Supplemental Digital Content is available in the text.
Keywords: acute respiratory distress syndrome, coronavirus disease 2019, intensive care, outcomes, proning, ventilation
Abstract
Importance:
New treatments and increased experience are changing the management of hospitalized coronavirus disease 2019 patients but the impact on ICU management is unclear.
Objectives:
To examine characteristics, ventilatory management, and outcomes of critically ill patients in two distinct waves of the pandemic.
Design, Setting, and Participants:
Observational cohort study in an ICU in a single-center university-affiliated U.K. hospital. Two-hundred ten adults with coronavirus disease 2019 admitted to ICU between March 17, 2020, to May 31, 2020, and September 1, 2020, to December 10, 2020, with hourly data and 100% follow-up to ICU discharge.
Main Outcomes and Measures:
Data were extracted from the electronic medical record for patient characteristics and clinical data. Patients were classified into distinct waves of the pandemic and assessed for differences between the two waves.
Results:
The duration of noninvasive ventilation/nasal high flow increased in wave 2 versus wave 1, both in self-ventilating patients (107 vs 72 hr; p = 0.02), and in those ultimately requiring invasive mechanical ventilation (34 vs 10 hr; p = 0.02). The proportion of survivors treated without invasive mechanical ventilation increased in wave 2 (59% vs 39%; p = 0.01). In both waves, longer duration of noninvasive ventilation/nasal high flow prior to intubation was associated with higher ICU mortality (survivors 10 hr [4–21 hr] vs nonsurvivors 50 hr [23–124 hr]; p < 0.01). Proned invasive mechanical ventilation was common (54.7%) and prolonged. In wave 2, invasive mechanical ventilation patients were generally more hypoxic with proning initiated at lower Pao2/Fio2 ratios (81 vs 116 mm Hg; p = 0.02) and yielding smaller improvements in Fio2 requirements. Continued proning episodes despite poor responses were commonplace and typically futile. Length of stay for patients requiring tracheostomy increased markedly in wave 2 (51.3 vs 33.7 d; p = 0.03). Overall survival remained similar in wave 2 (68.0% vs 60.9%; p = 0.31).
Conclusions and Relevance:
Our data suggest that management of critically ill coronavirus disease 2019 patients is changing with more survivors avoiding invasive mechanical ventilation. Duration of noninvasive ventilation/nasal high flow use is increasing, which may be associated with worsening outcomes for individuals who require invasive mechanical ventilation. Among invasively ventilated patients, changes in the use of and response to prone positioning and increased length of stay following tracheostomy may imply that the care of these patients is becoming more challenging.
Coronavirus disease 2019 (COVID-19) has placed significant demands on ICUs across the world in terms of capacity, resources, and staffing. While randomized controlled trials have identified new therapies such as dexamethasone and interleukin-6 (IL-6) antagonists that have improved mortality in critically ill patients, the daily ventilatory management of these patients remains challenging (1, 2). Although clinician experience has increased, it is likely that ICUs will increasingly receive patients who have failed to respond to standard COVID-19 treatment, and this may have implications for ongoing patient management.
Accordingly, we present the characteristics, ventilatory management, and ICU outcomes of over 200 critically ill patients with COVID-19 at our institution from two distinct waves in the U.K. pandemic. We describe hourly data for the duration of ICU stay in order to build a detailed report of our experiences. We hypothesized that more patients in the second wave would maintain their own airway throughout ICU admission and that we would observe changes in the use of noninvasive ventilation (NIV)/nasal high flow (NHF). We also hypothesized that the outcomes of individuals requiring invasive mechanical ventilation (IMV) in the second wave would be different, as evidenced by their Fio2 response to proning, their need for tracheostomy, and their ICU length of stay (LOS).
MATERIALS AND METHODS
Ethical approval was not required by our institution’s research and innovation department. Information governance safeguards for the project were approved by our institution’s Caldicott Guardian.
We reviewed ICU admission records to identify all patients admitted with COVID-19 in two peak periods of the U.K. pandemic (March 17, 2020, to May 31, 2020, and September 1, 2020, to December 10, 2020). We included patients in whom the primary admission indication was confirmed or probable severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (3). Routine practice was to test for SARS-CoV-2 infection by polymerase chain reaction analysis of nasopharyngeal throat swab. Repeat samples were obtained after negative results where clinical suspicion remained. We excluded patients with an incidentally positive swab who were admitted for another indication.
ICU data was extracted from the IntelliSpace critical care information system (Philips N.V, Amsterdam, Netherlands) used for documentation in our ICU. Where necessary, this was supplemented by manual review of the record. We recorded hourly data on airway management, use of high-flow (HF) oxygen therapy, NIV, IMV, and associated ventilatory parameters, use of neuromuscular blocker (NMB) infusions, and prone position ventilation. We also extracted arterial blood gas data. Resuscitation status and treatment limitations were collected electronically and verified manually. A treatment limitation precluding IMV was defined as present if recorded in the medical record on the first review by a senior ICU physician and within 24 hours of ICU admission.
Our specific outcomes of interest included the percentage of patients who maintained their own airway while in critical care, the frequency and duration of NHF/NIV use, the Fio2 response to proning in IMV patients, frequency of tracheostomy use, ICU LOS, and outcome. Outcome data were obtained from our ICU’s Intensive Care National Audit and Research Centre (ICNARC) dataset (4).
Critical Care Practices in Our Institution
Manchester Royal Infirmary has approximately 900 adult inpatient beds and is part of Manchester University NHS Foundation Trust. There are 40 adult ICU beds and 12 adult cardiac ICU beds. Eighteen paediatric ICU (PICU) beds are located within the same building in Royal Manchester Children’s Hospital. During the study period, our ICU footprint was escalated into three additional clinical areas including the PICU, although most patients were cared for in the main unit. We maintained a 1:1 nurse-patient ratio for level 3 patients and an overall patient to attending physician ratio of 1:12. Appendix 1 (http://links.lww.com/CCX/A862) provides an overview of our COVID-19 treatment guidelines.
During wave 1, we avoided using HF oxygen therapy due to concerns over increased aerosolization of viral particles (5). Dexamethasone and IL-6 antagonists were only used in the context of clinical trials (1, 6). By wave 2, dexamethasone was administered routinely, and HF oxygen therapy was used liberally.
NMB infusions and prone mechanical ventilation were instituted at the direction of the treating clinicians with a recommendation that proning and NMB be considered at a Pao2/Fio2 (PF) ratio of less than 150 mm Hg in line with previous trial protocols (7, 8).
We assessed response to prone positioning in IMV patients using absolute changes in Fio2. Our routine practice is to titrate Fio2 according to target oxygen saturations (Sao2) rather than a target Pao2 or PF ratio. Our Sao2 target in acute hypoxemic respiratory failure is 90% (Appendix 1, http://links.lww.com/CCX/A862) and the Fio2 is adjusted frequently by our nursing staff and recorded hourly. In contrast, our guidance at the start of the pandemic was for “routine” arterial blood gas analysis every 12 hours with no additional measurements unless there was a clinical need. We therefore chose to use Fio2 to assess response to proning due to the hourly data that better reflected contemporary adjustments by our nursing staff. We recorded the absolute change in inspired Fio2 between the last Fio2 reading before proning and the median value between 11 and 13 hours (termed “12 hr”) post-initiation of a proning episode. To facilitate comparison with other studies, we also noted PF ratios at 12 hours after the first prone episode.
Statistical Analysis
Analysis was conducted using the R statistical computing package v3.6.1 (R Foundation for Statistical Computing, Vienna, Austria). All data were assessed for normality. Group comparisons were performed using Fisher exact test for equal proportion, Student t test for normally distributed data, and Wilcoxon rank-sum tests otherwise. Counts are reported as n (%), continuous variables are reported as means with sds, or medians with interquartile ranges as appropriate.
In exploratory analysis, statistically significant results may occur which do not have clinical importance. It is unclear what represents an important difference in vital signs and outcomes in critical care but for the ease of the reader, we highlight results felt to have clinical importance.
RESULTS
We identified 110 patients with COVID-19 during wave 1 and 100 patients in wave 2. A total of 77,708 hours of ICU patient data were analyzed. During the same time periods, our hospital treated approximately 650 COVID-19 inpatients in wave 1 and 763 inpatients in wave 2.
Overall Characteristics
Patient characteristics are displayed in Table 1, stratified by treatment wave. Characteristics stratified by survival are presented in Appendix 2 (http://links.lww.com/CCX/A862). Admission characteristics in waves 1 and 2 were similar except for ethnicity, in which we observed a higher proportion of Asian (Indian, Pakistani, or Bangladeshi heritage) patients in the second wave (p < 0.01).
TABLE 1.
Baseline Characteristics of Coronavirus Disease 2019 Patients in Wave 1 and 2
| Variable | All Patients (n = 210) | Wave 1 (n = 110) | Wave 2 (n = 100) | p |
|---|---|---|---|---|
| Age, mean (sd) | 57.6 (13.8) | 57.2 (13.2) | 57.9 (14.4) | 0.72 |
| Male sex, n (%) | 139 (66.2) | 72 (65.5) | 67 (67.0) | 0.88 |
| Female sex, n (%) | 71 (33.8) | 38 (34.5) | 33 (33.0) | |
| Body mass index (kg/m2), n (%) | ||||
| < 18.5 | 1 (0.5) | 1 (0.9) | 0 (0.0) | |
| 18.5–25 | 44 (21.0) | 24 (21.8) | 20 (20.0) | |
| 25–30 | 78 (37.1) | 44 (40.0) | 34 (34.0) | 0.06 |
| 30–35 | 43 (20.5) | 26 (23.6) | 17 (17.0) | |
| 35+ | 44 (21.0) | 15 (13.6) | 29 (29.0) | |
| Ethnicity, n (%) | ||||
| Caucasian | 85 (40.5) | 50 (45.5) | 35 (35.0) | |
| Asian | 66 (31.4) | 21 (19.1) | 45 (45.0) | |
| Afro-Caribbean | 38 (18.1) | 23 (20.9) | 15 (15.0) | < 0.01 |
| Other | 9 (4.3) | 6 (5.5) | 3 (3.0) | |
| Not known | 12 (5.7) | 10 (9.1) | 2 (2.0) | |
| Prehospital dependency, n (%) | ||||
| Able to live without assistance in daily activities | 161 (76.7) | 83 (75.5) | 78 (78.0) | |
| Minor assistance with some daily activities | 28 (13.3) | 19 (17.3) | 9 (9.0) | 0.11 |
| Major assistance with majority of/all daily activities | 21 (10.0) | 8 (7.3) | 13 (13.0) | |
| Admitted from, n (%) | ||||
| Emergency department: | 65 (30.1) | 30 (27.3) | 35 (35.0) | |
| Ward/theaters | 119 (56.7) | 65 (59.1) | 54 (54.0) | 0.47 |
| Other ICU | 26 (12.4) | 15 (13.6) | 11 (11.0) | |
| Treatment limitation at ICU admission, n (%) | 40 (19.0) | 21 (19.1) | 19 (19.0) | 1.00 |
| APACHE II, median (IQR) | 15 (12–19) | 15 (12–19) | 14.5 (12–18) | 0.61 |
| APACHE II probability of death (%), median (IQR) | 22.6 (14.5–34.4) | 22.6 (14.0–34.4) | 20.2 (15.9–31.2) | 0.60 |
APACHE = Acute Physiology and Chronic Health Evaluation, IQR = interquartile range.
Overall, 41.4% of patients (87/210) were obese (body mass index > 30 kg/m2), 23.3% of patients required minor or major assistance at home prior to admission, and 19.0% of patients had a treatment limitation at admission to ICU precluding them from IMV. The proportion of patients with treatment limitations was very similar between waves.
Patients admitted from the wards or transferred to our institution from another ICU (i.e., having already received some hospital treatment prior to transfer to the ICU) comprised 69.0% of the total cohort.
Noninvasive Ventilation
The proportion of patients who received any NIV/NHF was similar in waves 1 and 2, and this was regardless of whether the patients progressed to IMV (Table 2). However, we observed that the duration of NIV/NHF treatment increased significantly in wave 2 both as a sole treatment modality and in patients for whom it was a precursor to IMV.
TABLE 2.
Summary of Airway and Ventilatory Management
| Variable | All Patients (n = 210) | Wave 1 (n = 110) | Wave 2 (n = 100) | p | Clinically Important |
|---|---|---|---|---|---|
| Airway status at the point of arrival to the ICU | |||||
| Own airway, n (%) | 165 (78.6) | 81 (73.6) | 84 (84.0) | 0.04 | a |
| Cuffed oral endotracheal tube, n (%) | 41 (19.5) | 28 (25.5) | 13 (13.0) | ||
| Tracheostomy, n (%) | 4 (1.9) | 1 (0.9) | 3 (3.0) | ||
| Maintained own airway for entire ICU admission | (n = 100) | (n = 49) | (n = 51) | ||
| NIV/HF during ICU admission, n (%) | 89 (89) | 42 (86) | 47 (92) | 0.35 | |
| With treatment limitation, n (%) | 32 (32) | 19 (39) | 15 (29) | 0.27 | |
| Without treatment limitation, n (%) | 54 (54) | 23 (47) | 32 (63) | ||
| Duration of NIV/HF (hr), mean (sd) | 91 (73) | 72 (72) | 107 (71) | 0.02 | a |
| Management of patients requiring intubation and invasive mechanical ventilation | (n = 106) | (n = 60) | (n = 46) | ||
| Intubated prior to ICU admission, n (%) | 41 (38.7) | 28 (47) | 13 (28) | 0.02 | a |
| Total intubated ≤ 24 hr of ICU admission, n (%) | 74 (69.8) | 45 (75) | 29 (63) | 0.21 | |
| NIV prior to intubation, n (%) | 58 (54.7) | 29 (48) | 29 (63) | 0.17 | |
| NIV duration prior to intubation (hr), median (IQR) | 19 (7–61) | 10 (5–38) | 34 (14–104) | 0.02 | a |
| Neuromuscular blockade infusion, n (%) | 77 (72.6) | 44 (73.3) | 33 (72) | 1.0 | |
| Cumulative duration per patient (hr), median (IQR) | 111.0 (46–185) | 88 (47–162) | 139 (46–237) | 0.15 | |
| Prone ventilation, n (%) | 58 (54.7) | 30 (50) | 28 (61) | 0.33 | |
| Duration of each episode (hr), median (IQR) | 19 (15–23) | 18 (13–20) | 22 (17–25) | < 0.01 | |
| Cumulative duration per patient proned (hr), median (IQR) | 43 (22–76) | 41 (23–71) | 52 (22–87) | 0.61 | |
| Number of episodes per patient proned, median (IQR) | 2 (1–5) | 2 (1–6) | 2 (1–4) | 0.19 | |
| Interval from Intubation to first prone event (hr), median (IQR) | 41 (5–111) | 32 (3–87) | 66 (5–163) | 0.19 | |
| Fio2 prior to first episode of proning (%), mean (sd) | 78 (16.4) | 72 (16.6) | 84 (14.0) | < 0.01 | a |
| Pao2/Fio2 prior to first episode proning (mm Hg), mean (sd) | 99 (58) | 116 (76) | 81 (16) | 0.02 | a |
| Fio2 at 12 hr post first episode of proning (%), mean (sd) | 55 (17.2) | 49 (14.7) | 60 (18.2) | 0.02 | a |
| Pao2/Fio2 12 hr post first prone episode (mm Hg), mean (sd)b | 139 (60) | 154 (66) | 122 (50) | 0.05 | a |
| Inhaled nitric oxide therapy at any point, n (%) | 6 (5.7) | 6 (10) | 0 (0) | 0.03 | |
| Transferred for extracorporeal membrane oxygenation, n (%) | 3 (2.8) | 3 (5.0) | 0 (0) | 0.26 | |
| Overall duration of invasive ventilation (d), mean (sd) | 20.7 (19.6) | 18.3 (13.4) | 23.7 (25.4) | 0.20 | |
| Tracheostomy management | (n = 39) | (n = 25) | (n = 14) | ||
| Day of ICU admission performed, mean (sd) | 19.7 (9.1) | 17.6 (4.9) | 23.5 (13.1) | 0.12 | |
| Days to decannulation after insertion, mean (sd) | 21.9 (16.5) | 15.5 (8.6) | 45.1 (18.5) | 0.01 | a |
HF = high-flow oxygen, IQR = interquartile range, NIV = noninvasive ventilation.
aConsidered to be clinically important as set out in the method.
bPao2/Fio2 12 hr post first prone episode defined as the measurement obtained closest to 12 hr post-initiation of prone ventilation.
Considering both waves together, for patients who did not require IMV, NIV/NHF duration was similar in ICU survivors and nonsurvivors (Appendix 3, http://links.lww.com/CCX/A862). For patients requiring IMV, ICU survivors received a significantly shorter duration of NIV/NHF prior to intubation (10 hr [4–21 hr] vs 50 hr [23–124 hr]; p < 0.01).
Invasive Mechanical Ventilation
One-hundred patients maintained their own airway throughout the entire ICU admission but 106 patients required IMV. Most patients (69.8%) were intubated within 24 hours of arrival to ICU. Four patients were transferred to the ICU with tracheostomies already in situ (Table 2) and a further 39 patients received tracheostomies during their admission. ICU survival was higher in IMV patients who were intubated within 24 hours of arrival versus those intubated after 24 hours (Appendix 3, http://links.lww.com/CCX/A862).
The proportion of all patients who had been intubated prior to ICU admission fell from 25.0% to 13.0% in the second wave (p = 0.02) but ultimately a similar proportion required intubation (54.5% vs 46.0%; p = 0.33).
We observed no difference in airway management amongst patients who were obese body mass index (BMI) ≥ 35 kg/m2 versus those who were not (BMI < 35kg/m2) - Appendix 6 (http://links.lww.com/CCX/A862).
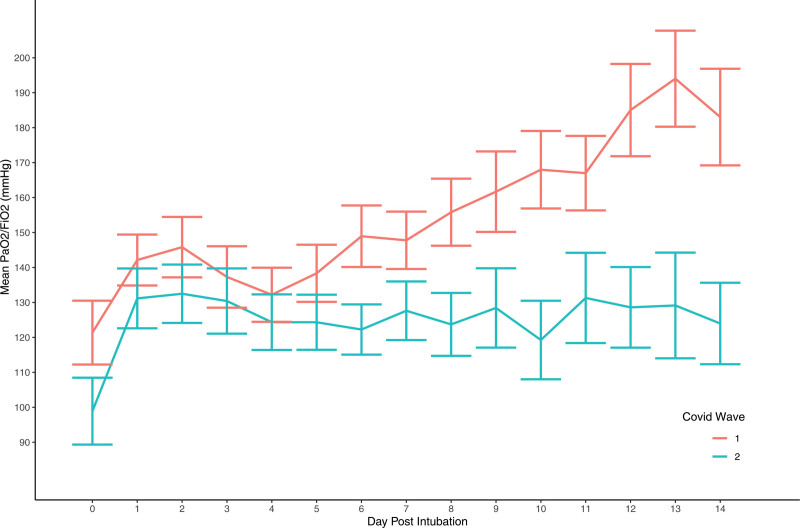
Figure 1 compares daily PF ratios in each cohort of IMV patients. In wave 2, PF ratios were lower and did not show the same trajectory of improvement observed in wave 1 over the first 14 days of IMV. Appendix 4 (http://links.lww.com/CCX/A862) provides a summary of additional ventilatory parameters for IMV patients in each wave.
Figure 1.
Mean Pao2/Fio2 (PF) ratios (mm Hg) per day post-intubation (using lowest daily PF ratio per patient). Error bars represent the sem. COVID = coronavirus disease.
Proning and Neuromuscular Blockade
NMB infusion was used commonly and similarly in both waves (Table 2) (72.6% of IMV patients). The median overall duration of NMB infusion was 111 hours (46–185 hr).
Prone ventilation was employed in 54.7% (58/106) of IMV patients. Those who required proning were less likely to survive to ICU discharge than those who did not (43.1% vs 73.2%; p < 0.01). We observed that proning occurred at a mean PF ratio of 99 mm Hg (sd 58 mm Hg) but that it was instigated at a significantly lower PF ratio in wave 2 (Table 2).
The absolute change in Fio2 at 12 hours compared with the preproning Fio2 was similar in both waves after the first episode of proning (23% in wave 1 vs 24% in wave 2). However, patients in wave 2 started from a significantly higher Fio2 and lower PF ratio prior to proning and had a significantly lower PF ratio at 12 hours after proning.
In the first wave, an Fio2 less than or equal to 55% 12 hours after pronation was common. Overall, 67% of patients (20/30) achieved this response in both their first and second episodes of proning (where required). However, this was not associated with survival to ICU discharge. In wave 2, the same degree of response was less common (36%; 10/28 patients) but those who achieved it were more likely to survive (Appendix 5, http://links.lww.com/CCX/A862). Importantly, we did not observe any survivors in either wave in whom the Fio2 requirement 12 hours after pronation remained greater than 65% in both their first and second episodes and who still went on to be proned on a third occasion (Appendix 5, http://links.lww.com/CCX/A862).
Outcomes
Table 3 shows LOS on ICU according to the level of airway intervention required. Overall, 64.3% patients (135/210) survived to ICU discharge. Survival in wave 2 was higher (68.0% vs 60.9%), although this did not reach statistical significance. In the subgroup of patients with a treatment limitation placed at admission, overall survival was 25% (10/40) (Appendix 2, http://links.lww.com/CCX/A862).
TABLE 3.
ICU Outcomes According to Treatment Wave and Airway Management
| Overall Outcomes | All Patients (n = 210) | Wave 1 (n = 110) | Wave 2 (n = 100) | p | Clinically Important |
|---|---|---|---|---|---|
| ICU nonsurvivors, n (%) | 75 (35.7) | 43 (39.1) | 32 (32.0) | 0.31 | |
| ICU survivors, n (%) | 135 (64.3) | 67 (60.9) | 68 (68.0) | ||
| Own airway throughout (treatment limitation) | 10 (4.8) | 4 (3.6) | 6 (6.0) | ||
| Own airway throughout (no treatment limitation) | 56 (26.7) | 22 (20.0) | 34 (34.0) | 0.01 | a |
| Intubation required | 65 (31.0) | 40 (36.4) | 25 (25.0) | ||
| Tracheostomy already sited on arrival | 4 (1.9) | 1 (0.9) | 3 (3.0) | 0.62 | |
| Overall ICU length of stay (d), median (interquartile range) | 8.6 (4.0–19.5) | 8.6 (3.4–20.8) | 8.8 (4.4–19.1) | 0.49 | |
| Own airway for entire ICU admission | 4.4 (2.6–7.0) | 4.0 (2.5–6.0) | 4.9 (3.2–7.8) | 0.18 | |
| Intubated, no tracheostomy | 13.7 (7.8–18.7) | 13.5 (7.8–17.8) | 14.9 (7.6–22.7) | 0.21 | |
| Intubated then tracheostomy | 33.9 (28.4–49.3) | 33.7 (28.0–38.5) | 51.3 (30.4–74.4) | 0.03 | a |
| Destination on discharge from ICU, n (%) | |||||
| Hospital ward | 124 (59.0) | 62 (56.4) | 62 (62.0) | — | |
| Transferred to other critical care | 9 (4.3) | 3 (2.7) | 6 (6.0) | 0.33 | |
| Transferred to other hospital | 2 (1.0) | 2 (1.8) | 0 (0.0) | ||
aConsidered to be clinically important as set out in the method.
Excluding patients who were transferred to our institution with a tracheostomy in situ, we observed that the proportion of ICU survivors who maintained their own airway throughout ICU admission increased from 39% in the first wave to 59% in the second wave (p = 0.01).
LOS was similar between wave 1 and wave 2 in patients who maintained their own airway for the duration of admission or for patients who required intubation but did not need a tracheostomy.
LOS for the patients who required a tracheostomy was significantly longer in the second wave (51.3 d [30.4–74.4 d] vs 33.7 d [28.0–38.5 d]; p = 0.03).
DISCUSSION
Published studies comparing first and second waves of the pandemic have indicated that the proportion of hospitalized patients requiring ICU admission are falling (9). However, detailed data on the management of critically ill patients in ICU across distinct waves is limited to small cohorts (10, 11). Here, we describe in detail the changing ventilatory care of patients in distinct waves of the pandemic in a large ICU. Reassuringly, the broad picture is in keeping with the U.K.’s ICNARC (12). Our observation that more ICU survivors have been managed without IMV is encouraging. This group is likely to need less intensive post ICU therapy with positive implications for pressurized rehabilitation services.
The changes we observed may reflect evolving clinical experience or the impact of novel therapies introduced during the study, such as routine dexamethasone use. Changes in attitudes to ICU admission may have played a role, although there was no change in admission policy and baseline characteristics remained similar throughout our study. We draw attention to several notable differences between waves that are worthy of highlight.
First, although the proportion of patients receiving NIV/NHF in each wave was similar, the duration of NIV/NHF use was much longer in wave 2. In general, IMV was also initiated later in wave 2. We observed fewer patients intubated prior to ICU admission, a trend toward lower overall proportions intubated within 24 hours and a smaller proportion of ICU survivors who received IMV.
It is possible that during wave 1, driven by uncertainty about a new disease and concerns over effectiveness of personal protective equipment (PPE), clinicians opted to intubate early. Some of these patients may have been managed with NIV/NHF only. In wave 2, where clinicians had greater confidence in management and in the efficacy of PPE, they may have felt more comfortable to persist with noninvasive respiratory support for longer (13). This trend toward lower rates of IMV in the second wave is consistent with larger datasets, and may highlight the benefits of new therapies utilized in the second wave, that had not been proven in the first wave (9, 14).
Although use of NIV/NHF appears encouraging and is supported by recent results from Recovery-Respiratory Support (RS), it is notable that the use of continuous positive airway pressure in Recovery-RS was found to reduce the need for intubation but not 30-day mortality (15). Taken in this context, our results imply optimal timing of intubation is key, particularly as worse outcome attributed to delay in intubation in patients failing NHF and NIV have previously been reported (16, 17). In our view, predicting NIV failure remains challenging and few studies have attempted to predict NIV failure in COVID-19 (18). Here, we highlight the challenge facing clinicians when applying NIV/NHF in COVID-19 patients. In wave 2, a greater proportion of patients survived to ICU discharge having only received NIV/NHF, and yet in both waves, a longer duration of NIV/NHF on ICU prior to intubation was associated with higher ICU mortality. This finding has also been observed by the NHS extracorporeal membrane oxygenation service (19). Additional research is needed to distinguish which patients might benefit from continued NIV, from those where the chance of failure is high, thus prompting consideration to earlier intubation.
Our second observation, in intubated patients, was that those in wave 2 had clinical parameters indicating IMV was more difficult. We observed worse PF ratios following intubation, a reduction in the rates of patients who displayed a good response to proning, and longer tracheostomy weans. Possible explanations include longer durations of NIV/NHF therapy utilized prior to intubation as well as change in clinician practice as to when proning was initiated. Immunosuppressive therapies may have increased susceptibility to secondary infection, and this is worthy of further exploration. Finally, routine dexamethasone therapy in addition to NMB and deep sedation may have increased rates of critical illness weakness, which we have already been noted to be problematic in COVID-19 ICU survivors (20).
Our third observation is that in both waves, NMB and proning were frequently and recurrently required. NMB was used for longer than in previous trial protocols (7, 21). This reflects our anecdotal experience that patients frequently exhibited acute desaturation episodes after withdrawal of NMB. Proning was undertaken in greater than or equal to 50% of IMV patients in both waves, but we observed that proning was initiated at significantly lower PF ratios in the second wave and in general was initiated at lower PF ratios than anticipated. The reasons for this are unclear but may reflect the high burden of care required for IMV proned patients and the impact this has on ICU resources.
We acknowledge several limitations of our work. This is a single-center retrospective study that limits generalizability. However, the management we describe is based on accepted practices, and our outcomes are consistent with U.K. national data (12). Some patients in wave 2 were transferred for capacity reasons with tracheostomies in situ but the LOS elsewhere for these patients merely adds weight to our argument that these patients had a notably increased LOS compared with wave 1.
Taken together, our findings have several implications. They prompt us to consider cautiously the role of prolonged NIV/NHF, particularly when the acute respiratory failure is typically of weeks duration, and longer than most other forms of pneumonia, including those in the FLORALI study (22). Particular caution should be applied in patients with features of rising work of breathing. Utilizing NHF/NIV early in those with better lung compliance, with a switch to IMV as work of breathing increases and compliance falls is a goal, but challenges remain as to identifying when this cross-over occurs.
Our findings should also encourage clinicians to evaluate the effectiveness of each proning episode. We advise careful consideration prior to repeated proning in order to avoid futile episodes in patients who fail to show a good response particularly after two attempts. However, our results also support a redoubling of efforts to undertake timely prone positioning as earlier proning in wave 1 typically achieved a lower Fio2 and failure to achieve this in wave 2 was associated with mortality.
Last, given the marked increase in LOS for tracheostomy patients in wave 2, there is a need to deliver coordinated multidisciplinary tracheostomy care and ensure effective physical rehabilitation for longer stay IMV/tracheostomized COVID-19 patients.
CONCLUSIONS
The care of patients with COVID-19 in critical care will continue to evolve as new therapies become available and clinicians develop greater experience. Such changes will undoubtedly impact the ventilatory management of patients and indeed the cohort who progress to require IMV.
We have compared two waves of patients cared for at our institution, coinciding with a pre- and post-dexamethasone era and observed that in wave 2, a greater proportion of patients survived to ICU discharge without the need for IMV and that trials of NIV and HF oxygen therapy were continued for significantly longer periods. Patients who were ultimately intubated remained more hypoxic and showed poorer responses to proning, although this may be due to proning initiation occurring when more hypoxic. Importantly, prolonged trials of NIV/HF prior to IMV were associated with higher ICU mortality in both waves.
While it is encouraging that more patients are surviving to ICU discharge without the need for IMV, clinicians should carefully consider the benefits of prolonged trials of NIV/HF and be prepared for challenging management in patients who are ultimately intubated.
Supplementary Material
Footnotes
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccejournal).
The authors have disclosed that they do not have any potential conflicts of interest.
This work was performed at Adult Critical Care Department, Manchester Royal Infirmary, Manchester University NHS Foundation Trust, Manchester, United Kingdom.
REFERENCES
- 1.Horby P, Lim WS, Emberson JR, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021; 384:693–704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gordon AC, Mouncey PR, Al-Beidh F, et al. Interleukin-6 receptor antagonists in critically ill patients with Covid-19. N Engl J Med. 2021; 384:1491–1502 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.European Centre for Disease Prevention and Control. Case Definition for Coronavirus Disease 2019 (COVID-19), As of 29 May 2020. Available at: https://www.ecdc.europa.eu/en/covid-19/surveillance/case-definition. Accessed August 11, 2020
- 4.Intensive Care National Audit & Research Centre. Available at: https://www.icnarc.org/. Accessed July 26, 2020
- 5.Elshof J, Hebbink RHJ, Duiverman ML, et al. High-flow nasal cannula for COVID-19 patients: Risk of bio-aerosol dispersion. Eur Respir J. 2020; 56:2003004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abani O, Abbas A, Abbas F, et al. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet. 2021; 397:1637–1645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Papazian L, Forel J-M, Gacouin A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010; 363:1107–1116 [DOI] [PubMed] [Google Scholar]
- 8.Guérin C, Reignier J, Richard JC, et al. ; PROSEVA Study Group. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013; 368:2159–2168 [DOI] [PubMed] [Google Scholar]
- 9.Karagiannidis C, Windisch W, McAuley DF, et al. Major differences in ICU admissions during the first and second COVID-19 wave in Germany. Lancet Respir Med. 2021; 9:e47–e48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Contou D, Fraissé M, Pajot O, et al. Comparison between first and second wave among critically ill COVID-19 patients admitted to a French ICU: No prognostic improvement during the second wave? Crit Care. 2021; 25:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Taboada M, González M, Alvarez A, et al. First, second and third wave of COVID-19. What have we changed in the ICU management of these patients? J Infect. 2021; 82:e14–e15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Intensive Care National Audit & Research Centre. Reports. Available at: https://www.icnarc.org/Our-Audit/Audits/Cmp/Reports. Accessed May 7, 2021
- 13.El Bouzidi K, Pirani T, Rosadas C, et al. ; COVID-STOICS (Serological Testing Of Intensive Care Staff). Severe acute respiratory syndrome coronavirus-2 infections in critical care staff: Beware the risks beyond the bedside. Crit Care Med. 2021; 49:428–436 [DOI] [PubMed] [Google Scholar]
- 14.Intensive Care National Audit & Research Centre. Reports. 2021. Available at: https://www.icnarc.org/Our-Audit/Audits/Cmp/Reports. Accessed April 16, 2021
- 15.Perkins GD, Ji C, Connolly BA, et al. An adaptive randomized controlled trial of non-invasive respiratory strategies in acute respiratory failure patients with COVID-19. medRxiv. Preprint posted online August 4, 2021. doi: 10.1101/2021.08.02.21261379 [Google Scholar]
- 16.Kang BJ, Koh Y, Lim CM, et al. Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med. 2015; 41:623–632 [DOI] [PubMed] [Google Scholar]
- 17.Bellani G, Laffey JG, Pham T, et al. ; LUNG SAFE Investigators; ESICM Trials Group. Noninvasive ventilation of patients with acute respiratory distress syndrome. Insights from the LUNG SAFE study. Am J Respir Crit Care Med. 2017; 195:67–77 [DOI] [PubMed] [Google Scholar]
- 18.Arina P, Baso B, Moro V, et al. ; UCL Critical Care COVID-19 Research Group. Discriminating between CPAP success and failure in COVID-19 patients with severe respiratory failure. Intensive Care Med. 2021; 47:237–239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Camporota L, Meadows C, Ledot S, et al. ; NHS England ECMO Service. Consensus on the referral and admission of patients with severe respiratory failure to the NHS ECMO service. Lancet Respir Med. 2021; 9:e16–e17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Parker AJ, Humbir A, Tiwary P, et al. Recovery after critical illness in COVID-19 ICU survivors. Br J Anaesth. 2021; 126:e217–e219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moss M, Huang DT, Brower RG, et al. Early neuromuscular blockade in the acute respiratory distress syndrome. N Engl J Med. 2019; 380:1997–2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Frat JP, Thille AW, Mercat A, et al. ; FLORALI Study Group; REVA Network. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015; 372:2185–2196 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.