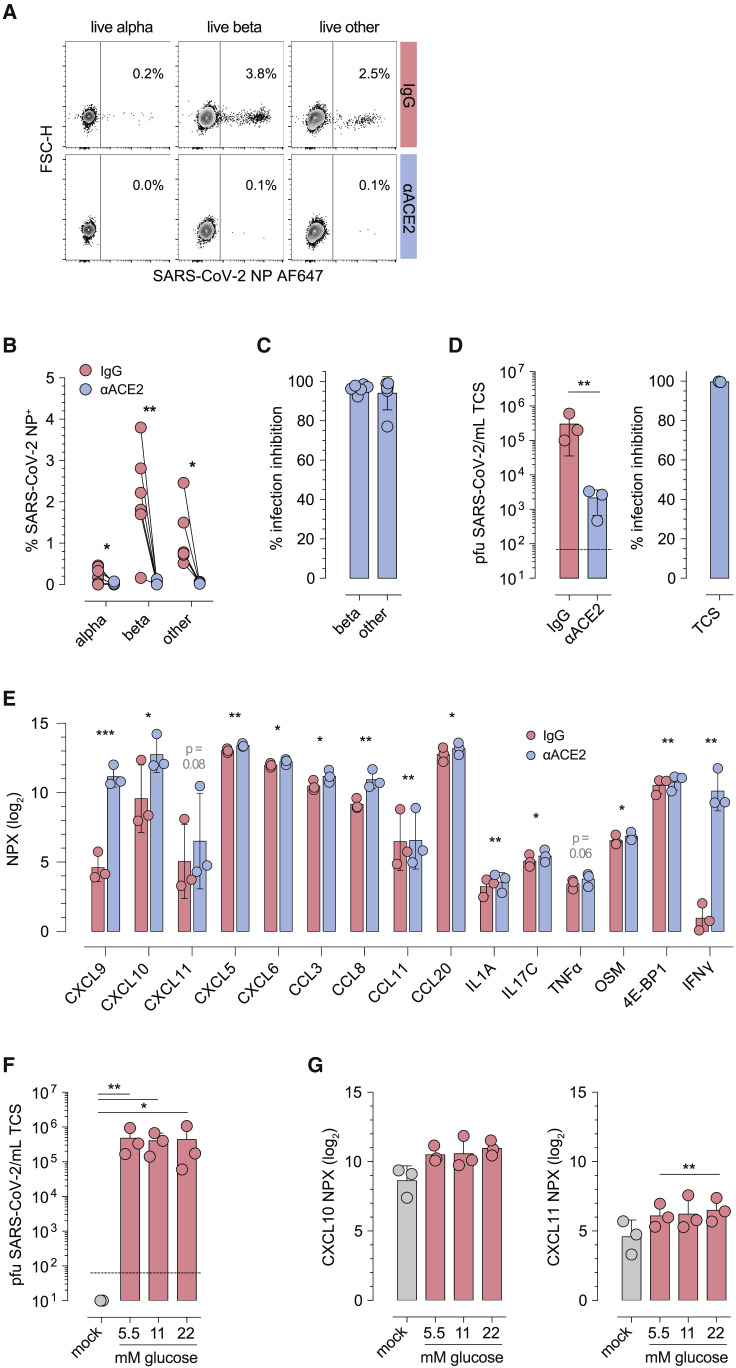
Figure 2.
Stringent ACE2 requirement for pancreatic islet cell infection with SARS-CoV-2
(A) Representative contour plots gated on live α, β, and “other” cells pre-treated with IgG (irrelevant polyclonal goat antibody AF7197) or the anti-ACE2 blocking antibody AF933 prior to SARS-CoV-2 infection (48 h).
(B) Summary of SARS-CoV-2 NP expression by live islet cell subsets as a function of IgG treatment or ACE2 blockade (n = 6 donors).
(C) Percent infection inhibition for β and “other” cells (inhibition for α cells is not shown because the very low extent of α cell infection in IgG-treated cultures for 2 of 6 donors substantially skews such calculations).
(D) Infectious SARS-CoV-2 titers and extent of infection inhibition following ACE2 blockade (n = 3 donors).
(E) Quantification of chemokines and cytokines in UV-inactivated TCS of SARS-CoV-2-infected islet cell cultures under conditions of IgG treatment or ACE2 blockade (48-h infection, n = 3 donors).
(F) Infectious SARS-CoV-2 titers in TCS as a function of glucose concentration in islet culture medium (n = 3 donors).
(G) Quantification of CXCL10 and CXCL11 in TCS as a function of glucose concentration. All summary bar diagrams represent mean ± SD and scatter for the indicated number of donors; statistical analyses were conducted by paired t test or repeated-measures ANOVA with Tukey’s multiple comparisons where applicable (∗, p < 0.05; ∗∗, p < 0.01; ∗∗∗, p < 0.001).
All summary bar diagrams represent mean ± SD and scatter for the indicated number of donors; statistical analyses were conducted by paired t test or repeated-measures ANOVA with Tukey’s multiple comparisons where applicable.