injured alveolar progenitor type II cells is upregulated and decreases the expression of PTEN.556 This promotes cell proliferation and re-epithelialization of injured alveoli through activating phosphorylation of Akt and its substrates GSK3β and FOXOs. Importantly, the elevated levels of exosomal miR-371b-5p were also observed in lavage samples from patients with acute pneumonia, which may provide a potential therapeutic target for acute pneumonia.
Dysregulated immune reaction is a major contributing factor to the pathophysiology of pneumonia and is associated with the high incidence ratio of pneumonia. It was found that exosomal miR-221 and miR-222 levels derived from lipopolysaccharide (LPS)-induced macrophage increases and promotes the proliferation of epithelial cells.557 Besides, macrophages induced by LPS also releases high number of exosomes that deliver miR-223 and miR-142 hence inhibiting the LPS-triggered inflammatory responses in the lung.558 However, the expression of exosomal miR-103a-3p from LPS-induced lung cells and from circulation of pneumonia patients were decreased. Overexpression of exosomal miR-103a-3p attenuates inflammation by regulating immune response via transducing β-like 1X related protein 1/NF-κB signaling pathway.559
Exosomal ncRNAs in viral hepatitis
Viral hepatitis is an infectious disease caused by a variety of hepatitis viruses. If not treated, it can develop into liver cirrhosis or even liver cancer which is very dangerous for human health. Therefore, exploration of the underlying mechanisms of viral hepatitis is of great significance. Accumulating evidence demonstrates that exosomal ncRNAs are involved in the pathogenesis of viral hepatitis.560
Exosomal miRNAs has been demonstrated to be involved in the regulation of multiple signaling pathways during the development of viral hepatitis.561,562 Globally, hepatitis B virus (HBV) is the most common hepatitis and causes very heavy economic and heathy burden. According to Li et al. some exosomal miRNAs, such as miR-221-3p and miR-25-3p are upregulated in patients with chronic HBV infection, whereas other such as miR-372-3 and miR-10a-5p are downregulated.563 Besides, miR-122, miR-204, miR-let7c, miR-23b, and miR150 are also altered and are correlated with HBV-related liver diseases.562
Hepatitis C is also common in the human population. The fibrosis of the liver is correlated with the chronic infection of hepatitis C virus (HCV). However, the specific mechanisms remain poorly understood. Recently, Devhare et al. reported that the exosomal miR-19a released by HCV-infected hepatocytes is upregulated and increases the expression of fibrosis marker genes in hepatic stellate cells (HSC).564 Further analysis revealed that the exosomal miR-19a aggravates the liver fibrosis by activating the SOC3/STAT3/TGF-β signaling pathway.
Relatively, there are few studies that evaluated exosomal lncRNAs in the development of viral hepatitis.565 It was found that the expression of runt-related transcription factor 1 (RUNX1) and RUNX1 overlapping RNA (RUNXOR) are significantly upregulated in myeloid-derived suppressor cells (MDSCs) during chronic HCV infection. These were positively correlated with the immunosuppressive molecule levels.565 Mechanistically, it has been demonstrated that HCV-associated exosomes deliver RUNXOR and RUNX1 to MDSCs and promote the function of differentiation and suppressive functions of MDSCs through regulation of STAT3 signaling pathway. These results indicate that RUNXOR and RUNX1 may become a promising target for immunomodulation with antiviral treatment during HCV infection.
In summary, the roles of exosomal miRNAs in pneumonia has been studied most intensively, among which miR-155 and miR-371B-5p are related to the occurrence and development of pneumonia, while miR-221, miR-222, miR-223, miR-142, and miR-103a-3p are associated with the immune response of pneumonia. As for viral hepatitis, exosomal miRNAs have been shown to regulate multiple signaling pathways, whereas few exosomal lncRNAs (RUNXOR and RUNX1) have been demonstrated to be involved in the development of viral hepatitis.
Clinical applications of exosomal ncRNAs in human diseases
Recently, exosomal ncRNAs have emerged as novel players in the occurrence and development of various human diseases. Circulating ncRNA profiles in exosomes are altered for a wide range of diseases, suggesting that exosomal ncRNAs may serve as cycling indicators of an individual’s physiological status and targeted therapies tools in precision medicine. Therefore, exosomal ncRNAs exert potential to be candidate biomarkers and therapeutic targets for the diseases. This section summarizes the present knowledge on the potential clinical implication of exosomal ncRNAs in biomarker identification and therapy exploration in diseases.
Exosomal ncRNAs as diagnostic biomarkers in human diseases
Biomarker are a series of molecules that can be utilized for disease detection and/or prognosis prediction. Sensitivity, specificity, stability, and relatively non-invasive are most important and necessary characteristics of a good biomarker. Notably, emerging research is targeting exosomes as potential biomarkers because they can be detected using simple and inexpensive methods in many body fluids and are stable against enzyme degradation by providing a protective transportation system. It is worth noting that exosomal ncRNAs are differentially expressed in human lifestyle activities and various pathological diseases, including cardiovascular, metabolic, neurodegenerative, autoimmune, infectious, and cancerous disorders, which underlies the potential of ncRNAs as promising biomarkers for early detection.360,364,566–569 This study compiles the circulating exosomal ncRNAs that have been reported to be altered during the course of human diseases, as detailed in Table 1.
Table 1.
Exosomal ncRNAs as potential diagnostic biomarkers in human diseases
| Diseases | Cargoes | Expression | Effects | Refs |
|---|---|---|---|---|
| Lung cancer | miR-23a | ↑ | Inducing phenotypic changes, increasing vascular permeability and cancer migration | 137,150 |
| miR-126 | ↑ | Inducing loss of malignancy of NSCLC cells | 144 | |
| miR-193a-3p, miR-210-3p, miR-5100 | ↑ | Activating STAT3 signaling and increasing the expression of mesenchymal related molecules | 138 | |
| miR-499a-5p | ↑ | Promoting cell proliferation, migration and EMT | 139 | |
| GAS5 | ↑ | Function as an ideal noninvasive biomarker for NSCLC | 154 | |
| SOX2-OT | ↑ | Serving as a promising non-invasive plasma-based tumor biomarker for LSCC | 155 | |
| circSATB2 | ↑ | Acting as a biomarker for the diagnosis of NSCLC | 156 | |
| circ-0007761, circ-0047921, circ-0056285 | ↑ | Acting as promising biomarkers for NSCLC diagnosis | 159 | |
| BC | miR-9 | ↑ | Inducing tumor growth | 174 |
| miR-10b | ↑ | Modulating tumor microenvironment | 170 | |
| miR-1246 | ↑ | Regulating breast tumor progression | 171 | |
| miR-20a-5p | ↑ | Inducing proliferation and differentiation of osteoclasts | 183 | |
| miR-134 | ↓ | Acting as a potential biomarker for BC | 172 | |
| MALAT1 | ↑ | Inducing BC progression | 188 | |
| H19 | ↑ | Acting as a biomarker for the diagnosis of BC | 190 | |
| circHIF1A | ↑ | Regulating stem cell properties of BC | 196 | |
| HCC | miR-10b-5p | ↑ | Acting as a biomarker for early-stage HCC | 211 |
| miR-125b | ↓ | Associated with tumor number, encapsulation, and TNM stage | 214 | |
| miR-18a, miR-221, miR-222, miR-224 | ↑ | Function as novel serological biomarkers for HCC | 217 | |
| miR-101, miR-106b, miR-122, miR-195 | ↓ | |||
| FAL1 | ↑ | Promoting cell proliferation and metastasis | 220 | |
| H19 | ↑ | Promoting cell proliferation, migration, and invasion | 221 | |
| LINC00161 | ↑ | Serving as a novel biomarker for HCC | 227 | |
| HEIH | ↑ | Serving as a novel biomarker for HCC | 230 | |
| SENP3-EIF4A1 | ↓ | Inducing tumor growth | 231 | |
| FAM138B | ↓ | Inhibiting HCC growth | 232 | |
| circUHRF1 | ↑ | Inducing immunosuppression | 240 | |
| CRC | miR-1229, miR-1246, miR-150, miR-21, miR-223, miR-23a | ↑ | Promising biomarkers for non-invasive diagnosis of CRC | 256 |
| miR-23a, miR-301a | ↑ | Promising biomarkers for non-invasive diagnosis of CRC | 257 | |
| miR-6803-5p | ↑ | Serving as a diagnostic and prognostic biomarker | 259 | |
| LINC02418 | ↑ | Inducing tumorigenesis | 267 | |
| NNT-AS1 | ↑ | Inducing the proliferation, migration and invasion of CRC cells | 578 | |
| LINC02418 | ↑ | Involvement in CRC tumorigenesis | 267 | |
| CCAT2 | ↑ | Serving as a novel potential predictor in CRC | 274 | |
| LNCV6-116109, LNCV6-98390, LNCV6-38772, LNCV-108266, LNCV6-84003, LNCV6-98602 | ↑ | Serving as potential non-invasive biomarkers for early diagnosis of CRC | 275 | |
| circPACRGL | ↑ | Inducing cell proliferation, migration and invasion | 278 | |
| circFMN2 | ↑ | Mediating cell proliferation and migration | 279 | |
| hsa-circ-0004771 | ↑ | Serving as a novel diagnostic biomarker of CRC | 283 | |
| GC | miR-34 | ↓ | Inducing GC cell proliferation and invasion and tumor growth | 289 |
| miR-1246 | ↑ | Serving as potential biomarker for the early diagnosis of GC | 296 | |
| miR-1290 | ↑ | Inducing GC cell proliferation and invasion | 285 | |
| UEGC1 | ↑ | Acting as a promising biomarker in the development of GC | 305 | |
| GC1 | ↑ | Serving as a noninvasive biomarker for detecting early-stage GC | 307 | |
| lnc-SLC2A12-10:1 | ↑ | Serving as a noninvasive biomarker for the diagnosis of GC | 310 | |
| CEBPA-AS1 | ↑ | Promoting cell proliferation, inhibiting apoptosis, and inducing GC progression | 311 | |
| lnc-GNAQ-6:1 | ↓ | Serving as a potential biomarker for the detection of GC | 312 | |
| circNEK9 | ↑ | Promoting the proliferation, migration, and invasion of GC cells | 316 | |
| circ29 | ↑ | Involved in the occurrence and development of GC | 317 | |
| has-circ-0065149 | ↓ | Acting as a novel biomarker for diagnosis | 320 | |
| PCa | miR-141 | ↑ | Function as a useful biomarker for the diagnosis of metastatic PCa | 331 |
| circ-0044516 | ↑ | Inducing tumor cell metastasis | 337 | |
| T2DM | miR-20b-5p | ↑ | Attenuating insulin-stimulated glycogen accumulation | 363 |
| Obesity | miR-26a | ↓ | Inversely correlated with BMI | 360 |
| MetS | MALAT1 | ↓ | Serving as a potential epigenetic biomarker of diabetes risk or MetS | 366 |
| OP | miR-214 | ↑ | Repressing osteoblasts activity | 395 |
| hsa-circ-0006859 | ↑ | Serving as a high sensitivity and specificity biomarker | 404 | |
| Hypertension | miR-155-5p | ↓ | Promoting VSMCs proliferation and vascular remodeling | 422 |
| miR-27a | ↑ | Promoting vasodilation and causing hypertension | 423 | |
| miR-425-5p, miR-17-5p | ↑ | Potentially serving as biomarkers | 421 | |
| miR-17 | ↓ | Promoting ECs inflammation | 424 | |
| AS | miR-223 | ↑ | Inhibiting inflammation and VSMCs proliferation | 434,435 |
| miR-92a | ↑ | Promoting ECs inflammation and the formation of atherosclerotic plaque | 437 | |
| miR-342-5p | ↑ | Promoting H2O2-induced ECs apoptosis | 438 | |
| miR-155 | ↑ | Inhibiting ECs proliferation and migration | 430 | |
| miR-21-3p | ↑ | Promoting VSMCs proliferation and migration | 432 | |
| miR-106a-3p | ↑ | Promoting proliferation and inhibiting apoptosis in VSMCs | 439 | |
| GAS5 | ↑ | Promoting the apoptosis of macrophages and ECs | 440 | |
| MALAT1 | ↑ | Promoting the M2 macrophage polarization, the formation of neutrophil extracellular traps in neutrophils | 442,443 | |
| circRNA-0006896 | ↑ | Promoting proliferation and migration of ECs | 445 | |
| AMI | miR-125b | ↑ | Ameliorating cardiomyocytes apoptosis and facilitating ischemic cardiac repair | 450 |
| miR-22 | ↑ | Inhibiting apoptosis of cardiomyocytes | 452 | |
| miR-301 | ↑ | Inhibiting myocardial autophagy | 454 | |
| miR-25-3p | ↑ | Reducing myocardial apoptosis and inflammation | 458 | |
| miR-144 | ↑ | Ameliorating hypoxia-induced cardiomyocyte apoptosis | 459 | |
| miR-146a | ↑ | Inhibiting myocardial apoptosis, inflammatory response, and fibrosis | 461 | |
| MALAT1 | ↑ | Enhancing neovascularization | 448 | |
| H19 | ↓ | Reducing infarct size and cardiomyocyte apoptosis | 449 | |
| lncAK139128 | ↓ | Promoting the apoptosis and inhibiting the proliferation, migration, and invasion of cardiac fibroblasts | 463 | |
| HF | miR-425, miR-744 | ↓ | Protecting against myocardial remodeling | 466 |
| AF | miR-320d | ↓ | Promoting viability and inhibiting apoptosis of cardiomyocytes | 469 |
| miR-107 | ↑ | Suppressing viability and migration of ECs, enhancing cell apoptosis | 470 | |
| AD | miR-34a | ↑ | Promoting amyloid processing of APP | 486,487 |
| miR-141-3p | ↑ | Damaging the antioxidant defense system and up-regulating oxidative stress | 496 | |
| miR125b-5p | ↑ | Promoting neuronal apoptosis and Tau phosphorylation | 498 | |
| PD | let-7 | ↑ | Leading to neurodegenerative changes | 511 |
| RA | miR-548a-3p | ↓ | Regulating macrophage mediated inflammation | 528 |
| Hotair | ↑ | Inducing an immune response | 529 | |
| SLE | miR-155 | ↑ | Regulating autoimmune inflammation in SLE | 536 |
| miR-21 | ↑ | Inducing the production of type I interferon | 532 | |
| Pneumonia | miR-155 | ↑ | Promoting inflammation and proliferation | 555 |
| miR-371b-5p | ↑ | Promoting proliferation and re-epithelialization of injured alveoli | 556 | |
| miR-221/222 | ↑ | Promoting epithelial cell proliferation | 557 | |
| miR-223/142 | ↑ | Inhibiting LPS-induced lung inflammation | 558 | |
| miR-103a-3p | ↓ | Inhibiting inflammation | 559 | |
| Viral hepatitis | miR-221-3p, miR-25-3p | ↑ | Associated with liver fibrosis and inflammation | 563 |
| miR-372-3, miR-10a-5p | ↓ | Associated with liver fibrosis and inflammation | 563 | |
| miR-19a | ↑ | Promoting liver fibrosis | 564 |
NSCLC non-small cell lung cancer, STAT3 signal transducer and activator of transcription 3, EMT epithelial-mesenchymal transition, LSCC lung squamous cell carcinoma, BC breast cancer, HCC hepatocellular carcinoma, CRC colorectal cancer, GC gastric cancer, PCa prostate cancer, T2DM type 2 diabetes mellitus, BMI body mass index, MetS metabolic syndrome, OP osteoporosis, AS atherosclerosis, VSMCs vascular smooth muscle cells, ECs endothelial cells, AMI acute myocardial infarction, HF heart failure, AF atrial fibrillation, AD Alzheimer’s disease, APP amyloid precursor proteins, PD Parkinson’s disease, RA rheumatoid arthritis, SLE systemic lupus erythematosus, LPS lipopolysaccharide
Exosomal ncRNAs-based therapeutics in human diseases
Considering the various advantages of exosomes, such as natural availability, considerate biocompatibility, biological barrier permeability, low immunogenicity, and toxicity, exosomes are poised to become a promising tool for therapeutic vehicles.7,570,571 Genetic engineering has been explored to modify exosomes for therapeutic utilization. The ability to modify isolated and purificatory exosomes loaded with mimics or inhibitors of ncRNAs and target molecules on their surface, offers the possibility of using exosomes as vectors to transport specific ncRNAs to specific tissues or organs (Fig. 7). This gene regulation proposes exosomal ncRNAs as promising approach for the treatment of various human diseases.
Fig. 7.
Exosomal ncRNA-based therapeutics in human diseases. Endogenous (pre-loading cargoes into donor cells followed by exosomal cargo release) or exogenous (directly loading cargoes into exosomes after their production or isolation) loading of ncRNAs or their inhibitors into exosomes exhibits significant therapeutic potential. ASO antisense oligonucleotide. This figure was created with the aid of Servier Medical Art (https://smart.servier.com/)
Therapies that upregulate exosomal ncRNAs
Exosomes can load ncRNAs release of which may present promising therapeutic effects. Several studies have explored important therapeutic interventions of exosome-based ncRNAs. By modifying exosomes with ncRNA mimics or inhibitors, exosomes can serve as delivery system of specific ncRNA sequences.
Elevated expression levels of beneficial ncRNAs carried by exosomes have been described to have various therapeutic implications. The levels of ncRNAs in exosomes will enhance by loading ncRNAs into exosomes endogenously or exogenously. Exogenous loading is performed after exosome production or isolation with cargo of interest encapsulated to exosomes by co-incubation, chemical transfection, or physical approaches such as electroporation and dialysis.572 For example, Lv et al. used electroporation to package miR-21-5p mimics into ADSC-derived exosomes. The results of their study suggested that the miR-21-5p mimics-enriched exosomes accelerate diabetic wound healing in vivo through promoting re-epithelialization, collagen remodeling, angiogenesis, and vessel maturation.573
In addition, cargoes can be loaded in an endogenous way by pre-loading donor cells with therapeutic ncRNA, which will then be encapsulated in exosome. For instance, Zhuo et al. transfected lncRNA FAM138 into HCC cells and then created exosomes enriched in FAM138, which alleviated the progression of HCC by regulating miR-765.232 Therefore, endogenous or exogenous loading of beneficial ncRNAs into exosomes exhibits significant therapeutic potential.
Therapies that downregulate exosomal ncRNAs
It may also be therapeutic to decrease the amount of detrimental ncRNAs in exosomes. This effect can be achieved through endogenous or exogenous loading of small interfering RNAs (siRNAs) or ncRNA inhibitors such as antisense oligonucleotides (ASO) into exosomes. For instance, exosome-delivered circRNA has-circ-0005963 promotes drug resistance in CRC, whereas siRNA of has-circ-0005963 transported by exosomes can reverse resistance in vivo.281 Moreover, miR-21 is an oncogenic miRNA, while delivery of anti-miRNA oligonucleotides miR-21 using modified exosomes can reduce miR-21 level in the glioblastoma, contributing to reduction of tumor size.574
Above all, these studies provide new strategies for potential clinical diagnosis and therapeutic management for exosome-mediated human diseases. Exosome-based delivery of curative ncRNAs, as well as siRNAs or inhibitors of harmful ncRNAs has the potential to be used in development of disease therapies. However, the transition of exosomal ncRNAs from basic laboratory research to clinical application remains challenging. Further development of biomedical materials and molecular targeted therapies is needed to elucidate the functions of exosomal ncRNAs.
Perspectives and conclusions
Knowledge of the roles of exosomal ncRNAs in physiological and pathological conditions has advanced considerably over the past few decades. In this review, we discuss the structures of exosomes and their cargo ncRNAs, and compile the underlying regulatory mechanisms. In order to distinguish between ncRNAs in exosomes and non-exosomes in health and disease, we also strive to study the differences in physiological homeostasis and pathological processes. Exosomes exert their roles in the pathophysiology of multiple disorders by modulating immune response, oxidative stress, autophagy, gut microbe, and cell cycle dysregulation. The impact of exosomes on cell biology is greater than originally expected, which makes related research rather complex. The temporal and spatial expression patterns, precise roles and mechanisms of specific miRNAs, lncRNAs, and circRNAs encapsulated in exosomes remain largely unknown in different system development and diseases.
Interest in the contribution of exosomal ncRNAs to the progression of various diseases, including cancers, metabolic diseases, neurodegenerative diseases, cardiovascular diseases, autoimmune diseases, and infectious diseases, is booming. Tissue-specific alterations of exosomal ncRNAs play an important role in the initiation and development of human diseases as well as their complications. First, exosomal ncRNAs are involved in pathological cellular processes including cell proliferation, migration, invasion, metastasis, angiogenesis, and EMT associated with diverse cancers. Second, exosomal ncRNAs promote cell-to-cell and tissue-to-tissue crosstalk in an autocrine, paracrine or endocrine manner, and hence applying pleiotropic activities in metabolic diseases such as T2DM, obesity, and primary osteoporosis. Third, exosomal ncRNAs serve as regulators of dyslipidemia and myocardial structure and function in cardiovascular diseases including hypertension, AS, AMI, HF, and AF. Fourth, Exosomes carry ncRNAs to regulate communication between neurons and act as mediators involved in the pathogenesis and progression of neurodegenerative diseases. Fifth, exosomal ncRNAs modulate autoimmune diseases mainly by regulating inflammation and immune imbalance. Besides, the roles of exosomal ncRNAs in infectious diseases have been shown to be involved in the development of pneumonia and viral hepatitis. However, much effort remains to be done to understand the full extent of exosomal ncRNAs exerting their pathological effects.
Although exosomal ncRNAs have been identified for a relatively short time, there have been massive progression in clinical and therapeutic applications. In the field of diagnostics, it is noteworthy that exosomal ncRNAs are differentially expressed in human lifestyle activities and a variety of pathological diseases, suggesting that exosomal ncRNAs have the potential to be biomarkers for early detection. Exosomal ncRNAs exert advantages as biomarkers over non-exosomal ncRNAs. NcRNAs harbored in exosomes can be protected from degradation by RNase thus increasing their stability. Exosomal ncRNAs can present a high concentration in body fluid.575 Several studies have compared exosomal and non-exosomal miRNAs as biomarkers, and 75% of the studies reported that exosomal pattern of miRNAs are more important in biological processes compared with non-exosomal ones. This can be attributed to the advantages of exosomal miRNAs including quantity, quality, and stability.576 In the area of therapeutics, the use of exosomal ncRNAs have many potential benefits, so increasing exosomal ncRNAs therapies will enter a formal drug development process. Exosomes bearing a specific cargo can function as a drug delivery system. Exosomes have several advantages when delivering ncRNAs compared with traditional delivery methods, such as virus and liposome. Use of exosomes as endogenous vehicles can evade immune response, thus minimizing their toxicity. An in vivo study conducted by Mendt et al. reported no side effects including adverse immune response even after repeated administration of exosomes.570 Furthermore, exosomes can target specific tissues and cells through specific ligands on the surface.577 Moreover, exosomes can readily penetrate biological barriers such as the blood-brain barrier (BBB). For instance, an in vivo study reported that exosomes can cross the BBB and deliver siRNAs into the brain.571 Despite exosomal ncRNAs have great potential as biomarkers and therapeutics for a wide range of human diseases, there are many hurdles to bring them to clinic. First of all, we need to determine the extent to which purified exosomes are likely to be sufficient to confer the positive effects. Secondly, the dynamics and pharmacokinetics, as well as toxic studies of potential exosomal ncRNAs drugs require to be repeatedly tested. Besides, there is urgent need for experiments using animal and clinical models to identify whether exosomal ncRNAs exert regulatory homeostasis or pathological functions. Overcoming these obstacles will take the field to another unprecedented level.
The purpose of this study is to explore and strengthen the understanding of the mechanisms and roles of exosomal ncRNAs in human health and diseases, and to provide the basis for new clinical diagnosis and therapy strategies.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 82071593, 81770833, 81974223, and 82101663); the Fundamental Research Funds For the Central Universities of Central South University (NO. 2019zzts354).
Author contributions
C.L. and Y.-Q.N. wrote the manuscript and drew the figures. H.X., Q.-Y.X., and Y.Z. collected literature and summarized the table. J.-K.Z. supervised the manuscript and modified the figures. J.-Y.H. and S.L. polished language and corrected grammar errors. Y.-S.L. conceived the idea and directed the writing. All authors have read and approved the article.
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Chen Li, Yu-Qing Ni
References
- 1.Lakshmi S, Hughes TA, Priya S. Exosomes and exosomal RNAs in breast cancer: a status update. Eur. J. Cancer. 2021;144:252–268. doi: 10.1016/j.ejca.2020.11.033. [DOI] [PubMed] [Google Scholar]
- 2.Yang D, et al. Progress, opportunity, and perspective on exosome isolation - efforts for efficient exosome-based theranostics. Theranostics. 2020;10:3684–3707. doi: 10.7150/thno.41580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Safdar A, Saleem A, Tarnopolsky MA. The potential of endurance exercise-derived exosomes to treat metabolic diseases. Nat. Rev. Endocrinol. 2016;12:504–517. doi: 10.1038/nrendo.2016.76. [DOI] [PubMed] [Google Scholar]
- 4.Li S, et al. Exosomes from hyperglycemia-stimulated vascular endothelial cells contain versican that regulate calcification/senescence in vascular smooth muscle cells. Cell Biosci. 2019;9:1. doi: 10.1186/s13578-018-0263-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lin X, et al. Exosomal Notch3 from high glucose-stimulated endothelial cells regulates vascular smooth muscle cells calcification/aging. Life Sci. 2019;232:116582. doi: 10.1016/j.lfs.2019.116582. [DOI] [PubMed] [Google Scholar]
- 6.Couzin J. Cell biology: the ins and outs of exosomes. Science. 2005;308:1862–1863. doi: 10.1126/science.308.5730.1862. [DOI] [PubMed] [Google Scholar]
- 7.Ni Y-Q, Lin X, Zhan J-K, Liu Y-S. Roles and functions of exosomal non-coding RNAs in vascular aging. Aging Dis. 2020;11:164–178. doi: 10.14336/AD.2019.0402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jeppesen DK, et al. Reassessment of exosome composition. Cell. 2019;177:428–445.e418. doi: 10.1016/j.cell.2019.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Castaño C, Novials A, Párrizas M. Exosomes and diabetes. Diabetes Metab. Res. Rev. 2019;35:e3107. doi: 10.1002/dmrr.3107. [DOI] [PubMed] [Google Scholar]
- 10.Palade GE. A small particulate component of the cytoplasm. J. Biophys. Biochem Cytol. 1955;1:59–68. doi: 10.1083/jcb.1.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hoagland MB, et al. A soluble ribonucleic acid intermediate in protein synthesis. J. Biol. Chem. 1958;231:241–257. [PubMed] [Google Scholar]
- 12.Crick FH. On protein synthesis. Symp. Soc. Exp. Biol. 1958;12:138–163. [PubMed] [Google Scholar]
- 13.Tyc K, Steitz JA. U3, U8 and U13 comprise a new class of mammalian snRNPs localized in the cell nucleolus. EMBO J. 1989;8:3113–3119. doi: 10.1002/j.1460-2075.1989.tb08463.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Prestayko AW, Busch H. Low molecular weight RNA of the chromatin fraction from Novikoff hepatoma and rat liver nuclei. Biochim. Biophys. Acta. 1968;169:327–337. doi: 10.1016/0005-2787(68)90041-5. [DOI] [PubMed] [Google Scholar]
- 15.Zieve G, Penman S. Small RNA species of the HeLa cell: metabolism and subcellular localization. Cell. 1976;8:19–31. doi: 10.1016/0092-8674(76)90181-1. [DOI] [PubMed] [Google Scholar]
- 16.Jarroux J, Morillon A, Pinskaya M. History, discovery, and classification of lncRNAs. Adv. Exp. Med Biol. 2017;1008:1–46. doi: 10.1007/978-981-10-5203-3_1. [DOI] [PubMed] [Google Scholar]
- 17.Sanger HL, et al. Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc. Natl Acad. Sci. USA. 1976;73:3852–3856. doi: 10.1073/pnas.73.11.3852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brannan CI, Dees EC, Ingram RS, Tilghman SM. The product of the H19 gene may function as an RNA. Mol. Cell Biol. 1990;10:28–36. doi: 10.1128/mcb.10.1.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee JT, Davidow LS, Warshawsky D. Tsix, a gene antisense to Xist at the X-inactivation centre. Nat. Genet. 1999;21:400–404. doi: 10.1038/7734. [DOI] [PubMed] [Google Scholar]
- 20.Brown CJ, et al. A gene from the region of the human X inactivation centre is expressed exclusively from the inactive X chromosome. Nature. 1991;349:38–44. doi: 10.1038/349038a0. [DOI] [PubMed] [Google Scholar]
- 21.Lander ES, et al. Initial sequencing and analysis of the human genome. Nature. 2001;409:860–921. doi: 10.1038/35057062. [DOI] [PubMed] [Google Scholar]
- 22.The ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature. 489, 57–74, (2012). [DOI] [PMC free article] [PubMed]
- 23.Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75:843–854. doi: 10.1016/0092-8674(93)90529-y. [DOI] [PubMed] [Google Scholar]
- 24.Reinhart BJ, et al. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature. 2000;403:901–906. doi: 10.1038/35002607. [DOI] [PubMed] [Google Scholar]
- 25.Kapranov P, et al. Large-scale transcriptional activity in chromosomes 21 and 22. Science. 2002;296:916–919. doi: 10.1126/science.1068597. [DOI] [PubMed] [Google Scholar]
- 26.Okazaki Y, et al. Analysis of the mouse transcriptome based on functional annotation of 60,770 full-length cDNAs. Nature. 2002;420:563–573. doi: 10.1038/nature01266. [DOI] [PubMed] [Google Scholar]
- 27.Rinn JL, et al. The transcriptional activity of human Chromosome 22. Genes Dev. 2003;17:529–540. doi: 10.1101/gad.1055203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim JK, et al. Functional genomic analysis of RNA interference in C. elegans. Science. 2005;308:1164–1167. doi: 10.1126/science.1109267. [DOI] [PubMed] [Google Scholar]
- 29.Bhat AA, et al. Role of non-coding RNA networks in leukemia progression, metastasis and drug resistance. Mol. Cancer. 2020;19:57. doi: 10.1186/s12943-020-01175-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Anastasiadou E, Jacob LS, Slack FJ. Non-coding RNA networks in cancer. Nat. Rev. Cancer. 2018;18:5–18. doi: 10.1038/nrc.2017.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Valadi H, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007;9:654–659. doi: 10.1038/ncb1596. [DOI] [PubMed] [Google Scholar]
- 32.Huang X, et al. Characterization of human plasma-derived exosomal RNAs by deep sequencing. BMC Genomics. 2013;14:319. doi: 10.1186/1471-2164-14-319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.van Balkom BW, et al. Quantitative and qualitative analysis of small RNAs in human endothelial cells and exosomes provides insights into localized RNA processing, degradation and sorting. J. Extracell. Vesicles. 2015;4:26760. doi: 10.3402/jev.v4.26760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mori MA, et al. Extracellular miRNAs: from biomarkers to mediators of physiology and disease. Cell Metab. 2019;30:656–673. doi: 10.1016/j.cmet.2019.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang J, et al. Exosome and exosomal microRNA: trafficking, sorting, and function. Genomics Proteom. Bioinforma. 2015;13:17–24. doi: 10.1016/j.gpb.2015.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shan S-K, et al. Exosomes and bone disease. Curr. Pharm. Des. 2020;25:4536–4549. doi: 10.2174/1381612825666191127114054. [DOI] [PubMed] [Google Scholar]
- 37.Kita S, Maeda N, Shimomura I. Interorgan communication by exosomes, adipose tissue, and adiponectin in metabolic syndrome. J. Clin. Investig. 2019;129:4041–4049. doi: 10.1172/JCI129193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kamerkar S, et al. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature. 2017;546:498–503. doi: 10.1038/nature22341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lei, Y. T., Liu, X. G. & Xu, S. Exosomal CircRNAs in Aging and Aging-related Diseases. Chin. J. Biochem. Mol. Biol.10.13865/j.cnki.cjbmb.2021.03.1621 (2021).
- 40.Su T, et al. Bone marrow mesenchymal stem cells-derived exosomal MiR-29b-3p regulates aging-associated insulin resistance. ACS Nano. 2019;13:2450–2462. doi: 10.1021/acsnano.8b09375. [DOI] [PubMed] [Google Scholar]
- 41.Prattichizzo F, et al. Exosome-based immunomodulation during aging: A nano-perspective on inflamm-aging. Mech. Ageing Dev. 2017;168:44–53. doi: 10.1016/j.mad.2017.02.008. [DOI] [PubMed] [Google Scholar]
- 42.Sun Y, et al. Expression of miRNA-29 in Pancreatic β Cells Promotes Inflammation and Diabetes via TRAF3. Cell Rep. 2021;34:108576. doi: 10.1016/j.celrep.2020.108576. [DOI] [PubMed] [Google Scholar]
- 43.Kamalden TA, et al. Exosomal MicroRNA-15a transfer from the pancreas augments diabetic complications by inducing oxidative stress. Antioxid. Redox Signal. 2017;27:913–930. doi: 10.1089/ars.2016.6844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.He Q, et al. Mesenchymal stem cell-derived exosomes exert ameliorative effects in type 2 diabetes by improving hepatic glucose and lipid metabolism via enhancing autophagy. Stem Cell Res. Ther. 2020;11:223. doi: 10.1186/s13287-020-01731-6. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 45.Chelakkot C, et al. Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions. Exp. Mol. Med. 2018;50:e450. doi: 10.1038/emm.2017.282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gehrmann U, et al. Harnessing the exosome-induced immune response for cancer immunotherapy. Semin Cancer Biol. 2014;28:58–67. doi: 10.1016/j.semcancer.2014.05.003. [DOI] [PubMed] [Google Scholar]
- 47.Kurywchak P, Tavormina J, Kalluri R. The emerging roles of exosomes in the modulation of immune responses in cancer. Genome Med. 2018;10:23. doi: 10.1186/s13073-018-0535-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;7:367. doi: 10.1126/science.aau6977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li W, Jin L-Y, Cui Y-B, Xie N. Human umbilical cord mesenchymal stem cells-derived exosomal microRNA-17-3p ameliorates inflammatory reaction and antioxidant injury of mice with diabetic retinopathy via targeting STAT1. Int. Immunopharmacol. 2021;90:107010. doi: 10.1016/j.intimp.2020.107010. [DOI] [PubMed] [Google Scholar]
- 50.Venkat P, et al. CD133+ exosome treatment improves cardiac function after stroke in type 2 diabetic mice. Transl. stroke Res. 2021;12:112–124. doi: 10.1007/s12975-020-00807-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Govindappa PK, et al. Targeting exosome-associated human antigen R attenuates fibrosis and inflammation in diabetic heart. FASEB J.: 2020;34:2238–2251. doi: 10.1096/fj.201901995R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Li W, et al. Bone marrow mesenchymal stem cells-induced exosomal microRNA-486-3p protects against diabetic retinopathy through TLR4/NF-?B axis repression. J. Endocrinol. Invest. 2020;44:1193–1207. doi: 10.1007/s40618-020-01405-3. [DOI] [PubMed] [Google Scholar]
- 53.Gu C, Zhang H, Gao Y. Adipose mesenchymal stem cells-secreted extracellular vesicles containing microRNA-192 delays diabetic retinopathy by targeting ITGA1. J. Cell. Physiol. 2020;236:5036–5051. doi: 10.1002/jcp.30213. [DOI] [PubMed] [Google Scholar]
- 54.Cui X, et al. Circulating exosomes activate dendritic cells and induce unbalanced CD4+ T cell differentiation in hashimoto thyroiditis. J. Clin. Endocrinol. Metab. 2019;104:4607–4618. doi: 10.1210/jc.2019-00273. [DOI] [PubMed] [Google Scholar]
- 55.Zitvogel L, et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 1998;4:594–600. doi: 10.1038/nm0598-594. [DOI] [PubMed] [Google Scholar]
- 56.Cheng Y, Schorey JS. Exosomes carrying mycobacterial antigens can protect mice against Mycobacterium tuberculosis infection. Eur. J. Immunol. 2013;43:3279–3290. doi: 10.1002/eji.201343727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ding G, et al. Pancreatic cancer-derived exosomes transfer miRNAs to dendritic cells and inhibit RFXAP expression via miR-212-3p. Oncotarget. 2015;6:29877–29888. doi: 10.18632/oncotarget.4924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pegtel DM, et al. Functional delivery of viral miRNAs via exosomes. Proc. Natl Acad. Sci. USA. 2010;107:6328–6333. doi: 10.1073/pnas.0914843107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dini L, et al. Microvesicles and exosomes in metabolic diseases and inflammation. Cytokine Growth Factor Rev. 2020;51:27–39. doi: 10.1016/j.cytogfr.2019.12.008. [DOI] [PubMed] [Google Scholar]
- 60.Kranendonk MEG, et al. Human adipocyte extracellular vesicles in reciprocal signaling between adipocytes and macrophages. Obes. (Silver Spring, Md.) 2014;22:1296–1308. doi: 10.1002/oby.20679. [DOI] [PubMed] [Google Scholar]
- 61.Hirsova P, et al. Lipid-induced signaling causes release of inflammatory extracellular vesicles from hepatocytes. Gastroenterology. 2016;150:956–967. doi: 10.1053/j.gastro.2015.12.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Song M, et al. Adipocyte-derived exosomes carrying sonic hedgehog mediate M1 macrophage polarization-induced insulin resistance via Ptch and PI3K pathways. Cell. Physiol. Biochem. 2018;48:1416–1432. doi: 10.1159/000492252. [DOI] [PubMed] [Google Scholar]
- 63.Deng Z-b, et al. Adipose tissue exosome-like vesicles mediate activation of macrophage-induced insulin resistance. Diabetes. 2009;58:2498–2505. doi: 10.2337/db09-0216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Xie Z, et al. Adipose-derived exosomes exert proatherogenic effects by regulating macrophage foam cell formation and polarization. J. Am. Heart Assoc. 2018;7:e007442. doi: 10.1161/JAHA.117.007442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Xiao M, Zhang J, Chen W, Chen W. M1-like tumor-associated macrophages activated by exosome-transferred THBS1 promote malignant migration in oral squamous cell carcinoma. J. Exp. Clin. Cancer Res. 2018;37:143. doi: 10.1186/s13046-018-0815-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhao H, et al. Exosomes from adipose-derived stem cells attenuate adipose inflammation and obesity through polarizing M2 Macrophages and beiging in white adipose tissue. Diabetes. 2018;67:235–247. doi: 10.2337/db17-0356. [DOI] [PubMed] [Google Scholar]
- 67.Li J, et al. Exosomes derived from mesenchymal stem cells attenuate the progression of atherosclerosis in ApoE(−/−) mice via miR-let7 mediated infiltration and polarization of M2 macrophage. Biochem. Biophys. Res. Commun. 2019;510:565–572. doi: 10.1016/j.bbrc.2019.02.005. [DOI] [PubMed] [Google Scholar]
- 68.Zhao J, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019;115:1205–1216. doi: 10.1093/cvr/cvz040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Deng S, et al. Exosomes from adipose-derived mesenchymal stem cells ameliorate cardiac damage after myocardial infarction by activating S1P/SK1/S1PR1 signaling and promoting macrophage M2 polarization. Int. J. Biochem Cell Biol. 2019;114:105564. doi: 10.1016/j.biocel.2019.105564. [DOI] [PubMed] [Google Scholar]
- 70.López-Janeiro Á, et al. Prognostic value of macrophage polarization markers in epithelial neoplasms and melanoma. A systematic review and meta-analysis. Mod. Pathol. 2020;33:1458–1465. doi: 10.1038/s41379-020-0534-z. [DOI] [PubMed] [Google Scholar]
- 71.Wang LP, et al. Exosomal DLX6-AS1 from hepatocellular carcinoma cells induces M2 macrophage polarization to promote migration and invasion in hepatocellular carcinoma through microRNA-15a-5p/CXCL17 axis. J. Exp. Clin. Cancer Res. 2021;40:177. doi: 10.1186/s13046-021-01973-z. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 72.Zhang X, et al. Tumor-derived exosomes induce N2 polarization of neutrophils to promote gastric cancer cell migration. Mol. Cancer. 2018;17:146. doi: 10.1186/s12943-018-0898-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jiang M, et al. Keratinocyte exosomes activate neutrophils and enhance skin inflammation in psoriasis. FASEB J. 2019;33:13241–13253. doi: 10.1096/fj.201900642R. [DOI] [PubMed] [Google Scholar]
- 74.Piccard H, Muschel RJ, Opdenakker G. On the dual roles and polarized phenotypes of neutrophils in tumor development and progression. Crit. Rev. Oncol. Hematol. 2012;82:296–309. doi: 10.1016/j.critrevonc.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 75.Yao J, et al. Extracellular vesicles derived from human umbilical cord mesenchymal stem cells alleviate rat hepatic ischemia-reperfusion injury by suppressing oxidative stress and neutrophil inflammatory response. FASEB J. 2019;33:1695–1710. doi: 10.1096/fj.201800131RR. [DOI] [PubMed] [Google Scholar]
- 76.Liu J, et al. Clinical Potential of Extracellular Vesicles in Type 2 Diabetes. Front. Endocrinol. 2020;11:596811. doi: 10.3389/fendo.2020.596811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Silva ND, Samblas M, Martínez JA, Milagro FI. Effects of exosomes from LPS-activated macrophages on adipocyte gene expression, differentiation, and insulin-dependent glucose uptake. J. Physiol. Biochem. 2018;74:559–568. doi: 10.1007/s13105-018-0622-4. [DOI] [PubMed] [Google Scholar]
- 78.Castaño C, Kalko S, Novials A, Párrizas M. Obesity-associated exosomal miRNAs modulate glucose and lipid metabolism in mice. Proc. Natl Acad. Sci. USA. 2018;115:12158–12163. doi: 10.1073/pnas.1808855115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ding X, et al. MiR-21-5p in macrophage-derived extracellular vesicles affects podocyte pyroptosis in diabetic nephropathy by regulating A20. J. Endocrinol. Invest. 2020;44:1175–1184. doi: 10.1007/s40618-020-01401-7. [DOI] [PubMed] [Google Scholar]
- 80.Fabbri M, et al. MicroRNAs bind to Toll-like receptors to induce prometastatic inflammatory response. Proc. Natl Acad. Sci. USA. 2012;109:E2110–E2116. doi: 10.1073/pnas.1209414109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shi X, et al. Paeonol inhibits NLRP3 mediated inflammation in rat endothelial cells by elevating hyperlipidemic rats plasma exosomal miRNA-223. Eur.J, Pharmacol. 2020;885:173473. doi: 10.1016/j.ejphar.2020.173473. [DOI] [PubMed] [Google Scholar]
- 82.Valencia K, et al. miRNA cargo within exosome-like vesicle transfer influences metastatic bone colonization. Mol. Oncol. 2014;8:689–703. doi: 10.1016/j.molonc.2014.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Rani V, et al. Oxidative stress and metabolic disorders: pathogenesis and therapeutic strategies. Life Sci. 2016;148:183–193. doi: 10.1016/j.lfs.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 84.Zhang C, et al. Exosomes, the message transporters in vascular calcification. J. Cell. Mol. Med. 2018;22:4024–4033. doi: 10.1111/jcmm.13692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wen Z, et al. Hypertrophic adipocyte-derived exosomal miR-802-5p contributes to insulin resistance in cardiac myocytes through targeting HSP60. Obes. (Silver Spring, Md.). 2020;28:1932–1940. doi: 10.1002/oby.22932. [DOI] [PubMed] [Google Scholar]
- 86.Varela-López, A. et al. Age-related loss in bone mineral density of rats fed lifelong on a fish oil-based diet is avoided by coenzyme Q10 addition. Nutrients. 9, 176 (2017). [DOI] [PMC free article] [PubMed]
- 87.Xie Y, et al. Involvement of serum-derived exosomes of elderly patients with bone loss in failure of bone remodeling via alteration of exosomal bone-related proteins. Aging Cell. 2018;17:e12758. doi: 10.1111/acel.12758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Wang Y, et al. Exosomal circHIPK3 released from hypoxia-pretreated cardiomyocytes regulates oxidative damage in cardiac microvascular endothelial cells via the miR-29a/IGF-1 Pathway. Oxid. Med Cell Longev. 2019;2019:7954657. doi: 10.1155/2019/7954657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Xu, J., Camfield, R. & Gorski, S. M. The interplay between exosomes and autophagy—partners in crime. J. Cell Sci. 131, jcs215210 (2018). [DOI] [PubMed]
- 90.Wang F, Jia J, Rodrigues B. Autophagy, metabolic disease, and pathogenesis of heart dysfunction. Can J. Cardiol. 2017;33:850–859. doi: 10.1016/j.cjca.2017.01.002. [DOI] [PubMed] [Google Scholar]
- 91.Xu, J., Camfield, R. & Gorski, S. M. The interplay between exosomes and autophagy—partners in crime. J. Cell Sci. 131, jcs215210 (2018). [DOI] [PubMed]
- 92.Villarroya-Beltri, C. et al. ISGylation controls exosome secretion by promoting lysosomal degradation of MVB proteins. Nat. Commun. 7, 13588 (2016). [DOI] [PMC free article] [PubMed]
- 93.Kumar D, Gupta D, Shankar S, Srivastava RK. Biomolecular characterization of exosomes released from cancer stem cells: Possible implications for biomarker and treatment of cancer. Oncotarget. 2015;6:3280–3291. doi: 10.18632/oncotarget.2462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Qi M, et al. Autophagy maintains the function of bone marrow mesenchymal stem cells to prevent estrogen deficiency-induced osteoporosis. Theranostics. 2017;7:4498–4516. doi: 10.7150/thno.17949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Liu H, et al. Intermittent fasting preserves beta-cell mass in obesity-induced diabetes via the autophagy-lysosome pathway. Autophagy. 2017;13:1952–1968. doi: 10.1080/15548627.2017.1368596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yang L, et al. Defective hepatic autophagy in obesity promotes ER stress and causes insulin resistance. Cell Metab. 2010;11:467–478. doi: 10.1016/j.cmet.2010.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Li F, et al. Adipose-specific knockdown of Sirt1 results in obesity and insulin resistance by promoting exosomes release. Cell Cycle (Georget., Tex.) 2019;18:2067–2082. doi: 10.1080/15384101.2019.1638694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Hu J, et al. Exosomal Mst1 transfer from cardiac microvascular endothelial cells to cardiomyocytes deteriorates diabetic cardiomyopathy. Biochim. Biophys. Acta Mol. Basis Dis. 2018;1864:3639–3649. doi: 10.1016/j.bbadis.2018.08.026. [DOI] [PubMed] [Google Scholar]
- 99.Shi R, et al. Exosomes derived from mmu_circ_0000250-modified adipose-derived mesenchymal stem cells promote wound healing in diabetic mice by inducing miR-128-3p/SIRT1-mediated autophagy. Am. J. Physiol. Cell Physiol. 2020;318:C848–C856. doi: 10.1152/ajpcell.00041.2020. [DOI] [PubMed] [Google Scholar]
- 100.Jin J, et al. Exosome secreted from adipose-derived stem cells attenuates diabetic nephropathy by promoting autophagy flux and inhibiting apoptosis in podocyte. Stem Cell Res. Ther. 2019;10:95. doi: 10.1186/s13287-019-1177-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ebrahim, N. et al. Mesenchymal stem cell-derived exosomes ameliorated diabetic nephropathy by autophagy induction through the mTOR signaling pathway. Cells. 7, (2018). [DOI] [PMC free article] [PubMed]
- 102.Huang H, et al. M2 macrophage-derived exosomal miR-25-3p improves high glucose-induced podocytes injury through activation autophagy via inhibiting DUSP1 expression. IUBMB life. 2020;72:2651–2662. doi: 10.1002/iub.2393. [DOI] [PubMed] [Google Scholar]
- 103.Yang Y, et al. Secretory carrier membrane protein 5 is an autophagy inhibitor that promotes the secretion of α-synuclein via exosome. PLoS ONE. 2017;12:e0180892. doi: 10.1371/journal.pone.0180892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wang B, et al. Exosomal miR-1910-3p promotes proliferation, metastasis, and autophagy of breast cancer cells by targeting MTMR3 and activating the NF-κB signaling pathway. Cancer Lett. 2020;489:87–99. doi: 10.1016/j.canlet.2020.05.038. [DOI] [PubMed] [Google Scholar]
- 105.Colletti M, Ceglie D, Di Giannatale A, Nazio F. Autophagy and exosomes relationship in cancer: friends or foes? Front. Cell Dev. Biol. 2020;8:614178. doi: 10.3389/fcell.2020.614178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Yao W, Guo P, Mu Q, Wang Y. Exosome-derived Circ-PVT1 contributes to cisplatin resistance by regulating autophagy, invasion, and apoptosis via miR-30a-5p/YAP1 axis in gastric cancer cells. Cancer Biother. Radiopharm. 2021;36:347–359. doi: 10.1089/cbr.2020.3578. [DOI] [PubMed] [Google Scholar]
- 107.Zhang YJ, et al. Impacts of gut bacteria on human health and diseases. Int J. Mol. Sci. 2015;16:7493–7519. doi: 10.3390/ijms16047493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 2021;19:55–71. doi: 10.1038/s41579-020-0433-9. [DOI] [PubMed] [Google Scholar]
- 109.Baothman OA, et al. The role of gut microbiota in the development of obesity and Diabetes. Lipids Health Dis. 2016;15:108. doi: 10.1186/s12944-016-0278-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Gopalakrishnan V, et al. The influence of the gut microbiome on cancer, immunity, and cancer immunotherapy. Cancer Cell. 2018;33:570–580. doi: 10.1016/j.ccell.2018.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Liu S, et al. The host shapes the gut microbiota via fecal microRNA. Cell host microbe. 2016;19:32–43. doi: 10.1016/j.chom.2015.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Ashrafian F, et al. Akkermansia muciniphila-derived extracellular vesicles as a mucosal delivery vector for amelioration of obesity in mice. Front. Microbiol. 2019;10:2155. doi: 10.3389/fmicb.2019.02155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Choi Y, et al. Gut microbe-derived extracellular vesicles induce insulin resistance, thereby impairing glucose metabolism in skeletal muscle. Sci. Rep. 2015;5:15878. doi: 10.1038/srep15878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Luo Z, et al. CRIg+ Macrophages prevent gut microbial DNA-containing extracellular vesicle-induced tissue inflammation and insulin resistance. Gastroenterology. 2021;160:863–874. doi: 10.1053/j.gastro.2020.10.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Liu, J. H. et al. Extracellular vesicles from child gut microbiota enter into bone to preserve bone mass and strength. Adv. Sci.8, 2004831 (2021). [DOI] [PMC free article] [PubMed]
- 116.Williams GH, Stoeber K. The cell cycle and cancer. J. Pathol. 2012;226:352–364. doi: 10.1002/path.3022. [DOI] [PubMed] [Google Scholar]
- 117.Dai X, et al. Exosomal circRNA_100284 from arsenite-transformed cells, via microRNA-217 regulation of EZH2, is involved in the malignant transformation of human hepatic cells by accelerating the cell cycle and promoting cell proliferation. Cell Death Dis. 2018;9:454. doi: 10.1038/s41419-018-0485-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Pan L, et al. Exosomes-mediated transfer of long noncoding RNA ZFAS1 promotes gastric cancer progression. J. Cancer Res Clin. Oncol. 2017;143:991–1004. doi: 10.1007/s00432-017-2361-2. [DOI] [PubMed] [Google Scholar]
- 119.Reza A, Choi YJ, Yasuda H, Kim JH. Human adipose mesenchymal stem cell-derived exosomal-miRNAs are critical factors for inducing anti-proliferation signalling to A2780 and SKOV-3 ovarian cancer cells. Sci. Rep. 2016;6:38498. doi: 10.1038/srep38498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Arroyo JD, et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl Acad. Sci. USA. 2011;108:5003–5008. doi: 10.1073/pnas.1019055108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Wang K, et al. Export of microRNAs and microRNA-protective protein by mammalian cells. Nucleic Acids Res. 2010;38:7248–7259. doi: 10.1093/nar/gkq601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Tosar JP, Witwer K, Cayota A. Revisiting extracellular RNA release, processing, and function. Trends Biochem Sci. 2021;46:438–445. doi: 10.1016/j.tibs.2020.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Vickers KC, et al. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat. Cell Biol. 2011;13:423–433. doi: 10.1038/ncb2210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Tosar JP, et al. Fragmentation of extracellular ribosomes and tRNAs shapes the extracellular RNAome. Nucleic Acids Res. 2020;48:12874–12888. doi: 10.1093/nar/gkaa674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Nechooshtan G, Yunusov D, Chang K, Gingeras TR. Processing by RNase 1 forms tRNA halves and distinct Y RNA fragments in the extracellular environment. Nucleic Acids Res. 2020;48:8035–8049. doi: 10.1093/nar/gkaa526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Martínez MC, Andriantsitohaina R. Extracellular vesicles in metabolic syndrome. Circulation Res. 2017;120:1674–1686. doi: 10.1161/CIRCRESAHA.117.309419. [DOI] [PubMed] [Google Scholar]
- 127.Muraca, M. & Cappariello, A. The role of extracellular vesicles (EVs) in the epigenetic regulation of bone metabolism and osteoporosis. Int. J. Mol. Sci. 21, 8682 (2020). [DOI] [PMC free article] [PubMed]
- 128.Prattichizzo F, et al. Extracellular vesicle-shuttled miRNAs: a critical appraisal of their potential as nano-diagnostics and nano-therapeutics in type 2 diabetes mellitus and its cardiovascular complications. Theranostics. 2021;11:1031–1045. doi: 10.7150/thno.51605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Wu P, et al. Emerging role of tumor-related functional peptides encoded by lncRNA and circRNA. Mol. cancer. 2020;19:22. doi: 10.1186/s12943-020-1147-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Torre LA, et al. Global cancer statistics, 2012. CA: Cancer J. Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 131.Xie Y, et al. The role of exosomal noncoding RNAs in cancer. Mol. cancer. 2019;18:37. doi: 10.1186/s12943-019-0984-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Hu C, et al. Role of exosomal microRNAs in lung cancer biology and clinical applications. Cell Prolif. 2020;53:e12828. doi: 10.1111/cpr.12828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.He Y, et al. Exosomal microRNA: a novel biomarker for breast cancer. Biomark. Med. 2018;12:177–188. doi: 10.2217/bmm-2017-0305. [DOI] [PubMed] [Google Scholar]
- 134.Li S, et al. Exosomal miRNAs in hepatocellular carcinoma development and clinical responses. J. Hematol. Oncol. 2018;11:54. doi: 10.1186/s13045-018-0579-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Molina JR, et al. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin. Proc. 2008;83:584–594. doi: 10.4065/83.5.584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 137.Kim J, et al. Exosome cargo reflects TGF-β1-mediated epithelial-to-mesenchymal transition (EMT) status in A549 human lung adenocarcinoma cells. Biochem. Biophys. Res. Commun. 2016;478:643–648. doi: 10.1016/j.bbrc.2016.07.124. [DOI] [PubMed] [Google Scholar]
- 138.Zhang X, et al. Hypoxic BMSC-derived exosomal miRNAs promote metastasis of lung cancer cells via STAT3-induced EMT. Mol. Cancer. 2019;18:40. doi: 10.1186/s12943-019-0959-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.He S, et al. Exosomal miR-499a-5p promotes cell proliferation, migration and EMT via mTOR signaling pathway in lung adenocarcinoma. Exp. Cell Res. 2019;379:203–213. doi: 10.1016/j.yexcr.2019.03.035. [DOI] [PubMed] [Google Scholar]
- 140.Patterson JW. Recent advances in dermatopathology. Va. Med. 1988;115:572–577. [PubMed] [Google Scholar]
- 141.Wu H, et al. Circulating exosomal microRNA-96 promotes cell proliferation, migration and drug resistance by targeting LMO7. J. Cell. Mol. Med. 2017;21:1228–1236. doi: 10.1111/jcmm.13056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Cheng G, et al. Targeting lonidamine to mitochondria mitigates lung tumorigenesis and brain metastasis. Nat. Commun. 2019;10:2205. doi: 10.1038/s41467-019-10042-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Wei F, et al. Correction to: Exosomes derived from gemcitabine resistant cells transfer malignant phenotypic traits via delivery of miRNA-222-3p. Mol. Cancer. 2021;20:35. doi: 10.1186/s12943-021-01320-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Grimolizzi F, et al. Exosomal miR-126 as a circulating biomarker in non-small-cell lung cancer regulating cancer progression. Sci. Rep. 2017;7:15277. doi: 10.1038/s41598-017-15475-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Tang Y, et al. Erratum to: Radiation-induced miR-208a increases the proliferation and radioresistance by targeting p21 in human lung cancer cells. J. Exp. Clin. Cancer Res. 2016;35:20. doi: 10.1186/s13046-016-0299-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Adi Harel S, et al. Reactivation of epigenetically silenced miR-512 and miR-373 sensitizes lung cancer cells to cisplatin and restricts tumor growth. Cell Death Differ. 2015;22:1328–1340. doi: 10.1038/cdd.2014.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002;29:15–18. doi: 10.1053/sonc.2002.37263. [DOI] [PubMed] [Google Scholar]
- 148.Zhuang G, et al. Tumour-secreted miR-9 promotes endothelial cell migration and angiogenesis by activating the JAK-STAT pathway. EMBO J. 2012;31:3513–3523. doi: 10.1038/emboj.2012.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Liu Y, et al. STAT3-regulated exosomal miR-21 promotes angiogenesis and is involved in neoplastic processes of transformed human bronchial epithelial cells. Cancer Lett. 2016;370:125–135. doi: 10.1016/j.canlet.2015.10.011. [DOI] [PubMed] [Google Scholar]
- 150.Hsu YL, et al. Hypoxic lung cancer-secreted exosomal miR-23a increased angiogenesis and vascular permeability by targeting prolyl hydroxylase and tight junction protein ZO-1. Oncogene. 2017;36:4929–4942. doi: 10.1038/onc.2017.105. [DOI] [PubMed] [Google Scholar]
- 151.Cui H, et al. Tissue inhibitor of metalloproteinases-1 induces a pro-tumourigenic increase of miR-210 in lung adenocarcinoma cells and their exosomes. Oncogene. 2015;34:3640–3650. doi: 10.1038/onc.2014.300. [DOI] [PubMed] [Google Scholar]
- 152.Xu Z, et al. Lung adenocarcinoma cell-derived exosomal miR-21 facilitates osteoclastogenesis. Gene. 2018;666:116–122. doi: 10.1016/j.gene.2018.05.008. [DOI] [PubMed] [Google Scholar]
- 153.Poulet C, et al. Exosomal long non-coding RNAs in lung diseases. Int. J. Mol. Sci. 2020;21:3580. doi: 10.3390/ijms21103580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Li C, et al. Tumor-derived exosomal lncRNA GAS5 as a biomarker for early-stage non-small-cell lung cancer diagnosis. J. Cell. Physiol. 2019;234:20721–20727. doi: 10.1002/jcp.28678. [DOI] [PubMed] [Google Scholar]
- 155.Teng Y, Kang H, Chu Y. Identification of an exosomal long noncoding RNA SOX2-OT in plasma as a promising biomarker for lung squamous cell carcinoma. Genet. Test. Mol. Biomark. 2019;23:235–240. doi: 10.1089/gtmb.2018.0103. [DOI] [PubMed] [Google Scholar]
- 156.Zhang N, et al. Circular RNA circSATB2 promotes progression of non-small cell lung cancer cells. Mol. cancer. 2020;19:101. doi: 10.1186/s12943-020-01221-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Xu X, Tao R, Sun L, Ji X. Exosome-transferred hsa_circ_0014235 promotes DDP chemoresistance and deteriorates the development of non-small cell lung cancer by mediating the miR-520a-5p/CDK4 pathway. Cancer Cell Int. 2020;20:552. doi: 10.1186/s12935-020-01642-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Wang J, et al. circRNA-002178 act as a ceRNA to promote PDL1/PD1 expression in lung adenocarcinoma. Cell Death Dis. 2020;11:32. doi: 10.1038/s41419-020-2230-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Xian J, et al. Identification of three circular RNA cargoes in serum exosomes as diagnostic biomarkers of non-small-cell lung cancer in the Chinese population. J. Mol. Diagnostics. 2020;22:1096–1108. doi: 10.1016/j.jmoldx.2020.05.011. [DOI] [PubMed] [Google Scholar]
- 160.Shi, Q. et al. Serum exosomes-based biomarker circ_0008928 Regulates cisplatin sensitivity, tumor progression, and glycolysis metabolism by miR-488/HK2 axis in cisplatin-resistant nonsmall cell lung carcinoma. Cancer Biother. Radiopharm.10.1089/cbr.2020.4490 (2021). [DOI] [PubMed]
- 161.Yang B, et al. Tumor-derived exosomal circRNA_102481 contributes to EGFR-TKIs resistance via the miR-30a-5p/ROR1 axis in non-small cell lung cancer. Aging. 2021;13:13264–13286. doi: 10.18632/aging.203011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ding C, et al. Exosomal Circ-MEMO1 promotes the progression and aerobic glycolysis of non-small cell lung cancer through targeting MiR-101-3p/KRAS axis. Front. Genet. 2020;11:962. doi: 10.3389/fgene.2020.00962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Fang, K. et al. Serum-derived exosomes-mediated circular RNA ARHGAP10 modulates the progression of non-small-cell lung cancer through the miR-638/FAM83F Axis. Cancer Biother. Radiopharm.10.1089/cbr.2019.3534 (2020). [DOI] [PubMed]
- 164.Shao N, Song L, Sun X. Exosomal circ_PIP5K1A regulates the progression of non-small cell lung cancer and cisplatin sensitivity by miR-101/ABCC1 axis. Mol. Cell Biochem. 2021;476:2253–2267. doi: 10.1007/s11010-021-04083-8. [DOI] [PubMed] [Google Scholar]
- 165.Chen T, et al. Tumor-derived exosomal circFARSA mediates M2 macrophage polarization via the PTEN/PI3K/AKT pathway to promote non-small cell lung cancer metastasis. Cancer Treat. Res. Commun. 2021;28:100412. doi: 10.1016/j.ctarc.2021.100412. [DOI] [PubMed] [Google Scholar]
- 166.Chen F, et al. Circular RNAs expression profiles in plasma exosomes from early-stage lung adenocarcinoma and the potential biomarkers. J. Cell. Biochem. 2020;121:2525–2533. doi: 10.1002/jcb.29475. [DOI] [PubMed] [Google Scholar]
- 167.Harbeck N, et al. Breast cancer. Nat. Rev. Dis. Prim. 2019;5:66. doi: 10.1038/s41572-019-0111-2. [DOI] [PubMed] [Google Scholar]
- 168.Bray F, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J. Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 169.Liu Q, Peng F, Chen J. The role of exosomal MicroRNAs in the tumor microenvironment of breast cancer. Int. J. Mol. Sci. 2019;20:3884. doi: 10.3390/ijms20163884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Singh R, et al. Exosome-mediated transfer of miR-10b promotes cell invasion in breast cancer. Mol. cancer. 2014;13:256. doi: 10.1186/1476-4598-13-256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Li XJ, Ren ZJ, Tang JH, Yu Q. Exosomal microRNA MiR-1246 promotes cell proliferation, invasion and drug resistance by targeting CCNG2 in breast cancer. Cell. Physiol. Biochem. 2017;44:1741–1748. doi: 10.1159/000485780. [DOI] [PubMed] [Google Scholar]
- 172.O’Brien K, et al. miR-134 in extracellular vesicles reduces triple-negative breast cancer aggression and increases drug sensitivity. Oncotarget. 2015;6:32774–32789. doi: 10.18632/oncotarget.5192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Kong X, et al. MiR-130a-3p inhibits migration and invasion by regulating RAB5B in human breast cancer stem cell-like cells. Biochem. Biophys. Res. Commun. 2018;501:486–493. doi: 10.1016/j.bbrc.2018.05.018. [DOI] [PubMed] [Google Scholar]
- 174.Baroni S, et al. Exosome-mediated delivery of miR-9 induces cancer-associated fibroblast-like properties in human breast fibroblasts. Cell death Dis. 2016;7:e2312. doi: 10.1038/cddis.2016.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Wu HJ, Hao M, Yeo SK, Guan JL. FAK signaling in cancer-associated fibroblasts promotes breast cancer cell migration and metastasis by exosomal miRNAs-mediated intercellular communication. Oncogene. 2020;39:2539–2549. doi: 10.1038/s41388-020-1162-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Donnarumma E, et al. Cancer-associated fibroblasts release exosomal microRNAs that dictate an aggressive phenotype in breast cancer. Oncotarget. 2017;8:19592–19608. doi: 10.18632/oncotarget.14752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Kim KS, et al. ELK3 expressed in lymphatic endothelial cells promotes breast cancer progression and metastasis through exosomal miRNAs. Sci. Rep. 2019;9:8418. doi: 10.1038/s41598-019-44828-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Zhou W, et al. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer cell. 2014;25:501–515. doi: 10.1016/j.ccr.2014.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Fong MY, et al. Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat. cell Biol. 2015;17:183–194. doi: 10.1038/ncb3094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Le MT, et al. miR-200-containing extracellular vesicles promote breast cancer cell metastasis. J. Clin. Investig. 2014;124:5109–5128. doi: 10.1172/JCI75695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Di Modica M, et al. Breast cancer-secreted miR-939 downregulates VE-cadherin and destroys the barrier function of endothelial monolayers. Cancer Lett. 2017;384:94–100. doi: 10.1016/j.canlet.2016.09.013. [DOI] [PubMed] [Google Scholar]
- 182.Hashimoto K, et al. Cancer-secreted hsa-miR-940 induces an osteoblastic phenotype in the bone metastatic microenvironment via targeting ARHGAP1 and FAM134A. Proc. Natl Acad. Sci. USA. 2018;115:2204–2209. doi: 10.1073/pnas.1717363115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Guo L, et al. Breast cancer cell-derived exosomal miR-20a-5p promotes the proliferation and differentiation of osteoclasts by targeting SRCIN1. Cancer Med. 2019;8:5687–5701. doi: 10.1002/cam4.2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Ono M, et al. Exosomes from bone marrow mesenchymal stem cells contain a microRNA that promotes dormancy in metastatic breast cancer cells. Sci. Signal. 2014;7:ra63. doi: 10.1126/scisignal.2005231. [DOI] [PubMed] [Google Scholar]
- 185.Kosaka N, et al. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J. Biol. Chem. 2013;288:10849–10859. doi: 10.1074/jbc.M112.446831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Lee JK, et al. Exosomes derived from mesenchymal stem cells suppress angiogenesis by down-regulating VEGF expression in breast cancer cells. PLoS ONE. 2013;8:e84256. doi: 10.1371/journal.pone.0084256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Pakravan K, et al. MicroRNA-100 shuttled by mesenchymal stem cell-derived exosomes suppresses in vitro angiogenesis through modulating the mTOR/HIF-1α/VEGF signaling axis in breast cancer cells. Cell. Oncol. (Dordr.) 2017;40:457–470. doi: 10.1007/s13402-017-0335-7. [DOI] [PubMed] [Google Scholar]
- 188.Zhang P, et al. Exosome-mediated delivery of MALAT1 induces cell proliferation in breast cancer. OncoTargets Ther. 2018;11:291–299. doi: 10.2147/OTT.S155134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Lu Y, Chen L, Li L, Cao Y. Exosomes derived from brain metastatic breast cancer cells destroy the blood-brain barrier by carrying lncRNA GS1-600G8.5. Biomed. Res. Int. 2020;2020:7461727. doi: 10.1155/2020/7461727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Zhong G, et al. Determination of serum exosomal H19 as a noninvasive biomarker for breast cancer diagnosis. OncoTargets Ther. 2020;13:2563–2571. doi: 10.2147/OTT.S243601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Na-Er A, Xu YY, Liu YH, Gan YJ. Upregulation of serum exosomal SUMO1P3 predicts unfavorable prognosis in triple negative breast cancer. Eur. Rev. Med. Pharmacol. Sci. 2021;25:154–160. doi: 10.26355/eurrev_202101_24379. [DOI] [PubMed] [Google Scholar]
- 192.Lan F, et al. Serum exosomal lncRNA XIST is a potential non-invasive biomarker to diagnose recurrence of triple-negative breast cancer. J. Cell. Mole. Med. 2021;25:7602–7607. doi: 10.1111/jcmm.16009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Tang S, et al. Overexpression of serum exosomal HOTAIR is correlated with poor survival and poor response to chemotherapy in breast cancer patients. J. Biosci. 2019;44:37. [PubMed] [Google Scholar]
- 194.Wang YL, et al. Long non-coding RNA HOTAIR in circulatory exosomes is correlated with ErbB2/HER2 positivity in breast cancer. Breast (Edinb., Scotl.) 2019;46:64–69. doi: 10.1016/j.breast.2019.05.003. [DOI] [PubMed] [Google Scholar]
- 195.Yang SJ, et al. Tumor-derived exosomal circPSMA1 facilitates the tumorigenesis, metastasis, and migration in triple-negative breast cancer (TNBC) through miR-637/Akt1/β-catenin (cyclin D1) axis. Cell Death Dis. 2021;12:420. doi: 10.1038/s41419-021-03680-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Zhan Y, et al. Carcinoma-associated fibroblasts derived exosomes modulate breast cancer cell stemness through exonic circHIF1A by miR-580-5p in hypoxic stress. Cell Death Discov. 2021;7:141. doi: 10.1038/s41420-021-00506-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Chen T, et al. CircHIF1A regulated by FUS accelerates triple-negative breast cancer progression by modulating NFIB expression and translocation. Oncogene. 2021;40:2756–2771. doi: 10.1038/s41388-021-01739-z. [DOI] [PubMed] [Google Scholar]
- 198.Wang J, et al. Circular RNA expression in exosomes derived from breast cancer cells and patients. Epigenomics. 2019;11:411–421. doi: 10.2217/epi-2018-0111. [DOI] [PubMed] [Google Scholar]
- 199.Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet (Lond., Engl.) 2012;379:1245–1255. doi: 10.1016/S0140-6736(11)61347-0. [DOI] [PubMed] [Google Scholar]
- 200.Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet (Lond., Engl.) 2018;391:1301–1314. doi: 10.1016/S0140-6736(18)30010-2. [DOI] [PubMed] [Google Scholar]
- 201.Sorop A, et al. Exosomal microRNAs as biomarkers and therapeutic targets for hepatocellular carcinoma. Int. J. Mol. Sci. 2021;22:4997. doi: 10.3390/ijms22094997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Cui Y, et al. Mechanism of exosomal microRNA-224 in development of hepatocellular carcinoma and its diagnostic and prognostic value. World J. Gastroenterol. 2019;25:1890–1898. doi: 10.3748/wjg.v25.i15.1890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Xue X, et al. Exosomal miR-93 promotes proliferation and invasion in hepatocellular carcinoma by directly inhibiting TIMP2/TP53INP1/CDKN1A. Biochem. Biophys. Res. Commun. 2018;502:515–521. doi: 10.1016/j.bbrc.2018.05.208. [DOI] [PubMed] [Google Scholar]
- 204.Qu Z, et al. Exosomal miR-665 as a novel minimally invasive biomarker for hepatocellular carcinoma diagnosis and prognosis. Oncotarget. 2017;8:80666–80678. doi: 10.18632/oncotarget.20881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Fang T, et al. Tumor-derived exosomal miR-1247-3p induces cancer-associated fibroblast activation to foster lung metastasis of liver cancer. Nat. Commun. 2018;9:191. doi: 10.1038/s41467-017-02583-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Lin XJ, et al. Hepatocellular carcinoma cell-secreted exosomal microRNA-210 promotes angiogenesis in vitro and in vivo. Mol. Ther. Nucleic Acids. 2018;11:243–252. doi: 10.1016/j.omtn.2018.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Matsuura Y, et al. Exosomal miR-155 Derived from hepatocellular carcinoma cells under hypoxia promotes angiogenesis in endothelial cells. Digestive Dis. Sci. 2019;64:792–802. doi: 10.1007/s10620-018-5380-1. [DOI] [PubMed] [Google Scholar]
- 208.Lee YR, et al. Circulating exosomal noncoding RNAs as prognostic biomarkers in human hepatocellular carcinoma. Int. J. cancer. 2019;144:1444–1452. doi: 10.1002/ijc.31931. [DOI] [PubMed] [Google Scholar]
- 209.Wang H, et al. Expression of serum exosomal microRNA-21 in human hepatocellular carcinoma. Biomed. Res. Int. 2014;2014:864894. doi: 10.1155/2014/864894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Nakano T, et al. Circulating exosomal miR-92b: Its role for cancer immunoediting and clinical value for prediction of posttransplant hepatocellular carcinoma recurrence. Am. J. Transplant. 2019;19:3250–3262. doi: 10.1111/ajt.15490. [DOI] [PubMed] [Google Scholar]
- 211.Cho HJ, et al. Serum exosomal MicroRNA, miR-10b-5p, as a potential diagnostic biomarker for early-stage hepatocellular carcinoma. J. Clin. Med. 2020;9:281. doi: 10.3390/jcm9010281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Matboli M, et al. Exosomal miR-1298 and lncRNA-RP11-583F2.2 Expression in Hepato-cellular Carcinoma. Curr. genomics. 2020;21:46–55. doi: 10.2174/1389202920666191210111849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Tang J, et al. Exosomal miR-9-3p suppresses HBGF-5 expression and is a functional biomarker in hepatocellular carcinoma. Minerva Med. 2018;109:15–23. doi: 10.23736/S0026-4806.17.05167-9. [DOI] [PubMed] [Google Scholar]
- 214.Liu W, et al. Serum exosomal miR-125b is a novel prognostic marker for hepatocellular carcinoma. OncoTargets Ther. 2017;10:3843–3851. doi: 10.2147/OTT.S140062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Shi M, et al. Decreased levels of serum exosomal miR-638 predict poor prognosis in hepatocellular carcinoma. J. Cell. Biochem. 2018;119:4711–4716. doi: 10.1002/jcb.26650. [DOI] [PubMed] [Google Scholar]
- 216.Sugimachi K, et al. Identification of a bona fide microRNA biomarker in serum exosomes that predicts hepatocellular carcinoma recurrence after liver transplantation. Br. J. Cancer. 2015;112:532–538. doi: 10.1038/bjc.2014.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Sohn W, et al. Serum exosomal microRNAs as novel biomarkers for hepatocellular carcinoma. Exp. Mol. Med. 2015;47:e184. doi: 10.1038/emm.2015.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Wang G, et al. Exosomal MiR-744 inhibits proliferation and sorafenib chemoresistance in hepatocellular carcinoma by targeting PAX2. Med. Sci. Monit. 2019;25:7209–7217. doi: 10.12659/MSM.919219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Cao Q, et al. Exosomal long noncoding RNAs in aging and age-related diseases. IUBMB life. 2019;71:1846–1856. doi: 10.1002/iub.2141. [DOI] [PubMed] [Google Scholar]
- 220.Li B, et al. LncRNA FAL1 promotes cell proliferation and migration by acting as a CeRNA of miR-1236 in hepatocellular carcinoma cells. Life Sci. 2018;197:122–129. doi: 10.1016/j.lfs.2018.02.006. [DOI] [PubMed] [Google Scholar]
- 221.Wang D, et al. Exosomal lncRNA H19 promotes the progression of hepatocellular carcinoma treated with Propofol via miR-520a-3p/LIMK1 axis. Cancer Med. 2020;9:7218–7230. doi: 10.1002/cam4.3313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Conigliaro A, et al. CD90+ liver cancer cells modulate endothelial cell phenotype through the release of exosomes containing H19 lncRNA. Mol. Cancer. 2015;14:155. doi: 10.1186/s12943-015-0426-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Li X, Lei Y, Wu M, Li N. Regulation of macrophage activation and polarization by HCC-derived exosomal lncRNA TUC339. Int. J. Mol. Sci. 2018;19:2958. doi: 10.3390/ijms19102958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Takahashi K, et al. Involvement of extracellular vesicle long noncoding RNA (linc-VLDLR) in tumor cell responses to chemotherapy. Mol. cancer Res.: MCR. 2014;12:1377–1387. doi: 10.1158/1541-7786.MCR-13-0636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Takahashi K, et al. Extracellular vesicle-mediated transfer of long non-coding RNA ROR modulates chemosensitivity in human hepatocellular cancer. FEBS Open Bio. 2014;4:458–467. doi: 10.1016/j.fob.2014.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Ma D, et al. Exosome-transferred long non-coding RNA ASMTL-AS1 contributes to malignant phenotypes in residual hepatocellular carcinoma after insufficient radiofrequency ablation. Cell Prolif. 2020;53:e12795. doi: 10.1111/cpr.12795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Sun L, et al. Serum and exosome long non coding RNAs as potential biomarkers for hepatocellular carcinoma. J. Cancer. 2018;9:2631–2639. doi: 10.7150/jca.24978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Xu H, Chen Y, Dong X, Wang X. Serum exosomal long noncoding RNAs ENSG00000258332.1 and LINC00635 for the diagnosis and prognosis of hepatocellular carcinoma. Cancer Epidemiol., Biomark. Prev. 2018;27:710–716. doi: 10.1158/1055-9965.EPI-17-0770. [DOI] [PubMed] [Google Scholar]
- 229.Yao Z, et al. Serum exosomal long noncoding RNAs lnc-FAM72D-3 and lnc-EPC1-4 as diagnostic biomarkers for hepatocellular carcinoma. Aging. 2020;12:11843–11863. doi: 10.18632/aging.103355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Zhang C, et al. lncRNA-HEIH in serum and exosomes as a potential biomarker in the HCV-related hepatocellular carcinoma. Cancer Biomark. 2018;21:651–659. doi: 10.3233/CBM-170727. [DOI] [PubMed] [Google Scholar]
- 231.Wang J, et al. Exosome-transmitted long non-coding RNA SENP3-EIF4A1 suppresses the progression of hepatocellular carcinoma. Aging. 2020;12:11550–11567. doi: 10.18632/aging.103302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Zhuo C, et al. Exosomal linc-FAM138B from cancer cells alleviates hepatocellular carcinoma progression via regulating miR-765. Aging (Albany NY) 2020;12:26236–26247. doi: 10.18632/aging.202430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Huang XY, et al. Exosomal circRNA-100338 promotes hepatocellular carcinoma metastasis via enhancing invasiveness and angiogenesis. J. Exp. Clin. Cancer Res. 2020;39:20. doi: 10.1186/s13046-020-1529-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Su Y, et al. CircRNA Cdr1as functions as a competitive endogenous RNA to promote hepatocellular carcinoma progression. Aging. 2019;11:8183–8203. doi: 10.18632/aging.102312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Li Y, Zang H, Zhang X, Huang G. Exosomal Circ-ZNF652 promotes cell proliferation, migration, invasion and glycolysis in hepatocellular carcinoma via miR-29a-3p/GUCD1 axis. Cancer Manag. Res. 2020;12:7739–7751. doi: 10.2147/CMAR.S259424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Lai, Z. et al. Exosomal circFBLIM1 promotes hepatocellular carcinoma progression and glycolysis by regulating the miR-338/LRP6 axis. Cancer Biother. Radiopharm.10.1089/cbr.2020.3564 (2020). [DOI] [PubMed]
- 237.Zhu C, et al. Circular RNA hsa_circ_0004277 stimulates malignant phenotype of hepatocellular carcinoma and epithelial-mesenchymal transition of peripheral cells. Front. Cell Dev. Biol. 2020;8:585565. doi: 10.3389/fcell.2020.585565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Wang G, et al. Three isoforms of exosomal circPTGR1 promote hepatocellular carcinoma metastasis via the miR449a-MET pathway. EBioMedicine. 2019;40:432–445. doi: 10.1016/j.ebiom.2018.12.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Liu D, et al. Exosome-transmitted circ_MMP2 promotes hepatocellular carcinoma metastasis by upregulating MMP2. Mol. Oncol. 2020;14:1365–1380. doi: 10.1002/1878-0261.12637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Zhang PF, et al. Cancer cell-derived exosomal circUHRF1 induces natural killer cell exhaustion and may cause resistance to anti-PD1 therapy in hepatocellular carcinoma. Mol. Cancer. 2020;19:110. doi: 10.1186/s12943-020-01222-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Luo Y, Liu F, Gui R. High expression of circulating exosomal circAKT3 is associated with higher recurrence in HCC patients undergoing surgical treatment. Surgical Oncol. 2020;33:276–281. doi: 10.1016/j.suronc.2020.04.021. [DOI] [PubMed] [Google Scholar]
- 242.Sun XH, et al. Serum-derived three-circRNA signature as a diagnostic biomarker for hepatocellular carcinoma. Cancer Cell Int. 2020;20:226. doi: 10.1186/s12935-020-01302-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Yu Y, et al. Circular RNA hsa_circ_0061395 accelerates hepatocellular carcinoma progression via regulation of the miR-877-5p/PIK3R3 axis. Cancer cell Int. 2021;21:10. doi: 10.1186/s12935-020-01695-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Chen W, et al. Exosome-transmitted circular RNA hsa_circ_0051443 suppresses hepatocellular carcinoma progression. Cancer Lett. 2020;475:119–128. doi: 10.1016/j.canlet.2020.01.022. [DOI] [PubMed] [Google Scholar]
- 245.Wang Y, et al. Downregulation of hsa_circ_0074854 suppresses the migration and invasion in hepatocellular carcinoma via interacting with HuR and via suppressing exosomes-mediated macrophage M2 polarization. Int. J. Nanomed. 2021;16:2803–2818. doi: 10.2147/IJN.S284560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet (Lond., Engl.) 2014;383:1490–1502. doi: 10.1016/S0140-6736(13)61649-9. [DOI] [PubMed] [Google Scholar]
- 247.Zhang N, Hu X, Du Y, Du J. The role of miRNAs in colorectal cancer progression and chemoradiotherapy. Biomed. Pharmacother. . 2021;134:111099. doi: 10.1016/j.biopha.2020.111099. [DOI] [PubMed] [Google Scholar]
- 248.Shang A, et al. Exosomal miR-183-5p promotes angiogenesis in colorectal cancer by regulation of FOXO1. Aging. 2020;12:8352–8371. doi: 10.18632/aging.103145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Zeng Z, et al. Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat. Commun. 2018;9:5395. doi: 10.1038/s41467-018-07810-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Cheng WC, et al. RAB27B-activated secretion of stem-like tumor exosomes delivers the biomarker microRNA-146a-5p, which promotes tumorigenesis and associates with an immunosuppressive tumor microenvironment in colorectal cancer. Int. J. Cancer. 2019;145:2209–2224. doi: 10.1002/ijc.32338. [DOI] [PubMed] [Google Scholar]
- 251.Zhang Y, et al. Cancer-associated fibroblasts-derived exosomal miR-17-5p promotes colorectal cancer aggressive phenotype by initiating a RUNX3/MYC/TGF-β1 positive feedback loop. Cancer Lett. 2020;491:22–35. doi: 10.1016/j.canlet.2020.07.023. [DOI] [PubMed] [Google Scholar]
- 252.Bigagli E, Luceri C, Guasti D, Cinci L. Exosomes secreted from human colon cancer cells influence the adhesion of neighboring metastatic cells: Role of microRNA-210. Cancer Biol. Ther. 2016;17:1062–1069. doi: 10.1080/15384047.2016.1219815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Zhao S, et al. Tumor-derived exosomal miR-934 induces macrophage M2 polarization to promote liver metastasis of colorectal cancer. J. Hematol. Oncol. 2020;13:156. doi: 10.1186/s13045-020-00991-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Zhang X, et al. Exosomal miR-1255b-5p targets human telomerase reverse transcriptase in colorectal cancer cells to suppress epithelial-to-mesenchymal transition. Mol. Oncol. 2020;14:2589–2608. doi: 10.1002/1878-0261.12765. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 255.Liu H, et al. Colorectal cancer-derived exosomal miR-106b-3p promotes metastasis by down-regulating DLC-1 expression. Clin. Sci. (Lond., Engl.: 1979). 2020;134:419–434. doi: 10.1042/CS20191087. [DOI] [PubMed] [Google Scholar]
- 256.Ogata-Kawata H, et al. Circulating exosomal microRNAs as biomarkers of colon cancer. PLoS ONE. 2014;9:e92921. doi: 10.1371/journal.pone.0092921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Karimi N, et al. Serum overexpression of miR-301a and miR-23a in patients with colorectal cancer. J. Chin. Med. Assoc.: JCMA. 2019;82:215–220. doi: 10.1097/JCMA.0000000000000031. [DOI] [PubMed] [Google Scholar]
- 258.Fu F, Jiang W, Zhou L, Chen Z. Circulating exosomal miR-17-5p and miR-92a-3p predict pathologic stage and grade of colorectal cancer. Transl. Oncol. 2018;11:221–232. doi: 10.1016/j.tranon.2017.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Yan S, et al. Exosomal miR-6803-5p as potential diagnostic and prognostic marker in colorectal cancer. J. Cell. Biochem. 2018;119:4113–4119. doi: 10.1002/jcb.26609. [DOI] [PubMed] [Google Scholar]
- 260.Tang Y, et al. Tumor-derived exosomal miRNA-320d as a biomarker for metastatic colorectal cancer. J. Clin. Lab. Anal. 2019;33:e23004. doi: 10.1002/jcla.23004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Liu X, et al. Circulating exosomal miR-27a and miR-130a act as novel diagnostic and prognostic biomarkers of colorectal cancer. Cancer Epidemiol. Biomark. Prev. 2018;27:746–754. doi: 10.1158/1055-9965.EPI-18-0067. [DOI] [PubMed] [Google Scholar]
- 262.Zhang N, et al. Reduced serum exosomal miR-874 expression predicts poor prognosis in colorectal cancer. Eur. Rev. Med. Pharmacol. Sci. 2020;24:664–672. doi: 10.26355/eurrev_202001_20043. [DOI] [PubMed] [Google Scholar]
- 263.Sun Y, et al. Identification of serum miR-30a-5p as a diagnostic and prognostic biomarker in colorectal cancer. Cancer Biomark. 2019;24:299–305. doi: 10.3233/CBM-182129. [DOI] [PubMed] [Google Scholar]
- 264.Liu T, et al. Exosome-transmitted miR-128-3p increase chemosensitivity of oxaliplatin-resistant colorectal cancer. Mol. Cancer. 2019;18:43. doi: 10.1186/s12943-019-0981-7. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 265.Liang ZX, et al. Correction: LncRNA RPPH1 promotes colorectal cancer metastasis by interacting with TUBB3 and by promoting exosomes-mediated macrophage M2 polarization. Cell Death Dis. 2020;11:465. doi: 10.1038/s41419-020-2661-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Xu J, et al. Exosomal MALAT1 sponges miR-26a/26b to promote the invasion and metastasis of colorectal cancer via FUT4 enhanced fucosylation and PI3K/Akt pathway. J. Exp. Clin. Cancer Res. 2020;39:54. doi: 10.1186/s13046-020-01562-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Zhao Y, et al. Long noncoding RNA LINC02418 regulates MELK expression by acting as a ceRNA and may serve as a diagnostic marker for colorectal cancer. Cell Death Dis. 2019;10:568. doi: 10.1038/s41419-019-1804-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Ren J, et al. Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics. 2018;8:3932–3948. doi: 10.7150/thno.25541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Chen X, et al. Exosomal Long Non-coding RNA HOTTIP increases resistance of colorectal cancer cells to mitomycin via impairing MiR-214-mediated degradation of KPNA3. Front. Cell Dev. Biol. 2020;8:582723. doi: 10.3389/fcell.2020.582723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Oehme F, et al. Low level of exosomal long non-coding RNA HOTTIP is a prognostic biomarker in colorectal cancer. RNA Biol. 2019;16:1339–1345. doi: 10.1080/15476286.2019.1637697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Liu L, et al. Prognostic and predictive value of long non-coding RNA GAS5 and mircoRNA-221 in colorectal cancer and their effects on colorectal cancer cell proliferation, migration and invasion. Cancer Biomark. 2018;22:283–299. doi: 10.3233/CBM-171011. [DOI] [PubMed] [Google Scholar]
- 272.Liu T, et al. Exosomal long noncoding RNA CRNDE-h as a novel serum-based biomarker for diagnosis and prognosis of colorectal cancer. Oncotarget. 2016;7:85551–85563. doi: 10.18632/oncotarget.13465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Yu B, et al. Diagnostic potential of serum exosomal colorectal neoplasia differentially expressed long non-coding RNA (CRNDE-p) and microRNA-217 expression in colorectal carcinoma. Oncotarget. 2017;8:83745–83753. doi: 10.18632/oncotarget.19407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Wang L, et al. Circulating long non-coding RNA colon cancer-associated transcript 2 protected by exosome as a potential biomarker for colorectal cancer. Biomed. Pharmacother. . 2019;113:108758. doi: 10.1016/j.biopha.2019.108758. [DOI] [PubMed] [Google Scholar]
- 275.Hu D, et al. Plasma exosomal long non-coding RNAs serve as biomarkers for early detection of colorectal cancer. Cell. Physiol. Biochem. 2018;51:2704–2715. doi: 10.1159/000495961. [DOI] [PubMed] [Google Scholar]
- 276.Yu M, et al. Circulating serum exosomal long non-coding RNAs FOXD2-AS1, NRIR, and XLOC_009459 as diagnostic biomarkers for colorectal cancer. Front. Oncol. 2021;11:618967. doi: 10.3389/fonc.2021.618967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Barbagallo C, et al. LncRNA UCA1, upregulated in CRC biopsies and downregulated in serum exosomes, controls mRNA expression by RNA-RNA interactions. Mol. Ther. Nucleic Acids. 2018;12:229–241. doi: 10.1016/j.omtn.2018.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Shang A, et al. Exosomal circPACRGL promotes progression of colorectal cancer via the miR-142-3p/miR-506-3p- TGF-β1 axis. Mol. cancer. 2020;19:117. doi: 10.1186/s12943-020-01235-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Li Y, et al. A novel circFMN2 promotes tumor proliferation in CRC by regulating the miR-1182/hTERT signaling pathways. Clin. Sci. (Lond., Engl.: 1979). 2019;133:2463–2479. doi: 10.1042/CS20190715. [DOI] [PubMed] [Google Scholar]
- 280.Feng W, et al. circIFT80 functions as a ceRNA of miR-1236-3p to promote colorectal cancer progression. Mol. Ther. Nucleic Acids. 2019;18:375–387. doi: 10.1016/j.omtn.2019.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 281.Wang X, et al. Exosome-delivered circRNA promotes glycolysis to induce chemoresistance through the miR-122-PKM2 axis in colorectal cancer. Mol. Oncol. 2020;14:539–555. doi: 10.1002/1878-0261.12629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Yang H, et al. Hypoxia induced exosomal circRNA promotes metastasis of colorectal cancer via targeting GEF-H1/RhoA axis. Theranostics. 2020;10:8211–8226. doi: 10.7150/thno.44419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Pan B, et al. Identification of serum exosomal hsa-circ-0004771 as a novel diagnostic biomarker of colorectal cancer. Front. Genet. 2019;10:1096. doi: 10.3389/fgene.2019.01096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Smyth EC, et al. Gastric cancer. Lancet (Lond., Engl.) 2020;396:635–648. doi: 10.1016/S0140-6736(20)31288-5. [DOI] [PubMed] [Google Scholar]
- 285.Huang J, et al. Exosome-mediated transfer of miR-1290 promotes cell proliferation and invasion in gastric cancer via NKD1. Acta Biochim. Biophys. Sin. 2019;51:900–907. doi: 10.1093/abbs/gmz077. [DOI] [PubMed] [Google Scholar]
- 286.Wang M, et al. Deregulated microRNAs in gastric cancer tissue-derived mesenchymal stem cells: novel biomarkers and a mechanism for gastric cancer. Br. J. cancer. 2014;110:1199–1210. doi: 10.1038/bjc.2014.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Xia X, et al. Hypoxic gastric cancer-derived exosomes promote progression and metastasis via MiR-301a-3p/PHD3/HIF-1α positive feedback loop. Oncogene. 2020;39:6231–6244. doi: 10.1038/s41388-020-01425-6. [DOI] [PubMed] [Google Scholar]
- 288.Wei S, et al. Exosomal transfer of miR-15b-3p enhances tumorigenesis and malignant transformation through the DYNLT1/Caspase-3/Caspase-9 signaling pathway in gastric cancer. J. Exp. Clin. Cancer Res. 2020;39:32. doi: 10.1186/s13046-019-1511-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Shi L, et al. Exosomal miRNA-34 from cancer-associated fibroblasts inhibits growth and invasion of gastric cancer cells in vitro and in vivo. Aging. 2020;12:8549–8564. doi: 10.18632/aging.103157. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 290.Ohshima K, et al. Let-7 microRNA family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line. PLoS ONE. 2010;5:e13247. doi: 10.1371/journal.pone.0013247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Li Q, et al. Exosomal miR-21-5p derived from gastric cancer promotes peritoneal metastasis via mesothelial-to-mesenchymal transition. Cell death Dis. 2018;9:854. doi: 10.1038/s41419-018-0928-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Wang J, et al. Exosomal miR-27a derived from gastric cancer cells regulates the transformation of fibroblasts into cancer-associated fibroblasts. Cell. Physiol. Biochem. 2018;49:869–883. doi: 10.1159/000493218. [DOI] [PubMed] [Google Scholar]
- 293.Xu G, et al. Exosomal miRNA-139 in cancer-associated fibroblasts inhibits gastric cancer progression by repressing MMP11 expression. Int. J. Biol. Sci. 2019;15:2320–2329. doi: 10.7150/ijbs.33750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Yang H, et al. Exosome-derived miR-130a activates angiogenesis in gastric cancer by targeting C-MYB in vascular endothelial cells. Mol. Ther. 2018;26:2466–2475. doi: 10.1016/j.ymthe.2018.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 295.Wang N, et al. A serum exosomal microRNA panel as a potential biomarker test for gastric cancer. Biochem. Biophys. Res. Commun. 2017;493:1322–1328. doi: 10.1016/j.bbrc.2017.10.003. [DOI] [PubMed] [Google Scholar]
- 296.Shi Y, et al. Exosomal miR-1246 in serum as a potential biomarker for early diagnosis of gastric cancer. Int. J. Clin. Oncol. 2020;25:89–99. doi: 10.1007/s10147-019-01532-9. [DOI] [PubMed] [Google Scholar]
- 297.Ren W, et al. Exosomal miRNA-107 induces myeloid-derived suppressor cell expansion in gastric cancer. Cancer Manag. Res. 2019;11:4023–4040. doi: 10.2147/CMAR.S198886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Feng C, et al. Exosomal miR-196a-1 promotes gastric cancer cell invasion and metastasis by targeting SFRP1. Nanomed. (Lond., Engl.) 2019;14:2579–2593. doi: 10.2217/nnm-2019-0053. [DOI] [PubMed] [Google Scholar]
- 299.Zhu M, Zhang N, He S, Lu X. Exosomal miR-106a derived from gastric cancer promotes peritoneal metastasis via direct regulation of Smad7. Cell Cycle (Georget., Tex.) 2020;19:1200–1221. doi: 10.1080/15384101.2020.1749467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Shi SS, et al. Exosomal miR-155-5p promotes proliferation and migration of gastric cancer cells by inhibiting TP53INP1 expression. Pathol. Res. Pract. 2020;216:152986. doi: 10.1016/j.prp.2020.152986. [DOI] [PubMed] [Google Scholar]
- 301.Zhang J, et al. HOTAIR contributes to the carcinogenesis of gastric cancer via modulating cellular and exosomal miRNAs level. Cell death Dis. 2020;11:780. doi: 10.1038/s41419-020-02946-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Wang L, et al. Exosome-transferred LINC01559 promotes the progression of gastric cancer via PI3K/AKT signaling pathway. Cell Death Dis. 2020;11:723. doi: 10.1038/s41419-020-02810-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Lu Y, et al. Exosome-delivered LncHEIH promotes gastric cancer progression by upregulating EZH2 and stimulating methylation of the GSDME promoter. Front. Cell Dev. Biol. 2020;8:571297. doi: 10.3389/fcell.2020.571297. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 304.Wang J, et al. Exosome-mediated transfer of lncRNA HOTTIP promotes cisplatin resistance in gastric cancer cells by regulating HMGA1/miR-218 axis. OncoTargets Ther. 2019;12:11325–11338. doi: 10.2147/OTT.S231846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Lin LY, et al. Tumor-originated exosomal lncUEGC1 as a circulating biomarker for early-stage gastric cancer. Mol. cancer. 2018;17:84. doi: 10.1186/s12943-018-0834-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Zhao R, et al. Exosomal long noncoding RNA HOTTIP as potential novel diagnostic and prognostic biomarker test for gastric cancer. Mol. Cancer. 2018;17:68. doi: 10.1186/s12943-018-0817-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Guo X, et al. Circulating exosomal gastric cancer-associated long noncoding RNA1 as a biomarker for early detection and monitoring progression of gastric cancer: a multiphase study. JAMA Surg. 2020;155:572–579. doi: 10.1001/jamasurg.2020.1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Xu H, et al. Identification of serum exosomal lncRNA MIAT as a novel diagnostic and prognostic biomarker for gastric cancer. J. Clin. Lab. Anal. 2020;34:e23323. doi: 10.1002/jcla.23323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Zhou H, et al. Circulating exosomal long non-coding RNA H19 as a potential novel diagnostic and prognostic biomarker for gastric cancer. J. Int. Med. Res. 2020;48:300060520934297. doi: 10.1177/0300060520934297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Zheng P, et al. Plasma exosomal long noncoding RNA lnc-SLC2A12-10:1 as a novel diagnostic biomarker for gastric cancer. OncoTargets Ther. 2020;13:4009–4018. doi: 10.2147/OTT.S253600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Piao HY, Guo S, Wang Y, Zhang J. Exosomal long non-coding RNA CEBPA-AS1 inhibits tumor apoptosis and functions as a non-invasive biomarker for diagnosis of gastric cancer. OncoTargets Ther. 2020;13:1365–1374. doi: 10.2147/OTT.S238706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Li S, et al. Exosomal long noncoding RNA lnc-GNAQ-6:1 may serve as a diagnostic marker for gastric cancer. Clin. Chim. Acta. 2020;501:252–257. doi: 10.1016/j.cca.2019.10.047. [DOI] [PubMed] [Google Scholar]
- 313.Cai C, et al. Serum exosomal long noncoding RNA pcsk2-2:1 as a potential novel diagnostic biomarker for gastric cancer. OncoTargets Ther. 2019;12:10035–10041. doi: 10.2147/OTT.S229033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Xie M, et al. Exosomal circSHKBP1 promotes gastric cancer progression via regulating the miR-582-3p/HUR/VEGF axis and suppressing HSP90 degradation. Mol. Cancer. 2020;19:112. doi: 10.1186/s12943-020-01208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Zhang H, et al. Exosomal circRNA derived from gastric tumor promotes white adipose browning by targeting the miR-133/PRDM16 pathway. Int. J. Cancer. 2019;144:2501–2515. doi: 10.1002/ijc.31977. [DOI] [PubMed] [Google Scholar]
- 316.Yu, L. et al. Plasma exosomal CircNEK9 accelerates the progression of gastric cancer via miR-409-3p/MAP7 Axis. Dig, Dis. Sci.10.1007/s10620-020-06816-z (2021). [DOI] [PubMed]
- 317.Li S, et al. Gastric cancer derived exosomes mediate the delivery of circRNA to promote angiogenesis by targeting miR-29a/VEGF axis in endothelial cells. Biochem. Biophys. Res. Commun. 2021;560:37–44. doi: 10.1016/j.bbrc.2021.04.099. [DOI] [PubMed] [Google Scholar]
- 318.Hui C, Tian L, He X. Circular RNA circNHSL1 contributes to gastric cancer progression through the miR-149-5p/YWHAZ Axis. Cancer Manag. Res. 2020;12:7117–7130. doi: 10.2147/CMAR.S253152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Zhong Y, et al. Circular RNA circ_0032821 contributes to oxaliplatin (OXA) resistance of gastric cancer cells by regulating SOX9 via miR-515-5p. Biotechnol. Lett. 2021;43:339–351. doi: 10.1007/s10529-020-03036-3. [DOI] [PubMed] [Google Scholar]
- 320.Shao Y, et al. Hsa_circ_0065149 is an Indicator for Early Gastric Cancer Screening and Prognosis Prediction. Pathol. Oncol. Res. 2020;26:1475–1482. doi: 10.1007/s12253-019-00716-y. [DOI] [PubMed] [Google Scholar]
- 321.Ren S, et al. RNA-seq analysis of prostate cancer in the Chinese population identifies recurrent gene fusions, cancer-associated long noncoding RNAs and aberrant alternative splicings. Cell Res. 2012;22:806–821. doi: 10.1038/cr.2012.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Litwin MS, Tan HJ. The Diagnosis and Treatment of Prostate Cancer: A Review. JAMA. 2017;317:2532–2542. doi: 10.1001/jama.2017.7248. [DOI] [PubMed] [Google Scholar]
- 323.Wang J, et al. Exosomal microRNAs as liquid biopsy biomarkers in prostate cancer. Crit. Rev. Oncol. Hematol. 2020;145:102860. doi: 10.1016/j.critrevonc.2019.102860. [DOI] [PubMed] [Google Scholar]
- 324.Zhou C, et al. Functional implication of exosomal miR-217 and miR-23b-3p in the progression of prostate cancer. OncoTargets Ther. 2020;13:11595–11606. doi: 10.2147/OTT.S272869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Jiang S, et al. Human bone marrow mesenchymal stem cells-derived microRNA-205-containing exosomes impede the progression of prostate cancer through suppression of RHPN2. J. Exp. Clin. cancer Res. 2019;38:495. doi: 10.1186/s13046-019-1488-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Wang X, et al. Prostate carcinoma cell-derived exosomal MicroRNA-26a modulates the metastasis and tumor growth of prostate carcinoma. Biomed, Pharmacother. . 2019;117:109109. doi: 10.1016/j.biopha.2019.109109. [DOI] [PubMed] [Google Scholar]
- 327.Guan H, et al. Tumor-associated macrophages promote prostate cancer progression via exosome-mediated miR-95 transfer. J. Cell. Physiol. 2020;235:9729–9742. doi: 10.1002/jcp.29784. [DOI] [PubMed] [Google Scholar]
- 328.Dai Y, Gao X. Inhibition of cancer cell-derived exosomal microRNA-183 suppresses cell growth and metastasis in prostate cancer by upregulating TPM1. Cancer Cell Int. 2021;21:145. doi: 10.1186/s12935-020-01686-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Ye Y, et al. Exosomal miR-141-3p regulates osteoblast activity to promote the osteoblastic metastasis of prostate cancer. Oncotarget. 2017;8:94834–94849. doi: 10.18632/oncotarget.22014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 330.Huang X, et al. Exosomal miR-1290 and miR-375 as prognostic markers in castration-resistant prostate cancer. Eur. Urol. 2015;67:33–41. doi: 10.1016/j.eururo.2014.07.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Li Z, et al. Exosomal microRNA-141 is upregulated in the serum of prostate cancer patients. OncoTargets Ther. 2016;9:139–148. doi: 10.2147/OTT.S95565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Hessvik NP, Sandvig K, Llorente A. Exosomal miRNAs as biomarkers for prostate cancer. Front. Genet. 2013;4:36. doi: 10.3389/fgene.2013.00036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Wang J, et al. Long non-coding RNA MYU promotes prostate cancer proliferation by mediating the miR-184/c-Myc axis. Oncol. Rep. 2018;40:2814–2825. doi: 10.3892/or.2018.6661. [DOI] [PubMed] [Google Scholar]
- 334.Wang YH, et al. Tumor-derived exosomal long noncoding RNAs as promising diagnostic biomarkers for prostate cancer. Cell. Physiol. Biochem. 2018;46:532–545. doi: 10.1159/000488620. [DOI] [PubMed] [Google Scholar]
- 335.Ozgur E, Gezer U. Investigation of lncRNA H19 in prostate cancer cells and secreted exosomes upon androgen stimulation or androgen receptor blockage. Bratisl. Lek. Listy. 2020;121:362–365. doi: 10.4149/BLL_2020_058. [DOI] [PubMed] [Google Scholar]
- 336.Fang J, Qi J, Dong X, Luo J. Perspectives on circular RNAs as prostate cancer biomarkers. Front. Cell Dev. Biol. 2020;8:594992. doi: 10.3389/fcell.2020.594992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Li T, Sun X, Chen L. Exosome circ_0044516 promotes prostate cancer cell proliferation and metastasis as a potential biomarker. J. Cell. Biochem. 2020;121:2118–2126. doi: 10.1002/jcb.28239. [DOI] [PubMed] [Google Scholar]
- 338.Zhang H, et al. Exosomal Circ-XIAP promotes docetaxel resistance in prostate cancer by regulating miR-1182/TPD52 axis. Drug Des. Dev. Ther. 2021;15:1835–1849. doi: 10.2147/DDDT.S300376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Ariston Gabriel AN, et al. The involvement of exosomes in the diagnosis and treatment of pancreatic cancer. Mol. Cancer. 2020;19:132. doi: 10.1186/s12943-020-01245-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Vijan S. Type 2 diabetes. Ann. Intern. Med. 2019;171:ITC65–ITC80. doi: 10.7326/AITC201911050. [DOI] [PubMed] [Google Scholar]
- 341.Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018;14:88–98. doi: 10.1038/nrendo.2017.151. [DOI] [PubMed] [Google Scholar]
- 342.Li C-J, Fang Q-H, Liu M-L, Lin J-N. Current understanding of the role of adipose-derived extracellular vesicles in metabolic homeostasis and diseases: communication from the distance between cells/tissues. Theranostics. 2020;10:7422–7435. doi: 10.7150/thno.42167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Galic S, Oakhill JS, Steinberg GR. Adipose tissue as an endocrine organ. Mol. Cell. Endocrinol. 2010;316:129–139. doi: 10.1016/j.mce.2009.08.018. [DOI] [PubMed] [Google Scholar]
- 344.Thomou T, et al. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature. 2017;542:450–455. doi: 10.1038/nature21365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Gesmundo I, et al. Adipocyte-derived extracellular vesicles regulate survival and function of pancreatic β-cells. JCI insight. 2021;6:e141962. doi: 10.1172/jci.insight.141962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Pan Y, et al. Adipocyte-secreted exosomal microRNA-34a inhibits M2 macrophage polarization to promote obesity-induced adipose inflammation. J. Clin. Investig. 2019;129:834–849. doi: 10.1172/JCI123069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Li D, et al. Gonadal white adipose tissue-derived exosomal MiR-222 promotes obesity-associated insulin resistance. Aging. 2020;12:22719–22743. doi: 10.18632/aging.103891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Yu Y, et al. Adipocyte-derived exosomal MiR-27a induces insulin resistance in skeletal muscle through repression of PPARγ. Theranostics. 2018;8:2171–2188. doi: 10.7150/thno.22565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Dang S-Y, et al. Exosomal transfer of obesity adipose tissue for decreased miR-141-3p mediate insulin resistance of hepatocytes. Int. J. Biol. Sci. 2019;15:351–368. doi: 10.7150/ijbs.28522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Gan L, et al. Small extracellular microvesicles mediated pathological communications between dysfunctional adipocytes and cardiomyocytes as a novel mechanism exacerbating ischemia/reperfusion injury in diabetic mice. Circulation. 2020;141:968–983. doi: 10.1161/CIRCULATIONAHA.119.042640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Ying W, et al. Adipose tissue macrophage-derived exosomal miRNAs can modulate in vivo and in vitro insulin sensitivity. Cell. 2017;171:372–384.e312. doi: 10.1016/j.cell.2017.08.035. [DOI] [PubMed] [Google Scholar]
- 352.Liu T, Sun Y-C, Cheng P, Shao H-G. Adipose tissue macrophage-derived exosomal miR-29a regulates obesity-associated insulin resistance. Biochem. Biophys. Res. Commun. 2019;515:352–358. doi: 10.1016/j.bbrc.2019.05.113. [DOI] [PubMed] [Google Scholar]
- 353.Ying W, et al. MiR-690, an exosomal-derived miRNA from M2-polarized macrophages, improves insulin sensitivity in obese mice. Cell Metab. 2021;33:781–790.e5. doi: 10.1016/j.cmet.2020.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Tian F, et al. miR-210 in Exosomes Derived from Macrophages under High Glucose Promotes Mouse Diabetic Obesity Pathogenesis by Suppressing NDUFA4 Expression. J. Diabetes Res. 2020;2020:6894684. doi: 10.1155/2020/6894684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Wu J, et al. Hepatic exosome-derived miR-130a-3p attenuates glucose intolerance via suppressing PHLPP2 gene in adipocyte. Metab.: Clin. Exp. 2020;103:154006. doi: 10.1016/j.metabol.2019.154006. [DOI] [PubMed] [Google Scholar]
- 356.Fu Q, et al. Hepatocytes derived extracellular vesicles from high-fat diet induced obese mice modulate genes expression and proliferation of islet β cells. Biochem. Biophys. Res. Commun. 2019;516:1159–1166. doi: 10.1016/j.bbrc.2019.06.124. [DOI] [PubMed] [Google Scholar]
- 357.Jalabert A, et al. Exosome-like vesicles released from lipid-induced insulin-resistant muscles modulate gene expression and proliferation of beta recipient cells in mice. Diabetologia. 2016;59:1049–1058. doi: 10.1007/s00125-016-3882-y. [DOI] [PubMed] [Google Scholar]
- 358.Zhang A, et al. Islet β cell: An endocrine cell secreting miRNAs. Biochem. Biophys. Res. Commun. 2018;495:1648–1654. doi: 10.1016/j.bbrc.2017.12.028. [DOI] [PubMed] [Google Scholar]
- 359.Li J, et al. Pancreatic β cells control glucose homeostasis via the secretion of exosomal miR-29 family. J. Extracell. Vesicles. 2021;10:e12055. doi: 10.1002/jev2.12055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Xu H, et al. Pancreatic β cell microRNA-26a alleviates type 2 diabetes by improving peripheral insulin sensitivity and preserving β cell function. PLoS Biol. 2020;18:e3000603. doi: 10.1371/journal.pbio.3000603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Kamalden TA, et al. Exosomal MicroRNA-15a transfer from the pancreas augments diabetic complications by inducing oxidative stress. Antioxid. Redox Signal. 2017;27:913–930. doi: 10.1089/ars.2016.6844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Shen Z, et al. MicroRNA-127 inhibits cell proliferation via targeting Kif3b in pancreatic β cells. Aging. 2019;11:1342–1355. doi: 10.18632/aging.101835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Katayama M, et al. Circulating exosomal miR-20b-5p is elevated in type 2 diabetes and could impair insulin action in human skeletal muscle. Diabetes. 2019;68:515–526. doi: 10.2337/db18-0470. [DOI] [PubMed] [Google Scholar]
- 364.Santovito D, et al. Plasma exosome microRNA profiling unravels a new potential modulator of adiponectin pathway in diabetes: effect of glycemic control. J. Clin. Endocrinol. Metab. 2014;99:E1681–E1685. doi: 10.1210/jc.2013-3843. [DOI] [PubMed] [Google Scholar]
- 365.Ruan Y, et al. Circulating LncRNAs analysis in patients with type 2 diabetes reveals novel genes influencing glucose metabolism and Islet β-cell function. Cell. Physiol. Biochem. 2018;46:335–350. doi: 10.1159/000488434. [DOI] [PubMed] [Google Scholar]
- 366.Tello-Flores VA, et al. Altered levels of MALAT1 and H19 derived from serum or serum exosomes associated with type-2 diabetes. Non-coding RNA Res. 2020;5:71–76. doi: 10.1016/j.ncrna.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Li B, et al. The MSC-derived exosomal lncRNA H19 promotes wound healing in diabetic foot ulcers by upregulating PTEN via MicroRNA-152-3p. Mol. Ther. Nucleic Acids. 2020;19:814–826. doi: 10.1016/j.omtn.2019.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Tao S-C, et al. Extracellular vesicle-mimetic nanovesicles transport LncRNA-H19 as competing endogenous RNA for the treatment of diabetic wounds. Drug Deliv. 2018;25:241–255. doi: 10.1080/10717544.2018.1425774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Cao X, et al. MSC-derived exosomal lncRNA SNHG7 suppresses endothelial-mesenchymal transition and tube formation in diabetic retinopathy via miR-34a-5p/XBP1 axis. Life Sci. 2021;272:119232. doi: 10.1016/j.lfs.2021.119232. [DOI] [PubMed] [Google Scholar]
- 370.Wang S, et al. CircRNA-0077930 from hyperglycaemia-stimulated vascular endothelial cell exosomes regulates senescence in vascular smooth muscle cells. Cell Biochem Funct. 2020;38:1056–1068. doi: 10.1002/cbf.3543. [DOI] [PubMed] [Google Scholar]
- 371.Chen Z-J, et al. Serum and exosomal hsa_circ_0000907 and hsa_circ_0057362 as novel biomarkers in the early diagnosis of diabetic foot ulcer. Eur. Rev. Med. Pharmacol. Sci. 2020;24:8117–8126. doi: 10.26355/eurrev_202008_22498. [DOI] [PubMed] [Google Scholar]
- 372.Liu C, et al. Targeting pericyte-endothelial cell crosstalk by circular RNA-cPWWP2A inhibition aggravates diabetes-induced microvascular dysfunction. Proc. Natl Acad. Sci. USA. 2019;116:7455–7464. doi: 10.1073/pnas.1814874116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Bai S, et al. Exosomal circ_DLGAP4 promotes diabetic kidney disease progression by sponging miR-143 and targeting ERBB3/NF-κB/MMP-2 axis. Cell Death Dis. 2020;11:1008. doi: 10.1038/s41419-020-03169-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.WHO. Obesity. Available online at: https://www.who.int/health-topics/obesity#tab=tab_1 (2021).
- 375.Afshin A, et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017;377:13–27. doi: 10.1056/NEJMoa1614362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Larabee CM, Neely OC, Domingos AI. Obesity: a neuroimmunometabolic perspective. Nat. Rev. Endocrinol. 2020;16:30–43. doi: 10.1038/s41574-019-0283-6. [DOI] [PubMed] [Google Scholar]
- 377.Santamaria-Martos F, et al. Comparative and functional analysis of plasma membrane-derived extracellular vesicles from obese vs. nonobese women. Clin. Nutr. (Edinb., Scotl.) 2020;39:1067–1076. doi: 10.1016/j.clnu.2019.04.008. [DOI] [PubMed] [Google Scholar]
- 378.Wang J, et al. Elafin inhibits obesity, hyperglycemia, and liver steatosis in high-fat diet-treated male mice. Sci. Rep. 2020;10:12785. doi: 10.1038/s41598-020-69634-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Mendonça MD, et al. Aerobic exercise training regulates serum extracellular vesicle miRNAs linked to obesity to promote their beneficial effects in mice. Am. J. Physiol. Endocrinol. Metab. 2020;319:E579–E591. doi: 10.1152/ajpendo.00172.2020. [DOI] [PubMed] [Google Scholar]
- 380.Hubal MJ, et al. Circulating adipocyte-derived exosomal MicroRNAs associated with decreased insulin resistance after gastric bypass. Obesity (Silver Spring, Md.) 2017;25:102–110. doi: 10.1002/oby.21709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Bae Y-U, et al. Bariatric surgery alters microRNA content of circulating exosomes in patients with obesity. Obesity (Silver Spring, Md.) 2019;27:264–271. doi: 10.1002/oby.22379. [DOI] [PubMed] [Google Scholar]
- 382.Ferrante SC, et al. Adipocyte-derived exosomal miRNAs: a novel mechanism for obesity-related disease. Pediatr. Res. 2014;77:447–454. doi: 10.1038/pr.2014.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Yang Z, Wei Z, Wu X, Yang H. Screening of exosomal miRNAs derived from subcutaneous and visceral adipose tissues: Determination of targets for the treatment of obesity and associated metabolic disorders. Mol. Med. Rep. 2018;18:3314–3324. doi: 10.3892/mmr.2018.9312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Kim Y, Kim O-K. Potential roles of adipocyte extracellular vesicle-derived miRNAs in obesity-mediated insulin resistance. Adv. Nutr. 2020;12:566–574. doi: 10.1093/advances/nmaa105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Kim H, et al. Effect of diabetes on exosomal miRNA profile in patients with obesity. BMJ Open Diabetes Res. Care. 2020;8:e001403. doi: 10.1136/bmjdrc-2020-001403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Compston JE, McClung MR, Leslie WD. Osteoporosis. Lancet. 2019;393:364–376. doi: 10.1016/S0140-6736(18)32112-3. [DOI] [PubMed] [Google Scholar]
- 387.Hendrickx G, Boudin E, van Hul W. A look behind the scenes: the risk and pathogenesis of primary osteoporosis. Nat. Rev. Rheumatol. 2015;11:462–474. doi: 10.1038/nrrheum.2015.48. [DOI] [PubMed] [Google Scholar]
- 388.Li G, et al. An overview of osteoporosis and frailty in the elderly. BMC Musculoskelet. Disord. 2017;18:46. doi: 10.1186/s12891-017-1403-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Fuggle NR, et al. Fracture prediction, imaging and screening in osteoporosis. Nat. Rev. Endocrinol. 2019;15:535–547. doi: 10.1038/s41574-019-0220-8. [DOI] [PubMed] [Google Scholar]
- 390.Xu T, et al. Exosomal miRNA-128-3p from mesenchymal stem cells of aged rats regulates osteogenesis and bone fracture healing by targeting Smad5. J. Nanobiotechnol. 2020;18:47. doi: 10.1186/s12951-020-00601-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Takafuji Y, et al. MicroRNA-196a-5p in extracellular vesicles secreted from myoblasts suppresses osteoclast-like cell formation in mouse cells. Tissue Int. 2021;108:364–376. doi: 10.1007/s00223-020-00772-6. [DOI] [PubMed] [Google Scholar]
- 392.Wang X, et al. Mesenchymal stem cell-derived exosomes have altered microRNA profiles and induce osteogenic differentiation depending on the stage of differentiation. PLoS ONE. 2018;13:e0193059. doi: 10.1371/journal.pone.0193059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Xu J-F, et al. Altered MicroRNA expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS ONE. 2014;9:e114627. doi: 10.1371/journal.pone.0114627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Jiang L-B, Tian L, Zhang C-G. Bone marrow stem cells-derived exosomes extracted from osteoporosis patients inhibit osteogenesis via microRNA-21/SMAD7. Eur. Rev. Med. Pharmacol. Sci. 2018;22:6221–6229. doi: 10.26355/eurrev_201810_16028. [DOI] [PubMed] [Google Scholar]
- 395.Sun W, et al. Osteoclast-derived microRNA-containing exosomes selectively inhibit osteoblast activity. Cell Discov. 2016;2:16015. doi: 10.1038/celldisc.2016.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Li D, et al. Osteoclast-derived exosomal miR-214-3p inhibits osteoblastic bone formation. Nat. Commun. 2016;7:10872. doi: 10.1038/ncomms10872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.Yang J-X, et al. Osteoclast-derived miR-23a-5p-containing exosomes inhibit osteogenic differentiation by regulating Runx2. Cell. Signal. 2020;70:109504. doi: 10.1016/j.cellsig.2019.109504. [DOI] [PubMed] [Google Scholar]
- 398.Fulzele S, et al. Muscle-derived miR-34a increases with age in circulating extracellular vesicles and induces senescence of bone marrow stem cells. Aging. 2019;11:1791–1803. doi: 10.18632/aging.101874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Zainabadi K, Liu CJ, Guarente L. SIRT1 is a positive regulator of the master osteoblast transcription factor, RUNX2. PLoS ONE. 2017;12:e0178520. doi: 10.1371/journal.pone.0178520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Chen Y, et al. SIRT1, a promising regulator of bone homeostasis. Life Sci. 2021;269:119041. doi: 10.1016/j.lfs.2021.119041. [DOI] [PubMed] [Google Scholar]
- 401.Qin Y, et al. Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA-218: A novel mechanism in muscle-bone communication. J. Biol. Chem. 2017;292:11021–11033. doi: 10.1074/jbc.M116.770941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Teng Z, et al. Osteoporosis is characterized by altered expression of exosomal long non-coding RNAs. Front. Genet. 2020;11:566959. doi: 10.3389/fgene.2020.566959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Cui Y, et al. EPC-derived exosomes promote osteoclastogenesis through LncRNA-MALAT1. J. Cell. Mol. Med. 2019;23:3843–3854. doi: 10.1111/jcmm.14228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 404.Zhi F, et al. Exosomal hsa_circ_0006859 is a potential biomarker for postmenopausal osteoporosis and enhances adipogenic versus osteogenic differentiation in human bone marrow mesenchymal stem cells by sponging miR-431-5p. Stem Cell Res. Ther. 2021;12:157. doi: 10.1186/s13287-021-02214-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Liu Z, et al. CircHmbox1 targeting miRNA-1247-5p is involved in the regulation of bone metabolism by TNF-α in postmenopausal osteoporosis. Front. Cell Dev. Biol. 2020;8:594785. doi: 10.3389/fcell.2020.594785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Zheng DH, et al. Erythropoietin attenuates high glucose-induced oxidative stress and inhibition of osteogenic differentiation in periodontal ligament stem cell (PDLSCs) Chem. Biol. Interact. 2019;305:40–47. doi: 10.1016/j.cbi.2019.03.007. [DOI] [PubMed] [Google Scholar]
- 407.Libby P, Ridker PM, Hansson GK, Leducq Transatlantic Network on Atherothrombosis Inflammation in atherosclerosis: from pathophysiology to practice. J. Am. Coll. Cardiol. 2009;54:2129–2138. doi: 10.1016/j.jacc.2009.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473:317–325. doi: 10.1038/nature10146. [DOI] [PubMed] [Google Scholar]
- 409.Barile L, et al. Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc Res. 2014;103:530–541. doi: 10.1093/cvr/cvu167. [DOI] [PubMed] [Google Scholar]
- 410.Pfeifer P, Werner N, Jansen F. Role and function of microRNAs in extracellular vesicles in cardiovascular biology. Biomed. Res Int. 2015;2015:161393. doi: 10.1155/2015/161393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Rezaie J, et al. Cardioprotective role of extracellular vesicles: a highlight on exosome beneficial effects in cardiovascular diseases. J. Cell Physiol. 2019;234:21732–21745. doi: 10.1002/jcp.28894. [DOI] [PubMed] [Google Scholar]
- 412.Qiao S, et al. Extracellular vesicles derived from Kruppel-Like Factor 2-overexpressing endothelial cells attenuate myocardial ischemia-reperfusion injury by preventing Ly6C(high) monocyte recruitment. Theranostics. 2020;10:11562–11579. doi: 10.7150/thno.45459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.Davidson SM, et al. Circulating blood cells and extracellular vesicles in acute cardioprotection. Cardiovasc Res. 2019;115:1156–1166. doi: 10.1093/cvr/cvy314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Kopin L, Lowenstein C. Dyslipidemia. Ann. Intern. Med. 2017;167:ITC81–ITC96. doi: 10.7326/AITC201712050. [DOI] [PubMed] [Google Scholar]
- 415.Fuchs FD, Whelton PK. High blood pressure and cardiovascular disease. Hypertension. 2020;75:285–292. doi: 10.1161/HYPERTENSIONAHA.119.14240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Bress AP, et al. Potential cardiovascular disease events prevented with adoption of the 2017 American College of Cardiology/American Heart Association Blood Pressure Guideline. Circulation. 2019;139:24–36. doi: 10.1161/CIRCULATIONAHA.118.035640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Brunström M, Carlberg B. Association of blood pressure lowering with mortality and cardiovascular disease across blood pressure levels: a systematic review and meta-analysis. JAMA Intern. Med. 2018;178:28–36. doi: 10.1001/jamainternmed.2017.6015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Tan PPS, et al. Exosomal microRNAs in the development of essential hypertension and its potential as biomarkers. Am. J. Physiol. Heart Circ. Physiol. 2021;320:H1486–h1497. doi: 10.1152/ajpheart.00888.2020. [DOI] [PubMed] [Google Scholar]
- 419.Jayaseelan VP, Arumugam P. Exosomal microRNAs as a promising theragnostic tool for essential hypertension. Hypertens. Res. 2020;43:74–75. doi: 10.1038/s41440-019-0343-2. [DOI] [PubMed] [Google Scholar]
- 420.Xu F, et al. Melatonin alleviates vascular calcification and ageing through exosomal miR-204/miR-211 cluster in a paracrine manner. J. Pineal. Res. 2020;68:e12631. doi: 10.1111/jpi.12631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Liu X, et al. miRNA profiling of exosomes from spontaneous hypertensive rats using next-generation sequencing. J. Cardiovasc. Transl. Res. 2019;12:75–83. doi: 10.1007/s12265-017-9784-7. [DOI] [PubMed] [Google Scholar]
- 422.Ren XS, et al. MiR155-5p in adventitial fibroblasts-derived extracellular vesicles inhibits vascular smooth muscle cell proliferation via suppressing angiotensin-converting enzyme expression. J. Extracell. Vesicles. 2020;9:1698795. doi: 10.1080/20013078.2019.1698795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Zou X, et al. Secreted monocyte miR-27a, via mesenteric arterial mas receptor-eNOS pathway, causes hypertension. Am. J. Hypertens. 2020;33:31–42. doi: 10.1093/ajh/hpz112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Osada-Oka M, et al. Macrophage-derived exosomes induce inflammatory factors in endothelial cells under hypertensive conditions. Hypertens. Res. 2017;40:353–360. doi: 10.1038/hr.2016.163. [DOI] [PubMed] [Google Scholar]
- 425.Oh J, et al. Macrophage secretion of miR-106b-5p causes renin-dependent hypertension. Nat. Commun. 2020;11:4798. doi: 10.1038/s41467-020-18538-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.O’Malley PG, et al. Management of dyslipidemia for cardiovascular disease risk reduction: synopsis of the 2020 updated U.S. Department of Veterans Affairs and U.S. Department of Defense Clinical Practice Guideline. Ann. Intern Med. 2020;173:822–829. doi: 10.7326/M20-4648. [DOI] [PubMed] [Google Scholar]
- 427.Plakht Y, Shiyovich A, Gilutz H. Predictors of long-term (10-year) mortality postmyocardial infarction: age-related differences. Soroka Acute Myocardial Infarction (SAMI) Project. J. Cardiol. 2015;65:216–223. doi: 10.1016/j.jjcc.2014.06.001. [DOI] [PubMed] [Google Scholar]
- 428.Roth GA, et al. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017;70:1–25. doi: 10.1016/j.jacc.2017.04.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Benjamin EJ, et al. Heart disease and stroke statistics-2017 update: a report from the American Heart Association. Circulation. 2017;135:e146–e603. doi: 10.1161/CIR.0000000000000485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Zheng B, et al. Exosome-mediated miR-155 transfer from smooth muscle cells to endothelial cells induces endothelial injury and promotes atherosclerosis. Mol. Ther. 2017;25:1279–1294. doi: 10.1016/j.ymthe.2017.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 431.Zhang Z, et al. Exosomal LINC01005 derived from oxidized low-density lipoprotein-treated endothelial cells regulates vascular smooth muscle cell phenotypic switch. Biofactors. 2020;46:743–753. doi: 10.1002/biof.1665. [DOI] [PubMed] [Google Scholar]
- 432.Zhu J, et al. Exosomes from nicotine-stimulated macrophages accelerate atherosclerosis through miR-21-3p/PTEN-mediated VSMC migration and proliferation. Theranostics. 2019;9:6901–6919. doi: 10.7150/thno.37357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Iaconetti C, Sorrentino S, De Rosa S, Indolfi C. Exosomal miRNAs in heart disease. Physiol. (Bethesda) 2016;31:16–24. doi: 10.1152/physiol.00029.2015. [DOI] [PubMed] [Google Scholar]
- 434.Tan M, et al. Thrombin stimulated platelet-derived exosomes inhibit platelet-derived growth factor receptor-beta expression in vascular smooth muscle cells. Cell Physiol. Biochem. 2016;38:2348–2365. doi: 10.1159/000445588. [DOI] [PubMed] [Google Scholar]
- 435.Li J, et al. Thrombin-activated platelet-derived exosomes regulate endothelial cell expression of ICAM-1 via microRNA-223 during the thrombosis-inflammation response. Thromb. Res. 2017;154:96–105. doi: 10.1016/j.thromres.2017.04.016. [DOI] [PubMed] [Google Scholar]
- 436.Liu J, et al. The role of exosomal non-coding RNAs in coronary artery disease. Front. Pharm. 2020;11:603104. doi: 10.3389/fphar.2020.603104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Loyer X, et al. Inhibition of microRNA-92a prevents endothelial dysfunction and atherosclerosis in mice. Circ. Res. 2014;114:434–443. doi: 10.1161/CIRCRESAHA.114.302213. [DOI] [PubMed] [Google Scholar]
- 438.Xing X, et al. Adipose-derived mesenchymal stem cells-derived exosome-mediated microRNA-342-5p protects endothelial cells against atherosclerosis. Aging (Albany NY) 2020;12:3880–3898. doi: 10.18632/aging.102857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Liu Y, et al. Exosome-mediated miR-106a-3p derived from ox-LDL exposed macrophages accelerated cell proliferation and repressed cell apoptosis of human vascular smooth muscle cells. Eur. Rev. Med Pharm. Sci. 2020;24:7039–7050. doi: 10.26355/eurrev_202006_21697. [DOI] [PubMed] [Google Scholar]
- 440.Chen L, et al. Exosomal lncRNA GAS5 regulates the apoptosis of macrophages and vascular endothelial cells in atherosclerosis. PLoS ONE. 2017;12:e0185406. doi: 10.1371/journal.pone.0185406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Li H, et al. Loss of exosomal MALAT1 from ox-LDL-treated vascular endothelial cells induces maturation of dendritic cells in atherosclerosis development. Cell Cycle. 2019;18:2255–2267. doi: 10.1080/15384101.2019.1642068. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 442.Huang C, et al. Exosomal MALAT1 derived from oxidized low-density lipoprotein-treated endothelial cells promotes M2 macrophage polarization. Mol. Med. Rep. 2018;18:509–515. doi: 10.3892/mmr.2018.8982. [DOI] [PubMed] [Google Scholar]
- 443.Gao H, et al. Exosomal MALAT1 derived from ox-LDL-treated endothelial cells induce neutrophil extracellular traps to aggravate atherosclerosis. Biol. Chem. 2020;401:367–376. doi: 10.1515/hsz-2019-0219. [DOI] [PubMed] [Google Scholar]
- 444.Yu Z, et al. CircRNAs open a new era in the study of cardiovascular disease (Review) Int. J. Mol. Med. 2021;47:49–64. doi: 10.3892/ijmm.2020.4792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Wen Y, et al. circRNA0006896miR1264DNMT1 axis plays an important role in carotid plaque destabilization by regulating the behavior of endothelial cells in atherosclerosis. Mol. Med. Rep. 2021;23:311. doi: 10.3892/mmr.2021.11950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 446.Feld S, Kjellgren O, Smalling RW. Aggressive interventional treatment of acute myocardial infarction. Lessons from the animal laboratory applied to the catheterization suite. Cardiology. 1995;86:365–373. doi: 10.1159/000176903. [DOI] [PubMed] [Google Scholar]
- 447.Ong SG, et al. Cross talk of combined gene and cell therapy in ischemic heart disease: role of exosomal microRNA transfer. Circulation. 2014;130:S60–S69. doi: 10.1161/CIRCULATIONAHA.113.007917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Shyu KG, et al. Hyperbaric oxygen-induced long non-coding RNA MALAT1 exosomes suppress MicroRNA-92a expression in a rat model of acute myocardial infarction. J. Cell Mol. Med. 2020;24:12945–12954. doi: 10.1111/jcmm.15889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 449.Huang P, et al. Atorvastatin enhances the therapeutic efficacy of mesenchymal stem cells-derived exosomes in acute myocardial infarction via up-regulating long non-coding RNA H19. Cardiovasc Res. 2020;116:353–367. doi: 10.1093/cvr/cvz139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Zhu LP, et al. Hypoxia-elicited mesenchymal stem cell-derived exosomes facilitates cardiac repair through miR-125b-mediated prevention of cell death in myocardial infarction. Theranostics. 2018;8:6163–6177. doi: 10.7150/thno.28021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 451.De Rosa S, et al. Transcoronary concentration gradients of circulating microRNAs. Circulation. 2011;124:1936–1944. doi: 10.1161/CIRCULATIONAHA.111.037572. [DOI] [PubMed] [Google Scholar]
- 452.Feng Y, et al. Ischemic preconditioning potentiates the protective effect of stem cells through secretion of exosomes by targeting Mecp2 via miR-22. PLoS ONE. 2014;9:e88685. doi: 10.1371/journal.pone.0088685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 453.Wang K, et al. Enhanced cardioprotection by human endometrium mesenchymal stem cells driven by exosomal microRNA-21. Stem Cells Transl. Med. 2017;6:209–222. doi: 10.5966/sctm.2015-0386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Li Y, et al. Exosomal miR-301 derived from mesenchymal stem cells protects myocardial infarction by inhibiting myocardial autophagy. Biochem Biophys. Res Commun. 2019;514:323–328. doi: 10.1016/j.bbrc.2019.04.138. [DOI] [PubMed] [Google Scholar]
- 455.Sahoo S, Losordo DW. Exosomes and cardiac repair after myocardial infarction. Circ. Res. 2014;114:333–344. doi: 10.1161/CIRCRESAHA.114.300639. [DOI] [PubMed] [Google Scholar]
- 456.Khan M, et al. Embryonic stem cell-derived exosomes promote endogenous repair mechanisms and enhance cardiac function following myocardial infarction. Circ. Res. 2015;117:52–64. doi: 10.1161/CIRCRESAHA.117.305990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Saparov A, et al. Role of the immune system in cardiac tissue damage and repair following myocardial infarction. Inflamm. Res. 2017;66:739–751. doi: 10.1007/s00011-017-1060-4. [DOI] [PubMed] [Google Scholar]
- 458.Peng Y, et al. Exosomal miR-25-3p from mesenchymal stem cells alleviates myocardial infarction by targeting pro-apoptotic proteins and EZH2. Cell Death Dis. 2020;11:317. doi: 10.1038/s41419-020-2545-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 459.Wen Z, et al. Mesenchymal stem cell-derived exosomes ameliorate cardiomyocyte apoptosis in hypoxic conditions through microRNA144 by targeting the PTEN/AKT pathway. Stem Cell Res Ther. 2020;11:36. doi: 10.1186/s13287-020-1563-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Wang W, et al. Exosomes derived miR-126 attenuates oxidative stress and apoptosis from ischemia and reperfusion injury by targeting ERRFI1. Gene. 2019;690:75–80. doi: 10.1016/j.gene.2018.12.044. [DOI] [PubMed] [Google Scholar]
- 461.Pan J, et al. Exosomes derived from miR-146a-modified adipose-derived stem cells attenuate acute myocardial infarction-induced myocardial damage via downregulation of early growth response factor 1. J. Cell Biochem. 2019;120:4433–4443. doi: 10.1002/jcb.27731. [DOI] [PubMed] [Google Scholar]
- 462.Chen Z, et al. Expression level and diagnostic value of exosomal NEAT1/miR-204/MMP-9 in acute ST-segment elevation myocardial infarction. IUBMB Life. 2020;72:2499–2507. doi: 10.1002/iub.2376. [DOI] [PubMed] [Google Scholar]
- 463.Wang L, Zhang J. Exosomal lncRNA AK139128 derived from hypoxic cardiomyocytes promotes apoptosis and inhibits cell proliferation in cardiac fibroblasts. Int J. Nanomed. 2020;15:3363–3376. doi: 10.2147/IJN.S240660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Liew CC, Dzau VJ. Molecular genetics and genomics of heart failure. Nat. Rev. Genet. 2004;5:811–825. doi: 10.1038/nrg1470. [DOI] [PubMed] [Google Scholar]
- 465.Melman YF, Shah R, Das S. MicroRNAs in heart failure: is the picture becoming less miRky? Circ. Heart Fail. 2014;7:203–214. doi: 10.1161/CIRCHEARTFAILURE.113.000266. [DOI] [PubMed] [Google Scholar]
- 466.Wang L, et al. Reduced exosome miR-425 and miR-744 in the plasma represents the progression of fibrosis and heart failure. Kaohsiung J. Med Sci. 2018;34:626–633. doi: 10.1016/j.kjms.2018.05.008. [DOI] [PubMed] [Google Scholar]
- 467.Wijffels MC, Kirchhof CJ, Dorland R, Allessie MA. Atrial fibrillation begets atrial fibrillation. a study in awake chronically instrumented goats. Circulation. 1995;92:1954–1968. doi: 10.1161/01.cir.92.7.1954. [DOI] [PubMed] [Google Scholar]
- 468.Ausma J, et al. Structural changes of atrial myocardium due to sustained atrial fibrillation in the goat. Circulation. 1997;96:3157–3163. doi: 10.1161/01.cir.96.9.3157. [DOI] [PubMed] [Google Scholar]
- 469.Liu L, et al. Exosomal miR-320d derived from adipose tissue-derived MSCs inhibits apoptosis in cardiomyocytes with atrial fibrillation (AF) Artif. Cells Nanomed. Biotechnol. 2019;47:3976–3984. doi: 10.1080/21691401.2019.1671432. [DOI] [PubMed] [Google Scholar]
- 470.Wang S, et al. Effects of atrial fibrillation-derived exosome delivery of miR-107 to human umbilical vein endothelial cells. DNA Cell Biol. 2021;40:568–579. doi: 10.1089/dna.2020.6356. [DOI] [PubMed] [Google Scholar]
- 471.Li S, et al. Myofibroblast-derived exosomes contribute to development of a susceptible substrate for atrial fibrillation. Cardiology. 2020;145:324–332. doi: 10.1159/000505641. [DOI] [PubMed] [Google Scholar]
- 472.Cogswell JP, et al. Identification of miRNA changes in Alzheimer’s disease brain and CSF yields putative biomarkers and insights into disease pathways. J. Alzheimers Dis. 2008;14:27–41. doi: 10.3233/jad-2008-14103. [DOI] [PubMed] [Google Scholar]
- 473.Faghihi MA, et al. Expression of a noncoding RNA is elevated in Alzheimer’s disease and drives rapid feed-forward regulation of beta-secretase. Nat. Med. 2008;14:723–730. doi: 10.1038/nm1784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 474.Hébert SS, et al. Loss of microRNA cluster miR-29a/b-1 in sporadic Alzheimer’s disease correlates with increased BACE1/beta-secretase expression. Proc. Natl Acad. Sci. USA. 2008;105:6415–6420. doi: 10.1073/pnas.0710263105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Packer AN, et al. The bifunctional microRNA miR-9/miR-9* regulates REST and CoREST and is downregulated in Huntington’s disease. J. Neurosci. 2008;28:14341–14346. doi: 10.1523/JNEUROSCI.2390-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 476.Saba R, et al. A miRNA signature of prion induced neurodegeneration. PLoS ONE. 2008;3:e3652. doi: 10.1371/journal.pone.0003652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 477.Sonntag K-C. MicroRNAs and deregulated gene expression networks in neurodegeneration. Brain Res. 2010;1338:48–57. doi: 10.1016/j.brainres.2010.03.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 478.Wang W-X, et al. The expression of microRNA miR-107 decreases early in Alzheimer’s disease and may accelerate disease progression through regulation of β-site amyloid precursor protein-cleaving enzyme 1. J. Neurosci. 2008;28:1213–1223. doi: 10.1523/JNEUROSCI.5065-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Sun B-L, et al. Clinical research on Alzheimer’s disease: progress and perspectives. Neurosci. Bull. 2018;34:1111–1118. doi: 10.1007/s12264-018-0249-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Chen J-J, et al. Exosome-encapsulated microRNAs as promising biomarkers for Alzheimer’s disease. Rev. Neurosci. 2019;31:77–87. doi: 10.1515/revneuro-2019-0001. [DOI] [PubMed] [Google Scholar]
- 481.Leidinger P, et al. A blood based 12-miRNA signature of Alzheimer disease patients. Genome Biol. 2013;14:R78. doi: 10.1186/gb-2013-14-7-r78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 482.Chen J-J, Zhao B, Zhao J, Li S. Potential roles of exosomal microRNAs as diagnostic biomarkers and therapeutic application in Alzheimer’s disease. Neural Plast. 2017;2017:7027380. doi: 10.1155/2017/7027380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 483.Miya Shaik M, et al. The Role of microRNAs in Alzheimer’s disease and their therapeutic potentials. Genes (Basel). 2018;9:174. doi: 10.3390/genes9040174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 484.Zhu H-C, et al. MicroRNA‐195 downregulates Alzheimer’s disease amyloid‐β production by targeting BACE1. Brain Res Bull. 2012;88:596–601. doi: 10.1016/j.brainresbull.2012.05.018. [DOI] [PubMed] [Google Scholar]
- 485.Absalon S, Kochanek DM, Raghavan V, Krichevsky AM. MiR‐26b, upregulated in Alzheimer’s disease, activates cell cycle entry, tau‐phosphorylation, and apoptosis in postmitotic neurons. J. Neurosci. 2013;33:14645–14659. doi: 10.1523/JNEUROSCI.1327-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Jian C, et al. miR-34a knockout attenuates cognitive deficits in APP/PS1 mice through inhibition of the amyloidogenic processing of APP. Life Sci. 2017;182:104–111. doi: 10.1016/j.lfs.2017.05.023. [DOI] [PubMed] [Google Scholar]
- 487.Xu Y, et al. miR-34a deficiency in APP/PS1 mice promotes cognitive function by increasing synaptic plasticity via AMPA and NMDA receptors. Neurosci. Lett. 2018;670:94–104. doi: 10.1016/j.neulet.2018.01.045. [DOI] [PubMed] [Google Scholar]
- 488.Sarkar S, et al. Expression of microRNA-34a in Alzheimer’s disease brain targets genes linked to synaptic plasticity, energy metabolism, and resting state network activity. Brain Res. 2016;1646:139–151. doi: 10.1016/j.brainres.2016.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 489.Liu C-G, Song J, Zhang Y-Q, Wang P-C. MicroRNA-193b is a regulator of amyloid precursor protein in the blood and cerebrospinal fluid derived exosomal microRNA-193b is a biomarker of Alzheimer’s disease. Mol. Med Rep. 2014;10:2395–2400. doi: 10.3892/mmr.2014.2484. [DOI] [PubMed] [Google Scholar]
- 490.Santos JR, Gois AM, Mendonça DMF, Freire MAM. Nutritional status, oxidative stress and dementia: The role of selenium in Alzheimer’s disease. Front Aging Neurosci. 2014;6:206. doi: 10.3389/fnagi.2014.00206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 491.Kanamaru T, et al. Oxidative stress accelerates amyloid deposition and memory impairment in a double-transgenic mouse model of Alzheimer’s disease. Neurosci. Lett. 2015;587:126–131. doi: 10.1016/j.neulet.2014.12.033. [DOI] [PubMed] [Google Scholar]
- 492.Kamat PK, et al. Mechanism of oxidative stress and synapse dysfunction in the pathogenesis of Alzheimer’s disease: Understanding the therapeutics strategie. Mol. Neurobiol. 2016;53:648–661. doi: 10.1007/s12035-014-9053-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 493.Cai Z-Y, Xiao M, Quazi SH, Ke Z-Y. Exosomes: a novel therapeutic target for Alzheimer’s disease. Neural Regen. Res. 2018;13:930–935. doi: 10.4103/1673-5374.232490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 494.Lugli G, et al. Plasma exosomal miRNAs in persons with and without Alzheimer disease: altered expression and prospects for biomarkers. PLoS ONE. 2015;10:e0139233. doi: 10.1371/journal.pone.0139233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 495.Gayen M, Bhomia M, Balakathiresan N, Knollmann-Ritschel B. Exosomal microRNAs released by activated astrocytes as potential neuroinflammatory biomarkers. Int J. Mol. Sci. 2020;21:2312. doi: 10.3390/ijms21072312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 496.Ji J, et al. Mitochondria-related miR-141-3p contributes to mitochondrial dysfunction in HFD-induced obesity by inhibiting PTEN. Sci. Rep. 2015;5:16262. doi: 10.1038/srep16262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 497.McKeever PM, et al. MicroRNA expression levels are altered in the cerebrospinal fluid of patients with young-onset Alzheimer’s disease. Mol. Neurobiol. 2018;55:8826–8841. doi: 10.1007/s12035-018-1032-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 498.Banzhaf-Strathmann J, et al. MicroRNA-125b induces tau hyperphosphorylation and cognitive deficits in Alzheimer’s disease. EMBO J. 2014;33:1667–1680. doi: 10.15252/embj.201387576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 499.Shen Y, et al. MiR-125b-5p is involved in oxygen and glucose deprivation injury in PC-12 cells via CBS/H 2 S pathway. Nitric Oxide. 2018;78:11–21. doi: 10.1016/j.niox.2018.05.004. [DOI] [PubMed] [Google Scholar]
- 500.Fotuhi SN, Khalaj-Kondori M, Hoseinpour Feizi MA, Talebi M. Long non-coding RNA BACE1-AS may serve as an Alzheimer’s disease blood-based biomarker. J. Mol. Neurosci. 2019;69:351–359. doi: 10.1007/s12031-019-01364-2. [DOI] [PubMed] [Google Scholar]
- 501.Kalia LV, Lang AE. Parkinson’s disease. Lancet. 2015;386:896–912. doi: 10.1016/S0140-6736(14)61393-3. [DOI] [PubMed] [Google Scholar]
- 502.Pagonabarraga J, Kulisevsky J, Strafella AP, Krack P. Apathy in Parkinson’s disease: clinical features, neural substrates, diagnosis, and treatment. Lancet Neurol. 2015;14:518–531. doi: 10.1016/S1474-4422(15)00019-8. [DOI] [PubMed] [Google Scholar]
- 503.Mirzaei H, et al. Diagnostic and therapeutic potential of exosomes in cancer: the beginning of a new tale? J. Cell Physiol. 2017;232:3251–3260. doi: 10.1002/jcp.25739. [DOI] [PubMed] [Google Scholar]
- 504.Li D, et al. Effect of regulatory network of exosomes and microRNAs on neurodegenerative diseases. Chin. Med J. (Engl.) 2018;131:2216–2225. doi: 10.4103/0366-6999.240817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 505.Saeedi Borujeni MJ, et al. Molecular aspects of diabetes mellitus: resistin, microRNA, and exosome. J. Cell Biochem. 2018;119:1257–1272. doi: 10.1002/jcb.26271. [DOI] [PubMed] [Google Scholar]
- 506.Pourhanifeh MH, et al. MicroRNAs and exosomes: small molecules with big actions in multiple myeloma pathogenesis. IUBMB Life. 2020;72:314–333. doi: 10.1002/iub.2211. [DOI] [PubMed] [Google Scholar]
- 507.Harischandra DS, et al. Environmental neurotoxicant manganese regulates exosome-mediated extracellular miRNAs in cell culture model of Parkinson’s disease: relevance to α-synuclein misfolding in metal neurotoxicity. Neurotoxicology. 2018;64:267–277. doi: 10.1016/j.neuro.2017.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 508.Gui Y, et al. Altered microRNA profiles in cerebrospinal fluid exosome in Parkinson disease and Alzheimer disease. Oncotarget. 2015;6:37043–37053. doi: 10.18632/oncotarget.6158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 509.Asikainen S, et al. Global microRNA expression profiling of Caenorhabditis elegans Parkinson’s disease models. J. Mol. Neurosci. 2010;41:210–218. doi: 10.1007/s12031-009-9325-1. [DOI] [PubMed] [Google Scholar]
- 510.Leggio L, et al. microRNAs in Parkinson’s disease: from pathogenesis to novel diagnostic and therapeutic approaches. Int J. Mol. Sci. 2017;18:2698. doi: 10.3390/ijms18122698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Winkler CW, Taylor KG, Peterson KE. Location is everything: let-7b microRNA and TLR7 signaling results in a painful TRP. Sci. Signal. 2014;7:pe14. doi: 10.1126/scisignal.2005407. [DOI] [PubMed] [Google Scholar]
- 512.Shamsuzzama Kumar,L, Nazir A. Modulation of alpha-synuclein expression and associated effects by MicroRNA Let-7 in transgenic C. elegans. Front Mol. Neurosci. 2017;10:328. doi: 10.3389/fnmol.2017.00328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 513.Cao X-Y, et al. MicroRNA biomarkers of Parkinson’s disease in serum exosome-like microvesicles. Neurosci. Lett. 2017;644:94–99. doi: 10.1016/j.neulet.2017.02.045. [DOI] [PubMed] [Google Scholar]
- 514.Yao YF, et al. Circulating exosomal miRNAs as diagnostic biomarkers in Parkinson’s disease. Eur. Rev. Med Pharm. Sci. 2018;22:5278–5283. doi: 10.26355/eurrev_201808_15727. [DOI] [PubMed] [Google Scholar]
- 515.Quinlan S, et al. MicroRNAs in neurodegenerative diseases. Int Rev. Cell Mol. Biol. 2017;334:309–343. doi: 10.1016/bs.ircmb.2017.04.002. [DOI] [PubMed] [Google Scholar]
- 516.Li N, et al. Plasma levels of miR-137 and miR-124 are associated with Parkinson’s disease but not with Parkinson’s disease with depression. Neurol. Sci. 2017;38:761–767. doi: 10.1007/s10072-017-2841-9. [DOI] [PubMed] [Google Scholar]
- 517.Jiang Y, et al. Serum secreted miR-137-containing exosomes affects oxidative stress of neurons by regulating OXR1 in Parkinson’s disease. Brain Res. 2019;1722:146331. doi: 10.1016/j.brainres.2019.146331. [DOI] [PubMed] [Google Scholar]
- 518.Wang Q, et al. Integrated analysis of exosomal lncRNA and mRNA expression profiles reveals the involvement of lnc-MKRN2-42:1 in the pathogenesis of Parkinson’s disease. CNS Neurosci. Ther. 2020;26:527–537. doi: 10.1111/cns.13277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Zou J, et al. Long noncoding RNA POU3F3 and α-synuclein in plasma L1CAM exosomes combined with β-glucocerebrosidase activity: potential predictors of Parkinson’s disease. Neurotherapeutics. 2020;17:1104–1119. doi: 10.1007/s13311-020-00842-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 520.Elkouris M, et al. Long non-coding RNAs associated with neurodegeneration-linked genes are reduced in Parkinson’s Disease patients. Front Cell Neurosci. 2019;13:58. doi: 10.3389/fncel.2019.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.Scott DL, Wolfe F, Huizinga TWJ. Rheumatoid arthritis. Lancet. 2010;376:1094–1108. doi: 10.1016/S0140-6736(10)60826-4. [DOI] [PubMed] [Google Scholar]
- 522.Karsdal MA, et al. Cartilage degradation is fully reversible in the presence of aggrecanase but not matrix metalloproteinase activity. Arthritis Res Ther. 2008;10:R63. doi: 10.1186/ar2434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 523.Wang C-h, et al. CD147 induces angiogenesis through a vascular endothelial growth factor and hypoxia-inducible transcription factor 1α-mediated pathway in rheumatoid arthritis. Arthritis Rheum. 2012;64:1818–1827. doi: 10.1002/art.34341. [DOI] [PubMed] [Google Scholar]
- 524.Sounni NE, et al. Stromal regulation of vessel stability by MMP14 and TGFbeta. Dis. Model Mech. 2010;3:317–332. doi: 10.1242/dmm.003863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 525.Ghisi M, et al. Modulation of microRNA expression in human T-cell development: targeting of NOTCH3 by miR-150. Blood. 2011;117:7053–7062. doi: 10.1182/blood-2010-12-326629. [DOI] [PubMed] [Google Scholar]
- 526.Li J, et al. Microvesicle-mediated transfer of microRNA-150 from monocytes to endothelial cells promotes angiogenesis. J. Biol. Chem. 2013;288:23586–23596. doi: 10.1074/jbc.M113.489302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 527.Chen Z, et al. Therapeutic potential of mesenchymal cell-derived miRNA-150-5p-expressing exosomes in rheumatoid arthritis mediated by the modulation of MMP14 and VEGF. J. Immunol. 2018;201:2472–2482. doi: 10.4049/jimmunol.1800304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 528.Wang Y, et al. MiR-548a-3p regulates inflammatory response via TLR4/NF-κB signaling pathway in rheumatoid arthritis. J Cell Biochem. 2018;120:1113–1140. doi: 10.1002/jcb.26659. [DOI] [PubMed] [Google Scholar]
- 529.Song J, et al. PBMC and exosome-derived Hotair is a critical regulator and potent marker for rheumatoid arthritis. Clin. Exp. Med. 2015;15:121–126. doi: 10.1007/s10238-013-0271-4. [DOI] [PubMed] [Google Scholar]
- 530.Long H, et al. Dysregulation of microRNAs in autoimmune diseases: pathogenesis, biomarkers and potential therapeutic targets. Cancer Lett. 2018;428:90–103. doi: 10.1016/j.canlet.2018.04.016. [DOI] [PubMed] [Google Scholar]
- 531.Le X, Yu X, Shen N. Novel insights of microRNAs in the development of systemic lupus erythematosus. Curr. Opin. Rheumatol. 2017;29:450–457. doi: 10.1097/BOR.0000000000000420. [DOI] [PubMed] [Google Scholar]
- 532.Salvi V, et al. Exosome-delivered microRNAs promote IFN-α secretion by human plasmacytoid DCs via TLR7. JCI Insight. 2018;3:e98204. doi: 10.1172/jci.insight.98204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 533.Wang P, et al. Inducible microRNA-155 feedback promotes type I IFN signaling in antiviral innate immunity by targeting suppressor of cytokine signaling 1. J. Immunol. 2010;185:6226–6233. doi: 10.4049/jimmunol.1000491. [DOI] [PubMed] [Google Scholar]
- 534.Aboelenein HR, et al. Reduction of CD19 autoimmunity marker on B cells of paediatric SLE patients through repressing PU.1/TNF-α/BAFF axis pathway by miR-155. Growth Factors. 2017;35:49–60. doi: 10.1080/08977194.2017.1345900. [DOI] [PubMed] [Google Scholar]
- 535.Tyrrell DJ, et al. Age-associated mitochondrial dysfunction accelerates atherogenesis. Circ. Res. 2020;126:298–314. doi: 10.1161/CIRCRESAHA.119.315644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 536.Xin Q, et al. miR-155 deficiency ameliorates autoimmune inflammation of systemic lupus erythematosus by targeting S1pr1 in Faslpr/lpr mice. J. Immunol. 2015;194:5437–5445. doi: 10.4049/jimmunol.1403028. [DOI] [PubMed] [Google Scholar]
- 537.Tang ZM, et al. Clinical relevance of plasma miR-21 in new-onset systemic lupus erythematosus patients. J. Clin. Lab Anal. 2014;28:446–451. doi: 10.1002/jcla.21708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 538.Stagakis E, et al. Identification of novel microRNA signatures linked to human lupus disease activity and pathogenesis: miR-21 regulates aberrant T cell responses through regulation of PDCD4 expression. Ann. Rheum. Dis. 2011;70:1496–1506. doi: 10.1136/ard.2010.139857. [DOI] [PubMed] [Google Scholar]
- 539.Tan L, et al. Downregulated serum exosomal miR-451a expression correlates with renal damage and its intercellular communication role in systemic lupus erythematosus. Front Immunol. 2021;12:630112. doi: 10.3389/fimmu.2021.630112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 540.Dong C, et al. Circulating exosomes derived-miR-146a from systemic lupus erythematosus patients regulates senescence of mesenchymal stem cells. Biomed. Res. Int. 2019;2019:6071308. doi: 10.1155/2019/6071308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 541.Perez-Hernandez, J. et al. Urinary exosomal miR-146a as a marker of albuminuria, activity changes and disease flares in lupus nephritis. J Nephrol. -, Aug 16. Online ahead of print, (2020). [DOI] [PubMed]
- 542.Tsokos GC, Lo MS, Costa Reis P, Sullivan KE. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat. Rev. Rheumatol. 2016;12:716–730. doi: 10.1038/nrrheum.2016.186. [DOI] [PubMed] [Google Scholar]
- 543.Santiago-Dieppa DR, et al. Extracellular vesicles as a platform for ‘liquid biopsy’ in glioblastoma patients. Expert Rev. Mol. Diagn. 2014;14:819–825. doi: 10.1586/14737159.2014.943193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 544.Chun-Yan L, et al. Liquid biopsy biomarkers of renal interstitial fibrosis based on urinary exosome. Exp. Mol. Pathol. 2018;105:223–228. doi: 10.1016/j.yexmp.2018.08.004. [DOI] [PubMed] [Google Scholar]
- 545.Gholaminejad A, Abdul Tehrani H, Gholami Fesharaki M. Identification of candidate microRNA biomarkers in renal fibrosis: a meta-analysis of profiling studies. Biomarkers. 2018;23:713–724. doi: 10.1080/1354750X.2018.1488275. [DOI] [PubMed] [Google Scholar]
- 546.Perez-Hernandez J, et al. Increased urinary exosomal micrornas in patients with systemic lupus erythematosus. PLoS ONE. 2015;10:e0138618. doi: 10.1371/journal.pone.0138618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 547.Zhou H, et al. miR-150 promotes renal fibrosis in lupus nephritis by downregulating SOCS1. J. Am. Soc. Nephrol. 2013;24:1073–1087. doi: 10.1681/ASN.2012080849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 548.Liu D, et al. miR-410 suppresses the expression of interleukin-6 as well as renal fibrosis in the pathogenesis of lupus nephritis. Clin. Exp. Pharm. Physiol. 2016;43:616–625. doi: 10.1111/1440-1681.12576. [DOI] [PubMed] [Google Scholar]
- 549.Solé C, et al. miR-29c in urinary exosomes as predictor of early renal fibrosis in lupus nephritis. Nephrol. Dial. Transpl. 2015;30:1488–1496. doi: 10.1093/ndt/gfv128. [DOI] [PubMed] [Google Scholar]
- 550.Tangtanatakul P, et al. Down-regulation of let-7a and miR-21 in urine exosomes from lupus nephritis patients during disease flare. Asian Pac. J. Allergy Immunol. 2019;37:189–197. doi: 10.12932/AP-130318-0280. [DOI] [PubMed] [Google Scholar]
- 551.Hashemian SM, et al. Non-coding RNAs and exosomes: their role in the pathogenesis of sepsis. Mol. Ther. Nucleic Acids. 2020;21:51–74. doi: 10.1016/j.omtn.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 552.Wu X, et al. Serum exosomal MicroRNAs predict acute respiratory distress syndrome events in patients with severe community-acquired pneumonia. Biomed. Res. Int. 2019;2019:3612020. doi: 10.1155/2019/3612020. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 553.Huang F, et al. Identification of potential diagnostic biomarkers for pneumonia caused by adenovirus infection in children by screening serum exosomal microRNAs. Mol. Med Rep. 2019;19:4306–4314. doi: 10.3892/mmr.2019.10107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 554.Torres A, et al. Challenges in severe community-acquired pneumonia: a point-of-view review. Intensive Care Med. 2019;45:159–171. doi: 10.1007/s00134-019-05519-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 555.Jiang K, et al. Peripheral circulating exosome-mediated delivery of miR-155 as a novel mechanism for acute lung inflammation. Mol. Ther. 2019;27:1758–1771. doi: 10.1016/j.ymthe.2019.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 556.Quan Y, et al. Exosome miR-371b-5p promotes proliferation of lung alveolar progenitor type II cells by using PTEN to orchestrate the PI3K/Akt signaling. Stem Cell Res Ther. 2017;8:138. doi: 10.1186/s13287-017-0586-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 557.Zhu Z, et al. Macrophage-derived apoptotic bodies promote the proliferation of the recipient cells via shuttling microRNA-221/222. J. Leukoc. Biol. 2017;101:1349–1359. doi: 10.1189/jlb.3A1116-483R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 558.Zhang D, et al. A potential role of microvesicle-containing miR-223/142 in lung inflammation. Thorax. 2019;74:865–874. doi: 10.1136/thoraxjnl-2018-212994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 559.Li R, Liang P, Yuan J, He F. Exosomal miR-103a-3p ameliorates lipopolysaccharide-induced immune response in BEAS-2B cells via NF-kappaB pathway by targeting transducin beta-like 1X related protein 1. Clin. Exp. Pharm. Physiol. 2020;47:620–627. doi: 10.1111/1440-1681.13241. [DOI] [PubMed] [Google Scholar]
- 560.Jiao Y, et al. Advances on liver cell-derived exosomes in liver diseases. J. Cell Mol. Med. 2021;25:15–26. doi: 10.1111/jcmm.16123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 561.Khatun M, Ray RB. Mechanisms underlying hepatitis C virus-associated hepatic fibrosis. Cells. 2019;8:1249. doi: 10.3390/cells8101249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 562.Bandopadhyay M, Bharadwaj M. Exosomal miRNAs in hepatitis B virus related liver disease: a new hope for biomarker. Gut Pathog. 2020;12:23. doi: 10.1186/s13099-020-00353-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 563.Li R, et al. Expression profiles of the exosomal miRNAs in the chronic hepatitis B patients with persistently normal ALT. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2018;43:475–480. doi: 10.11817/j.issn.1672-7347.2018.05.003. [DOI] [PubMed] [Google Scholar]
- 564.Devhare PB, et al. xosome-mediated intercellular communication between hepatitis C virus-infected hepatocytes and hepatic stellate Cells. J Virol. 2017;91:e02225–16. doi: 10.1128/JVI.02225-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 565.Thakuri BKC, et al. HCV-Associated exosomes upregulate RUNXOR and RUNX1 expressions to promote MDSC expansion and suppressive functions through STAT3-miR124 Axis. Cells. 2020;9:2715. doi: 10.3390/cells9122715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 566.Liu, C., Gao, Y., Wu, J. & Zou, J. Exosomal miR-23a and miR-192, potential diagnostic biomarkers for Type 2 diabetes. Clin. Laboratory. 67, 10.7754/clin.lab.2020.200612 (2021). [DOI] [PubMed]
- 567.Fu Q, et al. Injury factors alter miRNAs profiles of exosomes derived from islets and circulation. Aging. 2018;10:3986–3999. doi: 10.18632/aging.101689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 568.Shao J-L, et al. Identification of serum exosomal MicroRNA expression profiling in menopausal females with osteoporosis by high-throughput sequencing. Curr. Med. Sci. 2020;40:1161–1169. doi: 10.1007/s11596-020-2306-x. [DOI] [PubMed] [Google Scholar]
- 569.Karolina DS, et al. Circulating miRNA profiles in patients with metabolic syndrome. J. Clin. Endocrinol. Metab. 2012;97:E2271–E2276. doi: 10.1210/jc.2012-1996. [DOI] [PubMed] [Google Scholar]
- 570.Mendt, M. et al. Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI Insight. 3, e99263 (2018). [DOI] [PMC free article] [PubMed]
- 571.Alvarez-Erviti L, et al. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011;29:341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 572.Tang TT, Wang B, Lv LL, Liu BC. Extracellular vesicle-based nanotherapeutics: emerging frontiers in anti-inflammatory therapy. Theranostics. 2020;10:8111–8129. doi: 10.7150/thno.47865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 573.Lv Q, et al. Engineered human adipose stem-cell-derived exosomes loaded with miR-21-5p to promote diabetic cutaneous wound healing. Mol. pharmaceutics. 2020;17:1723–1733. doi: 10.1021/acs.molpharmaceut.0c00177. [DOI] [PubMed] [Google Scholar]
- 574.Kim G, et al. Systemic delivery of microRNA-21 antisense oligonucleotides to the brain using T7-peptide decorated exosomes. J. Control Release. 2020;317:273–281. doi: 10.1016/j.jconrel.2019.11.009. [DOI] [PubMed] [Google Scholar]
- 575.Cheng L, et al. Characterization and deep sequencing analysis of exosomal and non-exosomal miRNA in human urine. Kidney Int. 2014;86:433–444. doi: 10.1038/ki.2013.502. [DOI] [PubMed] [Google Scholar]
- 576.Nik Mohamed Kamal N, Shahidan WNS. Non-exosomal and exosomal circulatory MicroRNAs: which are more valid as biomarkers? Front Pharm. 2019;10:1500. doi: 10.3389/fphar.2019.01500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 577.Zhao C, Busch DJ, Vershel CP, Stachowiak JC. Multifunctional transmembrane protein ligands for cell-specific targeting of plasma membrane-derived vesicles. Small. 2016;12:3837–3848. doi: 10.1002/smll.201600493. [DOI] [PMC free article] [PubMed] [Google Scholar]