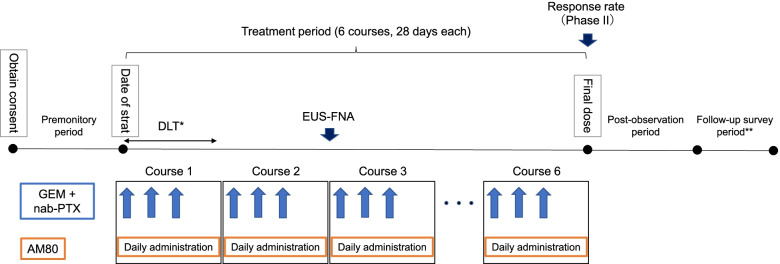
Fig. 2.
Protocol of the present study. In the present study, AM80 will be administered daily for 4 weeks and repeated for up to six courses. Each course consists of gemcitabine (GEM) (1,000 mg/m2) and nab-paclitaxel (nab-PTX) (125 mg/m2) administered intravenously over 30 min on Days 1, 8, and 15 without administration at Week 4. After completing Course 6, if no disease progression is noted based on the Response Evaluation Criteria in Solid Tumors version 1.1 or if the patient has no intolerable toxicity, GEM + nab-PTX will be continued as a usual treatment. In such a case, continuous GEM/nab-PTX will be considered as post-treatment. *Dose-limiting toxicity will be evaluated in a 4-week period only in the phase I trial. **The follow-up period will be set in all cases until the date of completion of the post-observation period (cut-off) in all cases