ABSTRACT
Guanylyl cyclase C (GUCY2C) is a tumor-suppressing receptor silenced by loss of expression of the luminocrine hormones guanylin and uroguanylin early in colorectal carcinogenesis. This observation suggests oral replacement with a GUCY2C agonist may be an effective targeted chemoprevention agent. Previous studies revealed that linaclotide, an oral GUCY2C agonist formulated for gastric release, did not persist to activate guanylyl cyclase signaling in the distal rectum. Dolcanatide is an investigational oral uroguanylin analog, substituted with select D amino acids, for enhanced stability and extended persistence to activate GUCY2C in small and large intestine. However, the ability of oral dolcanatide to induce a pharmacodynamic (PD) response by activating GUCY2C in epithelial cells of the colorectum in humans remains undefined. Here, we demonstrate that administration of oral dolcanatide 27 mg daily for 7 d to healthy volunteers did not activate GUCY2C, quantified as accumulation of its product cyclic GMP, in epithelial cells of the distal rectum. These data reveal that the enhanced stability of dolcanatide, with persistence along the rostral-caudal axis of the small and large intestine, is inadequate to regulate GUCY2C across the colorectum to prevent tumorigenesis. These results highlight the importance of developing a GUCY2C agonist for cancer prevention formulated for release and activity targeted to the colorectum.
KEYWORDS: Colorectum, colorectal cancer chemoprevention, dolcanatide, guanylyl cyclase C, cyclic GMP
Introduction
Colorectal cancer is the fourth most commonly diagnosed cancer in the United States, with approximately 150,000 new cases recorded each year.1 Over the course of a lifetime, approximately one in 20 U.S. residents will be diagnosed with this disease. Despite advances in early detection and treatment, the 5 y survival rate for colorectal cancer is less than 70%.2 Although screening and surveillance continue to be the cornerstone of colorectal cancer prevention, chemoprevention has emerged as a complementary approach among higher risk participants. To date, acetylsalicylic acid (ASA) in the form of aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs) represent the most thoroughly investigated class of colorectal cancer chemoprevention agents. However, given the established risk/benefit profile of these agents, the widespread use of ASA or other NSAIDs strictly for colorectal cancer chemoprevention seems unlikely in the average-risk population.
Guanylyl cyclase C (GUCY2C) is the intestinal epithelial cell receptor for the endogenous hormones guanylin and uroguanylin.3 Hormone–receptor interaction activates the intracellular catalytic domain, which converts guanosine triphosphate to cyclic guanosine monophosphate (cGMP). This cyclic nucleotide activates signaling intermediates, including cGMP-dependent protein kinase, which phosphorylates downstream effectors, including vasodilator-stimulated phosphoprotein (VASP) and cystic fibrosis transmembrane conductance regulator (CFTR). Phosphorylation of CFTR opens the chloride channel, resulting in fluid and electrolyte secretion. This mechanism has been co-opted by bacteria that secrete heat-stable enterotoxins (STs), which are structural and functional homologs of guanylin and uroguanylin and induce GUCY2C-dependent diarrhea.4–6 Beyond secretion, GUCY2C and its ligands also regulate intestinal homeostasis along the crypt-villus axis by restricting proliferative dynamics and coordinating cell cycle, differentiation, and metabolic circuits.7–9 Interestingly, guanylin and uroguanylin are the most commonly lost gene products in colorectal cancer in animals and humans.10–12 Of significance, epithelial cells undergoing transformation continue to express GUCY2C, and colon cancer cells over-express GUCY2C compared to normal adjacent mucosa.13,14 Moreover, delivery of GUCY2C ligands to intestines opposes intestinal tumorigenesis in mice.15–17
Taken together, these data suggest that GUCY2C is a tumor-suppressing receptor that, when silenced by loss of expression of guanylin and uroguanylin, universally contributes to early development of colorectal cancer. These properties highlight the potential value of oral ligand replacement with GUCY2C agonists as targeted prevention for colorectal cancer. Moreover, oral GUCY2C agonists have impressive safety profiles in pre-clinical through late-stage clinical trials for the treatment of chronic constipation syndromes. Given the paucity of compounds proven safe and effective for colorectal cancer chemoprevention, this class of agent warrants further investigation.
Linaclotide is an FDA-approved GUCY2C agonist formulated for immediate gastric release, with bioactivity in the small intestine. It is approved to treat constipation-type irritable bowel syndrome (IBS-C) and chronic idiopathic constipation (CIC). Analysis of chemopreventive-relevant PD responses in the human colon revealed that oral linaclotide at a dose of 0.87 mg, while active in the proximal colon, did not produce activation of GUCY2C signaling in the distal rectal mucosa of normal volunteers. This result likely reflects the pharmacokinetics of linaclotide in the intestine, as up to 99% of the peptide degrades before reaching the distal rectum.18,19 To overcome this pharmacokinetic barrier, the present study explored the use of oral dolcanatide, a uroguanylin analog formulated for enhanced stability and persistence in the gastrointestinal (GI) tract,20,21 to activate GUCY2C and stimulate cGMP production in epithelial cells of the distal rectum in healthy volunteers.
Materials and methods
Study design
The study was approved by the Cancer Prevention and Control Central Institutional Review Board (CPC CIRB) and registered on ClinicalTrials.gov (NCT03300570). Mayo Clinic in Rochester, Mn, served as the research base. The Data and Safety Monitoring Board of the Mayo Clinic Cancer Center reviewed safety data 6 months after study activation. Healthy volunteers enrolled were 18–65 y old, without personal or family (first-degree relative) histories of colorectal neoplasia, inflammatory bowel disease or other recent (≤3 months prior to day 0) or ongoing diseases producing acute or chronic diarrhea. Following informed consent, willing subjects completed a focused interview and physical examination. Women of childbearing potential were required to document a negative pregnancy test prior to enrollment. Following an overnight fast, subjects received up to three polyethylene glycol (PEG) enemas until evacuation was clear. PEG enemas were used because they are optimum for preserving epithelial cell integrity for biopsy and subsequent analyses.22 Only subjects who had a satisfactory intestinal preparation and who had no significant intestinal pathology advanced to drug intervention (Figure 1). During the intervention phase, subjects were randomly assigned to receive dolcanatide 27 mg or matching placebo for seven consecutive days, a duration associated with biological effects of linaclotide in the colorectum previously.18 Subjects returned each day to the clinical research unit for witnessed dosing. On day 7, following an overnight fast, subjects returned for their final dose of dolcanatide/placebo. Within 8 hours after dosing, subjects were given up to three PEG enemas, after which the post-intervention flexible sigmoidoscopy procedure was performed. During both the screening and post-intervention colonoscopies, quality of the preparation was described using the Boston bowel preparation scale.23 Eight biopsies were taken from eight discrete areas in the rectum at or just above the inferior valve of Houston, separated by no less than 0.5 cm in any direction. The biopsies from each section were flash-frozen in liquid nitrogen and stored at −80°C for analyses.
Figure 1.
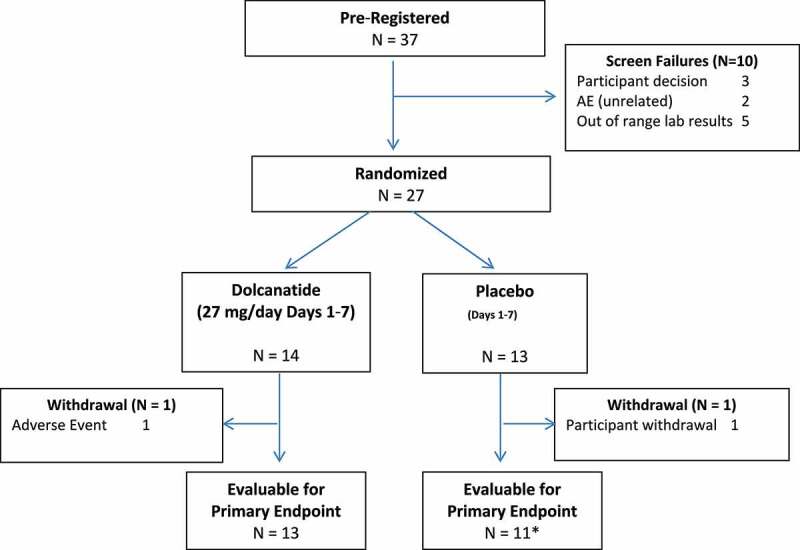
CONSORT flow diagram of subject progress through the clinical trial
Primary endpoint
Cyclic GMP
The primary endpoint of the study was a direct comparison of the pharmacological effect of dolcanatide on cGMP levels versus placebo. This was defined as the arithmetic difference in mean cGMP levels in rectal biopsies before and after 7 d of dolcanatide. The mean cGMP value was calculated based on the eight biopsies collected from the rectum during a flexible sigmoidoscopy procedure. Each biopsy was analyzed in triplicate using a Cayman Practice EIA enzyme-linked immunosorbent assay kit (Cayman Chemical, Ann Arbor, MI). Each patient analysis included a cGMP standard curve to quantify cGMP levels and ensure assay performance. With a sample size of 12 per arm, the study had 80% power to detect a statistically significant pharmacological effect for dolcanatide versus placebo, based on continuous cGMP results and assuming a one-sided significance level of 5%. The study was powered to detect an effect size of 1 (i.e., [difference in means]/standard deviation (SD) = 1) (two-sample t-test).
Safety
To confirm the safety and tolerability of dolcanatide and placebo, all subjects were evaluated for toxicity from the time of their first dose of dolcanatide/placebo. Common terminology criteria for adverse events (CTCAE) v4.0 was used to summarize adverse events. Attribution of drug-related adverse events was performed by the designated medical monitor (GDZ), blinded at the time of attribution.
Secondary endpoint
The PD response rate was assessed across all subjects and compared between intervention arms. This calculation was based on the standardized difference in means for the pharmacological effect on cGMP levels at the participant level, calculated as the z value = [(mean difference in cGMP levels after and before treatment) divided by (the combined standard deviation of those means)]. Subjects with a z value ≥1.645 were considered PD responders, while those with a z value <1.645 were considered non-responders. PD response rates were compared between intervention arms using a Fisher’s exact test.
Cyclic GMP quantification
The primary endpoint for this study was the ability of oral dolcanatide to increase cGMP accumulation in colorectal mucosae. The technique for cGMP quantification by immunoassay is well defined.18,24 Upon collection, mucosal biopsies were placed in cryogenic tubes, frozen in liquid nitrogen and archived in a − 80°C freezer. For analysis, samples underwent cryopulverization before thawing in 200 µL of pre-cooled passive lysis 5X buffer (Promega Corporation, Madison, WI) with 1 mM 3-isobutyl-1-methylxanthine/IBMX (MilliporeSigma, St. Louis, MO) on ice for 30 minutes followed by centrifugation (18,000 rpm, 30 minutes, 0–4°C). Fifty (50) μL of the supernatant was diluted in 450 μL of 50 mM sodium acetate (pH 4.0), and cGMP was quantified using a cGMP EIA kit, a validated enzyme-linked immunoassay (Cayman Chemical Company, Ann Arbor, MI). Tissue residues were dissolved in 0.2 N sodium hydroxide at 4°C overnight, and protein concentrations were determined using the Pierce BCA Protein Assay kit (Thermo Fisher Scientific, Waltham, MA). Cyclic GMP levels were normalized to the protein content from individual samples.
Messenger RNA quantification
Tissue samples were flash-frozen in liquid nitrogen and stored at −80°C until use. RNA was extracted and purified using the RNeasy Micro kit (Qiagen, Germantown, MD). Following isolation, RNA concentration and purity were measured using the Nanodrop 1000 Spectrophotometer (Thermo Fisher Scientific, Waltham, MA), and two-step quantitative RT-qPCR tests were used to interrogate gene expression. Complementary DNA was produced using the TaqMan RT-PCR kit according to the manufacturer’s specifications and then quantified by PCR using TaqMan primer probes (Thermo Fisher Scientific, Waltham, MA).25 Every qRT-PCR analysis was accompanied by buffer (negative) controls and a GUCY2C standard for routine quality control to ensure assay performance.
Statistical analyses
Frequency tables and percentages summarized baseline and clinical characteristics, treatment data, and AE data for each treatment arm. Descriptive statistics, including mean, standard deviation, median, range, and frequencies (percentages), were used to summarize these data. Graphs, including boxplots and bar graphs, were used to visually depict these data. Fisher’s exact and Wilcoxon rank-sum tests were used to test for associations between treatment arms and categorical and continuous data, using p < .05 as the threshold for significance. All statistical tests were two-sided and performed using the Base SAS web application server, version 9.4M5 (SAS Institute, Inc., 2017).
Results
Subject characteristics
For this study, 37 subjects were pre-registered and evaluated for inclusion, and 10 were considered screening failures (Figure 1). Two subjects did not complete the study. One subject completed study intervention but did not undergo the post-intervention endoscopy procedure with biopsy collection. Thus, 24 subjects were evaluated for the primary and secondary endpoints based on tissue analysis, while 27 subjects were evaluated for safety/toxicity and non-tissue-based secondary endpoints. These 27 subjects had a mean age of 47 ± 9 y; 85% were male, 89% were non-Hispanic, 33% were white, and 67% were black (Table 1). Physical exams and laboratory studies revealed no clinically remarkable findings for this cohort of normal healthy participants. Subjects randomized to dolcanatide and placebo groups had similar baseline physical exam findings (Table 2).
Table 1.
Baseline demographics for randomized subjects, by intervention arm
|
Arm |
|||
|---|---|---|---|
| Placebo (N = 13) |
Dolcanatide (N = 14) |
Total (N = 27) |
|
| Age | |||
| Mean (SD) | 44.2 (11.67) | 48.7 (5.72) | 46.6 (9.19) |
| Median | 49.0 | 50.0 | 49.0 |
| Range | 25.0, 57.0 | 39.0, 56.0 | 25.0, 57.0 |
| Gender, n (%) | |||
| Female | 4 (30.8%) | 0 (0.0%) | 4 (14.8%) |
| Male | 9 (69.2%) | 14 (100.0%) | 23 (85.2%) |
| Ethnicity, n (%) | |||
| Hispanic or Latino | 2 (15.4%) | 1 (7.1%) | 3 (11.1%) |
| Not Hispanic or Latino | 11 (84.6%) | 13 (92.9%) | 24 (88.9%) |
| Race, n (%) | |||
| Black or African American | 10 (76.9%) | 8 (57.1%) | 18 (66.7%) |
| White | 3 (23.1%) | 6 (42.9%) | 9 (33.3%) |
Table 2.
Baseline physical exam findings for randomized subjects, by intervention arm
| Arm |
|
|||
|---|---|---|---|---|
| Placebo (N = 13) |
Dolcanatide (N = 14) |
Total (N = 27) |
P-value | |
| BMI (kg/m2) | .30811 | |||
| Mean (SD) | 29.24 (3.97) | 27.93 (3.41) | 28.56 (3.68) | |
| Median | 29.97 | 27.70 | 28.50 | |
| Range | 21.30, 34.90 | 22.40, 33.30 | 21.30, 34.90 | |
| ECG result, n (%) | 1.00002 | |||
| Normal | 6 (46.2%) | 7 (50.0%) | 13 (48.1%) | |
| Abnormal – Not Clinically Significant | 7 (53.8%) | 7 (50.0%) | 14 (51.9%) | |
| ECOG performance status, n (%) | - | |||
| 0 | 13 (100.0%) | 14 (100.0%) | 27 (100.0%) | |
|
Does the participant have any allergies? n (%) |
1.00002 | |||
| Yes | 1 (7.7%) | 2 (14.3%) | 3 (11.1%) | |
| No | 12 (92.3%) | 12 (85.7%) | 24 (88.9%) | |
Abbreviations: BMI = body mass index; ECG = electrocardiogram; ECOG = Eastern Cooperative Oncology Group.
1Wilcoxon rank-sum p-value.
2Fisher’s exact p-value.
GUCY2C activation
The efficacy of enemas to produce bowel preparations suitable for biopsy before and after the treatment periods was generally similar for volunteers receiving dolcanatide and placebo (Tables 3, 4). The baseline endoscopic results between arms were similar (Table 3), except for the Boston bowel preparation scores (Boston), which were significantly higher in the dolcanatide arm compared to placebo (p = .0295). Specifically, 79% of the participants treated with dolcanatide had a Boston score of 3, compared to only 31% treated with placebo. The post-intervention sigmoidoscopy findings and biopsy results were similar across intervention arms (all p > .28; Table 4).
Table 3.
Baseline endoscopy findings for randomized subjects, by intervention arm
|
Arm |
|
|||
|---|---|---|---|---|
| Placebo (N = 13) |
Dolcanatide (N = 14) |
Total (N = 27) |
P-value | |
| Endoscopy completed, n (%) | - | |||
| Yes | 13 (100.0%) | 14 (100.0%) | 27 (100.0%) | |
| Was bowel preparation adequate? n (%) | - | |||
| Yes | 13 (100.0%) | 14 (100.0%) | 27 (100.0%) | |
| BBPSS, n (%) | .02951 | |||
| 1 | 2 (15.4%) | 0 (0.0%) | 2 (7.4%) | |
| 2 | 7 (53.8%) | 3 (21.4%) | 10 (37.0%) | |
| 3 | 4 (30.8%) | 11 (78.6%) | 15 (55.6%) | |
| Results of endoscopy, n (%) | 1.00001 | |||
| Normal | 13 (100.0%) | 13 (92.9%) | 26 (96.3%) | |
| Abnormal | 0 (0.0%) | 1 (7.1%) | 1 (3.7%) | |
| Number of 2–4 mm polyps, n (%) | 1.00001 | |||
| 0 | 13 (100.0%) | 13 (92.9%) | 26 (96.3%) | |
| 1 | 0 (0.0%) | 1 (7.1%) | 1 (3.7%) | |
| Number of 5–9 mm polyps, n (%) | - | |||
| 0 | 13 (100.0%) | 14 (100.0%) | 27 (100.0%) | |
| Number of >10 mm polyps, n (%) | - | |||
| 0 | 13 (100.0%) | 14 (100.0%) | 27 (100.0%) | |
| Were all rectal polyps >2 mm removed? n (%) | - | |||
| No | 13 (100.0%) | 14 (100.0%) | 27 (100.0%) | |
Abbreviations: BBPSS = Boston bowel preparation scale score.
1Fisher’s exact p-value.
Table 4.
Post-intervention endoscopy findings for randomized subjects, by intervention arm
|
Arm |
|
|||
|---|---|---|---|---|
| Placebo (N = 13) |
Dolcanatide (N = 14) |
Total (N = 27) |
P-value | |
| Endoscopy completed, n (%) | 1.00001 | |||
| Yes | 12 (92.3%) | 13 (92.9%) | 25 (92.6%) | |
| No | 1 (7.7%) | 1 (7.1%) | 2 (7.4%) | |
| Was bowel preparation adequate? n (%) | .48001 | |||
| Yes | 11 (91.7%) | 13 (100.0%) | 24 (96.0%) | |
| No | 1 (8.3%) | 0 (0.0%) | 1 (4.0%) | |
| Missing | 1 | 1 | 2 | |
| BBPSS, n (%) | .28781 | |||
| 0 | 1 (8.3%) | 0 (0.0%) | 1 (4.0%) | |
| 2 | 5 (41.7%) | 3 (23.1%) | 8 (32.0%) | |
| 3 | 6 (50.0%) | 10 (76.9%) | 16 (64.0%) | |
| Missing | 1 | 1 | 2 | |
| Results of endoscopy, n (%) | 1.00001 | |||
| Normal | 11 (100.0%) | 12 (92.3%) | 23 (95.8%) | |
| Abnormal | 0 (0.0%) | 1 (7.7%) | 1 (4.2%) | |
| Missing | 2 | 1 | 3 | |
| Number of 2–4 mm polyps, n (%) | 1.00001 | |||
| 0 | 11 (100.0%) | 12 (92.3%) | 23 (95.8%) | |
| 1 | 0 (0.0%) | 1 (7.7%) | 1 (4.2%) | |
| Missing | 2 | 1 | 3 | |
| Number of 5–9 mm polyps, n (%) | - | |||
| 0 | 11 (100.0%) | 13 (100.0%) | 24 (100.0%) | |
| Missing | 2 | 1 | 3 | |
| Number of >10 mm polyps, n (%) | - | |||
| 0 | 11 (100.0%) | 13 (100.0%) | 24 (100.0%) | |
| Missing | 2 | 1 | 3 | |
| Were all rectal polyps >2 mm removed? n (%) | 1.00001 | |||
| Yes | 0 (0.0%) | 1 (7.7%) | 1 (4.0%) | |
| No | 11 (91.7%) | 12 (92.3%) | 23 (92.0%) | |
| Not Applicable | 1 (8.3%) | 0 (0.0%) | 1 (4.0%) | |
| Missing | 1 | 1 | 2 | |
Abbreviations: BBPSS = Boston bowel preparation scale score.
1Fisher’s exact p-value.
Primary endpoint and key secondary endpoint results
Biopsies revealed that the mean differences in cGMP levels of the rectal mucosa before and after 7 d of treatment, which is a measure of GUCY2C stimulation, were similar in participants receiving dolcanatide and those receiving placebo (Figure 2; Table 5). Specifically, there was no significant difference in cGMP levels found between the arms after 7 d of treatment (p = .15; Table 5). In addition, dolcanatide did not produce a PD response in the rectal mucosa of any participant in the active arm of the trial (Figure 3). The failure of dolcanatide to activate GUCY2C did not reflect a target receptor deficiency in the rectal mucosa of the subjects. Indeed, there were no significant differences in GUCY2C mRNA expression in subjects before, or 7 d after, treatment with dolcanatide or placebo (Figure 4).
Figure 2.
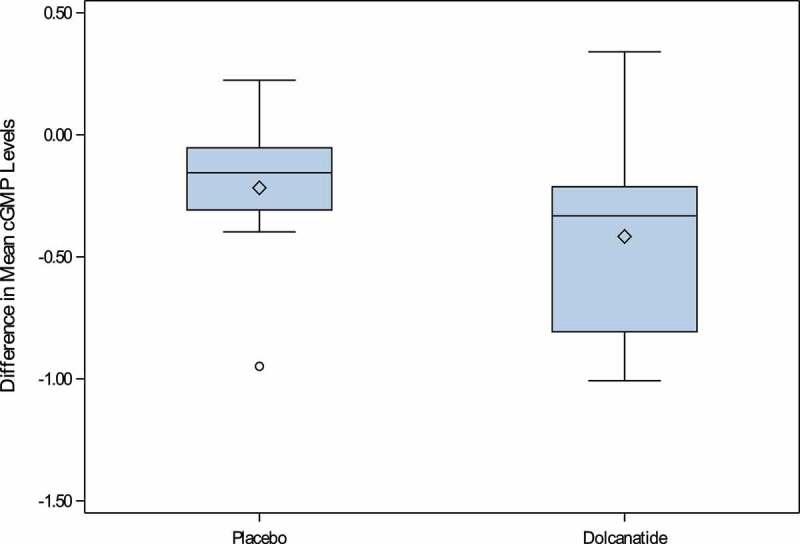
Differences in mean rectal cGMP following seven days of treatment. Cyclic GMP was quantified, and differences calculated, as described in Methods
Table 5.
Differences in mean cGMP levels (cGMP/mg protein) after 7 days of intervention
|
Arm |
|
|||
|---|---|---|---|---|
| Placebo (N = 11) |
Dolcanatide (N = 13) |
Total (N = 24) |
P-value | |
| Difference in Mean cGMP Levels (Day 7 – Day 0) | .14751 | |||
| Mean (SD) | −0.22 (0.29) | −0.42 (0.43) | −0.33 (0.38) | |
| Median | −0.16 | −0.33 | −0.27 | |
| Range | −0.95, 0.22 | −1.01, 0.34 | −1.01, 0.34 | |
Abbreviations: cGMP = cyclic guanosine monophosphate.
1Wilcoxon rank-sum p-value.
Figure 3.
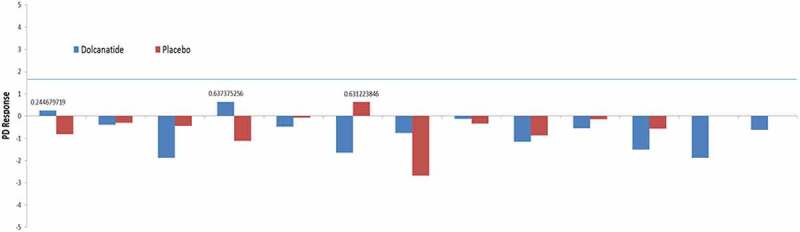
Pharmacodynamic (PD) Response Rate for 24 subjects evaluable for the primary endpoint. Cyclic GMP was quantified, and PD responses calculated, as described in Methods. A threshold rate of 1.645 (horizontal blue line) was taken as a positive response to dolcanatide
Figure 4.
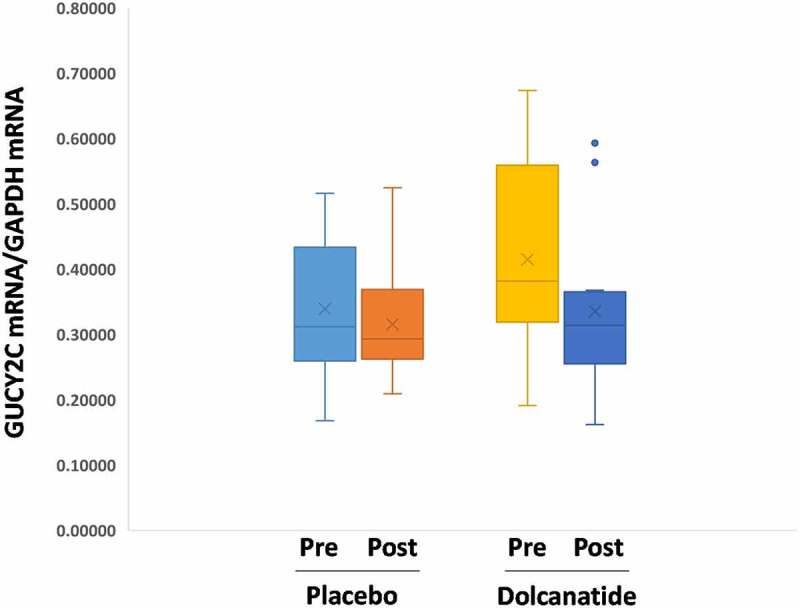
GUCY2C Expression Before and After Treatment. GUCY2C mRNA was quantified by RT-PCR as outlined in Materials and Methods and normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA expression
Safety
The 27 mg dose of dolcanatide employed in this study was well tolerated, all randomized subjects were treated, and 25 subjects completed the full 7 d of dosing without discontinuation. All 27 randomized subjects were able to be evaluated for adverse events (AEs). Fifteen of the 27 randomized subjects reported AEs during the intervention. A total of 74 AEs were reported by these 15 subjects. Twelve subjects, nine of whom were in the placebo arm, reported no AEs during the intervention. There was one report of a grade 3 AE (diarrhea). Two participants reported grade 2 AEs (one participant reported diarrhea, and one reported vomiting and anorexia). The remainder of reported AEs were all grade 1. All grade 2 and 3 AEs occurred in the dolcanatide treatment arm. Maximum grade AE rates were similar between arms (all p > .48), except for participants reporting grade 1 diarrhea, which occurred at a significantly higher rate in the dolcanatide arm than the placebo arm (64% vs. 0%; p = .00064; Table 6). In general, the GI-related AE rate was significantly higher in the dolcanatide arm than the placebo arm (79% vs. 15%; p = .002). Of the GI-related adverse events reported by the participants in the placebo arm, two were deemed probably related to study intervention by the blinded study team.
Table 6.
Maximum grade adverse events
| Grade 1 |
Grade 2 |
Grade 3 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Placebo (N = 13) |
Dolcanatide (N = 14) |
p-value1 | Placebo (N = 13) |
Dolcanatide (N = 14) |
P-value1 | Placebo (N = 13) |
Dolcanatide (N = 14) |
p-value1 | ||
| MedDRA SOC (v12.0) | CTCAE Term (v4.0) | |||||||||
| Gastrointestinal disorders | Bloating | 1 (7.7%) | 0 (0.0%) | .4815 | - | - | - | - | - | - |
| Constipation | 1 (7.7%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - | |
| Diarrhea | 0 (0.0%) | 9 (64.3%) | p=.0006 | 0 (0.0%) | 1 (7.1%) | 1.0000 | 0 (0.0%) | 1 (7.1%) | 1.0000 | |
| Dry mouth | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - | |
| Dyspepsia | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - | |
| Flatulence | 1 (7.7%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - | |
| Vomiting | 0 (0.0%) | 1 (7.1%) | 1.0000 | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | |
| General disorders and administration site conditions | Fatigue | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - |
| Metabolism and nutrition disorders | Anorexia | - | - | - | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - |
| Musculoskeletal and connective tissue disorders | Back pain | 1 (7.7%) | 0 (0.0%) | .4815 | - | - | - | - | - | - |
| Generalized weakness | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - | |
| Nervous system disorders | Dizziness | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - |
| Psychiatric disorders | Anxiety | 1 (7.7%) | 0 (0.0%) | .4815 | - | - | - | - | - | - |
| Respiratory, thoracic and mediastinal disorders | Cough | 1 (7.7%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - |
| Voice alteration | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - | |
| Skin/subcutaneous tissue disorders | Skin/subcutaneous tissue disorders – Other | 0 (0.0%) | 1 (7.1%) | 1.0000 | - | - | - | - | - | - |
Abbreviations: SOC = system organ class; CTCAE = common terminology criteria for adverse events.
1Fisher’s exact p-value.
Discussion
GUCY2C plays a key role in regulating proliferative and metabolic processes that oppose tumorigenesis. The near universal over-expression of GUCY2C in human colorectal cancers, coupled with the loss of endogenous ligands (guanylin and uroguanylin), highlights a potential targeted prevention strategy for colorectal cancer involving oral hormone replacement therapy. This strategy presumes that GUCY2C is a dormant tumor-suppressing receptor during colorectal carcinogenesis, whose re-engagement by exogenous ligands opposes dysregulated cell growth. In that context, GUCY2C signaling inhibits the cell cycle of normal human intestinal cells and human colon carcinoma cells in vitro and ex vivo.8,26 Similarly, GUCY2C signaling reverses the tumorigenic metabolic phenotype in human colon cancer cells.8,9 Mice receiving oral GUCY2C agonist replacement demonstrated a decrease in small and large intestine adenoma formation compared to controls.15–17 Taken together, these observations suggest that oral hormone replacement with GUCY2C agonists could prevent colorectal cancer initiation and progression.
Initially, linaclotide was explored as an oral GUCY2C agonist that might be suitable as a chemoprevention agent.18 Linaclotide is a chemically synthesized 14-amino acid analog of bacterial heat-stable enterotoxins that are potent GUCY2C agonists.27 This drug is approved by the FDA under the trade name Linzess™ to treat IBS-C and CIC.28,29 Linaclotide enhances bowel function by activating GUCY2C and inducing fluid and electrolyte secretion in the small intestine, improving frequency and stool consistency. Generally, linaclotide is well tolerated, with side effects primarily reflecting on-target activity of GUCY2C mediation of fluid and electrolyte secretion, resulting in diarrhea.28,29 The robust safety of this agent is underscored by the negligible bioavailability of orally administered linaclotide.19,30 While treatment of chronic constipation syndromes usually involves daily oral linaclotide doses of 0.072–0.290 mg,28,29 daily doses up to 1 mg were safe. A study dose of 0.87 mg was selected to determine whether linaclotide-activated GUCY2C signaling in the distal rectum. There is minimal systemic absorption following oral administration of linaclotide. Its pharmacological effects are local in the GI tract. Linaclotide and its metabolite are activated by oxidation in the proximal GI tract. They are deactivated by reduction of its disulfide bonds and are further degraded into smaller peptides and amino acids; only 3% to 5% of the intact active drug or metabolite is excreted in the feces.19,30 Indeed, oral linaclotide 0.87 mg administered for 7 d increased cGMP, which was associated with changes in downstream signaling pathways, including VASP phosphorylation and Ki67 staining.18 Biopsy specimens from the cecum, transverse colon, and rectum were obtained by colonoscopy from healthy participants following administration of oral MoviPrepTM bowel preparation.18 However, linaclotide failed to increase rectal mucosa cGMP in participants for which rectal stool was cleared by enema, in the absence of bowel preparation with MoviPrepTM.18 These data suggest that sufficient peptide is delivered to rectal mucosa to produce GUCY2C activation when the bowel has been treated with MoviPrepTM and the GI transit time was decreased.18 However, in the native bowel, in the absence of a laxative preparation, degradation of linaclotide during intestinal transit results in insufficient concentrations of peptide reaching the distal rectal epithelium to produce GUCY2C activation.18,19
These observations suggested that to advance GUCY2C as a target for colorectal cancer chemoprevention an oral GUCY2C agonists with pharmacokinetic properties that support activation in the distal small bowel is needed. Dolcanatide (SP-333) is a chemically synthesized hexadecapeptide analog of uroguanylin composed of naturally occurring L-amino acids, except at amino and carboxyl terminals, which contain D-asparagine and D-leucine, respectively. Incorporation of D-amino acids is one strategy to improve the stability of the peptide and its persistence in the intestinal lumen and enhance delivery to the colorectum.20,21 Indeed, 10% to 20% of an oral dose of dolcanatide is estimated to be delivered to the distal colon, compared to 3% to 5% of an oral dose of linaclotide.19 Because of that delivery, dolcanatide is being evaluated for the treatment of disorders involving the colorectum, including inflammatory bowel diseases.31
In this study, dolcanatide 27 mg administered to healthy participants in a daily oral dose for 7 d produced no statistically significant increase in GUCY2C activation and cGMP accumulation compared to placebo. Study conditions were optimized to ensure the greatest opportunity to achieve receptor activation and biochemical signaling in the distal rectum. A dose of 27 mg is near the maximum tolerated dose safely administered to subjects for 14 consecutive days. All agent doses were witnessed daily by trained staff in a clinical research center. The last dose (day 7) was administered within 8 hours of sigmoidoscopy to maximize the opportunity for administered doses to interact with epithelial receptors in the distal rectum. Moreover, the incidence of diarrhea in subjects receiving the drug was eight-fold greater than in those receiving placebo (Table 6), confirming administration, delivery to the site of intestinal secretion, and bioactivity of dolcanatide.
We can only speculate about mechanisms that prevented dolcanatide from activating rectal GUCY2C and cGMP production. First, it is noteworthy that 11 of the 14 subjects (79%) who received dolcanatide experienced diarrhea (Table 6). Previous clinical studies revealed that approximately 20% to 30% of patients did not experience changes in bowel movements when administered linaclotide.32 Moreover, there was an association between the absence of changes in bowel movements and the inability of linaclotide to activate mucosal cGMP production.18 This variability in response could reflect genetic factors affecting the PD of GUCY2C peptide agonists in epithelial cells in some individuals. In that context, GUCY2C expression in normal epithelia varies about two orders of magnitude in the population.25 Those observations notwithstanding failure of dolcanatide to induce cGMP accumulation in the present study does not reflect a selective deficiency of GUCY2C in rectal mucosa. Indeed, GUCY2C mRNA expression was nearly identical in rectal mucosa samples from patients in both arms of the study, before and after treatments. It is noteworthy that future studies could extend these observations by assessing levels of GUCY2C protein by immunohistochemistry, to correlate with mRNA results. Further, mutations that alter GUCY2C activity have been described.33–36 Alternatively, there may be pharmacokinetic polymorphisms, with differences in metabolic clearance of the peptide in the intestine that limit the availability of active drug in some patients. Additionally, the contribution of environmental factors extrinsic to epithelial cells – for example, variations in the microbiome – may contribute to the variability of individual responses to GUCY2C agonist peptides. Moreover, inactivity of dolcanatide could indicate the presence of stool throughout the colorectum on day 7, which would prevent delivery of active peptide to the distal rectal mucosa, possibly reflecting established surface-active characteristics of GUCY2C peptides and the resulting immobilization of dolcanatide in the solid phase of the intestinal contents.
These observations suggest concrete steps for advancing GUCY2C as a target for colorectal cancer chemoprevention using oral hormone replacement therapy. Future studies should identify subjects who are biological responders to GUCY2C agonists to avoid enrolling subjects who are insensitive to this agent because of pharmacokinetic or PD differences.18 Further, molecular mechanisms underlying insensitivity to GUCY2C ligands in the population should be explored to better generalize the ultimate chemoprevention strategy for the greatest number of patients. In addition, exploring responses along the entire rostral-caudal axis of the colorectum could reveal pharmacokinetic differences in peptide luminal activities and persistence, and pharmacodynamic differences in GUCY2C responses in different segments of the large bowel. Moreover, it will be important to test sustained release formulations of oral GUCY2C agonists that are targeted specifically for delivery to the colorectum to maximize the PD effects of GUCY2C activation and downstream signaling that mediates chemoprevention.
Acknowledgments
The authors gratefully acknowledge the generous donation of dolcanatide and placebo for these studies by Synergy Pharmaceuticals, as well as the staffs of the Thomas Jefferson University Clinical Research Unit and Mayo Clinic Cancer Prevention Network for their assistance with study design, administration, and manuscript preparation.
Funding Statement
This work was sponsored by the National Cancer Institute, Division of Cancer Prevention [contract no. HHSN261201200042I].
Disclosure statement
SAW is the Chair of the Scientific Advisory Board, and a member of the Board of Directors, of Targeted Diagnostics & Therapeutics, Inc. Also, SAW is the Samuel MV Hamilton Professor of Medicine of Thomas Jefferson University. PJL serves as co-Chief Medical Officer for Exact Sciences, Inc. through a contracted services agreement with the Mayo Clinic in Rochester, MN. Dr. Limburg and Mayo Clinic have contractual rights to receive royalties through this agreement.
References
- 1.Jemal A, Siegel R, Xu J, Ward E.. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- 2.Society AC . Survival rates for colorectal cancer. In: Society AC , editor. Early detection, diagnosis, and staging;2019. https://www.cancer.org/cancer/colon-rectal-cancer/detection-diagnosis-staging/survival-rates.html [Google Scholar]
- 3.K A Lucas, G M Pitari, S Kazerounian, I Ruiz-Stewart, J Park, S Schulz, K P Chepenik, S A Waldman . Guanylyl cyclases and signaling by cyclic GMP. Pharmacol Rev. 2000;52:375–414. [PubMed] [Google Scholar]
- 4.Field M. Mechanisms of action of cholera and Escherichia coli enterotoxins. Am J Clin Nutr. 1979;32:189–196. doi: 10.1093/ajcn/32.1.189. [DOI] [PubMed] [Google Scholar]
- 5.Guerrant RL, Hughes JM, Chang B, Robertson DC, Murad F. Activation of intestinal guanylate cyclase by heat-stable enterotoxin of Escherichia coli: studies of tissue specificity, potential receptors, and intermediates. J Infect Dis. 1980;142:220–228. doi: 10.1093/infdis/142.2.220. [DOI] [PubMed] [Google Scholar]
- 6.Guarino A, Cohen M, Thompson M, Dharmsathaphorn K, Giannella R. T84 cell receptor binding and guanyl cyclase activation by Escherichia coli heat-stable toxin. Am J Physiol. 1987;253:G775–80. [DOI] [PubMed] [Google Scholar]
- 7.Li P, Lin JE, Snook AE, Gibbons AV, Zuzga DS, Schulz S, Pitari GM, Waldman SA. Colorectal cancer is a paracrine deficiency syndrome amenable to oral hormone replacement therapy. Clin Transl Sci. 2008;1:163–167. doi: 10.1111/j.1752-8062.2008.00040.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li P, Lin JE, Chervoneva I, Schulz S, Waldman SA, Pitari GM. Homeostatic control of the crypt-villus axis by the bacterial enterotoxin receptor guanylyl cyclase C restricts the proliferating compartment in intestine. Am J Pathol. 2007;171:1847–1858. doi: 10.2353/ajpath.2007.070198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lin JE, Li P, Snook AE, Schulz S, Dasgupta A, Hyslop TM, Gibbons AV, Marszlowicz G, Pitari GM, Waldman SA. The hormone receptor GUCY2C suppresses intestinal tumor formation by inhibiting AKT signaling. Gastroenterology. 2010;138:241–254. doi: 10.1053/j.gastro.2009.08.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.K A Steinbrecher, T M Tuohy, K Heppner Goss, M C Scott, D P Witte, J Groden, M B Cohen. Expression of guanylin is downregulated in mouse and human intestinal adenomas. Biochem Biophys Res Commun. 2000;273:225–230. doi: 10.1006/bbrc.2000.2917. [DOI] [PubMed] [Google Scholar]
- 11.Notterman DA, Alon U, Sierk AJ, Levine AJ. Transcriptional gene expression profiles of colorectal adenoma, adenocarcinoma, and normal tissue examined by oligonucleotide arrays. Cancer Res. 2001;61:3124–3130. [PubMed] [Google Scholar]
- 12.Karin Birkenkamp-Demtroder, Lise Lotte Christensen, Sanne Harder Olesen, Casper M Frederiksen, Päivi Laiho, Lauri A Aaltonen, Søren Laurberg, Flemming B Sørensen, Rikke Hagemann, Torben F ØRntoft . Gene expression in colorectal cancer. Cancer Res. 2002;62:4352–4363. [PubMed] [Google Scholar]
- 13.Waldman SA, Cagir B, Rakinic J, Fry RD, Goldstein SD, Isenberg G, Barber M, Biswas S, Minimo C, Palazzo J, et al. Use of guanylyl cyclase C for detecting micrometastases in lymph nodes of patients with colon cancer. Dis Colon Rectum. 1998;41:310–315. doi: 10.1007/BF02237484. [DOI] [PubMed] [Google Scholar]
- 14.Birbe R, Palazzo JP, Walters R, Weinberg D, Schulz S, Waldman SA. Guanylyl cyclase C is a marker of intestinal metaplasia, dysplasia, and adenocarcinoma of the gastrointestinal tract. Hum Pathol. 2005;36:170–179. doi: 10.1016/j.humpath.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 15.Li P, Lin JE, Snook AE, Waldman SA. ST-producing E. coli oppose carcinogen-induced colorectal tumorigenesis in mice. Toxins (Basel). 2017;9:279. doi: 10.3390/toxins9090279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.K Shailubhai, H H Yu, K Karunanandaa, J Y Wang, S L Eber, Y Wang, N S Joo, H D Kim, B W Miedema, S Z Abbas, S S Boddupalli, M G Currie, L R Forte. Uroguanylin treatment suppresses polyp formation in the Apc(Min/+) mouse and induces apoptosis in human colon adenocarcinoma cells via cyclic GMP. Cancer Res. 2000;60:5151–5157. [PubMed] [Google Scholar]
- 17.Sharman SK, Islam BN, Hou Y, Singh N, Berger FG, Sridhar S, Yoo W, Browning DD. Cyclic-GMP-elevating agents suppress polyposis in Apc(Min) Apc min mice by targeting the preneoplastic epithelium. Cancer Prev Res (Phila). 2018;11:81–92. doi: 10.1158/1940-6207.CAPR-17-0267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weinberg DS, Lin JE, Foster NR, Della’Zanna G, Umar A, Seisler D, Kraft WK, Kastenberg DM, Katz LC, Limburg PJ, et al. Bioactivity of oral Linaclotide in human colorectum for cancer chemoprevention. Cancer Prev Res (Phila). 2017;10(6):345–354. doi: 10.1158/1940-6207.CAPR-16-0286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Busby RW, Kessler MM, Bartolini WP, Bryant AP, Hannig G, Higgins CS, Solinga RM, Tobin JV, Wakefield JD, Kurtz CB, et al. Pharmacologic properties, metabolism, and disposition of linaclotide, a novel therapeutic peptide approved for the treatment of irritable bowel syndrome with constipation and chronic idiopathic constipation. J Pharmacol Exp Ther. 2013;344(1):196–206. doi: 10.1124/jpet.112.199430. [DOI] [PubMed] [Google Scholar]
- 20.Boulete IM, Thadi A, Beaufrand C, Patwa V, Joshi A, Foss JA, Eddy EP, Eutamene H, Palejwala VA, Theodorou V, et al. Oral treatment with plecanatide or dolcanatide attenuates visceral hypersensitivity via activation of guanylate cyclase-C in rat models. World J Gastroenterol. 2018;24:1888–1900. doi: 10.3748/wjg.v24.i17.1888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kunwar Shailubhai, Vaseem Palejwala, Krishna Priya Arjunan, Sayali Saykhedkar, Bradley Nefsky, John A Foss, Stephen Comiskey, Gary S Jacob, Scott E Plevy. Plecanatide and dolcanatide, novel guanylate cyclase-C agonists, ameliorate gastrointestinal inflammation in experimental models of murine colitis. World J Gastrointest Pharmacol Ther. 2015;6:213–222. doi: 10.4292/wjgpt.v6.i4.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schmelzer M, Schiller LR, Meyer R, Rugari SM, Case P. Safety and effectiveness of large-volume enema solutions. Appl Nurs Res. 2004;17:265–274. [PubMed] [Google Scholar]
- 23.Lai EJ, Calderwood AH, Doros G, Fix OK, Jacobson BC. The Boston bowel preparation scale: a valid and reliable instrument for colonoscopy-oriented research. Gastrointest Endosc. 2009;69:620–625. doi: 10.1016/j.gie.2008.05.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Steiner AL, Pagliara AS, Chase LR, Kipnis DM. Radioimmunoassay for cyclic nucleotides. II. Adenosine 3ʹ,5ʹ-monophosphate and guanosine 3ʹ,5ʹ-monophosphate in mammalian tissues and body fluids. J Biol Chem. 1972;247:1114–1120. doi: 10.1016/S0021-9258(19)45622-6. [DOI] [PubMed] [Google Scholar]
- 25.Scott A Waldman, Terry Hyslop, Stephanie Schulz, Alan Barkun, Karl Nielsen, Janis Haaf, Christine Bonaccorso, Yanyan Li, David S Weinberg. Association of GUCY2C expression in lymph nodes with time to recurrence and disease-free survival in pN0 colorectal cancer. Jama. 2009;301:745–752. doi: 10.1001/jama.2009.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pitari GM, Di Guglielmo MD, Park J, Schulz S, Waldman SA. Guanylyl cyclase C agonists regulate progression through the cell cycle of human colon carcinoma cells. Proc Natl Acad Sci U S A. 2001;98:7846–7851. doi: 10.1073/pnas.141124698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Waldman SA, Camilleri M. Guanylate cyclase-C as a therapeutic target in gastrointestinal disorders. Gut. 2018;67:1543–1552. doi: 10.1136/gutjnl-2018-316029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chey WD, Lembo AJ, Lavins BJ, Shiff SJ, Kurtz CB, Currie MG, MacDougall JE, Jia XD, Shao JZ, Fitch DA, et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am J Gastroenterol. 2012;107:1702–1712. doi: 10.1038/ajg.2012.254. [DOI] [PubMed] [Google Scholar]
- 29.Lembo AJ, Schneier HA, Shiff SJ, Kurtz CB, MacDougall JE, Jia XD, Shao JZ, Lavins BJ, Currie MG, Fitch DA, et al. Two randomized trials of linaclotide for chronic constipation. N Engl J Med. 2011;365:527–536. doi: 10.1056/NEJMoa1010863. [DOI] [PubMed] [Google Scholar]
- 30.Busby RW, Bryant AP, Bartolini WP, Cordero EA, Hannig G, Kessler MM, Mahajan-Miklos S, Pierce CM, Solinga RM, Sun LJ, et al. Linaclotide, through activation of guanylate cyclase C, acts locally in the gastrointestinal tract to elicit enhanced intestinal secretion and transit. Eur J Pharmacol. 2010;649:328–335. doi: 10.1016/j.ejphar.2010.09.019. [DOI] [PubMed] [Google Scholar]
- 31.Forte LR, Freeman RH, Krause WJ, London RM. Guanylin peptides: cyclic GMP signaling mechanisms. Braz J Med Biol Res. 1999;32:1329–1336. doi: 10.1590/S0100-879X1999001100002. [DOI] [PubMed] [Google Scholar]
- 32.Bharucha AE, Waldman SA. Taking a lesson from microbial diarrheagenesis in the management of chronic constipation. Gastroenterology. 2010;138:813–817. doi: 10.1053/j.gastro.2010.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fiskerstrand T, Arshad N, Haukanes BI, Tronstad RR, Pham KD, Johansson S, Håvik B, Tønder SL, Levy SE, Brackman D, et al. Familial diarrhea syndrome caused by an activating GUCY2C mutation. N Engl J Med. 2012;366:1586–1595. doi: 10.1056/NEJMoa1110132. [DOI] [PubMed] [Google Scholar]
- 34.Muller T, Rasool I, Heinz-Erian P, Mildenberger E, Hulstrunk C, Muller A, Michaud L, Koot BGP, Ballauff A, Vodopiutz J, et al. Congenital secretory diarrhoea caused by activating germline mutations in GUCY2C. Gut. 2016;65:1306–1313. doi: 10.1136/gutjnl-2015-309441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Romi H, Cohen I, Landau D, Alkrinawi S, Yerushalmi B, Hershkovitz R, Newman-Heiman N, Cutting G, Ofir R, Sivan S, et al. Meconium ileus caused by mutations in GUCY2C, encoding the CFTR-activating guanylate cyclase 2C. Am J Hum Genet. 2012;90:893–899. doi: 10.1016/j.ajhg.2012.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Smith A, Bulman DE, Goldsmith C, Bareke E, Majewski J, Boycott KM, Nikkel SM. Meconium ileus in a Lebanese family secondary to mutations in the GUCY2C gene. Eur J Hum Genet. 2015;23:990–992. doi: 10.1038/ejhg.2014.236. [DOI] [PMC free article] [PubMed] [Google Scholar]


