Introduction
Cutaneous adverse effects during and after treatment of breast cancer with radiation therapy (RT) are common, with radiation dermatitis occurring in 74% to 100% of patients.1,2 Other known cutaneous side effects include radiation-induced morphea, radiation-induced fibrosis, radiation recall dermatitis, secondary malignancies, erosions, and ulcerations.3 Blistering diseases such as bullous pemphigoid (BP) are rarely triggered by RT.4,5
Bullous pemphigoid is overall the most common autoimmune blistering disease and is characterized by subepidermal blistering and autoantibody formation against the dermal-epidermal junction.6 It is most frequently seen in elderly patients in the 8th decade of life.6 Generally, BP presents with a pruritic prebullous phase that may last for several months, followed by the formation of tense bullae and urticarial plaques, sometimes with mucosal involvement, that can be either generalized or localized.6 The pathophysiology involves tissue-bound and circulating autoantibodies to BP180 and BP230 proteins, components of the hemidesmosome.7 Only 15% of patients present with an identifiable inciting factor for BP, such as viral infections, various drugs (eg, furosemide or immune checkpoint inhibitors), and rarely, RT.8
We present a unique case of a patient who experienced a chronic generalized pruritic eruption for years and who developed severe generalized BP while undergoing RT for invasive ductal carcinoma of the breast.
Case Description
A 73-year-old postmenopausal woman with pT1cN0 well-differentiated invasive ductal carcinoma (ER/PR+, HER2–) of the breast who had undergone a right partial mastectomy was noted to have a chronic pruritic rash involving the proposed radiation field at the time of RT consultation. Thus, she was also referred to the dermatology department to assess the skin lesions. She had no history of prior RT or of any collagen vascular disease. At the time of the initial dermatologic consultation, she exhibited scattered pruritic erythematous papules with overlying erosions on the chest, back, arms, and legs that had been present for several years and had no specific triggers. There were also numerous angulated and linear erosions with crust (consistent with excoriations), a background of white atrophic scars (suggesting chronicity of the process), and xerosis. A diagnosis of prurigo simplex with nonspecific eczematous dermatitis was made. The patient was prescribed twice daily 0.1% triamcinolone cream. The generous use of emollients was recommended during her RT.
The patient was started on her whole-breast RT plan with standard tangents, without a boost. The original plan was for 4005 cGy in 15 fractions to the right breast. The patient initially tolerated treatment well, with some reported fatigue. Two weeks after starting treatment (after her 11th fraction), she reported new pruritic bullae and vesicles on the right breast, arms, and legs that developed synchronously. At this time, the patient was not receiving any systemic cancer therapy and had not started taking any new medications, including antibiotics, aside from mirtazapine. She was prescribed silver sulfadiazine initially for the new bullae. She continued to develop additional blistering with moist desquamation, worsening grade 4 dermatitis of the right breast (within the radiation field), and generalized bullae across the trunk and upper and lower extremities (all outside of the radiation field). Given the unexplained rapidly progressive skin changes both inside and outside of the radiation field, RT was discontinued after the patient received 13 (3471 cGy) of the originally planned 15 fractions (4005 cGy) because she had low-risk breast cancer and RT was intended for adjuvant treatment to reduce the risk of breast cancer recurrence. Risks of further RT in the setting of worsening dermatitis were felt to outweigh the small expected benefit of completing the final 2 fractions of RT.
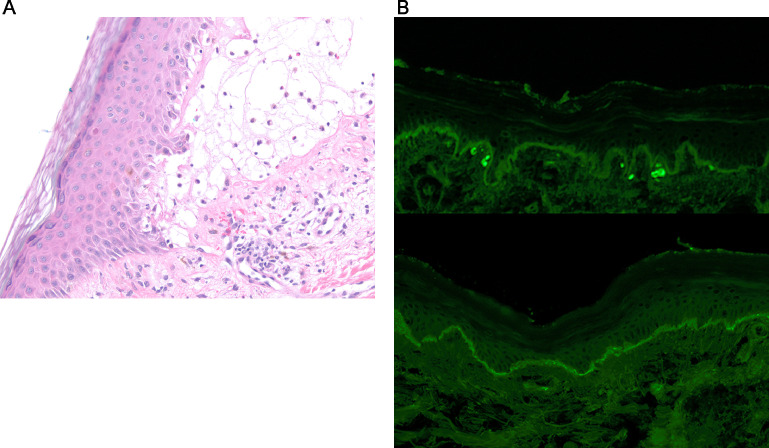
Dermatology was reconsulted for the patient's severe cutaneous eruption triggered by RT. On examination, the right breast had intensely erythematous patches with moist desquamation covering the entire breast (Fig 1A). The patient also had numerous urticarial plaques, tense vesicles, and bullae scattered on the torso, arms, and legs (Fig 1B and 1C). There was no involvement of the oral or anogenital mucosa and no generalized fever or chills. Given these findings, she was evaluated for an immunobullous disorder with skin biopsies for microscopic examination and direct immunofluorescence (DIF) of perilesional skin. On histopathology findings, there was a superficial perivascular and interstitial mixed cell infiltrate with numerous eosinophils under a subepidermal blister (Fig 2A). Staining with DIF showed IgG and C3 staining in a linear pattern at the dermal-epidermal junction, confirming the diagnosis of BP (Fig 2B). Serum BP180 (180 U/mL) and BP 230 IgG (10 U/mL) levels supported the diagnosis. Other laboratory findings, including a complete blood count, metabolic panel, and tuberculosis and hepatitis testing, were all within normal limits. Of note, the patient had no eosinophilia; at the time of admission to the hospital for her cutaneous eruption, the eosinophil absolute count was 300/μL and the eosinophil percentage was 3.7%.
Figure 1.
Patient presentation after 13 fractions (3471 cGy) of radiation. A, Grade 4 radiation dermatitis with erythema, erosions, and moist desquamation. B, Eroded pink plaques and scarring on the back. C, Urticarial plaques with tense vesicles and bullae on the right thigh.
Figure 2.
A, Subepidermal bulla containing eosinophils (magnification 200 ×, hematoxylin and eosin stain). B, Composite IgG (top) and C3 (bottom) direct immunofluorescence showing linear deposition along the dermal-epidermal junction (magnification 100 ×).
Given the severity of the patient's BP, she was admitted for initial treatment and wound care. She was treated with prednisone, 1 mg/kg (total dose, 85 mg), and prophylactically with a proton pump inhibitor for gastric ulcer, calcium and vitamin D for osteoporosis, and trimethoprim and sulfamethoxazole for Pneumocystis pneumonia. There was low concern for bacteremia and secondary infection of her skin lesions, and wound care was continued throughout her admission with emollients and triamcinolone cream. This was continued at her rehabilitation facility after discharge from the hospital. Mycophenolate mofetil, 500 mg, was started and titrated up to 2000 mg daily as a steroid-sparing agent, thus allowing for a slow taper of prednisone over 6 months to 5 mg twice daily. After 2 months of follow-up, the patient had a few erosions with crusts but no active bullae. At the 6-month follow-up, the patient had complete response of her skin disease with no active lesions.
Discussion
This case highlights a rare manifestation of BP triggered by RT in a patient with a chronic history of nonspecific eczematous dermatitis with lesions of prurigo simplex. Many theories have been proposed for how RT may trigger BP. For cases of localized BP after RT, it has been suggested that RT may change the antigenic properties of the basement membrane of skin cells and induce autoantibody formation.9, 10, 11 These autoantibodies may then trigger the complement cascade and hemidesmosome chemotaxis, resulting in bullae.9, 10, 11 In addition, since 1975, RT has been shown to cause an increase in basement membrane antibody binding, likely owing to vascular endothelial injury.12,13
The time course and generalized nature of this patient's BP during RT was unusual. A systematic review of 27 cases of radiation-associated BP concluded that in a large majority of cases (89%), blistering was confined to the site of RT and typically occurred months to years after RT.4,13 It was uncommon for patients to develop bullous lesions during RT (4 of 27 patients) or for their condition to progress to generalized skin disease (as seen in the current case) as opposed to just in the site of RT.4 Thus, given the time course, generalized nature, and history of preexisting dermatosis in our patient, we believe she had smoldering prebullous BP that flared during RT, manifesting as classic bullous lesions. She may have had circulating antibasement membrane antibodies BP180 and BP230 that could have been detected before RT; several atypical variants of BP that are positive for basement membrane autoantibodies, based on biopsy and/or serum titers, have been documented in the literature.14, 15, 16, 17, 18, 19
Given this patient's dramatic and unique presentation of BP during RT, another possibility to consider was whether the BP may have been a paraneoplastic syndrome associated with the patient's breast cancer. However, BP as a paraneoplastic syndrome is rare, and when it does arise, it is more often associated with lymphoproliferative diseases.20 There are 2 prior case reports in the literature of BP as a paraneoplastic syndrome of breast cancer, but in both cases, the patients had not received any RT that could have triggered the BP.21,22 Given our patient's BP development during RT and the severity of the cutaneous reaction at the radiation site, we believe it more likely that the BP was a generalized reaction to the RT.
Bullous pemphigoid generally responds well to systemic corticosteroids and immunosuppressive or anti-inflammatory agents such as mycophenolate mofetil, azathioprine, and methotrexate, often resulting in complete remission.4 High-potency topical corticosteroids may also be effective,23 and a combination of tetracycline and niacinamide has also been used with success.24 Severe cases that are recalcitrant to therapy may respond to intravenous immune globulin or rituximab.25 Treatment of radiation-induced BP does not differ from treatment of classic BP, but special considerations must be made regarding concurrent oncologic therapy and other comorbidities. Outcomes from treatment of radiation-induced BP are not uniformly reported in the literature but generally range from disease control to complete remission, even with nonsystemic corticosteroid therapy.13 Most patients experience BP after completing RT rather than during the course of treatment, but in a case similar to this one, in which a patient developed bullae during treatment, RT was also interrupted.26,27
Conclusions
In summary, we present a unique case of a patient who likely had a smoldering prebullous presentation of BP manifesting as generalized pruritus and excoriations and who then had a severe and generalized flare of her skin disease manifesting as classic BP after she received 3471 cGy of localized RT for breast carcinoma. The patient responded well to systemic corticosteroids and mycophenolate therapy, with complete response of her skin disease. Because of her generalized and severe BP and concurrent grade 4 radiation dermatitis, RT was discontinued prematurely. In future cases, we suggest that physicians managing patients with nonspecific eczematous eruptions or lesions of prurigo simplex and excoriations should consider a dermatology referral before beginning RT, for potential evaluation of BP with serum antibody titers as well as a skin biopsy with hematoxylin and eosin and DIF staining. This consideration should specifically be made in patients with risk factors for BP, such as older age, recalcitrant eruption to topical treatment, and unclear etiology of pruritus. This will help lead to heightened surveillance of such patients during and after RT by radiation oncologists and dermatologists.
Footnotes
Sources of support: This work had no specific funding.
Disclosures: The authors have no relevant conflicts of interest to declare for this work.
Research data are stored in an institutional repository and will be shared upon request to the corresponding author
References
- 1.Wengström Y, Häggmark C, Strander H, Forsberg C. Perceived symptoms and quality of life in women with breast cancer receiving radiation therapy. Eur J Oncol Nurs. 2000;4:78–88. doi: 10.1054/ejon.1999.0052. [DOI] [PubMed] [Google Scholar]
- 2.Schnur JB, Love B, Scheckner BL, et al. A systematic review of patient-rated measures of radiodermatitis in breast cancer radiotherapy. Am J Clin Oncol. 2011;34:529–536. doi: 10.1097/COC.0b013e3181e84b36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bray FN, Simmons BJ, Wolfson AH, Nouri K. Acute and chronic cutaneous reactions to ionizing radiation therapy. Dermatol Ther (Heidelb) 2016;6:185–206. doi: 10.1007/s13555-016-0120-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mul VEM, van Geest AJ, Pijls-Johannesma MC, et al. Radiation-induced bullous pemphigoid: A systematic review of an unusual radiation side effect. Radiother Oncol. 2007;82:5–9. doi: 10.1016/j.radonc.2006.11.014. [DOI] [PubMed] [Google Scholar]
- 5.Knoell KA, Patterson JW, Gampper TJ, Hendrix J, John D. Localized bullous pemphigoid following radiotherapy for breast carcinoma. Arch Dermatol. 1998;134:514–515. doi: 10.1001/archderm.134.4.514. [DOI] [PubMed] [Google Scholar]
- 6.Miyamoto D, Santi CG, Aoki V, Maruta CW. Bullous pemphigoid. An Bras Dermatol. 2019;94:133–146. doi: 10.1590/abd1806-4841.20199007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lo Schiavo A, Ruocco E, Brancaccio G, Caccavale S, Ruocco V, Wolf R. Bullous pemphigoid: Etiology, pathogenesis, and inducing factors: Facts and controversies. Clin Dermatol. 2013;31:391–399. doi: 10.1016/j.clindermatol.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 8.Venning VA, Wojnarowska F. Induced bullous pemphigold. Br J Dermatol. 1995;132:831–832. [PubMed] [Google Scholar]
- 9.Cliff S, Harland CC, Fallowfield ME, Mortimer PS. Localised bullous pemphigoid following radiotherapy. Acta Derm Venereol. 1996;76:330–331. doi: 10.2340/0001555576330331. [DOI] [PubMed] [Google Scholar]
- 10.Folliero G, Zurlo A, Amanti C, Tombolini V, Di Paola M. Bullous pemphigoid induced by radiation therapy. Clin Oncol. 1995;7:266–267. doi: 10.1016/s0936-6555(05)80618-1. [DOI] [PubMed] [Google Scholar]
- 11.Sheerin N, Bourke JF, Holder J, North J, Burns DA. Bullous pemphigoid following radiotherapy. Clin Exp Dermatol. 1995;20:80–82. doi: 10.1111/j.1365-2230.1995.tb01293.x. [DOI] [PubMed] [Google Scholar]
- 12.Remy W, Bockendahl H, Stüttgen G. The effects of X-ray, ultraviolet and infrared irradiation on the basement membrane zone antibody reaction of the human skin in vitro. Acta Derm Venereol. 1975;55:313–315. [PubMed] [Google Scholar]
- 13.Nguyen T, Kwan JM, Ahmed AR. Relationship between radiation therapy and bullous pemphigoid. DRM. 2014;229:88–96. doi: 10.1159/000362208. [DOI] [PubMed] [Google Scholar]
- 14.Schmidt E, Sitaru C, Schubert B, et al. Subacute prurigo variant of bullous pemphigoid: Autoantibodies show the same specificity compared with classic bullous pemphigoid. J Am Acad Dermatol. 2002;47:133–136. doi: 10.1067/mjd.2002.120445. [DOI] [PubMed] [Google Scholar]
- 15.Strohal R, Rappersberger K, Pehamberger H, Wolff K. Nonbullous pemphigoid: Prodrome of bullous pemphigoid or a distinct pemphigoid variant? J Am Acad Dermatol. 1993;29:293–299. doi: 10.1016/0190-9622(93)70179-w. [DOI] [PubMed] [Google Scholar]
- 16.Grattan CEH. Evidence of an association between bullous pemphigoid and psoriasis. Br J Dermatol. 1985;113:281–283. doi: 10.1111/j.1365-2133.1985.tb02079.x. [DOI] [PubMed] [Google Scholar]
- 17.Haber R, Fayad AM, Stephan F, Obeid G, Tomb R. Bullous pemphigoid associated with linagliptin treatment. JAMA Dermatol. 2016;152:224. doi: 10.1001/jamadermatol.2015.2939. [DOI] [PubMed] [Google Scholar]
- 18.Levine N. Localized pemphigoid simulating dyshidrosiform dermatitis. Arch Dermatol. 1979;115:320. [PubMed] [Google Scholar]
- 19.Lamb PM, Abell E, Tharp M, Frye R, Deng J-S. Prodromal bullous pemphigoid. Int J Dermatol. 2006;45:209–214. doi: 10.1111/j.1365-4632.2004.02457.x. [DOI] [PubMed] [Google Scholar]
- 20.DeVita VT, Lawrence TS, Rosenberg SA. Lippincott Williams & Wilkins; Philadelphia, PA: 2008. DeVita, Hellman, and Rosenberg's Cancer: Principles & Practice of Oncology. [Google Scholar]
- 21.Antal AS, Grieb S, Homey B, et al. Paraneoplastisches bullöses Pemphigoid bei einem Mann mit Mamma- und Prostatakarzinom. Hautarzt. 2007;58:833–834. doi: 10.1007/s00105-007-1398-6. [DOI] [PubMed] [Google Scholar]
- 22.Iuliano L, Micheletta F, Natoli S. Bullous pemphigoid: An unusual and insidious presentation of breast cancer. Clin Oncol. 2003;15:505. doi: 10.1016/j.clon.2003.08.006. [DOI] [PubMed] [Google Scholar]
- 23.Joly P, Roujeau JC, Benichou J, et al. A comparison of oral and topical corticosteroids in patients with bullous pemphigoid. N Engl J Med. 2002;346:321–327. doi: 10.1056/NEJMoa011592. [DOI] [PubMed] [Google Scholar]
- 24.Fivenson DP, Breneman DL, Rosen GB, Hersh CS, Cardone S, Mutasim D. Nicotinamide and tetracycline therapy of bullous pemphigoid. Arch Dermatol. 1994;130:753–758. [PubMed] [Google Scholar]
- 25.Ahmed AR, Shetty S, Kaveri S, Spigelman ZS. Treatment of recalcitrant bullous pemphigoid (BP) with a novel protocol: A retrospective study with a 6-year follow-up. J Am Acad Dermatol. 2016;74:700–708. doi: 10.1016/j.jaad.2015.11.030. e3. [DOI] [PubMed] [Google Scholar]
- 26.Srifi N, Benomar S, Zaghba N, et al. [Generalized bullous pemphigoid induced by radiotherapy] Ann Dermatol Venereol. 2011;138:311–314. doi: 10.1016/j.annder.2010.11.016. [DOI] [PubMed] [Google Scholar]
- 27.Kridin K, Ludwig RJ. The growing incidence of bullous pemphigoid: Overview and potential explanations. Front Med. 2018;5:220. doi: 10.3389/fmed.2018.00220. [DOI] [PMC free article] [PubMed] [Google Scholar]