Figure 3. Examples of SSR suggested changes in prednisone (or equivalent dose of another corticosteroid).
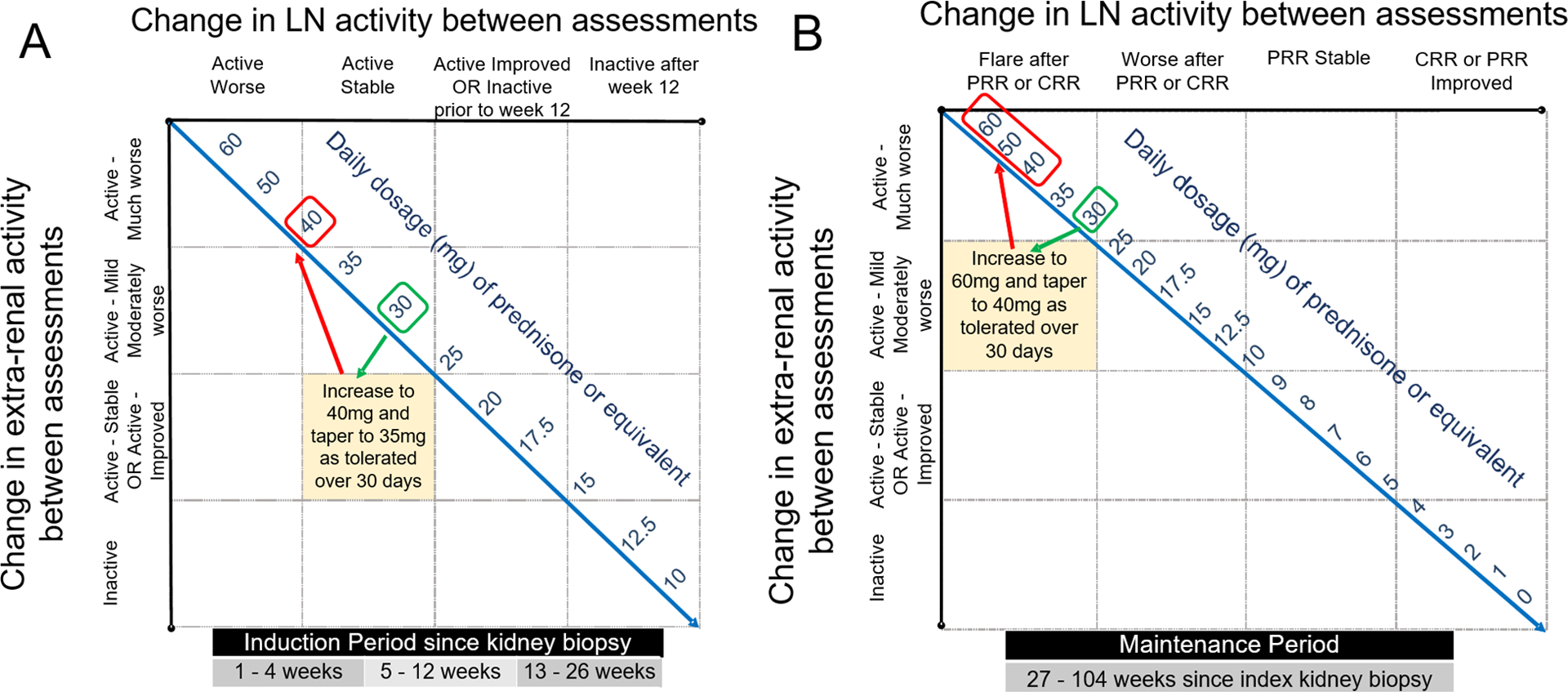
A: Suggested prednisone dose adjustment of a cSLE patient (≥ 40 kg) whose kidney biopsy showed proliferative lupus nephritis within the preceding 12 weeks. Upon re-assessment, the patient was taking prednisone 30 mg daily, his renal course was “Active-improved” and extra-renal disease course “Active-much worse”. The SSR recommends increasing daily prednisone dose to 40 mg. If tolerated, tapering oral-CS during the subsequent 4 weeks post assessment to 35 mg is proposed.
B: Suggested prednisone dose adjustment of a cSLE patient (≥ 40 kg) who completed induction therapy for LN, and achieved at least partial renal remission at week 26. Upon re-assessment, the patient was taking prednisone 30 mg daily and having a “LN flare after PRR”. The SSR recommends increasing the daily prednisone dose to 60 mg, irrespective of the extra-renal course. If renal response is improved with higher oral-CS doses, then oral-CS tapering can be initiated at day 10. The minimum allowable daily prednisone dose at day 30 following LN flare is 40 mg. If the patient has not improved by day 10, then intravenous pulse methylprednisolone (1–3 doses) should be considered.
