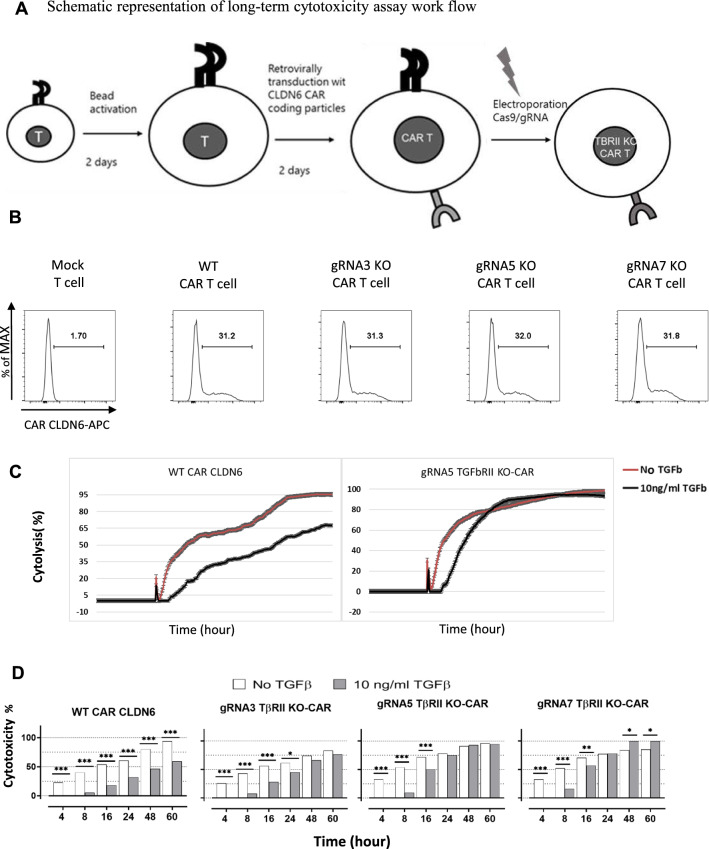
Fig. 6.
Genomic deletion of TGFβ receptor II restores long term cytotoxicity of TGFβRII KO CAR T-cells. A Schematic representation of long-term cytotoxicity assay work flow. Retroviral transduction of CD8+ T-cells with CAR CLDN6 supernatants is followed by CRISPR/Cas9 treatment to disrupt TGFβRII expression. B Equal CAR CLDN6 surface expression on retrovirally transduced WT and TGFβRII KO T-cells. C Impedance based cytotoxicity assay in the absence (red) and presence (black) of TGFβ. The averaged cell indexes of triplicates were normalized to the time shortly before seeding effector CAR T-cells, and then recalculated to %cytolysis according to the formula given in Material and methods. In the presence of TGFβ, WT CAR T-cells were compromised in cytotoxicity towards CLDN6+ tumor c.l. PA-1 lower the entire incubation time (> 3 days), while cytotoxicity of TGFβRII KO CAR T-cells recovered after 20 h. D Cytotoxicity bar chart of WT and TGFβRII KO CAR T-cells in the absence (white bars) and presence (grey bars) of 10 ng/ml TGFβ at designated time points taken from B. The initial gain in cytotoxicity for non-TGFβ treated KO CAR T-cells in comparison to the TGFβ-treated KO CAR T-cells levelled out over time (24 h) and led to comparable or even better cytolyses compared to WT CAR T-cells. P values were determined by two-way Anova using multiple comparison test. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001. In all experiments, mean ± SD of three technical replicates are given and experiments, involving T cells, are repeated for at least three donors