Abstract
Glioblastoma Multiforme (GBM) is the most common primary central nervous system (CNS) malignancy in adults. It is very aggressive and is notorious for its fast and local invasion of nearby brain parenchyma. Consequently, the overall survival (OS) of patients with GBM is short despite resection, radiotherapy and chemotherapy regimens. The most common sites of metastasis of GBM are the lungs and pleura, cervical lymph nodes, and bone. Metastasis to the skin is a rare event and to our knowledge, there are less than 30 cases of GBM metastasizing to cutaneous or subcutaneous tissue described in the literature. None of these cases were diagnosed and/or treated in the Middle East region; and the majority of the metastases found were adjacent to the site of surgery undergone to remove the primary malignancy.
We present the case of a 53-year-old male diagnosed with GBM and later showing signs of metastases at the anterio-auricular side of his face near-distant from the site of previous surgery done to remove the primary tumor.
Keywords: CNS (Central Nervous System), Glioblastoma Multiforme (GBM), Metastasis, Skin
Introduction
Glioblastoma (GBM) is known to be the most ubiquitous primary central nervous system tumor in the adult population, representing up to 25% of these tumors [1]. It's a malignant proliferation of astrocytes that carries a very high relapse and mortality rate and a median overall survival (OS) of 20 months regardless of the modality of treatment or surgeries used to eradicate it such as surgery, chemotherapy, and radiotherapy [2,3]. GBM being a very invasive tumor has the potential to rapidly occupy brain parenchyma around the primary site. Nonetheless, it's not very common for this tumor to spread to the bloodstream and beyond the central nervous system [4]. In the literature, the lungs and pleura were found to be the most common site of extra-cranial metastasis, accounting for 60% of reported cases [5],[17]. The next most frequent sites are the cervical lymph nodes described in 51% of cases [6], the bone marrow and bones seen in 30% of cases [5], the liver seen in 22% of cases [7] and skull[10] [24]. The spread to other organs including the skin is extremely rare [8],[13] and there are fewer than 30 cases of GBM cutaneous and/or subcutaneous metastases reported in the literature with the most common location of metastasis being the skull [8], one of them occurring in a child [9]. The majority of this patient population were found to have a skin metastasis at the exact site of previous surgery done to remove the primary tumor[15], whereas fewer patients have a more distant location [8], and none of these patients reside in the Middle East region.
In this report, we present the clinical, and pathologic features of the first case of GBM with metastasis to skin described in the Middle East region with the location of metastasis being distant to the site of primary surgery.
Case presentation
The patient is a 53-year-old Lebanese male who presented with left sided lower extremity weakness and heaviness. MRI of the brain showed ring enhancing lesion with a central enhancing solid component centered in the posterior limb of the right internal capsule and right thalamus measuring 2.4 × 2.4 × 2.3 cm (Fig. 1, Fig. 2). A biopsy was done and the patient was found to have a high-grade glioma (at least anaplastic astrocytoma WHO grade III) diffusely positive for GFAP and p53 with focal Olig2 expression and a high Ki-67 proliferation index. A Positron Emission Tomography (PET) scan showed a hypodense mass in the right cerebral hemisphere centered in the right internal capsule and thalamus, with decreased FDG uptake, concerning for a primary malignancy. One month later, he underwent subtotal resection of the mass and the pathology revealed IDH-wild type glioblastoma (WHO grade IV) with no residual disease. He then received concurrent radiation (60 Gy) and chemotherapy (Temozolomide) followed by adjuvant Temozolomide. Five months later, an MRI of the brain showed interval appearance of nodular enhancement along the medial aspect of the surgical cavity measuring 1.2 × 1.1 cm and interval appearance of a 1.5 × 1.2 nodular focus of enhancement in the right superior frontal gyrus with significant high FLAIR signal in the surrounding white matter, likely in part related to tumor infiltration and in part to vasogenic edema; as well as another adjacent tiny focus of enhancement in the right frontal lobe measuring 1 × 0.4 cm. The patient was subsequently started on Bevacizumab -later put on hold after minor bleeding- as well as temozolomide. An MRI of the brain 4 months later showed interval increase in the nodular enhancement in the surgical cavity and surrounding high FLAIR signal in keeping with tumor progression. Bevacizumab was then restarted, temozolomide was discontinued and the patient was shifted to lomustine. MRI of the brain was done 2 months later and showed increase in the size of the patient's known tumor as well as in the mass effect on the right cerebral hemisphere with associated midline shift and no herniation. Moreover, multiple new foci of increased FLAIR signal were evident on the left hemisphere that were indicative of disease progression. The patient also had a hard bulge on the right anterio-auricular side of his face on follow up with his oncologist who shifted him to irinotecan and bevacizumab. A month later, the patient underwent biopsy of that anterio-auricular lesion which was consistent with gliosacroma (with the following positive markers: Vimentin and SMA, S-100 and Desmin focally, Ki-67 60%; and the following characteristics on histology: proliferation of malignant spindle to oval cells with hyperchromatic pleiomorphic nuclei portrayed in Fig. 3). The patient's case was then raised to the tumor board of the hospital, and it was decided that the patient would undergo palliative treatment and receive bevacizumab since it was not possible for him to receive regorafenib at the time. Two months later, the patient was admitted to the hospital due to respiratory difficulty and decreased level of consciousness; brain imaging findings were suspicious of an acute stroke and showed disease progression with multifocal GBM as well as increase in the size of the lesion in the subcutaneous soft tissues overlying the right temporal bones. His hospital course was complicated by seizures, bacteremia, and multiple pneumonias (including resistant strains of Klebsiella and Acinetobacter) as well as sepsis requiring pressors. He eventually required intubation, mechanical ventilation, and tracheostomy placement. He subsequently had asystole and passed away a bit more than one month after being hospitalized.
Fig. 1.
MRI brain at diagnosis: T1 post-Gadolinium.
Fig. 2.
MRI brain at diagnosis: T2 FLAIR.
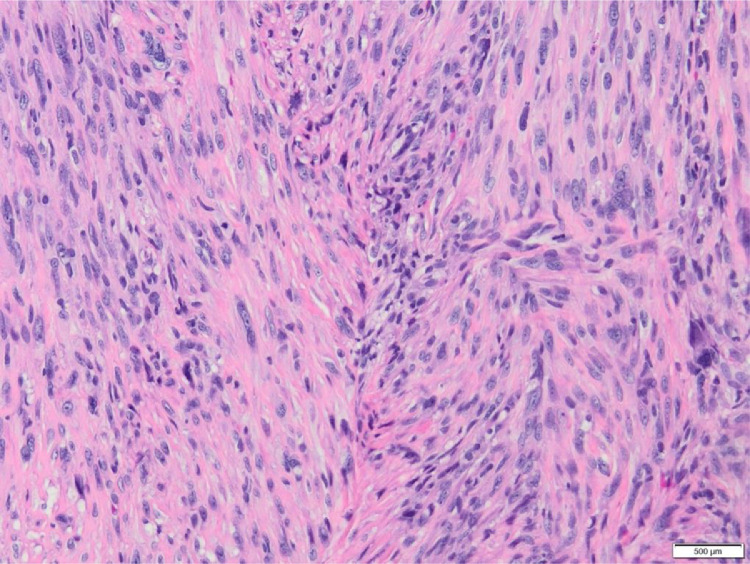
Fig. 3.
Histology section of the skin metastasis showing proliferation of malignant spindle to oval cells with hyperchromatic pleiomorphic nuclei. (H&E x 200).
Ring enhancing lesion with a central enhancing solid component centered in the posterior limb of the right internal capsule and right thalamus measuring 2.4 × 2.4 × 2.3 cm in the AP, transverse, and craniocaudal dimensions.
There is secondary 2 mm leftward midline shift.
Significant peri-lesional high FLAIR signal extending inferiorly to the right cerebral peduncle and involving the right optic tract and right aspect of the optic chiasm, also extending medially and significantly involving the right thalamus.
Discussion
Glioblastoma Multiforme, although very aggressive, does not have a propensity to metastasize to the skin, and soft tissue [4]. There have been less than 30 cases of GBM spreading to the skin in the literature and none of the adult patients described in these cases originated from the Middle East region. Most of these patients were adults aged 19 and above, and there was one patient aged 13.5 [9].
It's perplexing how such an aggressive tumor doesn't have a high tendency to metastasize outside the central nervous system [6]. A theory behind the scarcity of GBM cases metastasizing to the skin is the lethality of primary GBM leading to early death, thus not enough time to develop metastases [20]. It's also possible that the tumor cannot spread outside the CNS due to the blood brain barrier, strong basement membrane, and robust dura as well as the deprived lymphatic system in the brain [6].
A meta-analysis conducted by Pietschmann et al.[27] reviewing publications about GBM with extra-cranial metastasis noted an overall increased frequency of these cases. This was possibly attributed to better awareness in the medical field, improvement in medical diagnostic tools [26], and longer survival of GBM patients with improvement of treatment modalities. It was also observed in one of the studies that the cumulative effect of surgery, chemotherapy, radiotherapy as well as cerebrospinal fluid (CSF) shunting showed the longest average survival from time of metastasis detection to time of death [27]. This meta-analysis also distinguished that younger patients potentially have a tendency for extra-cerebrospinal metastasis due to longer survival [27].
Patients diagnosed with cutaneous or subcutaneous GBM invasion have seen a wide OS range (2 weeks-1 year) [4], and there haven't been studies focused on finding optimal treatment for such cases. As seen in the literature, these patients were either treated by surgical removal of the metastasis alone or in adjunction with chemotherapy and/or radiotherapy. Some cases have avoided surgery at the site of metastasis altogether and have focused on chemotherapy and/or local radiotherapy of the region [11]. Most patients in this category showed evidence of skin metastasis concomitantly with progression of primary CNS disease as evident in MRI reports showing increased size of primary tumor or increased edema around the mass. Similarly, most patients with GBM and skin metastasis described in the literature as well as our patient, developed skin metastasis at the site of previous surgery done to remove the primary tumor or around the facial region. Two cases described scalp metastasis along the area of stereotactic biopsy previously done for diagnostic purposes [5,11]. This implies that there may be iatrogenic tumor cell seeding or leakage along the procedure tract outward to the subcutaneous and cutaneous tissues [4,20]. Other proposed hypotheses are: glial defect formed after surgery ([6],[22]), extension of the primary tumor [20], post-operative neovascularization contributing to dissemination [18], or perineural invasion [14]. There's also the possibility of spread via the external jugular vein [16], and that the primitive neuronal component within primary tumor has a tendency to disseminate via CSF [4].
Interestingly, metastasis at the scalp or anywhere around the initial surgical site happens despite previous local radiotherapy administration, implying that radiation around the surgery scar does not confer a protective factor against future local cutaneous or subcutaneous invasion of the tumor. One can argue that radiation to the cutaneous and subcutaneous tissue is at a subtherapeutic dose, but increasing the dose is controversial due to the increased risk of toxicity to the area.
Furthermore, a review paper submitted by Sundahl et al [28] highlighted pre-clinical evidence showing pro-metastatic effects of radiotherapy through angiogenesis, motility, and invasion. However, this occurrence has not been confirmed clinically, and the long-term benefits of radiation therapy outweigh its negative effects and more data is still needed regarding this matter [28].
On the other hand, some papers have reported cutaneous or subcutaneous metastases that are distant from primary surgery site [12,21,23], and similarly to other GMB extracranial metastases, this could indicate hematogenic or lymphatic spread [19].
Conclusion
There are very few cases of Glioblastoma Multiforme metastasizing to extracranial locations and only a minor subset of them represents the subcutaneous and/or cutaneous tissue metastases. The scarcity of extracranial metastases might be due to the aggressive nature of the disease leading to early death of patients.
We described the case of a 53-year-old male diagnosed with GBM and later showing signs of metastases at the anterio-auricular side of his face. Our patient shares many similarities with the majority of the cased described in the literature like the appearance of the skin metastasis concomitantly with CNS disease progression, the combination of surgery and radiotherapy and/or chemotherapy to eradicate the primary tumor and the location of the metastasis at or near the previous site of surgery to remove the primary CNS tumor. This may indicate the role of tumor seeding along the procedure tract outward to the skin, glial defect formed after surgery, extension of primary tumor, perineural invasion or post-operative neovascularization possibly due to radiation therapy side effects. However, these hypotheses wouldn't explain the few cases of skin metastases that are distant to previous surgery sites and distant to the craniofacial area, which might be due to hematogenic or lymphatic spread. Therefore, more research should go into investigating the mechanism behind GBM tumors metastasizing to the cutaneous and/or subcutaneous tissues.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for profit sector.
Ethics approval
the authors complied with the ethical requirements of their institution
Patient consent
Since the patient was deceased, no consent was taken from the patient to proceed with this case report.
Footnotes
Competing Interests: The authors have no conflicts of interest to declare that are relevant to the content of this article.
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.radcr.2021.10.029.
Appendix. Supplementary materials
References
- 1.Bauchet L, Mathieu-Daudé H, Fabbro-Peray P, Rigau V, Fabbro M, Chinot O, et al. Oncological patterns of care and outcome for 952 patients with newly diagnosed glioblastoma in 2004. Neuro Oncol. 2010;12(7):725–735. doi: 10.1093/neuonc/noq030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jackson C, Choi J, Khalafallah AM, Price C, Bettegowda C, Lim M, et al. A systematic review and meta-analysis of supratotal versus gross total resection for glioblastoma. Journal of Neuro-Oncology. 2020;148(3):419–431. doi: 10.1007/s11060-020-03556-y. [DOI] [PubMed] [Google Scholar]
- 3.Socha J, Kepka L, Ghosh S, Roa W, Kumar N, Sinaika V, et al. Outcome of treatment of recurrent glioblastoma multiforme in elderly and/or frail patients. Journal of Neuro-Oncology. 2016;126(3):493–498. doi: 10.1007/s11060-015-1987-7. [DOI] [PubMed] [Google Scholar]
- 4.Lewis GD, Rivera AL, Tremont-Lukats IW, Ballester-Fuentes LY, Zhang YJ, Teh BS. GBM skin metastasis: a case report and review of the literature. CNS Oncol. 2017;6(3):203–209. doi: 10.2217/cns-2016-0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Houston SC, Crocker IR, Brat DJ, Olson JJ. Extraneural metastatic glioblastoma after interstitial brachytherapy. Int J Radiat Oncol Biol Phys. 2000;48(3):831–836. doi: 10.1016/s0360-3016(00)00662-3. [DOI] [PubMed] [Google Scholar]
- 6.Wallace CJ, Forsyth PA, Edwards DR. Lymph node metastases from glioblastoma multiforme. AJNR Am J Neuroradiol. 1996;17(10):1929–1931. [PMC free article] [PubMed] [Google Scholar]
- 7.Astner ST, Pihusch R, Nieder C, Rachinger W, Lohner H, Tonn JC, et al. Extensive local and systemic therapy in extraneural metastasized glioblastoma multiforme. Anticancer Res. 2006;26(6c):4917–4920. [PubMed] [Google Scholar]
- 8.Senetta R, Cassoni P. In: Tumors of the Central Nervous System. Volume 2: Gliomas: Glioblastoma (Part 2) Hayat MA, editor. Springer Science & Business Media; Dordrecht Heidelberg London New York: 2011. Skin metastases of glioblastoma; pp. 143–149. [Google Scholar]
- 9.Saad AG, Sachs J, Turner CD, Proctor M, Marcus KJ, Wang L, et al. Extracranial metastases of glioblastoma in a child: case report and review of the literature. J Pediatr Hematol Oncol. 2007;29(3):190–194. doi: 10.1097/MPH.0b013e31803350a7. [DOI] [PubMed] [Google Scholar]
- 10.Allan RS. Scalp metastasis from glioblastoma. J Neurol Neurosurg Psychiatry. 2004;75(4):559. [PMC free article] [PubMed] [Google Scholar]
- 11.Bouillot-Eimer S, Loiseau H, Vital A. Subcutaneous tumoral seeding from a glioblastoma following stereotactic biopsy: case report and review of the literature. Clin Neuropathol. 2005;24(6):247–251. [PubMed] [Google Scholar]
- 12.Anghileri E, Castiglione M, Nunziata R, Boffano C, Nazzi V, Acerbi F, et al. Extraneural metastases in glioblastoma patients: two cases with YKL-40-positive glioblastomas and a meta-analysis of the literature. Neurosurg Rev. 2016;39(1):37–45. doi: 10.1007/s10143-015-0656-9. [DOI] [PubMed] [Google Scholar]
- 13.Figueroa P, Lupton JR, Remington T, Olding M, Jones RV, Sekhar LN, et al. Cutaneous metastasis from an intracranial glioblastoma multiforme. J Am Acad Dermatol. 2002;46(2):297–300. doi: 10.1067/mjd.2002.104966. [DOI] [PubMed] [Google Scholar]
- 14.Forsyth TM, Bi WL, Abedalthagafi M, Dunn IF, Chiocca EA. Extracranial growth of glioblastoma multiforme. J Clin Neurosci. 2015;22(9):1521–1523. doi: 10.1016/j.jocn.2015.03.018. [DOI] [PubMed] [Google Scholar]
- 15.Ginat DT, Kelly HR, Schaefer PW, Davidson CJ, Curry W. Recurrent scalp metastasis from glioblastoma following resection. Clin Neurol Neurosurg. 2013;115(4):461–463. doi: 10.1016/j.clineuro.2012.05.038. [DOI] [PubMed] [Google Scholar]
- 16.Guo L, Qiu Y, Ge J, Zhou D. Glioblastoma multiforme with subcutaneous metastases, case report and literature review. J Korean Neurosurg Soc. 2012;52(5):484–487. doi: 10.3340/jkns.2012.52.5.484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hata N, Katsuta T, Inoue T, Arikawa K, Yano T, Takeshita M, et al. Extracranial metastasis of glioblastoma to the lung and heart with a histological resemblance to small cell carcinoma of the lung: an autopsy case. No Shinkei Geka. 2001;29(5):433–438. [PubMed] [Google Scholar]
- 18.Jain N, Mirakhur M, Flynn P, Choudhari KA. Cutaneous metastasis from glioblastoma. Br J Neurosurg. 2005;19(1):65–68. doi: 10.1080/02688690500081423. [DOI] [PubMed] [Google Scholar]
- 19.Magdaleno-Tapial J, Valenzuela-Oñate C, Pérez-Pastor G, Alegre de Miquel V. Skin metastasis of glioblastoma multiforme: a case report and literature review. Actas Dermosifiliogr. 2019;110(9):780–783. doi: 10.1016/j.ad.2018.05.017. [DOI] [PubMed] [Google Scholar]
- 20.Mentrikoski M, Johnson MD, Korones DN, Scott GA. Glioblastoma multiforme in skin: a report of 2 cases and review of the literature. Am J Dermatopathol. 2008;30(4):381–384. doi: 10.1097/DAD.0b013e31817532c4. [DOI] [PubMed] [Google Scholar]
- 21.Miliaras G, Tsitsopoulos PP, Markoula S, Kyritsis A, Polyzoidis KS, Malamou-Mitsi V. Multifocal glioblastoma with remote cutaneous metastasis: a case report and review of the literature. Cent Eur Neurosurg. 2009;70(1):39–42. doi: 10.1055/s-2008-1080941. [DOI] [PubMed] [Google Scholar]
- 22.Moon KS, Jung S, Lee MC, Kim IY, Kim HW, Lee JK, et al. Metastatic glioblastoma in cervical lymph node after repeated craniotomies: report of a case with diagnosis by fine needle aspiration. J Korean Med Sci. 2004;19(6):911–914. doi: 10.3346/jkms.2004.19.6.911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nguyen B, Samara J, Lee A, Fadia M, Ngu C, Pranavan G. A case of subgaleal metastasis from glioblastoma multiforme. Intern Med J. 2018;48(6):741–742. doi: 10.1111/imj.13818. [DOI] [PubMed] [Google Scholar]
- 24.Pérez-Bovet J, Rimbau-Muñoz J. Glioblastoma multiforme metastases to the masticator muscles and the scalp. J Clin Neurosci. 2018;53:237–239. doi: 10.1016/j.jocn.2018.04.021. [DOI] [PubMed] [Google Scholar]
- 26.Schultz S, Pinsky GS, Wu NC, Chamberlain MC, Rodrigo AS, Martin SE. Fine needle aspiration diagnosis of extracranial glioblastoma multiforme: Case report and review of the literature. Cytojournal. 2005;2:19. doi: 10.1186/1742-6413-2-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pietschmann S, von Bueren AO, Henke G, Kerber MJ, Kortmann RD, Müller K. An individual patient data meta-analysis on characteristics, treatments and outcomes of the glioblastoma/gliosarcoma patients with central nervous system metastases reported in literature until 2013. J Neurooncol. 2014;120(3):451–457. doi: 10.1007/s11060-014-1596-x. [DOI] [PubMed] [Google Scholar]
- 28.Sundahl N, Duprez F, Ost P, De Neve W, Mareel M. Effects of radiation on the metastatic process. Mol Med. 2018;24(1):16. doi: 10.1186/s10020-018-0015-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.