ABSTRACT
Objective
To evaluate the performance of lung ultrasound to determine short-term outcomes of patients with COVID-19 admitted to the intensive care unit.
Methods
This is a Prospective, observational study. Between July and November 2020, 59 patients were included and underwent at least two LUS assessments using LUS score (range 0-42) on day of admission, day 5th, and 10th of admission.
Results
Age was 66.5±15 years, APACHE II was 8.3±3.9, 12 (20%) patients had malignancy, 46 (78%) patients had a non-invasive ventilation/high-flow nasal cannula and 38 (64%) patients required mechanical ventilation. The median stay in ICU was 12 days (IQR 8.5-20.5 days). ICU or hospital mortality was 54%. On admission, the LUS score was 20.8±6.1; on day 5th and day 10th of admission, scores were 27.6±5.5 and 29.4±5.3, respectively (P=0.007). As clinical condition deteriorated the LUS score increased, with a positive correlation of 0.52, P <0.001. Patients with worse LUS on day 5th versus better score had a mortality of 76% versus 33% (OR 6.29, 95%CI 2.01-19.65, p. 0.003); a similar difference was observed on day 10. LUS score of 5th day of admission had an area under the curve of 0.80, best cut-point of 27, sensitivity and specificity of 0.75 and 0.78 respectively.
Conclusion
These findings position LUS as a simple and reproducible method to predict the course of COVID-19 patients.
Keywords: Lund diseases, COVID-19, Ultrasonography
RESUMO
Objetivo
Avaliar o desempenho da ultrassonografia pulmonar para determinar a evolução em curto prazo de pacientes com COVID-19 internados na unidade de terapia intensiva.
Métodos
Este é um estudo prospectivo e observacional. Entre julho e novembro de 2020, 59 pacientes foram incluídos e submetidos a pelo menos duas avaliações LUS usando o escore LUS (variação de 0-42) no dia da admissão, dia 5 e 10 de admissão.
Resultados
A idade foi de 66,5 ± 15 anos, APACHE II foi de 8,3 ± 3,9, 12 (20%) pacientes tinham malignidade, 46 (78%) pacientes tinham ventilação não invasiva / cânula nasal de alto fluxo e 38 (64%) pacientes ventilação mecânica necessária. A mediana de permanência na UTI foi de 12 dias (IQR 8,5-20,5 dias). A mortalidade na UTI ou hospitalar foi de 54%. Na admissão, o escore LUS era de 20,8 ± 6,1; no dia 5 e no dia 10 de admissão, os escores foram 27,6 ± 5,5 e 29,4 ± 5,3, respectivamente (P = 0,007). Com a deterioração do quadro clínico, o escore LUS aumentou, com correlação positiva de 0,52, P <0,001. Pacientes com pior LUS no dia 5 versus melhor pontuação tiveram mortalidade de 76% versus 33% (OR 6,29, IC 95% 2,01-19,65, p. 0,003); uma diferença semelhante foi observada no dia 10. O escore LUS do 5º dia de admissão teve uma área sob a curva de 0,80, melhor ponto de corte de 27, sensibilidade e especificidade de 0,75 e 0,78, respectivamente.
Conclusão
Esses achados posicionam o LUS como um método simples e reprodutível para predizer a evolução de pacientes com COVID-19.
Palavras-chave: Doenças de Lund, COVID-19, Ultrassonografia
INTRODUCTION
The new coronavirus disease (COVID-19) can result in severe and even fatal respiratory diseases such as acute respiratory distress syndrome (ARDS).(1) Non-invasive diagnostic methods have gained ground in clinical practice, especially in critical and emergency medicine as rapid, reliable, and reproducible information at the patient's bedside has contributed to the more accurate etiological diagnosis of acute pathology and a more targeted treatment.(2,3) In the context of the COVID-19 pandemic, ultrasound has become a useful tool in intensive care, with a POCUS (Point of Care Ultrasound) approach.(4)
Lung ultrasound (LUS) is available in countries with different levels of development of their healthcare systems and offers advantages compared to computed tomography (CT), which requires transferring patients outside intensive care units or chest radiography, which has shown limitations for the diagnosis and monitoring of pulmonary pathology in critical patients.(5) Several studies have shown that LUS can detect interstitial disease, subpleural consolidations, and respiratory distress of any aetiology, with sensitivity and specificity superior to chest radiography and comparable to CT.(6-8) Indeed, LUS has already been recommended in past viral pandemics(9) and growing evidence demonstrates its effectiveness in patients with COVID-19,(10) which allows identifying the degree of lung involvement, its course, and the possible association between the initial lung involvement and its prognosis.(11-15)
Our objective was to evaluate the performance of LUS through the lung ultrasound score (LUS score) to determine the severity of pneumonia and the short-term outcomes of patients with COVID-19 admitted to the intensive care unit. We hypothesize that the Lung Ultrasound Score (LUS) correlates with clinical evolution and predicts mortality in critically ill COVID-19 pneumonia patients.
METHODS
This is an observational, analytical, prospective, single-centre study performed at the Intensive Care Unit (ICU) of the Hospital Alemán in Buenos Aires, Argentina; a teaching hospital with a 30-bed ICU, 15 of these were purposed to COVID-19 care. The study was approved by the independent ethics committee of Hospital Alemán. The informed consent of the patients or their representatives was obtained. The study was performed in compliance with Act 25.326/Habeas Data. We preserved the identity of the participants according to local and international standards and legislation.
We included all patients with a confirmed diagnosis of COVID-19 by polymerase chain reaction (PCR) techniques, admitted to the ICU with respiratory failure, between July 1, 2020, and October 31, 2020, and who had at least two LUS. LUS was performed at baseline (time of admission to ICU), on day fifth and day 10th of admission to ICU. We excluded patients under 18 years old, those whose reason for admission to the ICU was not respiratory failure despite a confirmed diagnosis of COVID-19, and those with advance directives and do not resuscitate orders. Patients with low supplemental oxygen requirements were admitted to the ICU they showed a rapid radiological progression, deterioration with tachypnoea, and/or poor ventilatory mechanics.
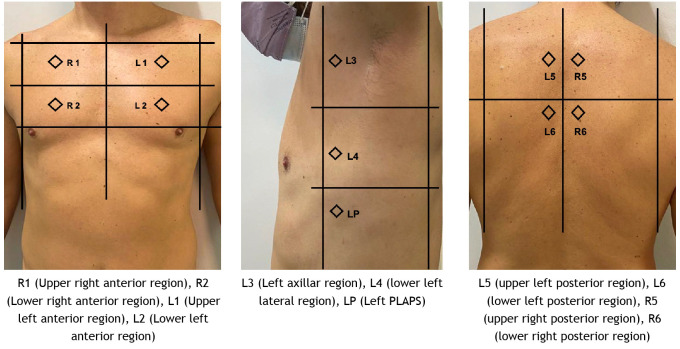
After obtaining informed consent and within 48 hours after admission to ICU, the first evaluation was performed using a Philips ultrasound machine, model CX 50, low-frequency convex transducer (2-6 MHz), with lung software (Philips Medical Systems, Bothell, WA, USA), at a pre-established depth of 11 to 14 cm. The LUS score was calculated according to methods described by Soldati et al. with a modification.(16) Instead of dividing the posterior areas into three regions, these were divided into two regions (upper and lower regions), and PLAPS point was added to lateral regions as pulmonary consolidation was observed. For this purpose, each hemithorax was divided into seven regions, right anterior between the clavicle upwards and the tenth intercostal space downwards, laterally between the right edge of the sternum and the anterior axillary line, and divided into upper and lower areas; right lateral between the axillary gap upwards and a line that continues from the tenth intercostal space downwards, between the anterior axillary lines and the posterior axillary line and divided into upper and lower; the posterior region was also divided into upper and lower, with lateral limits between the posterior axillary line and the spine and the superior region; and classified as follows: R1 (upper right anterior region) R2 (lower right anterior region), R3 (right axillary region), R4 (lower right lateral region), R5 (upper right posterior region), R6 (lower right posterior region), and R7 - PLAPS (posterior and/or lateral alveolar and/or pleural syndrome) (region between the union of the posterior axillary line and the intersection of the imaginary line that continues through the tenth intercostal space, requiring the visualization of the diaphragm for the correct exploration of the pleural fundus). In the same way and with equal limits, the left hemithorax was taken, naming each of the zones as L1 (upper left anterior region), L2 (lower left anterior region), L3 (left lateral region), L4 (lower left lateral region), L5 (upper left posterior region), L6 (lower left posterior region), and L7 – PLAPS. Figure 1 shows LUS regions.
Figure 1. Caption. Lung Ultrasound Score regions.
Each area was assigned a value according to the following assessment from 0 to 3 points. A-lines: normal pleural line, normal reverberation artifacts of the pleural line accompanied by lung sliding, corresponding to normal lung aeration = 0 points. B-lines: hyperechoic lines vertical to the pleural line rising from it and reaching the end of the screen, erasing the A-lines or in concert with them in the case of pulmonary pathology associated with COVID-19, representing reverberant artifacts of interlobar or oedematous alveolar septa, were divided into B1 separate B-lines corresponding to moderate loss of lung aeration = 1 point. Three or more lines define B1. B2 coalescent B-lines corresponding to a severe loss of pulmonary aeration, which was = 2 points, and pulmonary consolidation which was = 3 points.(17) Thus, the LUS score could have a normal value of 0 and the worst value of 42. This evaluation was performed by three intensive care physicians trained in performing LUS, at the bedside of the patient with confirmed COVID-19 using an ultrasound machine exclusively dedicated to these patients. Personal protective equipment was used for all studies and measurements were taken offline to reduce exposure time. The interobserver variability for LUS score measurement was determined by an independent expert operator who was blinded to the study.
At each assessment, we recorded the clinical condition measured by the level of ventilatory support: “mild” when the patient required a nasal O2 cannula; “moderate” when the patient required non-invasive ventilation/high-flow nasal cannula; “severe” when the patient required mechanical ventilation. In addition, the following parameters were measured: leukocytes, C-reactive protein, platelets, LDH, troponin-t, ferritin, and D-dimer. This evaluation was repeated on days fifth and 10th of ICU admission. Also, we recorded the number of days of ICU and hospital admission, ICU and hospital mortality, the total days of mechanical ventilation, and destination after hospital discharge (Home - Home Admission - 3rd level health centre).
For statistical analysis, continuous data were expressed as mean and standard deviation (SD), or as median and interquartile range [IQR 25-75], as appropriate. Normality analysis was performed using the Shapiro-Wilk test. Categorical data were expressed as absolute values and/or percentages. Student's t-test or Mann Whitney's U-test was used to comparing parametric and non-parametric continuous variables, respectively; and the Chi-square test and Fisher's exact tests were used to compare categorical variables. The relationship between the variables was initially determined through univariate analysis. For the multiple regression analysis, we selected the variables that we considered relevant (principle of parsimony) and/or those that in the univariate analysis resulted in a value p<0.1. Those patients with missing data on the variables of importance were excluded from the analysis. The statistical analyses were performed with the software R version 3.3.3.
RESULTS
During the four months of the study, 249 patients were admitted to ICU, of which 78 (31%) patients were diagnosed with COVID-19. A total of 19 patients were excluded for the following reasons: eight lacked ultrasound evaluations, five were admitted because of non-respiratory causes, five had a long hospital stay before the diagnosis of COVID-19 and one patient had a do-not-resuscitate/do-not intubate order. Finally, 59 patients were included in the final analysis. The mean age was 66.5±15 years and 43 (73%) of 59 patients were male. Severity at admission measured by APACHE II averaged 8.3±3.9 and Charlson's comorbidity score showed a mean of 3.2±2. Twelve (20%) patients had malignancy and 9 (15%) patients were fragile (Clinical Frailty Scale >4). In 46 (78%) patients, a non-invasive ventilation/high-flow nasal cannula was used, during a median of 3.5 days (IQR 2-6 days), and in 38 (64%) patients, mechanical ventilation was required, during a median of 12 days (IQR 9.25-19.75 days). The median stay in ICU was 12 days (IQR 8.5-20.5 days) and the median stay in hospital was 20 days (IQR 13.5-28.5 days). Mortality in ICU and hospital was 54%. The demographic characteristics of the patients are summarized in Table 1.
Table 1. Demographic and clinical characteristics.
| Characteristics | Value in total patients (n = 59) |
|---|---|
| Age, mean±SD | 66.5 ±15.0 |
| Male, n (%) | 43 (73) |
| APACHE II. mean±SD | 8.3±3.9 |
| Charlson Score. mean±SD | 3.2±2.0 |
| Cancer, n (%) | 12 (20) |
| Fragile patients (CFS >4). n (%) | 9 (15) |
| BMI, mean±SD | 27.6±4.5 |
| Support | |
| Non-invasive ventilation/high-flow nasal cannula, n (%) | 46 (78) |
| Days of the cannula, median (IQR) | 3.5 (2-6) |
| Mechanical ventilation, n (%) | 38 (64) |
| Days of MV. median (IQR) | 12 (9.25-19.75) |
| ECMO, n (%) | 1 (2) |
| Haemodialysis, n (%) | 9 (15) |
| Neuromuscular blocking agents, n (%) | 35 (59) |
| Days of neuromuscular blocking agents, median (IQR) | 8 (5-10) |
| Days from diagnosis to LUS, median (IQR) | 5 (3.5-9) |
| Length of ICU stay, days, median (IQR) | 12 (8.5-20.5) |
| Length of hospital stay, days, median (IQR) | 20 (13.5-28.5) |
| ICU mortality, n (%) | 32 (54) |
| Hospital mortality, n (%) | 32 (54) |
Abbreviations: CFS = Clinical Frailty Scale; BMI = body mass index; APACHE = Acute Physiology and Chronic Health Evaluation; ECMO = extracorporeal membrane oxygenation; ICU = intensive care unit; SD = standard deviation; IQR = interquartile range.
The median time from the diagnosis of COVID-19 to the first LUS was 5 days (IQR 3.5-9 days). The total median LUS score at admission was 20.8±6.1; at day 5, it was 27.6±5.5 and at day 10, 29.4±5.3 (p=0.007). As clinical condition deteriorated (according to the requirement for mechanical ventilation support), the LUS score increased, with a positive correlation of 0.52, p <0.001. On day 10 (n=41), LUS scores were 19.1±3.4, 23.3±4.5, 30.8±5.3 in the mild, moderate, and severe groups, respectively. When analysing the biochemical markers at the time of the first LUS evaluation, none of them showed a statistically significant association with the clinical condition, unlike the LUS score. The group of patients whose LUS score at day 5 was worse than baseline had a 76% mortality versus a 33% mortality in the group of patients whose LUS scores improved during the same period (OR 6.29, 95%CI 2.01-19.65, p=0.003). Table 2 shows ICU mortality according to changes observed with the LUS score.
Table 2. ICU mortality according to LUS score changes.
| LUS score change | n (%) | ICU mortality (%) | OR (CI 95%) | P-value |
|---|---|---|---|---|
| LUS on 5th day of admission | 0.003 | |||
| Improved score | 30 (51) | 33 | ||
| Deteriorated score | 29 (49) | 76 | 6.29 (2.01-19.65) | |
| LUS on the 10th day of admission | 0.023 | |||
| Improved score | 18 (44) | 33 | ||
| Deteriorated score | 23 (56) | 74 | 5.67 (1.47-21.89) |
Improved or deteriorated compared to the previous assessment. LUS = Lung ultrasound score; ICU = intensive care unit; OR = odds ratio; CI = confidence interval.
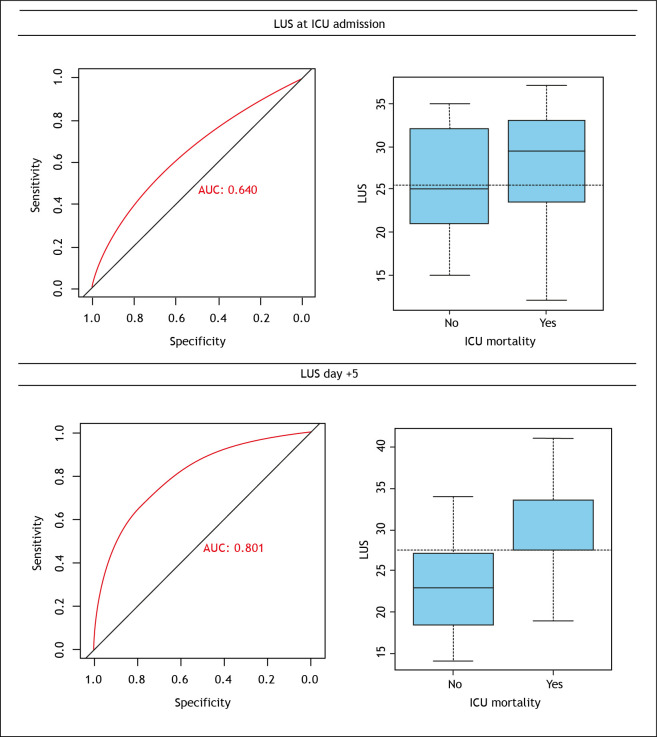
This difference was also observed on day 10 of admission to ICU. In multivariate analysis, adjusted by age, APACHE II, and Charlson, the LUS score was an independent predictor of mortality (OR 1.32, 95%CI 1.14 -1.60, p=0.001). In another regression model in which sex and body mass index were adjusted, the LUS score was also an independent predictor of mortality (OR 1.30, 95%CI 1.14-1.54, p<0.001). The sensitivity and specificity of the LUS score to predict patient mortality were evaluated. The LUS score at ICU admission showed an area under the curve (AUC) of 0.64, with 25 as the best cut-point, sensitivity of 0.63, and a specificity of 0.59. And on the fifth day after admission, the LUS score presented an AUC of 0.80, with the best cut-point of 27, sensitivity and specificity of 0.75 and 0.78, respectively (Table 3 and Figure 2). Figure 3 shows examples of LUS images of patients included in the study.
Table 3. Multivariate regression; mortality predicted by LUS.
| Model | OR (95%CI) | p-value | ||||
|---|---|---|---|---|---|---|
| Model 1 (adjusted by age, APACHE, Charlson) | 1.32 (1.14-1.60) | 0.001 | ||||
| Model 2 (adjusted by sex and BMI) | 1.30 (1.14-1.54) | < 0.001 | ||||
| ROC AUC | Sensitivity | Specificity | PPV | NPV | ||
| LUS at admission | ||||||
| LUS > 25 | 0.64 | 0.63 | 0.59 | 0.65 | 0.57 | |
| LUS on 5th day of admission | ||||||
| LUS > 27 | 0.80 | 0.75 | 0.78 | 0.80 | 0.72 | |
AUC = area under the curve; ROC = receiver operating characteristic curve; PPV = positive predictive value; NPV = negative predictive value; LUS = Lung ultrasound score.
Figure 2. Sensitivity and specificity of the LUS score to predict patient mortality at the intensive care unit.
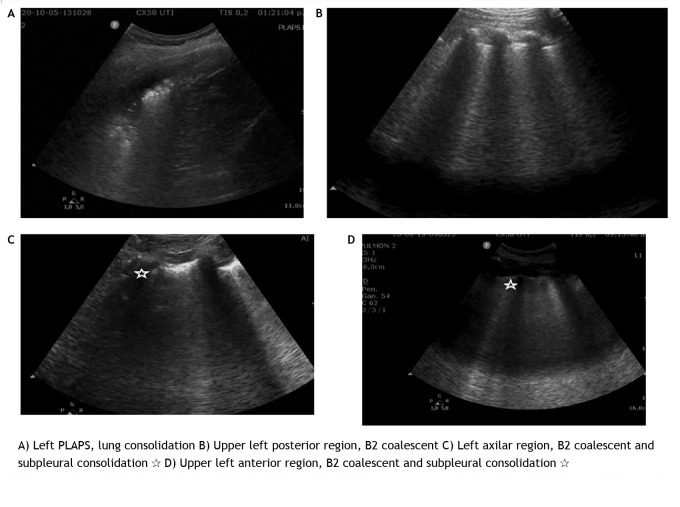
Figure 3. LUS images of patients included in the study.
DISCUSSION
Our findings show that lung ultrasound score is a feasible and easy method to predict the clinical course of critically ill patients with COVID-19. When patients’ clinical condition deteriorated, the score significantly increased. Indeed, the LUS score was an independent predictor of mortality and when assessed on day 5 of admission to ICU, the score presented an acceptable area under the curve.
The histopathologic features of COVID-19 pneumonia are characterized by alveolar damage, including alveolar oedema, while the inflammatory component is mild and patchy. Reparative processes with hyperplasia of pneumocytes and interstitial thickening may then occur; in advanced phases, there appear gravity-dependent consolidations similar to those of respiratory distress, as well as haemorrhagic necrosis, alveolar congestion, oedema, desquamation, and fibrosis.(18) Therefore, tools that can reliably assess lung involvement can also predict clinical deterioration. Our study suggests that the LUS score was useful to evaluate patients with COVID-19 and that, early after admission, could predict a higher risk of mortality.
The LUS score could define alterations that affect the relationship between tissue and air on the lung surface;(19) indeed, the higher score, the greater was the loss of pulmonary aeration. The median time between the diagnosis of COVID-19 and the first LUS evaluation was five days, and this may explain the difference between the first and the second LUS evaluation performance to predict unfavourable outcomes. In fact, the second evaluation was performed on average 10 days after diagnosis, when COVID-19 presents an inflammatory peak, mainly due to the increase of proinflammatory cytokines.(20,21) It has been shown that patients with respiratory distress from COVID-19 can retain near-normal lung compliance at the initial stages of the disease despite good oxygenation to later deteriorate or improve.(22,23)
The LUS score showed an association to the clinical severity of patients with COVID-19 at the baseline assessment on the day of admission to ICU; however, it was on the fifth day of hospitalization that the score reached its best AUC to predict mortality. In a comparable study, this difference in the score was already significant in the first LUS assessment.(24) Lichter et al also showed that the LUS score could identify the appearance of pleural thickening and effusion, predicting clinical deterioration which translated to the need for mechanical ventilation and mortality.(24) In their study, an LUS score higher than 18 indicated a significantly lower survival.(24)
Our study was directed to the most critical group of patients with COVID-19 at our hospital; those patients who required hospitalization in ICU because of acute respiratory failure. As a result, our LUS score values were higher than those in previous publications, which included patients being admitted to emergency departments or general wards, in their initial stages of the disease.(13) In addition, we decided to include the lung lateral region to the axilla and to the PLAPS points, where pleural effusion and larger consolidations are usually observed. The pleura is not described in detail because although it was found to be altered (pleural disruption, pleural thickening), the transducer was intended for the assessment of type A, type B lung patterns, and subpleural consolidations and in the PLAPS points. bilateral pathological characteristics were found in 100% of our population, in concordance with other LUS studies in patients with COVID-19(18,20) and the most common findings were B lines and subpleural consolidations in posterior segments.
Our study has several limitations that should be taken into consideration when interpreting our results. It was performed in a single centre, with a small number of patients; some of them had been first admitted into the general ward before being transferred to ICU, overestimating the score cut point to predict poor clinical evolution and mortality. The use of a low-frequency, high-penetrance transducer could have missed a more accurate assessment of pleural line characteristics. Other limitations were that the onset of symptoms was not recorded, other infections were not assessed, and that not all potential confounders and effect modifiers were measured, such as lung oedema, as a result of limited resources during the pandemic.
Our findings must be interpreted with caution. Apart from the limitations listed above, it is critical that the implementation of lung ultrasound must be supervised by trained personnel. Our study contributes to the literature supporting the use of ultrasound lung evaluation as a simple method to predict the course of COVID-19 patients, their need for mechanical ventilation, and death. LUS is an easily available tool that could help clinicians plan the management of patients with COVID-19.
Footnotes
Study carried out in the intensive care unit of Hospital Alemán (German Hospital), Buenos Aires, Argentina.
Financial support: None.
REFERENCES
- 1.Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lichtenstein DA. Ultrasound examination of the lungs in the intensive care unit. Pediatr Crit Care Med. 2009;10(6):693–698. doi: 10.1097/PCC.0b013e3181b7f637. [DOI] [PubMed] [Google Scholar]
- 3.Arbelot C, Ferrari F, Bouhemad B, Rouby JJ. Lung ultrasound in acute respiratory distress syndrome and acute lung injury. Curr Opin Crit Care. 2008;14(1):70–74. doi: 10.1097/MCC.0b013e3282f43d05. [DOI] [PubMed] [Google Scholar]
- 4.Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, et al. International evidence-based recommendations for point of care ultrasound. Intensive Care Med. 2012;38(4):577–591. doi: 10.1007/s00134-012-2513-4. [DOI] [PubMed] [Google Scholar]
- 5.Tierney DM, Huelster JS, Overgaard JD, Plunkett MB, Boland LL, St. Hill CA, et al. Comparative performance of pulmonary ultrasound, chest radiograph, and CT among patients with acute respiratory failure. Crit Care Med. 2020;48(2):151–157. doi: 10.1097/CCM.0000000000004124. [DOI] [PubMed] [Google Scholar]
- 6.Zieleskiewicz L, Markarian T, Lopez A, Taguet C, Mohammedi N, Boucekine M, et al. Comparative study of lung ultrasound and chest computed tomography scan in the assessment of severity of confirmed COVID-19 pneumonia. Intensive Care Med. 2020;46(9):1707–1713. doi: 10.1007/s00134-020-06186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lichtenstein D, Goldstein I, Mourgeon E, Cluzel P, Grenier P, Rouby JJ. Comparative diagnostic performances of auscultation, chest radiography, and lung ultrasonography in acute respiratory distress syndrome. Anesthesiology. 2004;100(1):9–15. doi: 10.1097/00000542-200401000-00006. [DOI] [PubMed] [Google Scholar]
- 8.Xirouchaki N, Magkanas E, Vaporidi K, Kondili E, Plataki M, Patrianakos A, et al. Lung ultrasound in critically ill patients: comparison with bedside chest radiography. Intensive Care Med. 2011;37(9):1488–1493. doi: 10.1007/s00134-011-2317-y. [DOI] [PubMed] [Google Scholar]
- 9.Testa A, Soldati G, Copetti R, Giannuzzi R, Portale G, Gentiloni-Silveri N. Early recognition of the 2009 pandemic influenza A (H1N1) pneumonia by chest ultrasound. Crit Care. 2012;16(1):R30. doi: 10.1186/cc11201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Buonsenso D, Pata D, Chiaretti A. COVID-19 outbreak: less stethoscope, more ultrasound. Lancet Respir Med. 2020;8(5):e27. doi: 10.1016/S2213-2600(20)30120-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bitar ZI, Shamsah M, Bamasood OM, Maadarani OS, Alfoudri H. Point-of-Care Ultrasound for COVID-19 Pneumonia Patients in the ICU. J Cardiovasc Imaging. 2021;29(1):60–68. doi: 10.4250/jcvi.2020.0138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hu Z, Liu Z, Dong Y, Liu J, Huang B, Liu A, et al. Evaluation of lung involvement in COVID-19 pneumonia based on ultrasound images. Biomed Eng Online. 2021;20(1):27. doi: 10.1186/s12938-021-00863-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Martinez Redondo J, Comas Rodriguez C, Pujol Salud J, Crespo Pons M, Garcia Serrano C, Ortega Bravo M, et al. Higher accuracy of lung ultrasound over chest X-ray for early diagnosis of COVID-19 pneumonia. Int J Environ Res Public Health. 2021;18(7):3481. doi: 10.3390/ijerph18073481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Volpicelli G, Gargani L, Perlini S, Spinelli S, Barbieri G, Lanotte A, et al. Lung ultrasound for the early diagnosis of COVID-19 pneumonia: an international multicenter study. Intensive Care Med. 2021;47(4):444–454. doi: 10.1007/s00134-021-06373-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heldeweg MLA, Lopez Matta JE, Haaksma ME, Smit JM, Elzo Kraemer CV, Grooth HJ, et al. Lung ultrasound and computed tomography to monitor COVID-19 pneumonia in critically ill patients: a two-center prospective cohort study. Intensive Care Med Exp. 2021;9(1):1. doi: 10.1186/s40635-020-00367-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Soldati G, Smargiassi A, Inchingolo R, Buonsenso D, Perrone T, Briganti DF, et al. Proposal for international standardization of the use of lung ultrasound for COVID-19 patients; a simple, quantitative, reproducible method. J Ultrasound Med. 2020;39(7):1413–1419. doi: 10.1002/jum.15285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lichter Y, Topilsky Y, Taieb P, Banai A, Hochstadt A, Merdler I, et al. Lung ultrasound predicts clinical course and outcomes in COVID-19 patients. Intensive Care Med. 2020;46(10):1873–1883. doi: 10.1007/s00134-020-06212-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tian S, Hu W, Niu L, Liu H, Xu H, Xiao SY. Pulmonary pathology of early phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J Thorac Oncol. 2020;15(5):700–704. doi: 10.1016/j.jtho.2020.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Soldati G, Smargiassi A, Inchingolo R, Sher S, Nenna R, Valente S, et al. Lung ultrasonography may provide an indirect estimation of lung porosity and airspace geometry. Respiration. 2014;88(6):458–468. doi: 10.1159/000368086. [DOI] [PubMed] [Google Scholar]
- 20.Clerkin KJ, Fried JA, Raikhelkar J, Sayer G, Griffin JM, Masoumi A, et al. COVID-19 and Cardiovascular Disease. Circulation. 2020;141(20):1648–1655. doi: 10.1161/CIRCULATIONAHA.120.046941. [DOI] [PubMed] [Google Scholar]
- 21.Kenny G, Mallon PW. COVID19- clinical presentation and therapeutic considerations. Biochem Biophys Res Commun. 2021;538:125–131. doi: 10.1016/j.bbrc.2020.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nouvenne A, Zani MD, Milanese G, Parise A, Baciarello M, Bignami EG, et al. Lung Ultrasound in COVID-19 pneumonia: correlations with Chest CT on hospital admission. Respiration. 2020;99(7):617–624. doi: 10.1159/000509223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tung-Chen Y, Martí de Gracia M, Díez-Tascón A, Alonso-González R, Agudo-Fernández S, Parra-Gordo ML, et al. Correlation between chest computed tomography and lung ultrasonography in patients with coronavirus disease 2019 (COVID-19) Ultrasound Med Biol. 2020;46(11):2918–2926. doi: 10.1016/j.ultrasmedbio.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lichter Y, Topilsky Y, Taieb P, Banai A, Hochstadt A, Merdler I, et al. Lung ultrasound predicts clinical course and outcomes in COVID-19 patients. Intensive Care Med. 2020;46(11):2128–2129. doi: 10.1007/s00134-020-06254-5. [DOI] [PMC free article] [PubMed] [Google Scholar]