Fig. 4 |. Activation of SARM1 by NMN.
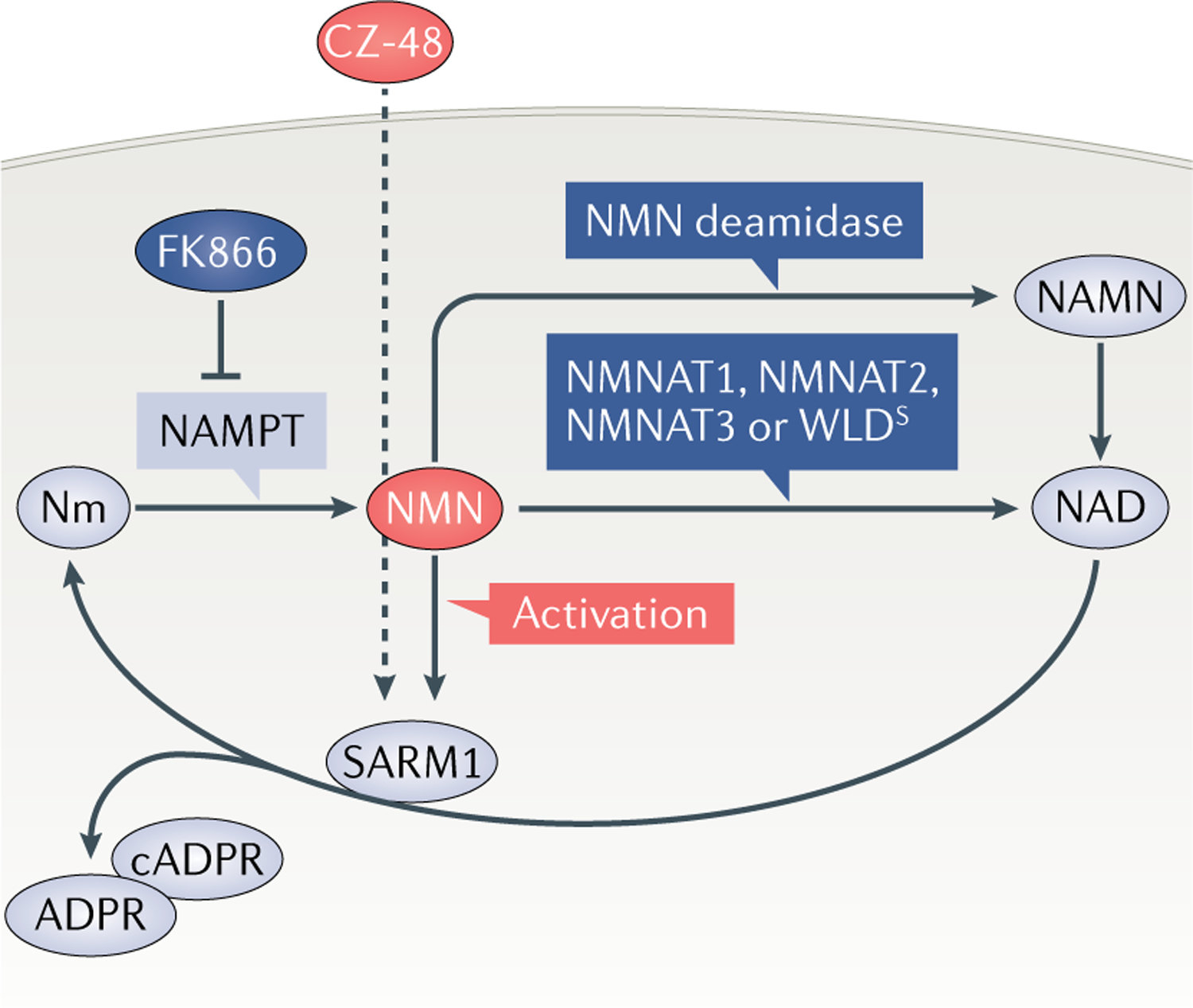
The figure summarizes evidence supporting activation of sterile-α and Toll/interleukin 1 receptor (TIR) motif containing protein 1 (SARM1) by nicotinamide mononucleotide (NMN). NMN accumulates after axon injury22 because the enzyme that normally removes it, nicotinamide mononucleotide adenylyltransferase 2 (NMNAT2), is quickly degraded12. If left unchecked, this activates SARM1, leading to axon degeneration27. However, NMN accumulation can be prevented in three ways (dark blue): through inhibition of its synthetic enzyme, nicotinamide phosphoribosyl transferase (NAMPT)22,29–31; by overexpression, or increased axonal targeting, of any isoform of NMNAT3,10, its normal processing enzyme in mammals; or by ectopic expression of bacterial NMN deamidase22,32. Conversely, its membrane-permeable analogue CZ-48 activates SARM1 directly35. Thus, NMN, or a close analogue, appears to be necessary and sufficient for SARM1 activation. ADPR, ADP-ribose; cADPR, cyclic ADP-ribose; NAD, nicotinamide adenine dinucleotide; NAMN, nicotinic acid mononucleotide; Nm, nicotinamide; WLDS, Wallerian degeneration slow.
