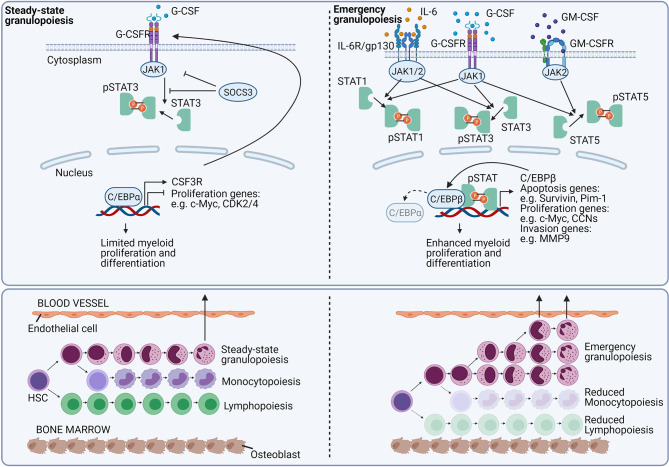
Figure 8.
Growth factor signaling regulates emergency granulopoiesis. In homeostatic conditions, the granulocyte colony-stimulating factor (G-CSF) is the main growth factor that regulates neutrophil development. G-CSF signals via the G-CSF receptor (G-CSFR) (encoded by the CSF3R gene), and signals via the Janus kinase (JAK)-signal transducer and activator of transcription (STAT) pathway. Via unknown mechanisms, G-CSFR signaling induces the CCAAT-enhancer-binding protein-α (C/EBPα). C/EBPα is the major transcription factor involved in myelopoiesis. The transcription factor enhances the expression CSF3R, whereas it inhibits genes required for the cell cycle, eventually causing a limited myeloid proliferation and differentiation. During excessive inflammation, neutrophils massively migrate to the site of inflammation. To counterbalance neutrophil depletion and to provide newly needed neutrophils, emergency granulopoiesis is initiated. Emergency granulopoiesis is characterised by a large-scale de novo generation of new neutrophils from neutrophil progenitor cells. Interleukin-6 (IL-6), G-CSF, and granulocyte-macrophage colony-stimulating factor (GM-CSF) are mainly stimulating the proliferation and differentiation of new neutrophils. IL-6 binds the IL-6 receptor (IL-6R)/gp130 and signals in a JAK-STAT1 or STAT3-dependent way. Excessive G-CSFR signalling induces both STAT1, STAT3, and STAT5 phosphorylation and signalling, whereas GM-CSF receptor (GM-CSFR) signalling mainly tempts STAT5 activation. Upon activation, the pSTAT molecules are translocated to the nucleus where they directly stimulate genes involved in the regulation of apoptosis, proliferation, and cellular translocation. Additionally, pSTAT signalling stimulates the expression of the transcription factor C/EBPβ. C/EBPβ replaces C/EBPα, and releases the brake on proliferative genes, cranking myelopoiesis. During emergency granulopoiesis, not only mature neutrophils are leaving the BM, but also more immature neutrophils are being released. This process is called “left shift”.