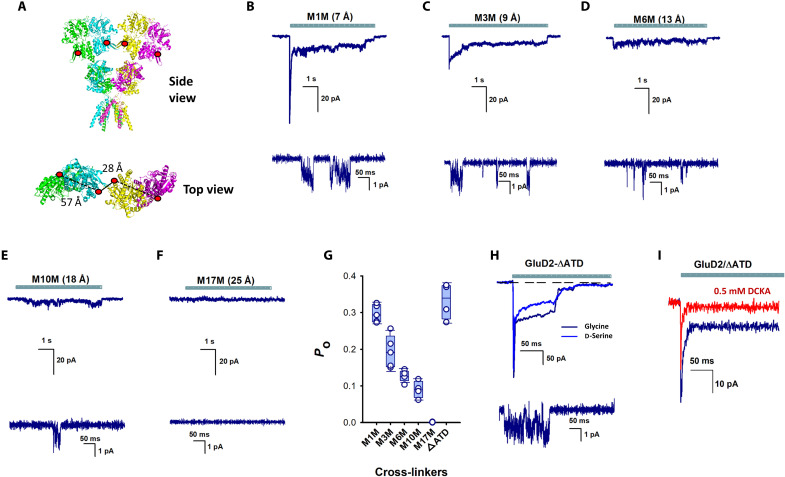
Fig. 5. N-terminal domain stabilization permits glycine activation of GluD2 receptors.
(A) Structure of GluD2 (modeled on GluD1; PDB: 6KSS) showing the incorporation of a cysteine residue at position 276. Side view (top) and close-up top-down view of 207 N-terminal domain residues and distance between dimers (bottom). Representative whole-cell (top) and single-channel (bottom) currents evoked by glycine application in the presence of (B) 1,1-methanediyl bis-MTS (M1M), (C) 1,3-propanediyl bis-MTS (M3M), (D) 1,6-hexanediyl bis-MTS (M6M), (E) 1,10-decadiyl bis-MTS (M10M), and (F) 3,6,9,12,15-pentaoxaheptadecane-1,17-diyl bis-MTS (M17M). (G) Bar graphs showing probability of opening from at least three patches for the different cross-linkers and GluD2-ΔATD. (H) Representative glycine- and d-serine–evoked currents in GluD2 receptors lacking N-terminal domains. (I) Representative currents showing the inhibition of 10 mM glycine-mediated currents by 500 μM DCKA. ATD, amino terminal domain.