Abstract
Background
Various factors related to the sensitivity of non-small cell lung carcinoma (NSCLC) to 5-fluorouracil (5-FU) have been reported, and some of them have been clinically applied. In this single-institutional prospective analysis, the mRNA expression level of five folic acid-associated enzymes was evaluated in surgical specimens of NSCLC. We investigated the correlation between the antitumor effect of 5-FU in NSCLC using an anticancer drug sensitivity test and the gene expression levels of five enzymes.
Materials and methods
Forty patients who underwent surgery for NSCLC were enrolled, and the antitumor effect was measured using an in vitro anticancer drug sensitivity test (histoculture drug response assay) using freshly resected specimens. In the same sample, the mRNA expression levels of five enzymes involved in the sensitivity to 5-FU were measured in the tumor using real-time PCR. The expression levels and the result of the sensitivity test were compared.
Results
No correlation was found between dihydropyrimidine dehydrogenase (DPD), orotate phosphoribosyltransferase (OPRT), or DPD/OPRT expression and the antitumor effects of 5-FU. On the other hand, a correlation was found between thymidylate synthase (TS), folylpoly-c-glutamate synthetase (FPGS), and dihydrofolate reductase (DHFR) expression and 5-FU sensitivity.
Conclusion
Expression of FPGS and DHFR may be useful for predicting the efficacy of 5-FU-based chemotherapy for NSCLC.
Keywords: Histoculture drug response assay, Reverse-transcriptase polymerase chain reaction, Thymidylate synthase, Dihydropyrimidine dehydrogenase, Orotate phosphoribosyltransferase
Introduction
5-fluorouracil (5-FU) is an antimetabolite that is widely used to treat various solid cancers including non-small cell lung cancer (NSCLC) [1, 2] In Japan, an oral 5-FU prodrug has been developed. At present, UFT® and TS-1® are indicated in Japan for NSCLC and are often used as one of the drugs in postoperative adjuvant chemotherapy or systemic chemotherapy [3, 4] Despite various measures to enhance the effects of 5-FU, the treatment is ineffective in many patients. In addition, 5-FU anticancer agents are less effective in patients with advanced or recurrent NSCLC [2] However, if the therapeutic effect of 5-FU anticancer drugs in cancer patients can be predicted before administration, the disadvantages of unnecessary administration can be prevented. Thus, identification of factors that can predict the effect of drugs before drug administration is important.
5-FU is activated only after it is converted to 5-fluorodeoxyuridine monophosphate (FdUMP). FdUMP inhibits DNA synthesis by forming a ternary complex with Thymidylate synthase (TS), which is an essential enzyme for DNA synthesis, together with 5,10-methylenetetrahydrofolate (5,10-CH2-THF). It has been reported that the main pathway for conversion of 5-FU to FdUMP in tumors is the pathway converted by Orotate phosphoribosyltransferase (OPRT) in the presence of phosphoribosylpyrophosphate (PRPP) [5] On the other hand, 5-FU is rapidly degraded by dihydropyrimidine dehydrogenase (DPD). Therefore, the expression levels of OPRT and DPD may be involved in the effects of 5-FU. In addition, 5,10-CH2-THF, which forms a ternary complex with FdUMP and TS, is one of the reduced folic acids and is regulated by dihydrofolate reductase (DHFR) and folylpoly-c-glutamate synthetase (FPGS) [6] Therefore, DHFR and FPGS may also be involved in the effects of 5-FU.
Several reports of NSCLC cases have described the sensitivity to 5-FU using clinical samples [7–9], but no clear findings have been obtained. In this study, the level of mRNA expression (TS, DPD, OPRT, DHFR, and FPGS) of factors associated with 5-FU sensitivity was measured with reverse-transcriptase polymerase chain reaction (RT-PCR), and we used the histoculture drug response assay (HDRA) method to assess sensitivity to 5-FU. We then investigated the correlation between the expression level of each factor and the chemosensitivity result.
Patients and methods
Patients
Tissue samples were collected at the time of surgery from 40 patients who underwent surgery at the Department of Surgery (II), University of Fukui Hospital for NSCLC with a tumor size > 20 mm between January 2012 and December 2015. The experimental use of the chemosensitivity test was approved by the Institutional Research Committee, and the trial was approved by the Institutional Review Board for University of Fukui Hospital. All patients were informed of the nature of this study, and written informed consent was obtained.
Fresh specimens were sampled from the primary lesion immediately after surgical resection, immersed in Hank’s solution, and used for in vitro chemosensitivity testing with HDRA. Ten-micrometer-thick slices from the paraffin-embedded specimens were later used for quantitative RT-PCR.
HDRA
The HDRA was used as an in vitro drug sensitivity test as previously reported. [10, 11] Collagen sponge gels (Gel foam®) manufactured from pig skin were purchased from Pfizer Japan Inc. (Tokyo, Japan). Cancerous portions of specimens were minced into pieces of approximately 10 mg, which were then placed on the prepared collagen surface in 24-well microplates. Plates were incubated for 7 days in the presence of drugs dissolved in RPMI 1640 medium containing 20% fetal calf serum in a humidified atmosphere containing 95% air/5% CO2 at 37 °C. 5-FU was provided by Tokyo Chemical Industry Co. Ltd. (Tokyo, Japan) and used at 300 μg/ml. After these specimens were histocultured, 100 μl Hank’s balanced salt solution containing 0.1 mg/ml type I collagenase (Sigma) and 100 μl 9.6 mg/ml 3-(4,5-dimetylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) solution dissolved in phosphate-buffered saline were added to each culture well and incubated for another 16 h. Following extraction with dimethysulfoxide, absorbance of the solution in each well was read at 540 nm (control 630 nm) using a microplate reader (Spectra Max M5; Molecular Device LLC, San Jose, CA). Absorbance per gram of cultured tumor tissue (OD/W) was calculated from the mean absorbance of tissue from three culture wells. The tumor tissue weight was determined prior to culture.
The inhibition rate was calculated using the following formula:
Laser-capture microdissection and real-time RT-PCR (the Danenberg tumor profile [DTP] method)
The quantitative assay of the five genes of interest (TS, DPD, OPRT, DHFR, and FPGS) using paraffin-embedded sections of resected NSCLC specimens was performed according to the DTP method of Response Genetics (New York, NY, USA) [12]. For every specimen, four sets of 10-µm-thick sections and one set of 5-µm-thick sections were prepared from formalin-fixed, paraffin-embedded tissues. The 5-µm-thick sections were stained with hematoxylin and eosin and examined histologically. The 10-µm-thick sections were stained with nuclear fast red (American Master Tech Scientific, Lodi, CA) and used for laser-capture microdissection (PALM Microsystem, Leica, Wetzlar, Germany) from the upper and lower thirds of tumors, separately. The dissected tissue samples were transferred to reaction tubes containing 400 µl RNA lysis buffer, and RNA was isolated. Finally, cDNA was prepared as described by Lord et al. [13, 14].
Quantification of the five genes of interest (TS, DPD, OPRT, DHFR, and FPGS) and an internal reference gene (β-actin) was performed using a fluorescence-based real-time PCR system (ABI PRISM 7900 Sequence Detection System, Applied Biosystems, Foster City, CA). The final volume of the reaction mixture was 20 µl. Cycling conditions and the primers and probes were described previously by Matsubara et al. [15] Gene expression was analyzed twice to confirm the reproducibility, and values (relative mRNA levels) were expressed as the ratio between the gene of interest and the internal reference gene (β-actin).
Examination of the necessity of Gimeracil (5-chloro-2, 4-dihydroxypyridine (CDHP))
NSCLC has high DPD activity [12] and requires the use of CDHP, an inhibitor of DPD. In an anticancer sensitivity test, the sensitivity effect of adding CDHP was also examined. Previous experiments were performed to determine the appropriate concentration of CDHP. The experiment was performed by dividing 5-FU alone into four groups at a concentration obtained by adding 200 μg/ml and 300 μg/ml CDHP to 200 μg/ml and 300 μg/ml 5-FU. At that concentration, 5-FU was used in combination with CDHP, and the results were observed.
Statistical analysis
Statistical analysis was performed on a personal computer with Stat Mate IV (Ver. IV, ATMS, Japan). The correlations between the mRNA expression of genes examined and clinico-pathological parameters were evaluated with the Pearson product moment correlation coefficient. To evaluate the correlation between two variables, linear regression was performed, and the Spearman rank correlation coefficient was calculated. Probability (P) values of less than 0.05 were considered statistically significant.
Results
Anticancer sensitivity test (HDRA) for 5-FU in our institution
The anticancer drug sensitivity test for 5-FU in NSCLC has been conducted in our department since 2006, and HDRA at the relevant 5-FU concentration of 300 μg/ml was successfully performed for 419 patients (289 males and 130 females, median age 71.3 years, range 39–82 years).
Examination of the necessity of CDHP
A test was conducted to examine the results of sensitivity to 5-FU by changing the concentration of CDHP in 29 cases of NSCLC. We found no significant difference in sensitivity between the 5-FU only group (suppression rate 62 ± 17%, n = 29) and the group to which CDHP was added (300 mg; 60 ± 18%, n = 29, 200 mg; 60 ± 17%, n = 23). Therefore, this study was performed using only 5-FU. We also examined the toxicity of CDHP by performing a CDHP alone sensitivity test without 5-FU. CDHP alone showed a very low inhibition rate and no antitumor effect (8 ± 10%, n = 29).
Patient characteristics
Forty patients (29 men (72.5%) and 11 women (27.5%)) who underwent surgery at our hospital for NSCLC were enrolled (Table 1). The median age was 70 years (range, 47–84 years). The postoperative pathological stage was stage IA in 10 patients (25%), stage IB in nine (22.5%), stage IIA in 10 (25%), stage IIB in four (10%), stage IIIA in five (12.5%), and stage IV in two (5%).
Table 1.
Patients characteristics
| Age(medial value) | |
|---|---|
| Sex | 70 (47–84) |
| Male | 29 (72.5%) |
| Female | 11 (27.5%) |
| Smoking | |
| Former and current | 30 (75%) |
| Never | 10 (25%) |
| Performance status | |
| 0 | 28 (70%) |
| 1 | 9 (22.5%) |
| 2 | 1 (2.5%) |
| 3 | 2 (5%) |
| Pathologocal stage | |
| IA | 10 (25%) |
| IB | 9 (22.5%) |
| IIA | 10 (25%) |
| IIB | 4 (10%) |
| IIIA | 5 (12.5%) |
| IV | 2 (5%) |
| Operative procedure | |
| Lobectomy | 35 (87.5%) |
| Segmentectomy | 2 (5%) |
| Partial resection | 3 (7.5%) |
| Pathologocal finding | |
| Adeno carcinoma | 23 (57.5%) |
| Squamous cell carcinoma | 15 (37.5%) |
| Others | 2 (5%) |
| Genetic status | |
| L858R: | 7 |
| exon19: | 3 |
| ALK Translocation: | 1 |
| Wild type: | 12 |
| Unexamined: | 17 |
The operative procedure was lobectomy in 35 patients (87.5%), segmental resection in two patients (5%), and partial wedge lung resection in three patients (7.5%). The histology included 23 cases of adenocarcinoma (57.5%), 15 cases of squamous cell carcinoma (37.5%), and two cases (5%) of other types of NSCLC. Regarding EGFR mutation and ALK translocation in adenocarcinoma, seven cases were L858R positive, three cases were exon 19 positive, one case was ALK positive, and 12 cases were wild type (Table 1).
Comparison of gene expression and the percent inhibition in the anticancer sensitivity test (HDRA) for 5-FU
Gene expression of TS, OPRT, DPD, FPGS, and DHFR was measured in 39 samples except for one that could not be measured successfully. However, 2 samples did not grow on the HDRA method, so they were excluded. Finally, 37 samples were considered. The mean levels of mRNA of TS, DPD, OPRT, FPGS, and DHFR in these specimens were 3.69 ± 2.89 (n = 37), 1.200 ± 0.637 (n = 37), 0.706 ± 1.23 (n = 37), 0.885 ± 0.477 (n = 37), and 2.10 ± 2.50 (n = 37), respectively (Table 2). Expression of these genes was not correlated with the age, sex, histopathological type, clinical stage, or driver gene mutation.
Table 2.
mRNA expression and inhibition and HDRA results
| mRNA (relative gene expression) | HDRA(%) | |||||
|---|---|---|---|---|---|---|
| TS | DPD | OPRT | FPGS | DHFR | Inhibition (%) | |
| N | 37 | 37 | 37 | 37 | 37 | 37 |
| Mean ± SD | 3.69 ± 2.89 | 1.20 ± 0.637 | 0.706 ± 1.23 | 0.885 ± 0.477 | 2.10 ± 2.50 | 55.4 ± 13.7 |
|
Median (range) |
2.56 (0.60–11.2) |
1.20 (0.25–2.68) |
0.371 (0.10–7.57) |
0.769 (0.42–1.54) |
1.49 (0.50–4.64) |
57 (35–66) |
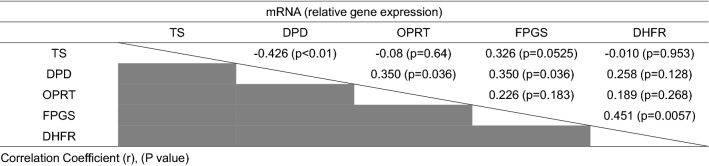
Table 3 shows the correlations among TS, OPRT, DPD, FPGS, and DHFR mRNA for all samples. The mRNA expression levels of TS were moderately correlated with those of DPD (Correlation Coefficient (r): -0.426, p < 0.01), and those of DPD were weakly correlated with those of OPRT (r: 0.350, p < 0.036) and FPGS (r: 0.350, p < 0.036). The mRNA expression levels of FPGS were moderately correlated with those of DHFR (r: 0.451, p = 0.0057).
Table 3.
Correlation of TS, DPD, OPRT, FPGS and DHFR
We found no significant correlation between expression of DPD, OPRT, or DPD/OPRT and the percent inhibition with 5-FU using the HDRA method (Fig. 1). We found a significant correlation between expression of TS, FPGS, and DHFR and the percent inhibition with 5-FU using the HDRA method (p < 0.05, r = 0.350, p < 0.01, r = 0.418, and p < 0.05, r = 0.331, respectively) (Fig. 2).
Fig. 1.
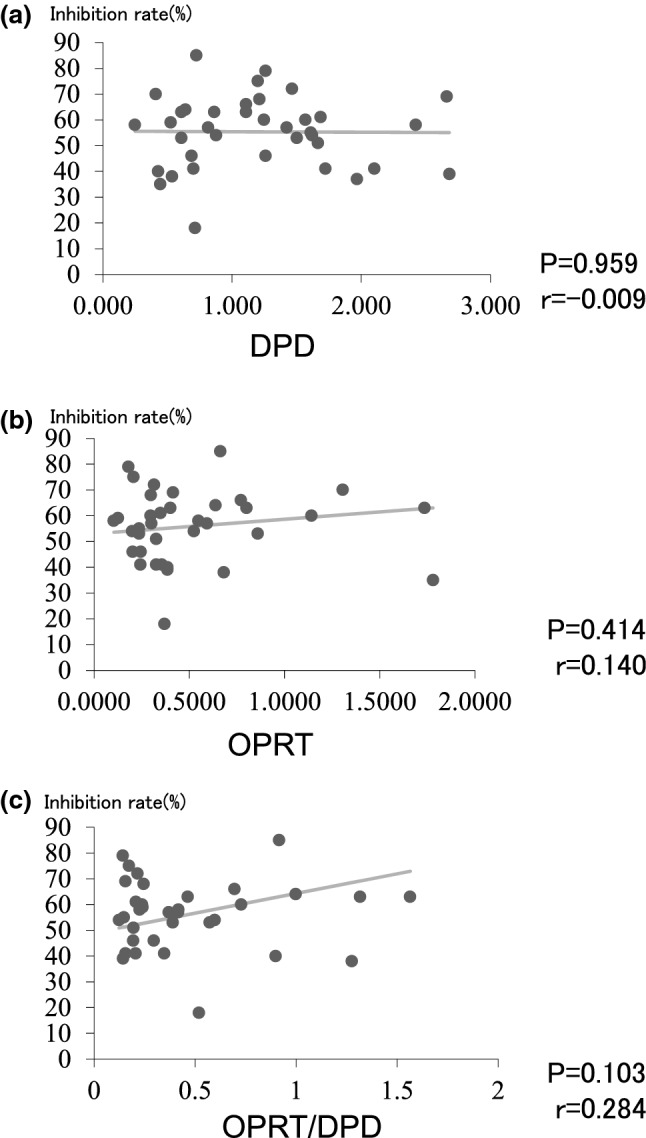
No significant correlation was found between expression of DPD (a), OPRT (b), and DPD/OPRT (c) and the percent inhibition with 5-FU using the HDRA method
Fig. 2.
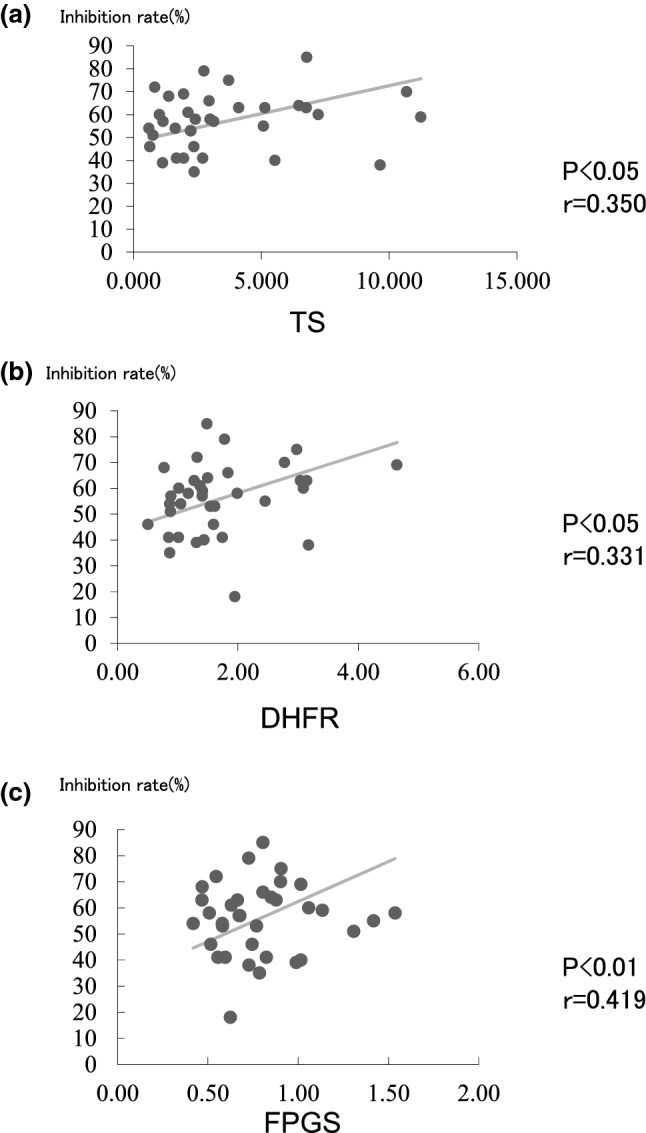
A significant correlation was found between expression of TS (a), DHFR (b), and FPGS (c) and the percent inhibition with 5-FU using the HDRA method
Discussion
To the best of our knowledge, this is the first study to show a correlation between FPGS and DHFR expression and 5-FU sensitivity results in NSCLC. FPGS showed a stronger correlation with 5-FU sensitivity than DHFR.
FPGS converts intracellular folic acid and folic acid antagonists, such as methotrexate, into polyglutamic acid, which is retained intracellularly for a long time. Polyglutamylation of intracellular 5,10-methylenetetrahydrofolate enables more efficient formation and stabilization of inhibitory ternary complexes involving TS and metabolites of 5-FU, and it may also increase the cytotoxicity of 5-FU. [6] Two reports have shown a correlation between 5-FU sensitivity and FPGS in colon cancer [6] and breast cancer. [16].
DHFR is the target enzyme of methotrexate, which enters the cell, tightly binds to DHFR, and inhibits the reduction of dihydrofolate to tetrahydrofolate. [17] Only one report has evaluated the activity of DHFR and the antitumor effect of 5-FU. [18] They reported that DHFR mRNA had a high value using a 5-FU-resistant mouse cell line.
5-FU is activated only after it is converted to 5-fluorodeoxyuridine monophosphate (FdUMP). TS, an enzyme that is essential for DNA synthesis, methylates deoxyuridine monophosphate (dUMP) and converts it to deoxythymidine monophosphate (dTMP). Therefore, FdUMP is covalently bonded to TS together with 5,10-methylenetetrahydrofolate (5,10-CH2-THF), which is reduced to folic acid, to form a strong ternary complex, which inhibits DNA synthesis. [19] FPGS acts on the pathway that converts 5,10-CH2-THF from monoglutamate to polyglutamates, the increase in which is indispensable for creating a ternary complex that may affect the sensitizing effect of 5-FU. The function of TS activity is inhibition, and the pool of dTTP, which is a precursor of DNA, is depleted, leading to inhibition of DNA synthesis and cell death. [19].
In this study, the positive correlation between TS expression and 5-FU sensitivity is the opposite of the results of other reports of NSCLC. [7, 20–22] Regarding the sensitivity of TS in gastric cancer and colorectal cancer, sporadic reports have shown no correlation or reverse correlation between the sensitivity to 5-FU and increased TS activity. [23, 24] In NSCLC, a positive correlation with TS was reported, but TS activity tends to be lower than in other carcinomas, [12] suggesting that even if TS activity is high, it falls within the range where the effect of 5-FU can be observed.
Regarding the sensitivity of 5-FU, the activities of OPRT and DPD have been well evaluated, and several reports show that they are involved in the sensitivity of NSCLC. [5, 25, 26] The results of this study did not show a correlation between OPRT and DPD expression and 5-FU sensitivity. Although the OPRT/DPD ratio has been reported to be an important predictor of the efficacy of fluoropyrimidine-based chemotherapy for metastatic colorectal cancer, [5] this report on DPD/OPRT showed no significant difference with the sensitivity of 5-FU.
Several reports have described the relationship between TS, DPD, and OPRT and 5-FU sensitivity in NSCLC. Eguchi et al. [20] evaluated the relationship between response to treatment and immunohistochemical expression levels in patients with advanced NSCLC. Low expression levels of DPD and TS were seen in patients not treated with S-1-carboplatin, which is associated with better response and longer survival in patients treated with paclitaxel-carboplatin. Tumor expression levels of TS and DPD predict the response to S-1-carboplatin chemotherapy in patients with advanced NSCLC. Nakano et al. [21] reported an immunohistochemical study on the clinical importance of TS, OPRT, and DPD expression using 151 NSCLC specimens resected from patients treated postoperatively with tegafur/uracil (UFT). Patients who had tumors with low TS expression (p = 0.0133), high OPRT expression (p = 0.0145), or low DPD expression (p = 0.0004) had significantly high of 5-year survival rates. Shintani et al. [7] investigated patients using RT-PCR for intratumoral expression and examined the correlation between gene expression and the efficacy of 5-FU in NSCLC. Patients receiving postoperative 5-FU alone (n = 30) comprised the 5-FU group, and those who had only surgery were included in the control group (n = 86). When dichotomized by mean TS and DPD mRNA levels, patients with low DPD tumors receiving 5-FU had significantly better prognosis than those who did not receive adjuvant treatment (p = 0.041). Based on these results, quantification of TS and DPD mRNA levels can predict the efficacy of 5-FU after surgery in patients with NSCLC.
5-FU is rapidly degraded by DPD. Due to the higher DPD activity in NSCLC compared to other carcinomas, [12] 5-FU alone is less effective, necessitating co-administration of CDHP, for which S-1 was developed. In view of the mechanism of action of 5-FU, the effects of 5-FU are expected to be reduced if expression of the target enzyme TS and the degrading enzyme DPD in tumor tissue is high. CDHP (Gimeracil), which is used in S-1, [27] inhibits DPD. In this study, we found no difference in the sensitivity results even if CDHP was added to 5-FU in the preliminary sensitivity test. This suggests that DPD may not affect the antitumor effect of 5-FU in vitro.
There are several reports that The HDRA method correlates well with the susceptibility of NSCLC to anticancer drugs and its clinical efficacy. [28–30] Further, the usefulness of HDRA has been documented for several other cancer types including gastric cancer [31] and colorectal cancer. [32] This histo-culture method has the advantage of culturing cancer cells while maintaining cell–cell contacts which has good cell viability, and the disadvantage of requiring a certain amount of tissue sample. In the present study, sufficient amount of sample could be obtained from the surgical specimens. Moreover, the high evaluability rate (n = 419, 96.5%) from previous tests for NSCLC conducted at our institution demonstrate that this method is a good alternative for testing the sensitivity of 5-FU in NSCLC.
The limitations of this study are the small number of cases, the single-institution design, and the in vitro results of the anticancer drug sensitivity test. This study pointed out the correaltionship between in vitro sensitivity of NSCLC samples to 5-FU and mRNA level of FPGS and DHFR, whether or not this can reflect in vivo drug effect needs more investigation. Furthermore, studying the relationship between anticancer effects in NSCLC patients who actually received 5-FU and the expression levels of various factors in the tumors is necessary in clinical study. Another limitation is the in vitro use of specimens obtained during surgery. For unresectable advanced NSCLC, small specimens such as those obtained from bronchoscopy should be used. The feasibility of such transbronchial lung biopsy samples is being investigated. Nakajima et al. [33] used a metastatic lymph node sample obtained with endo-bronchial ultrasound-guided transbronchial needle aspiration in patients with non-small cells to obtain TS, DPD, TP, and OPRT mRNA. The feasibility of expression analysis should be evaluated. Clinical application is also expected.
Few reports have examined the sensitivity of 5-FU in NSCLC. Our study provides results that will be useful for assessing the sensitivity of 5-FU in future clinical applications.
Conclusion
The mRNA levels of five folic acid-associated enzymes involved in the antitumor effect of 5-FU were examined in resected NSCLC tumor specimens with RT-PCR, and the in vitro anticancer sensitivity test was performed. In conclusion, FPGS and DHFR may be involved in 5-FU sensitivity.
Previous studies have reported that TS, OPRT, and DPD are associated with 5-FU sensitivity. Combined with this result, the folic acid metabolism pathway is also important, and it will be the basis for the development of new anticancer agents using both pathways.
Acknowledgements
The authors are indebted to Mio Kawakami for support as an excellent laboratory assistant.
Abbreviations
- HDRA
Histoculture Drug Response Assay,
- DPD
Dihydropyrimidine dehydrogenase
- OPRT
Orotate phosphoribosyltransferase
- TS
Thymidylate synthase
- CDHP
5-Chloro-2,4-dihydroxypyridine
- RT-PCR
Reverse-transcriptase polymerase chain reaction
- NSCLC
Non-small cell lung carcinoma
- 5-FU
5-Fluorouracil
- FPGS
Folylpoly-c-glutamate synthetase
- DHFR
Dihydrofolate reductase
- FdUMP
5-Fluorodeoxyuridine monophosphate
- dUMP
Deoxyuridine monophosphate
- dTMP
Deoxythymidine monophosphate
- dTTP
Deoxythymidine triphosphate
- 5,10-CH2-THF
5,10-Methylenetetrahydrofolate
Authors’ contributions
KS, MS, AO and TI are the attending physicians for these cases. KS is responsible for the concept and design of this study, and everything. The other co-authors contributed to the acquisition and interpretation of the reference. KS drafted the study and other co-authors revised it. All authors approve the publication of the manuscript and agree to take responsibility. All authors reviewed, edited, and agreed on the final draft of the manuscript. All authors read and approved the final manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability
The dataset is available upon request.
Code availability
Not applicable.
Declarations
Ethics approval and consent to participate
The authors confirm that the study has been approved by the Institutional Review Board for University of Fukui Hospital and certify that the study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Written informed consent was obtained from the patient for extracting his data from electronic medical chart system. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Kayo Sakon, Email: ksakon@u-fukui.ac.jp.
Masato Sasaki, Email: masato@u-fukui.ac.jp.
Kaede Tanaka, Email: kaedet@u-fukui.ac.jp.
Tae Mizunaga, Email: tae08@u-fukui.ac.jp.
Keita Yano, Email: yano3101@u-fukui.ac.jp.
Yuuko Kawamura, Email: yuko0131@u-fukui.ac.jp.
Akitoshi Okada, Email: lebleu@u-fukui.ac.jp.
Takeshi Ikeda, Email: tikeda@hosp.ncgm.go.jp.
Sawaka Tanabe, Email: tanabes@u-fukui.ac.jp.
Atsushi Takamori, Email: cookie@u-fukui.ac.jp.
Narihisa Yamada, Email: nyama@u-fukui.ac.jp.
Kouichi Morioka, Email: kmori@u-fukui.ac.jp.
Takaaki Koshiji, Email: koshiji@u-fukui.ac.jp.
References
- 1.Arkenau HT, Bermann A, Rettig K, Strohmeyer G, Porschen R. 5-Fluorouracil plus leucovorin is an effective adjuvant chemotherapy in curatively resected stage III colon cancer; long-term follow-up results of the adjCCA-01 trials. Ann Oncol. 2003;14:395–399. doi: 10.1093/annonc/mdg100. [DOI] [PubMed] [Google Scholar]
- 2.Takeda K. Clinical development of S-1 for non-small cell lung cancer: a Japanese perspective. Ther Adv Med Oncol. 2013;5(5):301–311. doi: 10.1177/1758834013500702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yoshioka H, Okamoto I, Morita S, et al. Efficacy and safety analysis according to histology for S-1 in combination with carboplatin as first-line chemotherapy in patients with advanced non-small-cell lung cancer: updated results of the West Japan Oncology Group LETS study. Ann Oncol. 2013;24(5):1326–1331. doi: 10.1093/annonc/mds629. [DOI] [PubMed] [Google Scholar]
- 4.Kato H, Ichinose Y, Ohta M, Hata E. A Randomized Trial of Adjuvant Chemotherapy with Uracil–Tegafur for Adenocarcinoma of the Lung. N Engl J Med. 2004;350:1713–1721. doi: 10.1056/NEJMoa032792. [DOI] [PubMed] [Google Scholar]
- 5.Ichikawa W, Uetake H, Shirota Y, et al. Both gene expression for orotate phosphoribosyltransferase and its ratio to dihydropyrimidine dehydrogenase influence outcome following fluoropyrimidine-based chemotherapy for metastatic colorectal cancer. Br J Cancer. 2003;89:1486–1492. doi: 10.1038/sj.bjc.6601335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sohn K-J, Smirnakis F, Moskovitz DN, et al. Effects of folylpolyglutamate synthetase modulation on chemosensitivity of colon cancer cells to 5-fluorouracil and methotrexate. Gut. 2004;53:1825–1831. doi: 10.1136/gut.2004.042713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shintani Y, Ohta M, Hirabayashi H, et al. Thymidylate synthase and dihydropyrimidine dehydrogenase mRNA levels in tumor tissues and the efficacy of 5-fluorouracil in patients with non-small-cell lung cancer. Lung Cancer. 2004;45(2):189–196. doi: 10.1016/j.lungcan.2004.01.015. [DOI] [PubMed] [Google Scholar]
- 8.Kimura M, Imamura F, Inoue T, et al. Protein and mRNA expression of folic acid-associated enzymes as biomarkers for the cytotoxicity of the thymidylate synthetase-targets drugs, pemetrexate and S-1, in non-small cell lung cancer. Mol Clin Oncol. 2017;7(1):15–23. doi: 10.3892/mco.2017.1262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Inoue K, Takao M, Watanabe F, et al. Role of dihydropyrimidine dehydrogenase inhibitory fluoropyrimidine against non-small cell lung cancer – in correlation with the tumoral expression of thymidylate synthase and dihydropyrimidine dehydrogenase. Lung Cancer. 2005;49(1):47–54. doi: 10.1016/j.lungcan.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 10.Vescio RA, Redfern CH, Nelson TJ, Ugoretz S, Stern PH, Hoffman RM. In vivo-like drug responses of human tumors growing in three-dimensional gel-supported primary culture. PNAS. 1987;84(14):5029–5033. doi: 10.1073/pnas.84.14.5029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Furukawa T, Kubota T, Hoffman RM. Clinical applications of the histoculture drug response assay. Clin Cancer Res. 1995;1:305–311. [PubMed] [Google Scholar]
- 12.Fukui Y, Oka T, Nagayama S, Danenberg PV, Danenberg KD, Fukushima M. Thymidylate synthase, dihydropyrimidine dehydrogenase, orotate phosphoribosyltransferase mRNA and protein expression levels in solid tumors in large scale population analysis. Int J Mol Med. 2008;22(6):709–716. doi: 10.3892/ijmm_00000076. [DOI] [PubMed] [Google Scholar]
- 13.Lord RVN, Salonga D, Danenberg KD, et al. Telomerase reverse transcriptase expression is increased early in the Barrett’s metaplasia, dysplasia, adenocarcinoma sequence. J Gastrointestinal Surg. 2000;4(2):135–142. doi: 10.1016/S1091-255X(00)80049-9. [DOI] [PubMed] [Google Scholar]
- 14.Botelho NK, Schneiders FL, Lord SJ, et al. Gene expression alterations in formalin-fixed paraffin-embedded Barrett esophagus and esophageal adenocarcinoma tissues. Can Biol Ther. 2010;10(2):172–179. doi: 10.4161/cbt.10.2.12166. [DOI] [PubMed] [Google Scholar]
- 15.Matsubara J, Nishina T, Yamada Y, et al. Impacts of excision repair cross-complementing gene 1 (ERCC1), dihydropyrimidine dehydrogenase, and epidermal growth factor receptor on the outcomes of patients with advanced gastric cancer. Brit J Cancer. 2008;98(4):832–839. doi: 10.1038/sj.bjc.6604211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cho RC, Cole PD, Sohn K-J, et al. Effects of folate and folylpolyglutamyl synthase modulation on chemosensitivity of breast cancer cells. Mol Cancer Ther. 2007;6(11):2909–2919. doi: 10.1158/1535-7163.MCT-07-0449. [DOI] [PubMed] [Google Scholar]
- 17.Schweitzer BI, Dicker AP, Bertino JR. Dihydrofolate reductase as a therapeutic target. FASEB J. 1990 doi: 10.1096/fasebj.4.8.2185970. [DOI] [PubMed] [Google Scholar]
- 18.Konishi Y, Kanamaru R, Ishioka C, et al. The changes in the levels of dihydrofolate reductase mRNA and its gene dosage in 5-fluorouracil-resistant L1210 cells. Tohoku J Exp Med. 1990;161(1):33–42. doi: 10.1620/tjem.161.33. [DOI] [PubMed] [Google Scholar]
- 19.Danenberg PV, Gustavsson B, Johnston P, et al. Folates as adjuvants to anticancer agents: chemical rationale and mechanism of action. Crit Rev Oncology Hematol. 2016;106:118–131. doi: 10.1016/j.critrevonc.2016.08.001. [DOI] [PubMed] [Google Scholar]
- 20.Eguchi K, Oyama T, Tajima A, et al. Intratumoral gene expression of 5-fluorouracil pharmacokinetics-related enzymes in stage I and II non-small cell lung cancer patients treated with uracil-tegafur after surgery: a prospective multi-institutional study in Japan. Lung Cancer. 2015;87(1):53–8. doi: 10.1016/j.lungcan.2014.10.013. [DOI] [PubMed] [Google Scholar]
- 21.Nakano J, Huang C, Liu D, et al. Evaluations of biomarkers associated with 5-FU sensitivity for non-small-cell lung cancer patients postoperatively treated with UFT. Br J Cancer. 2006;95:607–615. doi: 10.1038/sj.bjc.660329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Grimminger PP, Schneider PM, Metzger R, et al. Low thymidylate synthase, thymidine phosphorylase, and dihydropyrimidine dehydrogenase mRNA expression correlate with prolonged survival in resected non-small-cell lung cancer. Clin Lung Cancer. 2010;11(5):328–334. doi: 10.3816/CLC.2010.n.041. [DOI] [PubMed] [Google Scholar]
- 23.Ishikawa Y, Kubota T, Otani Y, et al. Dihydropyrimidine dehydrogenase and messenger RNA Levels in Gastric Cancer: Possible Predictor for Sensitivity to 5-Fluorouracil. Jpn J Cancer Res. 2000;91(1):105–112. doi: 10.1111/j.1349-7006.2000.tb00866.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Edler D, Glimelius B, Hallström M, Jakobsen A, Johnston PG, Magnusson I. Thymidylate synthase expression in colorectal cancer: a prognostic and predictive marker of benefit from adjuvant fluorouracil-based chemotherapy. J Clin Oncol. 2002;20(7):1721–28. doi: 10.1200/JCO.2002.07.039. [DOI] [PubMed] [Google Scholar]
- 25.Takeda M, Okamoto I, Hirabayashi N, Kitano M, Nakagawa K. Thymidylate synthase and dihydropyrimidine dehydrogenase expression levels are associated with response to S-1 plus carboplatin in advanced non-small cell lung cancer. Lung Cancer. 2011;73(1):103–109. doi: 10.1016/j.lungcan.2010.10.022. [DOI] [PubMed] [Google Scholar]
- 26.Takechi T, Okabe H, Ikeda K, et al. Correlations between antitumor activities of fluoropyrimidines and DPD activity in lung tumor xenografts. Oncol Rep. 2005;14(1):33–39. doi: 10.3892/or.14.1.33. [DOI] [PubMed] [Google Scholar]
- 27.Saif MW, Rosen LS, Saito K, Zergebel C, Ravage-Mass LD. A Phase I Study Evaluating the Effect of CDHP as a Component of S-1 on the Pharmacokinetics of 5-Fluorouracil. Anticancer Res. 2011;31:625–632. [PubMed] [Google Scholar]
- 28.Yoshimasu T, Oura S, Ohta F, Hashimoto S, Kawago M, Okamura Y. Epidermal growth factor receptor mutations are associated with docetaxel sensitivity in lung cancer. J Thoracic Oncol. 2011;6(10):1658–1662. doi: 10.1097/JTO.0b013e318221f71a. [DOI] [PubMed] [Google Scholar]
- 29.Tanahashi M, Niwa H, Yukiue H, Suzuki E, Haneda H, Yoshii N. Adjuvant chemotherapy based on the in vitro histoculture drug response assay for non-small cell lung cancer improves survival. J Thoracic Oncology. 2010;5(9):1376–1381. doi: 10.1097/JTO.0b013e3181e7d035. [DOI] [PubMed] [Google Scholar]
- 30.Ohashi T, Yoshimasu T, Oura S, et al. Class III Beta-tubulin expression in non-small cell lung cancer: a predictive factor for paclitaxel response. Anticancer Res. 2015;35:2669–2674. [PubMed] [Google Scholar]
- 31.Kubota T, Sasano N, Abe O, et al. Potential of the histoculture drug-response assay to contribute to cancer patient survival. Clin Cancer Res. 1995;1(12):1537–43. [PubMed] [Google Scholar]
- 32.Yoon YS, Kim CW, Roh SA, et al. Applicability of histoculture drug response assays in colorectal cancer chemotherapy. Anticancer Res. 2012;32:3581–3586. http://ar.iiarjournals.org/content/32/8/3581.full.pdf+html [PubMed]
- 33.Nakajima T, Yasufuku K, Suzuki M, et al. Thymidylate synthase, dihydropyrimidine dehydrogenase, thymidine phosphorylase, orotate phosphoribosyltransferase mRNA expression in lung cancer metastatic lymph node samples obtained by endobronchial ultrasound-guided transbronchial needle aspiration—a pilot study. Clin Lung Cancer. 2011;12(5):293–297. doi: 10.1016/j.cllc.2011.06.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The dataset is available upon request.
Not applicable.