Abstract
Digital health tools can promote disease self-management, but the association of smartphone app engagement and medication adherence is unclear. We assessed the relationship between objective smartphone app engagement and controller medication use in adults with asthma and COPD. We retrospectively analyzed data from participants enrolled in a digital self-management platform for asthma and COPD. Eligible adults had a smartphone and a paired electronic medication monitor (EMM). Longitudinal, mixed-effects logistic regressions estimated the relationship between daily app engagement (app opens, session duration) and daily controller medication use. Data from 2309 participants (71% asthma; 29% COPD) was analyzed. Opening the app (vs. not opening the app) was associated with significantly greater odds (OR (95% CI)) of using controller medications in asthma (2.08 (1.98, 2.19)) and COPD (1.61 (1.49, 1.75). Longer session duration was also associated with greater odds of using controller medications in asthma and COPD, but the odds of use attenuated with longer session duration in COPD. This study presents a novel assessment of the relationship between objectively-measured smartphone app engagement and controller medication use in asthma and COPD. Such insights may help develop targeted digital health tools and interventions.
Subject terms: Asthma, Chronic obstructive pulmonary disease, Human behaviour
Introduction
In the United States, it is estimated that 24.7 million children and adults have asthma1, and more than 16 million adults have COPD2. While distinct diseases, patients with asthma and COPD may experience similar symptoms like shortness of breath, chest discomfort and frequent cough3.
Inhaled controller medications help patients manage their asthma and COPD and may improve patient quality of life. Yet, adherence to these daily inhalers is frequently suboptimal, with estimates of controller adherence averaging 30–50% for patients with asthma4 and less than 50% for patients with COPD5,6. Medication non-adherence may lead to poor patient outcomes and be costly to patients and the healthcare system more broadly7–9.
Use of smartphone self-management applications (“apps”) have demonstrated value in helping people with asthma and COPD improve or maintain good medication adherence10–12 especially if behavior change techniques like feedback, education and self-monitoring are integrated and address core barriers to poor adherence13–15. A systematic review of digital interventions for asthma self-management found that digital programs integrating theory-based behavioral change approaches significantly improved medication adherence compared to those that did not16.
Despite this encouraging evidence, the impact of digital tools like smartphone apps can be limited by challenges not only in retention, but in engagement as well17. Further, self-reported engagement can be prone to bias and limited in what it can measure18,19. As such, it remains unclear what the relationship between app engagement and medication adherence is, especially in asthma and COPD. Thus, we aimed to explore the relationship between app engagement and medication adherence using objective data collected from a real-world sample of adults with asthma and COPD enrolled in a digital health platform.
Methods
Recruitment and eligibility
Data used in this retrospective analysis was collected from participants who enrolled in a digital self-management platform (Propeller Health, WI, USA) between January 2018 and March 2019. Participants who enrolled in the platform were recruited via social media campaigns (e.g., Facebook advertisements), and needed to own a smartphone and have a self-reported history of asthma and/or COPD to be eligible. All participants agreed to the platform’s Terms of Service.
Data was examined retrospectively using an aggregated dataset. To be included in the analysis, participants needed to be ≥ 18 years of age, reside in the United States, and have a controller inhaler compatible with an electronic medication monitor (EMM). Participants also needed to have at least 97 days of controller medication use data (the first seven days of participation was considered an onboarding period). The retrospective analysis plan was determined to be exempt and consent was waived by the Copernicus Independent Review Board (PRH1-18-132). A subset of data used in this retrospective analysis included data previously collected from an electronic survey to which patients provided consent (Protocol 20191728). All methods were carried out in accordance with relevant guidelines and regulations.
Description of the digital health platform
Propeller Health is an FDA-cleared digital platform that includes an EMM, and a paired smartphone app targeted to the user’s self-reported condition (asthma or COPD) (Fig. 1).
Figure 1.
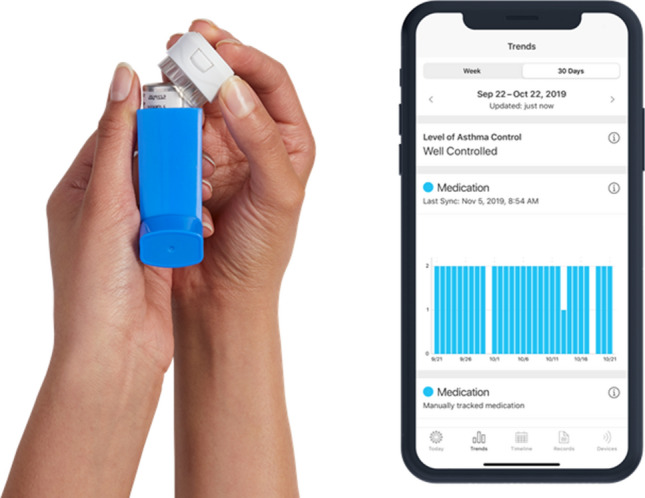
A small FDA-cleared electronic medication monitor (EMM) is attached to the user’s controller medication inhaler to capture the date and time of use. Data from the EMM is then transferred wirelessly via Bluetooth to a paired smartphone app providing feedback, insights and medication reminders.
Electronic medication monitors (EMMs) attach to a compatible inhaler medication to passively capture the date and time of use. Usage data is then wirelessly transferred via Bluetooth to a paired smartphone app. The EMM has a battery life of 12–18 months and does not require charging11. Compatible medications include inhaled corticosteroids (ICS), long-acting beta-agonists (LABA), long-acting muscarinic antagonists (LAMA), and combination therapies (ICS + LABA, LABA/LAMAs and triple therapy).
The paired smartphone app serves not only to collect data from the EMM, but also to engage the user through evidence-based asthma and/or COPD content, including relevant guideline content20,21, feedback on medication use and trends, and schedule-based medication reminders through the EMM and smartphone application. Patients with continued poor medication adherence may be presented with additional gamified features and challenges aimed at improving daily medication adherence. The app also prompts users to complete an in-app Asthma Control Test (ACT) or COPD Assessment Test (CAT) at baseline and monthly thereafter to better assess disease control and burden, respectively (Supplementary Fig. 1a,b).
Outcome and measures
Smartphone app engagement
We examined app engagement, defined by daily app opens and app active session duration, as the independent variable. Daily app engagement was defined differently for each model estimate to explore the varying associations of duration on controller medication adherence: Model 1 (no app open vs. any app open regardless of session duration) and Model 2 (no app open vs. at least one app open with < 1, 1–< 5, 5–10 and 10+ minutes of total daily app session duration).
Controller medication use
Controller inhaler use was determined using data collected from the EMM, which recorded the date and time of each inhaler actuation. Adherence was calculated by dividing the number of EMM-recorded actuations by the prescribed number of actuations (reported by the participant during enrollment) × 100 per day.
For the primary analyses, the outcome of controller medication use was dichotomously defined as having taken at least 1 actuation vs. no actuation per day. Controller adherence was dichotomized because daily adherence was either 0% or 100% on the majority of participant-days (74%). For secondary analyses, controller medication use was defined as being 100% adherent vs. < 100% per day. Being 100% adherent was defined as having EMM-recorded controller actuations that were greater than or equal to the total number of prescribed actuations for that day.
Self-reported measures of control and burden in asthma and COPD
Symptom control was assessed with the Asthma Control Test (ACT), a self-administered, validated questionnaire for patients with asthma. The 5-item assessment asks respondents to rate their symptoms on a scale of 1–5. A score > 19 indicates good symptom control, 15–19 indicates not well-controlled symptoms, and < 15 indicates very poorly controlled symptoms22.
Disease burden for participants with COPD was assessed with the COPD Assessment Test (CAT). The 8-item self-administered questionnaire asks respondents to rate their symptoms on a scale of 1–5. Summative scores < 20 indicate lower disease burden, and scores ≥ 20 indicate higher disease burden23.
Analyses
Longitudinal, mixed-effects logistic regressions were used to estimate the association between daily app engagement and daily controller medication adherence during days 8–97 of EMM use for asthma and COPD, adjusting for age, gender, smartphone type (iOS vs. Android), baseline disease status (defined as initial ACT or CAT score), rescue medication use, day on platform and US Census-derived neighborhood-level income and education. Analyses were also stratified by age and disease severity. Stratification by age was completed for participants 40 years and older to allow for comparisons between asthma and COPD. Odds ratios and 95% confidence intervals (CI) were presented, with alpha = 0.05 as the significance threshold.
Role of funding source
Representatives from the study sponsors (Propeller Health, Council of State and Territorial Epidemiologists) were involved in the study design, collection, analysis and interpretation of data, writing of the report, and in the decision to submit the paper for publication. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
Demographics
There were 2309 participants included in the analysis of whom 71% (N = 1629) had self-reported asthma, and 29% (N = 663) had self-reported COPD. Participants with asthma had a mean age (SD) of 39 (12) years and 80% were female. Participants with COPD had a mean (SD) age of 60 (9) years and 67% were female. At baseline, participants with asthma self-reported very poor control (mean ACT score: 13.2) and participants with COPD reported higher COPD burden (mean CAT score: 23.7) (Table 1).
Table 1.
Participant characteristics*
| Asthma n = 1629 |
COPD n = 663 |
|
|---|---|---|
| Age (mean (SD)); years | 39.4 (12.6) | 60.9 (8.3) |
| Female, n (%) | 1302 (80) | 443 (67) |
| Baseline CAT, mean (SD) | – | 23.8 (7.5) |
| Baseline ACT, mean (SD) | 13.3 (4.5) | – |
| Uncontrolled Asthma (ACT < 20), n (%) | 1448 (90.1) | |
| Higher Burden COPD (CAT > 20), n (%) | 449 (67.7) | |
| Android, n (%) | 860 (53) | 437 (66) |
| Rescue use (mean (SD)), puffs/day | 1.0 (1.8) | 1.74 (2.5) |
| Daily medication adherence (mean (SD)), % | 45 (32) | 62 (32) |
| Percent of days with 100% adherence (mean (SD)), % | 31 (33) | 50 (38) |
| Percent of days with app opens (mean (SD)), % | 16 (19) | 28 (27) |
| App opens/day (mean (SD)) | 0.2 (0.4) | 0.5 (0.8) |
| Daily app session duration (mean (SD)), mina | 4.4 (6.5) | 4.3 (5.0) |
*90 days of participant data included e.g., mean rescue puffs/day was calculated over 90 days.
aCapped at 60 min/day.
App opens, session duration and medication adherence
Over 90 days, unadjusted mean (SD) medication adherence was higher among participants with COPD than participants with asthma: 62 (32)% vs. 45 (32)%, respectively. On average, participants with asthma and COPD spent a similar amount of time in the app per day: 4.4 (6.5) min compared to 4.3 (5.0) min, respectively. Participants with COPD opened their app more often than participants with asthma (mean daily app opens (SD): 0.5 (0.8) vs. 0.2 (0.4)).
In participants with asthma (n = 1629), opening the app on a given day was associated with higher odds of using controller medication compared to participants who did not open the app; OR (95% CI) 2.08 (1.98, 2.19), p < 0.001. Participants who spent more time in the app were more likely to use their controller medication compared to those participants who spent no time in the app, and the odds of controller use was higher with greater app duration: < 1 min: OR (95% CI) 1.95 (1.82, 2.08) vs. > 10 min OR (95% CI) 2.70 (2.32, 3.14) (Table 2).
Table 2.
Odds ratios between any app open (reference = no app use) and any daily controller medication use, 90 days (n (asthma) = 1629, n (COPD) = 663).
| Model | Odds ratio (ref = no app open) | Odds ratio | Lower 95% CI | Upper 95% CI | p |
|---|---|---|---|---|---|
| Asthma | |||||
| 1 | Any app open | 2.08 | 1.98 | 2.19 | < 0.001 |
| 2 | < 1 min duration | 1.95 | 1.82 | 2.08 | < 0.001 |
| 1–< 5 min duration | 2.14 | 1.97 | 2.32 | < 0.001 | |
| 5–10 min duration | 2.30 | 1.93 | 2.75 | < 0.001 | |
| 10+ min duration | 2.70 | 2.32 | 3.14 | < 0.001 | |
| COPD | |||||
| 1 | Any app open | 1.61 | 1.49 | 1.75 | < 0.001 |
| 2 | < 1 min duration | 1.87 | 1.69 | 2.07 | < 0.001 |
| 1–< 5 min duration | 1.38 | 1.23 | 1.54 | < 0.001 | |
| 5–10 min duration | 1.58 | 1.26 | 1.98 | < 0.001 | |
| 10+ min duration | 1.34 | 1.10 | 1.61 | 0.003 | |
All generalized linear mixed effects logistic models adjusted for census-level income and education, age, gender, android (vs. IOs), ACT (for asthma) or CAT (for COPD) score, days since first controller EMM sync, and included a random intercept for participant to account for repeated measures.
In participants with COPD (n = 663), opening the app on a given day was also associated with higher odds of using controller medications compared to those participants who did not open the app (OR (95% CI) 1.61 (1.49, 1.75); p < 0.001). Participants who spent any amount of time in the app were more likely to use their controller medication compared to those who spent no time in the app; however, the magnitude of the odds of using controller medications per day attenuated with longer durations spent in the app: < 1 min OR (95% CI) 1.87 (1.69, 2.07) vs. > 10 min OR (95% CI) 1.34 (1.10, 1.61) (Table 2).
Similar patterns were observed in secondary analyses examining association with 100% controller adherence in participants with asthma and COPD (Supplementary Table 1).
Differences by age were observed for session durations of 5–10 min in both asthma and COPD, such that participants 40–60 years had higher odds of using controller medication than those > 60 years. Differences by disease severity were not observed for either asthma or COPD (Supplementary Tables 2, 3).
Discussion
This study demonstrates that in a large, real-world sample of adults with asthma or COPD using a digital health platform, greater smartphone app engagement is associated with higher absolute medication use. Specifically, we observed a significant relationship between controller medication use and daily app opens as well as the length of time spent in the app. Differences in app usage by disease type were also observed such that participants with COPD opened the app more often, but had diminishing benefit from prolonged session durations (10+ minutes) compared to participants with asthma.
At present, research on objective measures of smartphone app behavior and its relation to desired outcomes in respiratory disease management is relatively limited. However, research in other chronic conditions have demonstrated similar positive relationships between objective app engagement and positive behavior change. For example, in a small randomized trial promoting physical activity, Edney24 found that participants with high levels of app engagement, defined as daily app login and feature interaction, completed significantly more minutes of physical activity than those with lower levels of app engagement. In another study examining app engagement across multiple mobile applications for mental health, frequency of app use as well as lifetime app use was positively associated with improvements in depression and anxiety25. While these studies support our initial findings, more evidence is needed in asthma and COPD.
In our study, we examined outcomes by any medication use (< 100% of prescribed doses) (Table 2) and by 100% medication use (Supplementary Table 1). We observed that the odds of meeting 100% medication use were attenuated compared to odds of meeting < 100% medication use in both asthma and COPD when opening the app. The same trend was observed for session duration. This finding is not surprising, as patients who achieve their behavioral goal may be more motivated, and have greater self-efficacy26, and as such may not have relied on the app features to achieve their goals.
We also observed that the likelihood of using controller medications increased with longer session duration among participants with asthma, but the same did not hold true for those with COPD even when controlling for age. While we did not assess the change in app usage over time, it is possible that frequency of app use, not necessarily longer app sessions durations, could help explain the relationship observed, especially given that participants with COPD had a higher number of mean app opens per day. The literature also suggests that knowledge can play an important role in promoting medication adherence27. As such, it’s possible that participants with COPD were better educated about their condition (possibly due to more clinical visits and consequently, education) and may have relied more heavily on other features of the digital platform not requiring them to open the app, e.g., the audio-visual reminders on the EMM or the app-based push notifications. Further investigation is needed to confirm these hypotheses.
Finally, we observed some age-related variability among older adults (40–60 vs. 60 years of age) for both asthma and COPD, but its interpretation is unfortunately limited by smaller sample sizes. Further, while we did not observe a moderating effect by disease severity, there is evidence suggesting that patients who do not meet their behavioral goals are less likely to engage in that target behavior28. As such, it is possible that level of adherence to medication, rather than disease severity, may have moderated engagement so that those participants who met their adherence goals were more likely to engage with the app, than those who did not meet their adherence goals.
Limitations
This retrospective analysis is an important first step in better understanding objective measures of smartphone app engagement, but there are limitations. First, we did not have access to participant-level demographic information such as race or socioeconomic status, which may be important determinants of engagement and medication adherence. Second, while we measured two objective measures of engagement, other objective measures like app push notifications and reminders, daily and monthly active user ratios (DAU/ MAU)29, number of in-app clicks, opening of app content pages, etc. may also provide useful signals. Third, following examination of the data for the secondary analysis, we decided posteriori to assess medication use as a dichotomous outcome instead of a continuous variable due to the non-normal distribution. Fourth, the generalizability of the results may be limited given that the majority of participants were female (research suggests that women tend to be more engaged than men in digital platforms30), and that participants who enrolled in the platform were potentially more motivated to improve their health. Finally, data on behavior change determinants of medication adherence were not collected, limiting insight into the behavior change pathways leading to the outcomes observed in the analysis31. Further, we did not examine outcomes by dosing schedule, nor were participants asked to report if they had shared their data with their healthcare provider32 both of which could be important moderating factors.
Conclusion
Using objective app usage data, this retrospective analysis demonstrates how use of a smartphone app for asthma and COPD management is associated with medication adherence. Future work should explore additional measures of smartphone app engagement, as well as measure and assess behavior change pathways that may impact medication use over time.
Supplementary Information
Acknowledgements
This work was funded by Propeller Health, an affiliate of ResMed, and the Council of State and Territorial Epidemiologists, through their CDC cooperative agreement number, 5U38OT000143-05. The contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of CDC. With thanks to the manuscript reviewers for their comments and insights, as well as Vy Vuong for additional statistical consultation.
Abbreviations
- ACT
Asthma control test
- CAT
COPD assessment test
- COPD
Chronic obstructive pulmonary disease
- CI
Confidence interval
- EMM
Electronic medication monitor
- SD
Standard deviation
- OR
Odds ratio
Author contributions
All authors contributed to the conceptualization and design of the manuscript, data interpretation, and manuscript preparation and revision. Analyses were performed by RG.
Data availability
Individual participant data will not be available publicly or at request.
Competing interests
LK, RG and MAB are paid employees of ResMed and former employees of Propeller Health. DAS is a current employee of Propeller Health, an affiliate of ResMed. AT was formerly employed at the Council of State and Territorial Epidemiologists.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-03827-2.
References
- 1.Centers for Disease Control and Prevention (CDC), Most Recent National Asthma Data|CDC. 2020. https://www.cdc.gov/asthma/most_recent_national_asthma_data.htm. Accessed 06 Jul 2020.
- 2.CDC-Basics About COPD-Chronic Obstructive Pulmonary Disease (COPD). 2019. https://www.cdc.gov/copd/basics-about.html. Accessed 06 Jul 2020.
- 3.Chronic Obstructive Pulmonary Disease | AAAAI. The American Academy of Allergy, Asthma & Immunology. https://www.aaaai.org/conditions-and-treatments/related-conditions/chronic-obstructive-pulmonary-disease. Accessed 06 Jul 2020.
- 4.Engelkes M, Janssens HM, de Jongste JC, Sturkenboom MCJM, Verhamme KMC. Medication adherence and the risk of severe asthma exacerbations: A systematic review. Eur. Respir. J. 2015;45(2):396–407. doi: 10.1183/09031936.00075614. [DOI] [PubMed] [Google Scholar]
- 5.Bourbeau J, Bartlett SJ. Patient adherence in COPD. Thorax. 2008;63(9):831–838. doi: 10.1136/thx.2007.086041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gillespie CW, Morin PE, Tucker JM, Purvis L. Medication adherence, health care utilization, and spending among privately insured adults with chronic conditions in the United States, 2010–2016. Am. J. Med. 2020;133(6):690–704.e19. doi: 10.1016/j.amjmed.2019.12.021. [DOI] [PubMed] [Google Scholar]
- 7.Mäkelä MJ, Backer V, Hedegaard M, Larsson K. Adherence to inhaled therapies, health outcomes and costs in patients with asthma and COPD. Respir. Med. 2013;107(10):1481–1490. doi: 10.1016/j.rmed.2013.04.005. [DOI] [PubMed] [Google Scholar]
- 8.Otsuki-Clutter, M., Sutter, M. & Ewig, J. Promoting adherence to inhaled corticosteroid therapy in patients with asthma. Former USFSP Sch. 2011, [Online]. http://digital.usfsp.edu/former-pub/43.
- 9.George M. Adherence in asthma and COPD: New strategies for an old problem. Respir. Care. 2018;63(6):818–831. doi: 10.4187/respcare.05905. [DOI] [PubMed] [Google Scholar]
- 10.Van Sickle D, Barrett M, Humblet O, Henderson K, Hogg C. Randomized, controlled study of the impact of a mobile health tool on asthma SABA use, control and adherence. Eur. Respir. J. 2016;48:suppl 60. doi: 10.1183/13993003.congress-2016.PA1018. [DOI] [Google Scholar]
- 11.Mosnaim GS, et al. The impact of patient self-monitoring via electronic medication monitor and mobile app plus remote clinician feedback on adherence to inhaled corticosteroids: A randomized controlled trial. J. Allergy Clin. Immunol. Pract. 2020 doi: 10.1016/j.jaip.2020.10.064. [DOI] [PubMed] [Google Scholar]
- 12.Moore A, et al. A randomised controlled trial of the effect of a connected inhaler system on medication adherence in uncontrolled asthmatic patients. Eur. Respir. J. 2020 doi: 10.1183/13993003.03103-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Unni E, Gabriel S, Ariely R. A review of the use and effectiveness of digital health technologies in patients with asthma. Ann. Allergy. Asthma. Immunol. 2018;121(6):680–691.e1. doi: 10.1016/j.anai.2018.10.016. [DOI] [PubMed] [Google Scholar]
- 14.Ramsey RR, Plevinsky JM, Kollin SR, Gibler RC, Guilbert TW, Hommel KA. Systematic review of digital interventions for pediatric asthma management. J. Allergy Clin. Immunol. Pract. 2020;8(4):1284–1293. doi: 10.1016/j.jaip.2019.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yang F, Wang Y, Yang C, Hu H, Xiong Z. Mobile health applications in self-management of patients with chronic obstructive pulmonary disease: A systematic review and meta-analysis of their efficacy. BMC Pulm. Med. 2018 doi: 10.1186/s12890-018-0671-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lycett HJ, et al. Theory-based digital interventions to improve asthma self-management outcomes: Systematic review. J. Med. Internet Res. 2018;20(12):e293. doi: 10.2196/jmir.9666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Serrano KJ, Coa KI, Yu M, Wolff-Hughes DL, Atienza AA. Characterizing user engagement with health app data: A data mining approach. Transl. Behav. Med. 2017;7(2):277–285. doi: 10.1007/s13142-017-0508-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kosse RC, Bouvy ML, Belitser SV, De Vries TW, Van Der Wal PS, Koster ES. Effective engagement of adolescent asthma patients with mobile health–supporting medication adherence. JMIR mHealth uHealth. 2019;7(3):e12411. doi: 10.2196/12411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Osterberg L, Blaschke T. Adherence to medication. N. Engl. J. Med. 2005;353(5):487–497. doi: 10.1056/NEJMra050100. [DOI] [PubMed] [Google Scholar]
- 20.Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2016. http://www.ginasthma.org.
- 21.Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. https://goldcopd.org/2021-gold-reports/.
- 22.Nathan RA, et al. Development of the asthma control test: A survey for assessing asthma control. J. Allergy Clin. Immunol. 2004;113(1):59–65. doi: 10.1016/j.jaci.2003.09.008. [DOI] [PubMed] [Google Scholar]
- 23.Jones PW, Harding G, Berry P, Wiklund I, Chen W-H, Kline Leidy N. Development and first validation of the COPD assessment test. Eur. Respir. J. 2009;34(3):648–654. doi: 10.1183/09031936.00102509. [DOI] [PubMed] [Google Scholar]
- 24.Edney S, et al. User engagement and attrition in an app-based physical activity intervention: Secondary analysis of a randomized controlled trial. J. Med. Internet Res. 2019;21(11):e14645. doi: 10.2196/14645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kwasny MJ, Schueller SM, Lattie E, Gray EL, Mohr DC. Exploring the use of multiple mental health apps within a platform: Secondary analysis of the intellicare field trial. JMIR Ment. Health. 2019;6(3):e11572. doi: 10.2196/11572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Martos-Méndez MJ. Self-efficacy and adherence to treatment: The mediating effects of social support. J. Behav. Health Soc. Issues. 2015;7(2):19–29. doi: 10.5460/jbhsi.v7.2.52889. [DOI] [Google Scholar]
- 27.Kahwati L, Viswanathan M, Golin CE, Kane H, Lewis M, Jacobs S. Identifying configurations of behavior change techniques in effective medication adherence interventions: A qualitative comparative analysis. Syst. Rev. 2016 doi: 10.1186/s13643-016-0255-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tanenbaum ML, Ross KM, Wing RR. Overeat today, skip the scale tomorrow: An examination of caloric intake predicting nonadherence to daily self-weighing. Obesity. 2016;24(11):2341–2343. doi: 10.1002/oby.21650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Carlo AD, HosseiniGhomi R, Renn BN, Strong MA, Areán PA. Assessment of real-world use of behavioral health mobile applications by a novel stickiness metric. JAMA Netw. Open. 2020;3:8. doi: 10.1001/jamanetworkopen.2020.11978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Carroll JK, Moorhead A, Bond R, LeBlanc WG, Petrella RJ, Fiscella K. Who uses mobile phone health apps and does use matter? A secondary data analytics approach. J. Med. Internet Res. 2017;19(4):e125. doi: 10.2196/jmir.5604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bidargaddi N, et al. To prompt or not to prompt? A microrandomized trial of time-varying push notifications to increase proximal engagement with a mobile health app. JMIR MHealth UHealth. 2018;6(11):e10123. doi: 10.2196/10123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wilson SR, et al. Shared treatment decision making improves adherence and outcomes in poorly controlled asthma. Am. J. Respir. Crit. Care Med. 2010;181(6):566–577. doi: 10.1164/rccm.200906-0907OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Individual participant data will not be available publicly or at request.


