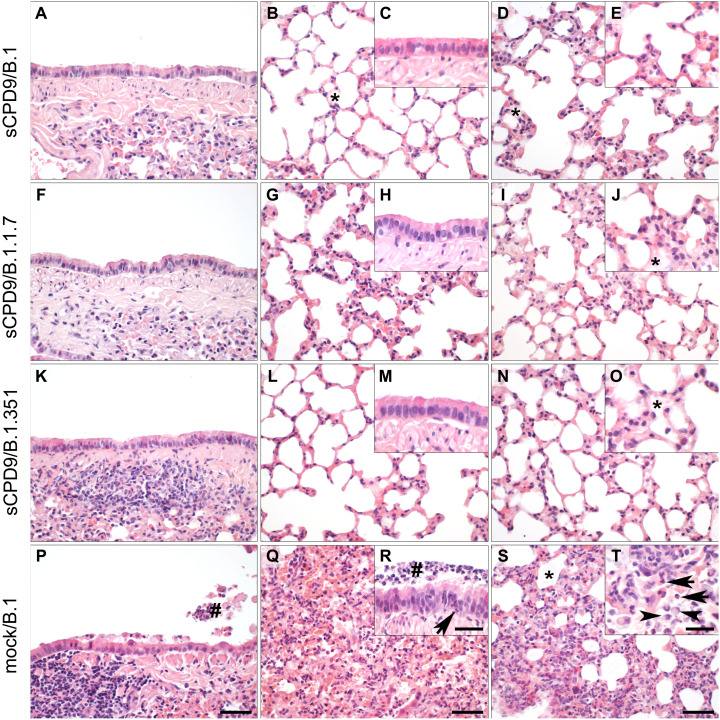
Fig. 2. Pulmonary histopathology of mock/sCPD9-vaccinated and challenged Roborovski dwarf hamsters on days 2 (left column), 3 (middle column), or 5 (right column) after challenge.
(A to T) Representative photomicrographs of hematoxylin and eosin (H&E)–stained, formalin-fixed, and paraffin-embedded lung tissue of infected hamsters. sCPD9-vaccinated hamsters were challenged with B.1, B.1.1.7, or B.1.351 virus on day 21 after vaccination. The lungs of sCPD9-vaccinated hamsters that were challenge-infected with three different SARS-CoV-2 variants showed very similar pulmonary morphology. Bronchioles had regular, columnar bronchiolar epithelium on days 2 and 3 after challenge (A, C, F, H, K, and M). Alveoli on days 3 (B, G, and L) and 5 after challenge (D, E, I, J, N, and O) showed mild to moderate infiltration by macrophages and few neutrophils with scattered mild alveolar edema (asterisk). In contrast, mock-vaccinated hamsters challenge-infected with the ancestral SARS-CoV-2 variant B.1 displayed typical lesions of COVID-19 at all times tested (P to T). After 2 days, the bronchiolar epithelium was flattened due to necrosis of bronchiolar epithelial cells and necrotic cellular debris, and degenerate neutrophils and macrophages were present in the bronchiolar lumen (hash) (P). In the course of disease, there was moderate to severe bronchiointerstitial pneumonia with necrosis of alveolar epithelial cells and massive infiltration by macrophages and neutrophils in the alveolar septa and alveolar spaces (Q). Similarly, bronchiolar epithelium was infiltrated by an increasing number of neutrophils (arrow) intraluminal cellular debris was accumulating (hash) (R). On day 5 after challenge, lungs showed predominantly interstitial pneumonia with remnants of alveolar edema fluid (asterisk) (S), neutrophils (T, arrow), and an increasing number of macrophages with prominently foamy cytoplasm (T, arrowhead). Scale bars, 50 μm (A, B, D, F, G, I, K, L, N, P, Q, and S) and 20 μm (C, E, H, J, M, O, R, and T).