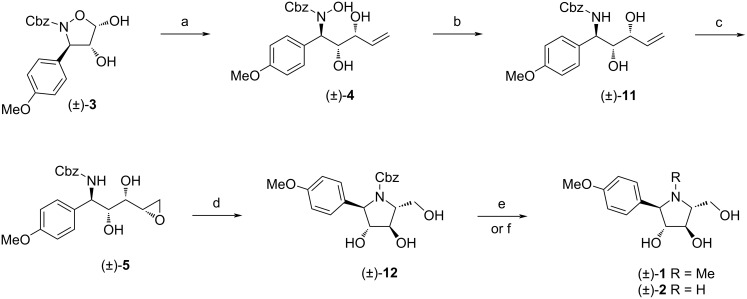
Scheme 3.
Synthesis of final pyrrolidines (±)-1 and (±)-2. Reagents and conditions: (a) vinyl-MgBr, CeCl3, THF, 0 °C to rt, 16 h, 73%; (b) Zn dust, AcOH, 40 °C, 24 h, 85%; (c) 12WO3·H3PO4×H2O, H2O2 (35 wt % in H2O), pyridine, ethyl acetate, rt, 48 h, 70%; (d) BF3·OEt2, CH2Cl2, 0 °C, 15 min, 69%; (e) H2 (1 atm), Pd(OH)2/C (5 wt %), MeOH, rt, 2 h, (±)-2, 71%; (f) H2 (1 atm), Pd(OH)2/C (5 wt %), MeOH, rt, 2 h; then formaldehyde (37 wt % in H2O), H2 (1 atm), Pd(OH)2/C (5 wt %), MeOH, rt, 16 h, (±)-1, 58% over two steps.