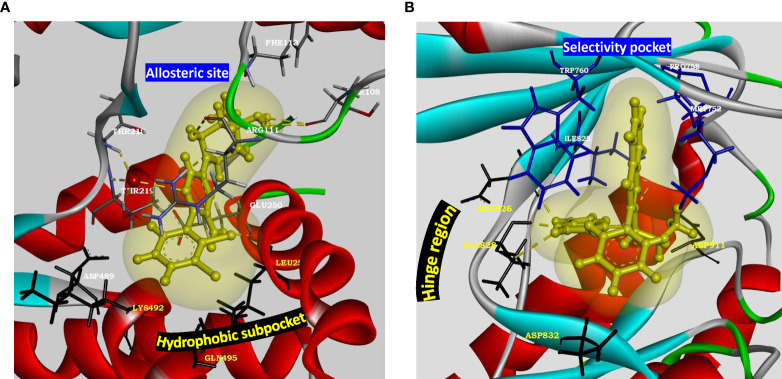
Figure 4.
Structural parameters of SHP2 and PI3Kδ for drug design. (A) Characterization of allosteric site present in SHP2 (PDB: 5EHR). Key residues of allosteric site (Thr218, Arg111, Phe113 and Thr108) are shown to form water mediated hydrogen bonds with the preclinical inhibitor PCC0208023. Hydrophobic subpocket presents residues Leu254 and Gln495 along with Asp489 and Lys492 for novel pharmacophore design and optimization. Allosteric site residues are rendered as sticks colored by element, hydrophobic pocket residues are rendered as black sticks, oxygen atom of water as red ball and inhibitor as yellow ball and stick model with its soft surface in light yellow and hydrogen bond interactions in yellow dashed lines. (B) Characterization of active site of p110 catalytic subunit of PI3Kδ (PDB: 4XE0). Hinge region residues (Glu826, Ala828, Asp832) provide hydrogen bond interactions. Hydrophobic pocket formed by gatekeeper residue Trp760 along with Met752, Pro758 and Ile825 may be targeted for adding selectivity in inhibitor design for delta isoform of PI3K. Kinase domain is rendered as solid ribbon colored according to secondary structure. Hinge region residues are rendered as black sticks, selectivity pocket residues as blue sticks, inhibitor as yellow ball and stick model with its soft surface depicted in light yellow and hydrogen bond interactions as yellow dashed lines.